Response of Bread Wheat Genotypes for Drought and Low Nitrogen Stress Tolerance
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plant Materials and Study Sites
2.2. Experimental Design and Crop Establishment
2.2.1. Greenhouse Experiment
2.2.2. Field Experiment
2.3. Data Collection
2.4. Data Analysis
3. Results
3.1. Genotype Responses and Environmental Impact
3.2. Mean Performance of Genotypes
3.3. Correlations between Agronomic Traits and Grain Yield
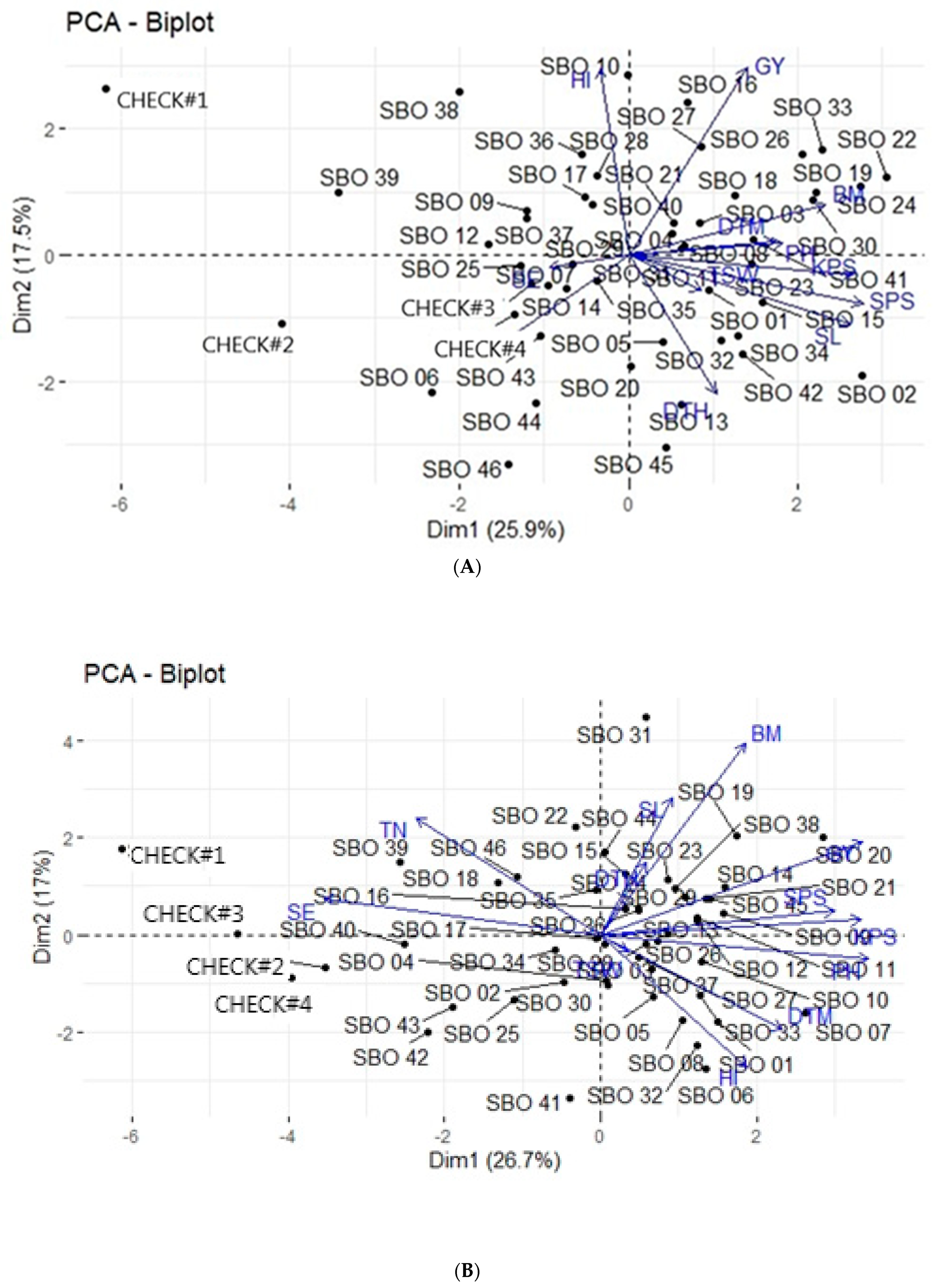
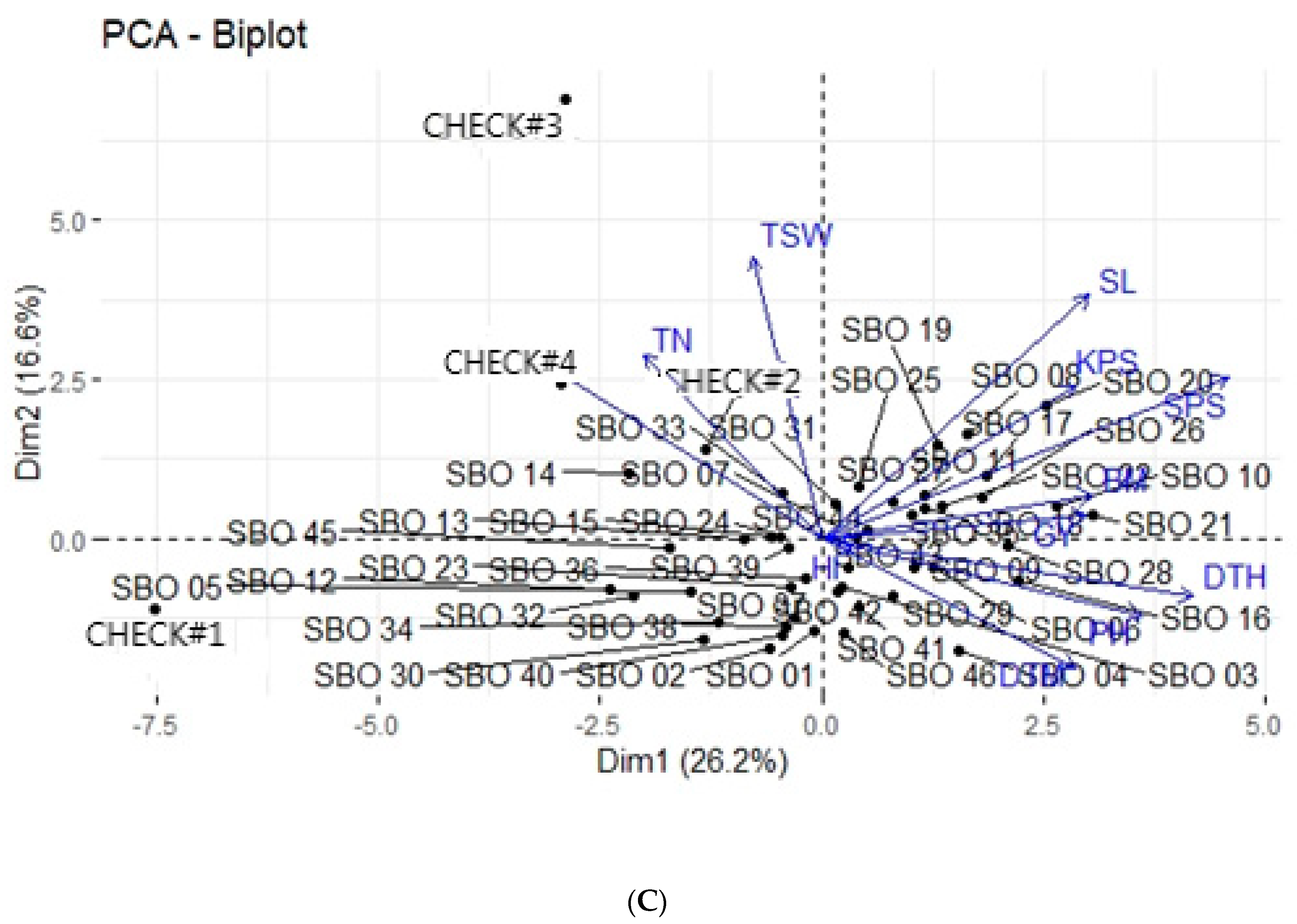
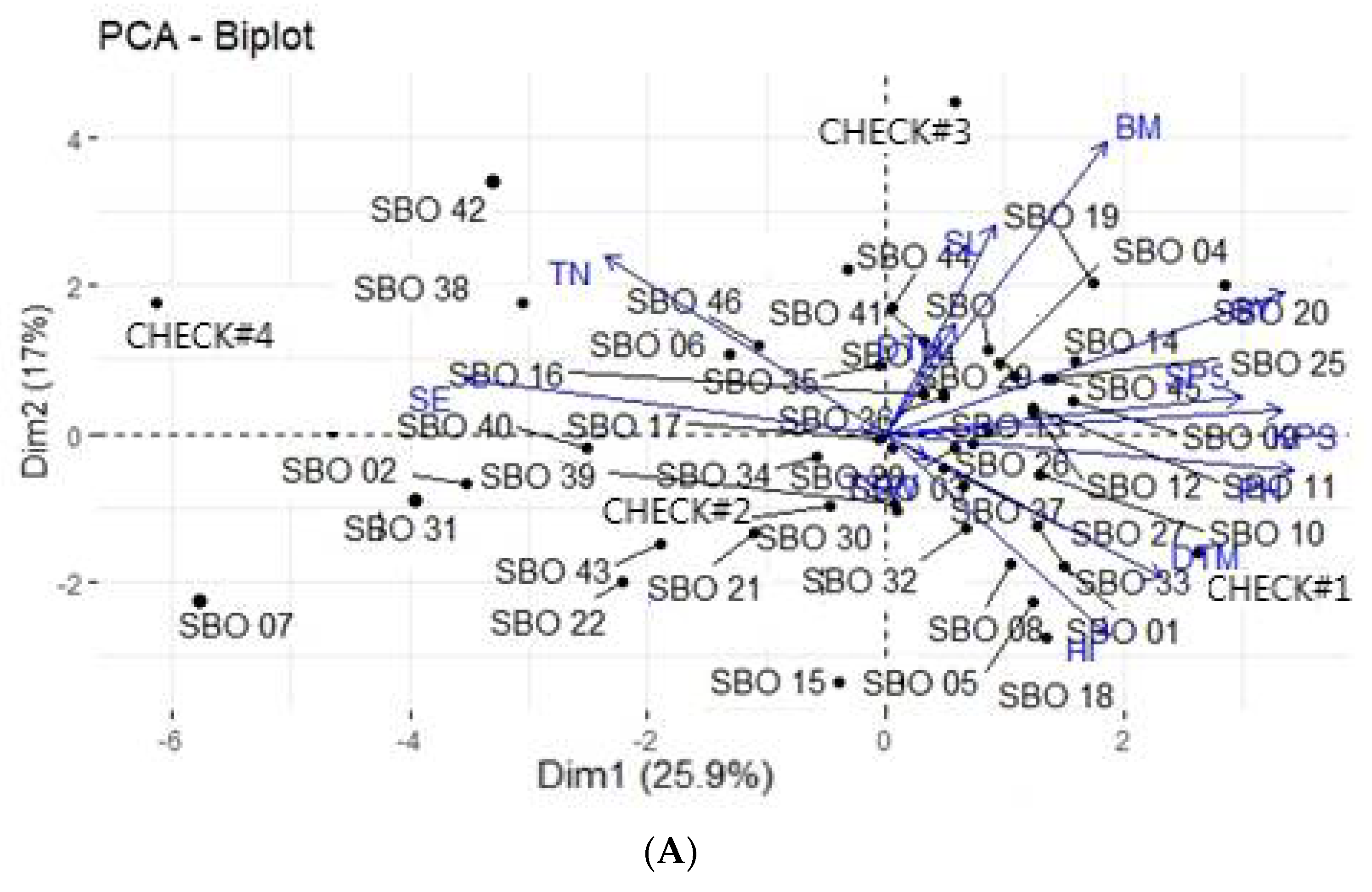
3.4. Principal Component Analysis
3.5. Principal Component Biplot Analysis
4. Discussion
4.1. Effect of Water Regime and Nitrogen Treatment on Genotype Performance and Grain Yield
4.2. Correlation of Agronomic Traits Tested under Variable Water Regimes and Nitrogen Treatments
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tadesse, W.; Bishaw, Z.; Assefa, S. Wheat production and breeding in Sub-Saharan Africa: Challenges and opportunities in the face of climate change. Int. J. Clim. Change Strateg. Manag. 2019, 11, 696–715. [Google Scholar] [CrossRef] [Green Version]
- Zörb, C.; Ludewig, U.; Hawkesford, M.J. Perspective on wheat yield and quality with reduced nitrogen supply. Trends Plant Sci. 2018, 23, 1029–1037. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ullah, M.I.; Mahpara, S.; Bibi, R.; Shah, R.U.; Ullah, R.; Abbas, S.; Ullah, M.I.; Hassan, A.M.; Elshehawi, A.M.; Brestic, M. Grain yield and correlated traits of bread wheat lines: Implications for yield improvement. Saudi J. Biol. Sci. 2021, 28, 5714–5719. [Google Scholar] [CrossRef] [PubMed]
- Lamaoui, M.; Jemo, M.; Datla, R.; Bekkaoui, F. Heat and drought stresses in crops and approaches for their mitigation. Front. Chem. 2018, 6, 26. [Google Scholar] [CrossRef] [PubMed]
- Yadav, M.R.; Choudhary, M.; Singh, J.; Lal, M.K.; Jha, P.K.; Udawat, P.; Gupta, N.K.; Rajput, V.D.; Garg, N.K.; Maheshwari, C. Impacts, Tolerance, Adaptation, and Mitigation of Heat Stress on Wheat under Changing Climates. Int. J. Mol. Sci. 2022, 23, 2838. [Google Scholar] [CrossRef]
- Ahmad, R.; Waraich, E.A.; Ashraf, M.; Ahmad, S.; Aziz, T. Does nitrogen fertilization enhance drought tolerance in sunflower? A review. J. Plant Nutr. 2014, 37, 942–963. [Google Scholar] [CrossRef]
- Abid, M.; Tian, Z.; Ata-Ul-Karim, S.T.; Cui, Y.; Liu, Y.; Zahoor, R.; Jiang, D.; Dai, T. Nitrogen nutrition improves the potential of wheat (Triticum aestivum L.) to alleviate the effects of drought stress during vegetative growth periods. Front. Plant Sci. 2016, 7, 981. [Google Scholar] [CrossRef] [Green Version]
- Haider, I.; Raza, M.A.S.; Iqbal, R.; Aslam, M.U.; Habib-ur-Rahman, M.; Raja, S.; Khan, M.T.; Aslam, M.M.; Waqas, M.; Ahmad, S. Potential effects of biochar application on mitigating the drought stress implications on wheat (Triticum aestivum L.) under various growth stages. J. Saudi Chem. Soc. 2020, 24, 974–981. [Google Scholar] [CrossRef]
- Weber, V.S.; Melchinger, A.E.; Magorokosho, C.; Makumbi, D.; Bänziger, M.; Atlin, G.N. Efficiency of managed-stress screening of elite maize hybrids under drought and low nitrogen for yield under rainfed conditions in Southern Africa. Crop Sci. 2012, 52, 1011–1020. [Google Scholar] [CrossRef] [Green Version]
- Fang, Y.; Du, Y.; Wang, J.; Wu, A.; Qiao, S.; Xu, B.; Zhang, S.; Siddique, K.H.; Chen, Y. Moderate drought stress affected root growth and grain yield in old, modern and newly released cultivars of winter wheat. Front. Plant Sci. 2017, 8, 672. [Google Scholar] [CrossRef] [Green Version]
- Hasanuzzaman, M.; Raihan, M.; Hossain, R.; Masud, A.A.C.; Rahman, K.; Nowroz, F.; Rahman, M.; Nahar, K.; Fujita, M. Regulation of Reactive Oxygen Species and Antioxidant Defense in Plants under Salinity. Int. J. Mol. Sci. 2021, 22, 9326. [Google Scholar] [CrossRef] [PubMed]
- Goufo, P.; Moutinho-Pereira, J.M.; Jorge, T.F.; Correia, C.M.; Oliveira, M.R.; Rosa, E.A.; António, C.; Trindade, H. Cowpea (Vigna unguiculata L. Walp.) metabolomics: Osmoprotection as a physiological strategy for drought stress resistance and improved yield. Front. Plant Sci. 2017, 8, 586. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, S.B.; Suh, M.C. Recent advances in cuticular wax biosynthesis and its regulation in Arabidopsis. Mol. Plant 2013, 6, 246–249. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- López-Maury, L.; Marguerat, S.; Bähler, J. Tuning gene expression to changing environments: From rapid responses to evolutionary adaptation. Nat. Rev. Genet. 2008, 9, 583–593. [Google Scholar] [CrossRef] [PubMed]
- Joshi, R.; Wani, S.H.; Singh, B.; Bohra, A.; Dar, Z.A.; Lone, A.A.; Pareek, A.; Singla-Pareek, S.L. Transcription factors and plants response to drought stress: Current understanding and future directions. Front. Plant Sci. 2016, 7, 1029. [Google Scholar] [CrossRef] [Green Version]
- Savin, R.; Slafer, G.A.; Cossani, C.M.; Abeledo, L.G.; Sadras, V.O. Cereal yield in Mediterranean-type environments: Challenging the paradigms on terminal drought, the adaptability of barley vs. wheat and the role of nitrogen fertilization. In Crop Physiology; Elsevier: LIeida, Spain, 2015; pp. 141–158. [Google Scholar]
- Qaseem, M.F.; Qureshi, R.; Shaheen, H. Effects of pre-anthesis drought, heat and their combination on the growth, yield and physiology of diverse wheat (Triticum aestivum L.) genotypes varying in sensitivity to heat and drought stress. Sci. Rep. 2019, 9, 6955. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zampieri, M.; Ceglar, A.; Dentener, F.; Toreti, A. Wheat yield loss attributable to heat waves, drought and water excess at the global, national and subnational scales. Environ. Res. Lett. 2017, 12, 064008. [Google Scholar] [CrossRef]
- Van der Westhuizen, L.; Trapnell, L. How do we compare with “down under”. In Measuring and Comparing the Competitiveness of South African Wheat Industry with Australia; SA Graan/Grain: Pretoria, South Africa, 2015; pp. 51–57. [Google Scholar]
- Sosibo, N.Z.; Muchaonyerwa, P.; Visser, L.; Barnard, A.; Dube, E.; Tsilo, T.J. Soil fertility constraints and yield gaps of irrigation wheat in South Africa. S. Afr. J. Sci. 2017, 113, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Leip, A.; Billen, G.; Garnier, J.; Grizzetti, B.; Lassaletta, L.; Reis, S.; Simpson, D.; Sutton, M.A.; De Vries, W.; Weiss, F. Impacts of European livestock production: Nitrogen, sulphur, phosphorus and greenhouse gas emissions, land-use, water eutrophication and biodiversity. Environ. Res. Lett. 2015, 10, 115004. [Google Scholar] [CrossRef]
- Mhike, X.; Okori, P.; Magorokosho, C.; Ndlela, T. Validation of the use of secondary traits and selection indices for drought tolerance in tropical maize (Zea mays L.). Afr. J. Plant Sci. 2012, 6, 96–102. [Google Scholar]
- Nazir, M.; Pandey, R.; Siddiqi, T.O.; Ibrahim, M.M.; Qureshi, M.I.; Abraham, G.; Vengavasi, K.; Ahmad, A. Nitrogen-deficiency stress induces protein expression differentially in low-N tolerant and low-N sensitive maize genotypes. Front. Plant Sci. 2016, 7, 298. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shahrasbi, S.; Emam, Y.; Ronaghi, A.; Pirasteh-Anosheh, H. Effect of drought stress and nitrogen fertilizer on grain yield and agronomic nitrogen use efficiency of wheat (Triticum aestivum L. cv. Sirvan) in Fars Province, Iran conditions. Iran. J. Crop Sci. 2015, 17, 349–363. [Google Scholar]
- Abdel-Motagally, F.; El-Zohri, M. Improvement of wheat yield grown under drought stress by boron foliar application at different growth stages. J. Saudi Soc. Agric. Sci. 2018, 17, 178–185. [Google Scholar] [CrossRef] [Green Version]
- Swain, E.Y.; Rempelos, L.; Orr, C.H.; Hall, G.; Chapman, R.; Almadni, M.; Stockdale, E.A.; Kidd, J.; Leifert, C.; Cooper, J.M. Optimizing nitrogen use efficiency in wheat and potatoes: Interactions between genotypes and agronomic practices. Euphytica 2014, 199, 119–136. [Google Scholar] [CrossRef]
- Hawkesford, M.J. Reducing the reliance on nitrogen fertilizer for wheat production. J. Cereal Sci. 2014, 59, 276–283. [Google Scholar] [CrossRef] [Green Version]
- Nehe, A.; Misra, S.; Murchie, E.; Chinnathambi, K.; Foulkes, M. Genetic variation in N-use efficiency and associated traits in Indian wheat cultivars. Field Crops Res. 2018, 225, 152–162. [Google Scholar] [CrossRef]
- Banziger, M.; Diallo, A. Progress in developing drought and N stress tolerant maize cultivars for eastern and southern Africa. In Proceedings of the 7th Eastern and Southern Africa Regional Maize Conference, Nairobi, Kenya, 5–11 February 2001. [Google Scholar]
- Nawaz, F.; Ahmad, R.; Waraich, E.; Naeem, M.; Shabbir, R. Nutrient uptake, physiological responses, and yield attributes of wheat (Triticum aestivum L.) exposed to early and late drought stress. J. Plant Nutr. 2012, 35, 961–974. [Google Scholar] [CrossRef]
- Naeem, M.K.; Ahmad, M.; Kamran, M.; Shah, M.K.N.; Iqbal, M.S. Physiological responses of wheat (Triticum aestivum L.) to drought stress. Int. J. Plant Soil Sci. 2015, 6, 1–9. [Google Scholar] [CrossRef]
- Pour-Aboughadareh, A.; Ahmadi, J.; Mehrabi, A.A.; Etminan, A.; Moghaddam, M.; Siddique, K.H. Physiological responses to drought stress in wild relatives of wheat: Implications for wheat improvement. Acta Physiol. Plant. 2017, 39, 106. [Google Scholar] [CrossRef]
- Lv, X.; Ding, Y.; Long, M.; Liang, W.; Gu, X.; Liu, Y.; Wen, X. Effect of Foliar Application of Various Nitrogen Forms on Starch Accumulation and Grain Filling of Wheat (Triticum aestivum L.) Under Drought Stress. Front. Plant Sci. 2021, 12, 463. [Google Scholar] [CrossRef]
- Shi, J.; Yasuor, H.; Yermiyahu, U.; Zuo, Q.; Ben-Gal, A. Dynamic responses of wheat to drought and nitrogen stresses during re-watering cycles. Agric. Water Manag. 2014, 146, 163–172. [Google Scholar] [CrossRef]
- Ahmad, Z.; Waraich, E.A.; Akhtar, S.; Anjum, S.; Ahmad, T.; Mahboob, W.; Hafeez, O.B.A.; Tapera, T.; Labuschagne, M.; Rizwan, M. Physiological responses of wheat to drought stress and its mitigation approaches. Acta Physiol. Plant. 2018, 40, 80. [Google Scholar] [CrossRef]
- Astaneh, N.; Bazrafshan, F.; Zare, M.; Amiri, B.; Bahrani, A. Nano-fertilizer prevents environmental pollution and improves physiological traits of wheat grown under drought stress conditions. Sci. Agropecu. 2021, 12, 41–47. [Google Scholar] [CrossRef]
- Monneveux, P.; Jing, R.; Misra, S. Phenotyping for drought adaptation in wheat using physiological traits. Front. Physiol. 2012, 3, 429. [Google Scholar] [CrossRef] [Green Version]
- Passioura, J. Phenotyping for drought tolerance in grain crops: When is it useful to breeders? Funct. Plant Biol. 2012, 39, 851–859. [Google Scholar] [CrossRef]
- Wasson, A.; Richards, R.; Chatrath, R.; Misra, S.; Prasad, S.S.; Rebetzke, G.; Kirkegaard, J.; Christopher, J.; Watt, M. Traits and selection strategies to improve root systems and water uptake in water-limited wheat crops. J. Exp. Bot. 2012, 63, 3485–3498. [Google Scholar] [CrossRef] [Green Version]
- Farooq, M.; Hussain, M.; Siddique, K.H. Drought stress in wheat during flowering and grain-filling periods. Crit. Rev. Plant Sci. 2014, 33, 331–349. [Google Scholar] [CrossRef]
- Hendriks, P.-W.; Kirkegaard, J.; Lilley, J.M.; Gregory, P.; Rebetzke, G. A tillering inhibition gene influences root–shoot carbon partitioning and pattern of water use to improve wheat productivity in rainfed environments. J. Exp. Bot. 2016, 67, 327–340. [Google Scholar] [CrossRef]
- Zahra, N.; Wahid, A.; Hafeez, M.B.; Ullah, A.; Siddique, K.H.; Farooq, M. Grain development in wheat under combined heat and drought stress: Plant responses and management. Environ. Exp. Bot. 2021, 188, 104517. [Google Scholar] [CrossRef]
- Payne, R.; Murray, D.; Harding, S. An Introduction to the GenStat Command Language; VSN International: Hemel Hempstead, UK, 2011. [Google Scholar]
- Tadesse, Z.; Habtemariam Zegeye, D.A.; Tafesse Solomon, Y.S.; Debele, T. Identification of Stable Bread Wheat (Triticum Aestivum L.) Genotypes using AMMI Analysis in Ethiopia. Int. J. Res. Agric. For. 2018, 5, 6–14. [Google Scholar]
- Yan, W.; Kang, M.S.; Ma, B.; Woods, S.; Cornelius, P.L. GGE biplot vs. AMMI analysis of genotype-by-environment data. Crop Sci. 2007, 47, 643–653. [Google Scholar] [CrossRef]
- Duma, S.W.; Shimelis, H.; Ramburan, S.; Shayanowako, A.I. Genotype-by-region interactions of released sugarcane varieties for cane yield in the South African sugar industry. J. Crop Improv. 2019, 33, 478–504. [Google Scholar] [CrossRef]
- Anwaar, H.A.; Perveen, R.; Mansha, M.Z.; Abid, M.; Sarwar, Z.M.; Aatif, H.M.; ud din Umar, U.; Sajid, M.; Aslam, H.M.U.; Alam, M.M. Assessment of grain yield indices in response to drought stress in wheat (Triticum aestivum L.). Saudi J. Biol. Sci. 2020, 27, 1818–1823. [Google Scholar] [CrossRef] [PubMed]
- Akcura, M.; Partigoç, F.; Kaya, Y. Evaluating of drought stress tolerance based on selection indices in Turkish bread wheat landraces. J. Anim. Plant Sci. 2011, 21, 700–709. [Google Scholar]
- Mäder, P.; Hahn, D.; Dubois, D.; Gunst, L.; Alföldi, T.; Bergmann, H.; Oehme, M.; Amadò, R.; Schneider, H.; Graf, U. Wheat quality in organic and conventional farming: Results of a 21 year field experiment. J. Sci. Food Agric. 2007, 87, 1826–1835. [Google Scholar] [CrossRef]
- Balouchi, H. Screening wheat parents of mapping population for heat and drought tolerance, detection of wheat genetic variation. Int. J. Nucl. Quantum Eng. 2010, 4, 63–73. [Google Scholar]
- Sallam, A.; Mourad, A.M.; Hussain, W.; Stephen Baenziger, P. Genetic variation in drought tolerance at seedling stage and grain yield in low rainfall environments in wheat (Triticum aestivum L.). Euphytica 2018, 214, 169. [Google Scholar] [CrossRef]
- Mwadzingeni, L.; Shimelis, H.; Tesfay, S.; Tsilo, T.J. Screening of bread wheat genotypes for drought tolerance using phenotypic and proline analyses. Front. Plant Sci. 2016, 7, 1276. [Google Scholar] [CrossRef] [Green Version]
- Barraclough, P.B.; Howarth, J.R.; Jones, J.; Lopez-Bellido, R.; Parmar, S.; Shepherd, C.E.; Hawkesford, M.J. Nitrogen efficiency of wheat: Genotypic and environmental variation and prospects for improvement. Eur. J. Agron. 2010, 33, 1–11. [Google Scholar] [CrossRef]
- Wang, R.; An, D.; Hu, C.; Li, L.; Zhang, Y.; Jia, Y.; Tong, Y. Relationship between nitrogen uptake and use efficiency of winter wheat grown in the North China Plain. Crop Pasture Sci. 2011, 62, 504–514. [Google Scholar] [CrossRef]
- Hawkesford, M.J. Genetic variation in traits for nitrogen use efficiency in wheat. J. Exp. Bot. 2017, 68, 2627–2632. [Google Scholar] [CrossRef] [PubMed]
- Sinha, S.K.; Kumar, A.; Tyagi, A.; Venkatesh, K.; Paul, D.; Singh, N.K.; Mandal, P.K. Root architecture traits variation and nitrate-influx responses in diverse wheat genotypes under different external nitrogen concentrations. Plant Physiol. Biochem. 2020, 148, 246–259. [Google Scholar] [CrossRef] [PubMed]
- Nehe, A.; Misra, S.; Murchie, E.; Chinnathambi, K.; Tyagi, B.S.; Foulkes, M. Nitrogen partitioning and remobilization in relation to leaf senescence, grain yield and protein concentration in Indian wheat cultivars. Field Crops Res. 2020, 251, 107778. [Google Scholar] [CrossRef] [PubMed]
- Ma, C.; Xie, P.; Zhang, K.; Yang, J.; Li, X.; Liu, F.; Lin, L.; Zhang, H. Contribution of the flag leaf to lead absorption in wheat grain at the grain-filling stage. Ecotoxicol. Environ. Saf. 2021, 225, 112722. [Google Scholar] [CrossRef] [PubMed]
- Mirosavljević, M.; Momčilović, V.; Mikić, S.; Trkulja, D.; Brbaklić, L.; Zorić, M.; Abičić, I. Changes in stay-green and nitrogen use efficiency traits in historical set of winter barley cultivars. Field Crops Res. 2020, 249, 107740. [Google Scholar] [CrossRef]
- Anderegg, J.; Aasen, H.; Perich, G.; Roth, L.; Walter, A.; Hund, A. Temporal trends in canopy temperature and greenness are potential indicators of late-season drought avoidance and functional stay-green in wheat. Field Crops Res. 2021, 274, 108311. [Google Scholar] [CrossRef]
- Bagrei, B.; Bybordi, A. Yield and yield components in bread wheat (Triticum aestivum L.) under non-stress and drought stress conditions. Int. J. Biosci. 2015, 6, 338–348. [Google Scholar]
- Foulkes, M.; Sylvester-Bradley, R.; Weightman, R.; Snape, J. Identifying physiological traits associated with improved drought resistance in winter wheat. Field Crops Res. 2007, 103, 11–24. [Google Scholar] [CrossRef]
- Ullah, H.; Santiago-Arenas, R.; Ferdous, Z.; Attia, A.; Datta, A. Improving water use efficiency, nitrogen use efficiency, and radiation use efficiency in field crops under drought stress: A review. Adv. Agron. 2019, 156, 109–157. [Google Scholar]
- Mojaddam, M. Effect of drought stress on physiological characteristics and seed yield of sunflower at different levels of nitrogen. Electron. J. Crop Prod. 2017, 9, 121–136. [Google Scholar]
- Basal, O.; Szabó, A. The combined effect of drought stress and nitrogen fertilization on soybean. Agronomy 2020, 10, 384. [Google Scholar] [CrossRef] [Green Version]
- Sedri, M.H.; Amini, A.; Golchin, A. Evaluation of nitrogen effects on yield and drought tolerance of rainfed wheat using drought stress indices. J. Crop Sci. Biotechnol. 2019, 22, 235–242. [Google Scholar] [CrossRef]
- Feiziasl, V.; Fotovat, A.; Astarae, A.; Lakzian, A.; Mousavi, S. Effect of optimized nitrogen application in reducing drought stress effect on grain yield of some rainfed bread wheat genotypes. Seed Plant Prod. J. 2014, 30–32, 169–198. [Google Scholar]
- Alvar-Beltrán, J.; Dao, A.; Dalla Marta, A.; Saturnin, C.; Casini, P.; Sanou, J.; Orlandini, S. Effect of drought, nitrogen fertilization, temperature and photoperiodicity on quinoa plant growth and development in the Sahel. Agronomy 2019, 9, 607. [Google Scholar] [CrossRef] [Green Version]
- Dodig, D.; Zorić, M.; Kandić, V.; Perović, D.; Šurlan-Momirović, G. Comparison of responses to drought stress of 100 wheat accessions and landraces to identify opportunities for improving wheat drought resistance. Plant Breed. 2012, 131, 369–379. [Google Scholar] [CrossRef]
- Sareen, S.; Tyagi, B.S.; Sarial, A.K.; Tiwari, V.; Sharma, I. Trait analysis, diversity, and genotype x environment interaction in some wheat landraces evaluated under drought and heat stress conditions. Chil. J. Agric. Res. 2014, 74, 135–142. [Google Scholar] [CrossRef] [Green Version]
- Blum, A. Plant Breeding for Water-Limited Environments; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2010. [Google Scholar]
- Khakwani, A.A.; Dennett, M.; Munir, M.; Abid, M. Growth and yield response of wheat varieties to water stress at booting and anthesis stages of development. Pak. J. Bot. 2012, 44, 879–886. [Google Scholar]
- Liu, Y.; Zhang, J.; Hu, Y.-G.; Chen, J. Dwarfing genes Rht4 and Rht-B1b affect plant height and key agronomic traits in common wheat under two water regimes. Field Crops Res. 2017, 204, 242–248. [Google Scholar] [CrossRef] [Green Version]
- Mursalova, J.; Akparov, Z.; Ojaghi, J.; Eldarov, M.; Belen, S.; Gummadov, N.; Morgounov, A. Evaluation of drought tolerance of winter bread wheat genotypes underdrip irrigation and rain-fed conditions. Turk. J. Agric. For. 2015, 39, 817–824. [Google Scholar] [CrossRef]
- Ballesta, P.; Mora, F.; Del Pozo, A. Association mapping of drought tolerance indices in wheat: QTL-rich regions on chromosome 4A. Sci. Agric. 2020, 77, e20180153. [Google Scholar] [CrossRef] [Green Version]
- Paul, K.; Pauk, J.; Deák, Z.; Sass, L.; Vass, I. Contrasting response of biomass and grain yield to severe drought in Cappelle Desprez and Plainsman V wheat cultivars. PeerJ 2016, 4, e1708. [Google Scholar] [CrossRef] [PubMed]
- Pennacchi, J.P.; Carmo-Silva, E.; Andralojc, P.J.; Feuerhelm, D.; Powers, S.J.; Parry, M.A. Dissecting wheat grain yield drivers in a mapping population in the UK. Agronomy 2018, 8, 94. [Google Scholar] [CrossRef] [Green Version]
- Thompson, J.P.; Clewett, T.G. Impacts of root-lesion nematode (Pratylenchus thornei) on plant nutrition, biomass, grain yield and yield components of susceptible/intolerant wheat cultivars determined by nematicide applications. Agronomy 2021, 11, 296. [Google Scholar] [CrossRef]
- Leilah, A.; Al-Khateeb, S. Statistical analysis of wheat yield under drought conditions. J. Arid. Environ. 2005, 61, 483–496. [Google Scholar] [CrossRef]
- Khodadadi, M.; Fotokian, M.H.; Miransari, M. Genetic diversity of wheat (Triticum aestivum L.) genotypes based on cluster and principal component analyses for breeding strategies. Aust. J. Crop Sci. 2011, 5, 17–24. [Google Scholar]
- Beheshtizadeh, H.; Rezaie, A.; Rezaie, A.; Ghandi, A. Principal component analysis and determination of the selection criteria in bread wheat (Triticum aestivum L.) genotypes. Int. J. Agric. Crop Sci. 2013, 5, 2024. [Google Scholar]
- Marcińska, I.; Czyczyło-Mysza, I.; Skrzypek, E.; Filek, M.; Grzesiak, S.; Grzesiak, M.T.; Janowiak, F.; Hura, T.; Dziurka, M.; Dziurka, K. Impact of osmotic stress on physiological and biochemical characteristics in drought-susceptible and drought-resistant wheat genotypes. Acta Physiol. Plant. 2013, 35, 451–461. [Google Scholar] [CrossRef] [Green Version]

| Entry Code | Pedigree/Name |
|---|---|
| Genotypes from CIMMYT’s Drought and Heat Nurseries | |
| SBO01 | ACHTAR*3//KANZ/KS85-8-5/4/MILAN/KAUZ//PRINIA/3/BAV92/5/MILAN/KAUZ//PRINIA/3/BAV92 |
| SBO02 | MILAN/KAUZ//PRINIA/3/BAV92/5/TRAP#1/BOW//VEE#5/SARA/3/ZHE JIANG 4/4/DUCULA |
| SBO03 | FRET2*2/4/SNI/TRAP#1/3/KAUZ*2/TRAP//KAUZ/5/ONIX |
| SBO04 | ONIX/4/MILAN/KAUZ//PRINIA/3/BAV92 |
| SBO05 | BAU/KAUZ//PASTOR |
| SBO06 | CNO79//PF70354/MUS/3/PASTOR/4/BAV92/5/FRET2/KUKUNA//FRET2/6/MILAN/KAUZ//PRINIA/3/BAV92 |
| SBO07 | CMSA04M00297S-040ZTP0Y-040ZTM-040SY-23ZTM-03Y-0B |
| SBO08 | SOKOLL*2/TROST |
| SBO09 | BUC/MN72253//PASTOR |
| SBO10 | MILAN/KAUZ//PRINIA/3/BABAX |
| SBO11 | SW89-5124*2/FASAN/3/ALTAR 84/AESQ//2*OPATA |
| SBO12 | SOKOLL/ROLF07 |
| SBO13 | ROLF07/3/T.DICOCCON PI94625/AE.SQUARROSA (372)//3*PASTOR |
| SBO14 | HD30/5/CNDO/R143//ENTE/MEXI75/3/AE.SQ/4/2*OCI |
| SBO15 | RL6043/4*NAC//PASTOR/3/BAV92/4/ATTILA/BAV92//PASTOR |
| SBO16 | CROC_1/AE.SQUARROSA (205)//KAUZ/3/SLVS |
| SBO17 | CROC_1/AE.SQUARROSA (224)//2*OPATA/3/2*RAC655 |
| SBO18 | GOUBARA-1/2*SOKOLL |
| SBO19 | SW89.5277/BORL95//SKAUZ |
| SBO20 | PBW343 |
| SBO21 | PRL/2*PASTOR |
| SBO22 | MUNAL #1 |
| SBO23 | QUAIU |
| SBO24 | WBLL1*2/BRAMBLING |
| SBO25 | WHEAR//2*PRL/2*PASTOR |
| SBO26 | FRET2/KUKUNA//FRET2/3/YANAC/4/FRET2/KIRITATI |
| SBO27 | YUNMAI 48//2*WBLL1*2/KURUKU |
| SBO28 | ATTILA/3*BCN//BAV92/3/TILHI/4/SHA7/VEE#5//ARIV92 |
| SBO29 | PRL/2*PASTOR*2//SKAUZ/BAV92 |
| SBO30 | C80.1/3*BATAVIA//2*WBLL1/3/ATTILA/3*BCN*2//BAV92/4/WBLL1*2/KURUKU |
| SBO31 | ATTILA*2/HUITES//FINSI/3/ATTILA*2/PBW65 |
| SBO32 | ATTILA*2//CHIL/BUC*2/3/KUKUNA |
| SBO33 | D67.2/P66.270//AE.SQUARROSA (320)/3/CUNNINGHAM |
| SBO34 | CNDO/R143//ENTE/MEXI_2/3/AEGILOPS SQUARROSA (TAUS)/4/WEAVER/5/2*FRAME |
| SBO35 | WBLL1//UP2338*2/VIVITSI |
| SBO36 | WBLL1*2/4/SNI/TRAP#1/3/KAUZ*2/TRAP//KAUZ/5/KACHU |
| SBO37 | HUW234+LR34/PRINIA*2//YANAC |
| SBO38 | SAUAL/3/MILAN/S87230//BAV92 |
| SBO39 | WBLL1*2/VIVITSI/6/CNDO/R143//ENTE/MEXI_2/3/AEGILOPS SQUARROSA |
| SBO40 | (TAUS)/4/WEAVER/5/2*JANZ |
| SBO41 | BABAX/3/PRL/SARA//TSI/VEE#5/4/CROC_1/AE.SQUARROSA (224)//2*OPATA |
| SBO42 | SW94.60002/4/KAUZ*2//DOVE/BUC/3/KAUZ/5/SW91-12331 |
| SBO43 | FRET2/KUKUNA//FRET2/3/PASTOR//HXL7573/2*BAU/5/FRET2*2/4/SNI/TRAP#1/3/KAUZ*2/TRAP//KAUZ |
| SBO44 | ROLF07/TUKURU/5/WBLL1*2/4/YACO/PBW65/3/KAUZ*2/TRAP//KAUZ |
| SBO45 | ROLF07/YANAC//TACUPETO F2001/BRAMBLING |
| SBO46 | FRET2/KUKUNA//FRET2/3/PARUS/5/FRET2*2/4/SNI/TRAP#1/3/KAUZ*2/TRAP//KAUZ |
| Leading commercial spring wheat cultivars as per the recommendation of the NWCET | |
| Check#1 | Local with PBR |
| Check#2 | Local with PBR |
| Check#3 | Local with PBR |
| Check#4 | Local with PBR |
| Soil Properties | Greenhouse Soil | Field Soil |
|---|---|---|
| Bulk density | 0.73 | 1.21 |
| pH (KCL) | 5.11 | 4.67 |
| Nitrogen (%) | 0.45 | 0.21 |
| Phosphorus (mg/L) | 125.00 | 38.00 |
| Potassium (mg/L) | 275.00 | 239.00 |
| Magnesium (mg/L) | 400.00 | 301.00 |
| Calcium (mg/L) | 1874.00 | 1378.00 |
| Organic carbon (%) | 5.43 | 3.21 |
| Clay (%) | 17.00 | 29.00 |
| Mean temperature (°C) | 26.89 | 21.54 |
| Month | Tmax (°C) | Tmin (°C) | RHmax (%) | Rhmin (%) | Rs (MJ/m2) | ET0 (mm) |
|---|---|---|---|---|---|---|
| March | 28.03 | 18.70 | 99.77 | 63.74 | 18.63 | 111.75 |
| April | 27.40 | 17.91 | 89.30 | 52.01 | 19.09 | 126.11 |
| May | 26.47 | 17.09 | 95.81 | 59.12 | 18.35 | 119.44 |
| June | 26.08 | 16.73 | 97.18 | 50.65 | 17.30 | 102.80 |
| July | 26.66 | 15.51 | 87.11 | 44.68 | 16.54 | 93.19 |
| August | 28.30 | 17.98 | 90.36 | 61.76 | 19.62 | 120.77 |
| Source of Variation | DF | Traits | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| SE | DTH | DTM | PH | TN | SL | SPS | KPS | TSW | GY | BM | HI | ||
| Incomplete block | 1 | 0.54 | 94.23 | 101.04 | 9.10 | 269.54 | 7.54 | 5.89 | 68.11 | 4.63 | 89.78 | 124.53 | 0.04 |
| Replication (Rep) | 1 | 0.09 | 7.36 | 12.81 | 1.47* | 1127.14 | 0.04 * | 16.49 | 4.42 * | 15.66 | 248.52 | 10,870.30 | 0.07 |
| Genotype (Gen) | 49 | 11.53 *** | 35.44 *** | 79.58 *** | 214.41 *** | 50.91 *** | 10.67 *** | 7.12 *** | 73.81 *** | 4.85 *** | 155.82 *** | 716.10 *** | 0.02 ** |
| Environment (Env) | 1 | 0.03 * | 187.23 | 1708.85 ** | 2144.74 ** | 333.91 | 1.27 ** | 7.64 * | 947.43 | 6.01 ** | 96,250.49 ** | 278,121.20 ** | 1.34 |
| Water Regime (WR) | 1 | 0.13 | 104.43 | 371.85 ** | 1650.48 ** | 2643.30 | 9.56 ** | 9.85 * | 155.51 * | 1.41 | 11,688.27 ** | 31,382.60 ** | 0.32 ** |
| Nitrogen rate (NT) | 2 | 19.57 | 1255.76 ** | 562.12 ** | 642.29 *** | 4503.29 ** | 46.49 *** | 30.09 ** | 123.14 | 28.78 * | 3615.80 ** | 76,065.60 *** | 0.47 ** |
| Gen*Env | 49 | 0.03 ** | 34.59 ** | 48.94 ** | 64.30 ** | 35.39 ** | 9.39 * | 3.78 ** | 32.75 ** | 1.75 * | 142.13 ** | 789.40 ** | 0.09 ** |
| Gen*WR | 49 | 1.28 ** | 3.72 | 41.66 * | 53.01 ** | 35.01 | 8.68 * | 3.41 * | 51.96 | 5.08 ** | 60.10 ** | 451.70 ** | 0.08 * |
| Gen*NT | 98 | 1.88 ** | 31.01 ** | 30.03 ** | 39.55 ** | 29.63 * | 5.41 ** | 3.72 | 43.29 * | 2.70 ** | 70.65 ** | 341.10 ** | 0.01 ** |
| Gen*Env*WR | 49 | 0.03 | 6.08 ** | 35.57 | 32.22 | 34.77 ** | 8.85 | 2.77 | 36.81 ** | 1.44 | 45.40 ** | 408.80 * | 0.01 |
| Gen*Env*NT | 98 | 0.03 | 18.26 ** | 24.91 ** | 37.75 ** | 29.66 | 4.99* | 3.73 *** | 34.07 ** | 4.56 | 64.76 ** | 375.20 ** | 0.02 ** |
| Gen*WR*NT | 98 | 1.39 ** | 7.76 ** | 25.68 ** | 39.50 ** | 27.70 ** | 5.22** | 4.03 * | 36.80 ** | 3.07 ** | 65.38 ** | 414.40 ** | 0.02 ** |
| Gen*Env*WR*NT | 98 | 0.03 | 5.68 ** | 27.62 ** | 32.22 ** | 37.70 ** | 5.43 | 3.30 ** | 44.80 ** | 4.35 ** | 63.11 ** | 433.00 ** | 0.12 ** |
| Residual | 679 | 0.70 | 7.70 | 33.08 | 43.48 | 35.10 | 6.29 | 3.38 | 48.06 | 3.94 | 70.92 | 447.20 | 0.01 |
| Genotype | SE | DTH | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| NS | DS | NS | DS | |||||||||||
| L | I | R | L | I | R | Mean | L | I | R | L | I | R | Mean | |
| Top ten genotypes | ||||||||||||||
| SBO 19 | 8.17 | 7.00 | 7.00 | 7.50 | 8.00 | 7.67 | 7.56 | 61.50 | 63.00 | 60.50 | 59.00 | 61.00 | 60.50 | 60.92 |
| SBO 16 | 7.33 | 8.00 | 8.00 | 8.00 | 6.50 | 7.00 | 7.47 | 63.30 | 61.50 | 59.00 | 59.00 | 62.00 | 59.00 | 60.63 |
| SBO 22 | 7.83 | 7.50 | 7.50 | 6.50 | 8.50 | 8.00 | 7.64 | 61.33 | 58.50 | 65.50 | 61.00 | 60.00 | 65.50 | 61.97 |
| SBO 04 | 7.83 | 7.50 | 7.50 | 6.00 | 7.50 | 7.33 | 7.28 | 62.83 | 58.50 | 63.50 | 61.50 | 60.50 | 63.50 | 61.72 |
| SBO 33 | 7.17 | 8.00 | 8.00 | 7.50 | 7.50 | 7.33 | 7.58 | 62.50 | 64.00 | 64.50 | 56.00 | 58.50 | 64.50 | 61.67 |
| SBO 24 | 7.17 | 8.50 | 8.00 | 6.00 | 6.50 | 7.14 | 7.22 | 60.33 | 59.50 | 66.00 | 59.00 | 61.50 | 66.00 | 62.06 |
| SBO 18 | 7.00 | 7.50 | 6.50 | 8.50 | 7.00 | 7.83 | 7.39 | 63.83 | 63.50 | 66.50 | 58.00 | 62.50 | 66.50 | 63.47 |
| SBO 03 | 7.50 | 6.50 | 8.00 | 6.50 | 7.50 | 7.33 | 7.22 | 63.00 | 63.50 | 63.50 | 58.00 | 62.50 | 63.50 | 62.33 |
| SBO 26 | 7.16 | 6.50 | 8.50 | 7.50 | 7.00 | 7.00 | 7.28 | 60.83 | 60.00 | 63.00 | 58.00 | 63.50 | 63.00 | 61.39 |
| SBO 27 | 6.33 | 8.00 | 8.00 | 7.50 | 6.50 | 7.33 | 7.28 | 61.66 | 64.00 | 63.50 | 57.00 | 63.50 | 63.50 | 62.19 |
| Bottom five genotypes | ||||||||||||||
| Check#1 | 10.00 | 7.50 | 8.00 | 9.67 | 9.50 | 8.50 | 8.86 | 57.50 | 61.50 | 65.50 | 50.66 | 50.00 | 62.00 | 60.36 |
| Check#4 | 7.67 | 8.00 | 7.50 | 7.67 | 8.00 | 7.50 | 7.72 | 61.17 | 61.00 | 64.81 | 53.60 | 61.50 | 64.50 | 62.30 |
| Check#2 | 9.50 | 6.50 | 7.50 | 9.17 | 9.50 | 7.50 | 8.28 | 59.66 | 62.50 | 64.50 | 52.00 | 58.50 | 63.50 | 61.01 |
| SBO 46 | 7.17 | 9.00 | 8.00 | 6.67 | 8.00 | 6.50 | 7.56 | 62.83 | 63.50 | 66.00 | 54.50 | 58.50 | 63.00 | 62.72 |
| Check#3 | 9.67 | 7.50 | 8.50 | 9.16 | 7.50 | 7.00 | 8.22 | 58.50 | 63.50 | 66.50 | 51.80 | 54.00 | 63.50 | 60.42 |
| Mean | 7.51 | 7.41 | 7.20 | 7.32 | 7.50 | 7.31 | 7.38 | 61.57 | 63.65 | 64.15 | 55.88 | 62.07 | 63.28 | 56.98 |
| SED | 0.88 | 0.74 | 0.84 | 0.73 | 0.74 | 0.64 | 0.76 | 3.14 | 4.21 | 3.86 | 2.45 | 2.74 | 3.50 | 3.32 |
| LSD (5%) | 1.73 | 2.01 | 1.84 | 1.83 | 1.94 | 1.36 | 1.79 | 6.18 | 5.45 | 6.62 | 4.82 | 4.05 | 4.12 | 5.21 |
| C.V (%) | 11.70 | 12.63 | 11.12 | 12.38 | 13.16 | 12.50 | 12.25 | 5.10 | 4.25 | 5.45 | 4.01 | 4.67 | 5.52 | 4.83 |
| Genotype | DTM | PH | ||||||||||||
| NS | DS | NS | DS | |||||||||||
| L | I | R | L | I | R | Mean | L | I | R | L | I | R | Mean | |
| Top ten genotypes | ||||||||||||||
| SBO 19 | 96.00 | 93.00 | 94.00 | 89.50 | 92.50 | 94.50 | 93.25 | 80.41 | 83.23 | 86.60 | 82.86 | 75.50 | 86.22 | 82.47 |
| SBO 16 | 94.50 | 94.50 | 95.00 | 93.50 | 97.50 | 95.50 | 93.25 | 82.81 | 81.53 | 78.88 | 78.33 | 87.85 | 81.85 | 81.88 |
| SBO 22 | 93.33 | 105.00 | 115.50 | 91.00 | 91.50 | 95.50 | 91.47 | 80.11 | 84.25 | 85.80 | 83.45 | 79.42 | 82.47 | 82.58 |
| SBO 04 | 90.50 | 94.50 | 98.00 | 95.66 | 91.50 | 100.50 | 91.94 | 86.67 | 80.68 | 78.80 | 78.94 | 86.82 | 83.77 | 82.61 |
| SBO 33 | 95.33 | 90.50 | 105.50 | 93.17 | 91.00 | 95.00 | 91.92 | 84.68 | 81.08 | 87.18 | 83.27 | 84.12 | 84.95 | 84.21 |
| SBO 24 | 93.33 | 100.00 | 97.00 | 96.17 | 98.00 | 91.50 | 94.33 | 82.54 | 86.22 | 76.66 | 73.90 | 81.50 | 82.75 | 80.60 |
| SBO 18 | 93.83 | 89.50 | 130.00 | 91.17 | 99.50 | 93.00 | 93.00 | 83.56 | 80.15 | 85.65 | 81.83 | 79.42 | 85.33 | 82.66 |
| SBO 03 | 94.00 | 100.50 | 93.00 | 94.33 | 94.50 | 110.00 | 92.56 | 84.69 | 82.57 | 82.05 | 80.36 | 82.27 | 76.57 | 81.42 |
| SBO 26 | 97.66 | 95.00 | 120.50 | 95.33 | 105.50 | 100.50 | 94.75 | 84.25 | 83.40 | 86.80 | 81.89 | 82.15 | 80.35 | 83.14 |
| SBO 27 | 96.83 | 91.00 | 92.50 | 94.33 | 87.00 | 93.00 | 92.44 | 79.60 | 83.40 | 80.17 | 81.63 | 76.60 | 85.72 | 81.19 |
| Bottom five genotypes | ||||||||||||||
| Check#1 | 90.49 | 91.25 | 92.52 | 85.33 | 92.55 | 94.45 | 91.22 | 67.05 | 75.45 | 75.22 | 69.13 | 76.23 | 76.66 | 76.42 |
| Check#4 | 93.49 | 96.50 | 97.20 | 90.67 | 92.50 | 86.41 | 93.30 | 83.58 | 76.28 | 81.85 | 76.14 | 81.53 | 85.65 | 80.84 |
| Check#2 | 92.66 | 92.00 | 94.00 | 91.83 | 91.50 | 94.05 | 92.34 | 72.98 | 79.67 | 82.47 | 70.02 | 84.25 | 85.05 | 79.07 |
| SBO 46 | 93.50 | 95.50 | 93.08 | 92.00 | 94.50 | 97.15 | 94.62 | 79.25 | 79.50 | 83.77 | 75.32 | 80.68 | 86.80 | 80.89 |
| Check#3 | 94.00 | 93.50 | 94.50 | 87.83 | 91.00 | 95.34 | 92.20 | 77.72 | 82.75 | 84.95 | 72.62 | 81.08 | 86.17 | 80.88 |
| Mean | 94.14 | 97.53 | 118.25 | 92.03 | 96.17 | 99.56 | 93.40 | 81.53 | 81.09 | 86.75 | 79.18 | 81.24 | 84.33 | 81.85 |
| SED | 5.85 | 5.25 | 4.44 | 5.81 | 5.55 | 5.80 | 5.45 | 6.58 | 5.15 | 6.04 | 6.87 | 6.30 | 5.56 | 6.08 |
| LSD (5%) | 11.50 | 12.41 | 11.00 | 11.43 | 11.45 | 11.60 | 11.57 | 12.96 | 12.78 | 11.47 | 13.52 | 12.56 | 12.70 | 12.67 |
| C.V (%) | 6.21 | 5.45 | 6.52 | 6.24 | 5.62 | 6.33 | 6.06 | 8.10 | 7.95 | 7.88 | 8.67 | 7.19 | 8.12 | 7.99 |
| Genotype | TN | SL | ||||||||||||
| NS | DS | NS | DS | |||||||||||
| L | I | R | L | I | R | Mean | L | I | R | L | I | R | Mean | |
| Top ten genotypes | ||||||||||||||
| SBO 19 | 18.83 | 20.00 | 24.50 | 18.66 | 16.75 | 20.00 | 19.79 | 8.54 | 8.00 | 8.50 | 9.06 | 8.23 | 8.76 | 8.52 |
| SBO 16 | 21.66 | 28.00 | 25.25 | 16.83 | 16.25 | 20.75 | 21.46 | 8.78 | 8.06 | 8.56 | 9.14 | 8.31 | 9.12 | 8.66 |
| SBO 22 | 19.17 | 19.00 | 27.75 | 17.99 | 20.00 | 20.25 | 20.69 | 8.59 | 8.75 | 8.88 | 9.17 | 8.21 | 8.32 | 8.65 |
| SBO 04 | 22.08 | 23.75 | 24.75 | 15.83 | 21.25 | 22.75 | 21.74 | 8.49 | 8.53 | 8.22 | 8.57 | 8.78 | 9.56 | 8.69 |
| SBO 33 | 22.99 | 24.25 | 26.00 | 19.66 | 21.25 | 19.75 | 22.32 | 8.38 | 8.63 | 8.81 | 8.76 | 9.20 | 9.19 | 8.83 |
| SBO 24 | 19.75 | 21.00 | 23.50 | 17.24 | 23.75 | 22.50 | 21.29 | 8.41 | 8.14 | 8.22 | 8.66 | 8.66 | 9.75 | 8.64 |
| SBO 18 | 21.49 | 28.00 | 39.75 | 19.91 | 19.75 | 23.25 | 25.36 | 8.46 | 8.47 | 8.55 | 8.76 | 9.06 | 9.13 | 8.74 |
| SBO 03 | 22.24 | 24.75 | 21.75 | 17.25 | 17.75 | 22.75 | 21.08 | 8.41 | 7.72 | 8.28 | 8.64 | 8.72 | 9.47 | 8.54 |
| SBO 26 | 21.75 | 25.75 | 28.75 | 17.41 | 16.50 | 25.50 | 22.61 | 8.54 | 8.23 | 9.09 | 9.25 | 9.25 | 12.72 | 9.51 |
| SBO 27 | 21.00 | 22.75 | 27.50 | 16.25 | 20.50 | 20.50 | 21.42 | 8.76 | 8.53 | 8.43 | 8.79 | 8.25 | 9.09 | 8.64 |
| Bottom five genotypes | ||||||||||||||
| Check#1 | 20.17 | 22.85 | 21.25 | 23.41 | 20.00 | 24.50 | 22.03 | 7.24 | 8.55 | 9.75 | 7.21 | 8.75 | 9.34 | 8.47 |
| Check#4 | 20.16 | 20.75 | 21.25 | 19.16 | 22.75 | 22.25 | 21.05 | 8.11 | 8.10 | 9.13 | 8.58 | 9.78 | 12.00 | 9.28 |
| Check#2 | 13.00 | 24.25 | 23.75 | 19.75 | 19.89 | 23.75 | 20.73 | 7.99 | 8.25 | 9.47 | 8.81 | 9.09 | 9.18 | 8.80 |
| SBO 46 | 20.91 | 22.75 | 19.75 | 20.41 | 23.00 | 24.50 | 21.89 | 8.63 | 8.33 | 12.72 | 10.41 | 11.01 | 11.25 | 10.39 |
| Check#3 | 20.75 | 22.75 | 17.75 | 21.50 | 20.00 | 24.50 | 21.21 | 8.35 | 8.98 | 9.09 | 9.04 | 9.47 | 9.38 | 9.05 |
| Mean | 21.33 | 23.15 | 30.25 | 18.36 | 21.68 | 22.20 | 22.83 | 8.62 | 8.94 | 9.19 | 8.80 | 7.75 | 9.62 | 8.82 |
| SED | 6.76 | 6.02 | 5.89 | 5.46 | 4.61 | 5.66 | 5.73 | 3.25 | 3.80 | 3.75 | 1.51 | 2.05 | 1.45 | 2.64 |
| LSD (5%) | 13.30 | 14.52 | 13.10 | 10.74 | 11.45 | 11.09 | 12.37 | 6.40 | 5.55 | 6.32 | 2.97 | 1.84 | 2.56 | 4.27 |
| C.V (%) | 31.70 | 29.00 | 30.56 | 29.71 | 30.35 | 29.34 | 30.11 | 37.70 | 34.02 | 37.15 | 17.17 | 16.46 | 17.75 | 26.71 |
| Genotype | SPS | KPS | ||||||||||||
| NS | DS | NS | DS | |||||||||||
| L | I | R | L | I | R | Mean | L | I | R | L | I | R | Mean | |
| Top ten genotypes | ||||||||||||||
| SBO 19 | 15.90 | 15.38 | 16.25 | 16.71 | 15.54 | 16.89 | 16.11 | 28.75 | 32.81 | 33.62 | 37.23 | 37.62 | 29.06 | 33.18 |
| SBO 16 | 16.98 | 16.00 | 16.75 | 16.39 | 15.92 | 16.81 | 16.48 | 35.73 | 32.50 | 33.56 | 34.42 | 35.94 | 33.81 | 34.33 |
| SBO 22 | 16.42 | 16.56 | 17.25 | 16.68 | 16.12 | 15.81 | 16.47 | 32.40 | 33.31 | 33.75 | 33.35 | 33.87 | 34.63 | 33.55 |
| SBO 04 | 16.46 | 16.37 | 16.38 | 15.55 | 16.81 | 15.94 | 16.25 | 34.17 | 34.63 | 29.31 | 31.73 | 37.44 | 42.31 | 34.93 |
| SBO 33 | 15.67 | 16.00 | 17.38 | 16.35 | 17.23 | 16.12 | 16.46 | 32.52 | 33.00 | 34.56 | 34.30 | 34.80 | 37.13 | 34.39 |
| SBO 24 | 15.85 | 15.94 | 16.00 | 17.04 | 16.94 | 16.75 | 16.42 | 31.50 | 32.56 | 25.44 | 35.42 | 39.25 | 37.31 | 33.58 |
| SBO 18 | 16.90 | 17.50 | 15.62 | 16.35 | 16.56 | 15.62 | 16.43 | 32.06 | 32.06 | 32.31 | 31.98 | 31.62 | 39.37 | 33.23 |
| SBO 03 | 15.83 | 15.38 | 15.88 | 16.02 | 17.56 | 17.50 | 16.36 | 33.37 | 29.44 | 33.01 | 32.54 | 30.08 | 32.19 | 31.77 |
| SBO 26 | 15.73 | 15.44 | 16.19 | 16.70 | 16.31 | 16.44 | 16.14 | 32.50 | 31.31 | 34.50 | 35.40 | 31.44 | 38.00 | 33.86 |
| SBO 27 | 15.11 | 15.19 | 18.50 | 16.07 | 15.67 | 16.00 | 16.09 | 30.83 | 33.44 | 33.94 | 33.19 | 27.81 | 34.50 | 32.29 |
| Bottom five genotypes | ||||||||||||||
| Check#1 | 14.62 | 16.00 | 14.75 | 12.70 | 15.12 | 16.94 | 15.02 | 26.35 | 36.69 | 34.56 | 26.58 | 38.62 | 39.31 | 33.69 |
| Check#4 | 16.65 | 17.75 | 15.00 | 14.85 | 16.24 | 18.12 | 16.44 | 30.27 | 33.45 | 25.44 | 28.61 | 35.44 | 39.37 | 32.10 |
| Check#2 | 14.16 | 18.25 | 16.50 | 15.51 | 18.23 | 19.75 | 17.07 | 29.41 | 34.56 | 32.31 | 29.06 | 35.87 | 36.19 | 32.90 |
| SBO 46 | 15.55 | 19.00 | 13.55 | 15.71 | 17.94 | 19.62 | 16.90 | 31.45 | 33.06 | 33.01 | 26.58 | 37.04 | 38.00 | 33.19 |
| Check#3 | 15.37 | 17.00 | 12.75 | 16.55 | 18.56 | 19.50 | 16.62 | 33.78 | 35.44 | 34.50 | 32.33 | 34.98 | 37.50 | 34.76 |
| Mean | 15.86 | 16.50 | 13.45 | 16.04 | 15.89 | 16.51 | 15.71 | 31.95 | 32.25 | 30.05 | 32.67 | 35.55 | 36.00 | 33.08 |
| SED | 1.95 | 1.43 | 1.65 | 1.75 | 1.49 | 1.25 | 1.59 | 6.12 | 6.38 | 5.79 | 7.73 | 7.80 | 6.58 | 6.73 |
| LSD (5%) | 3.84 | 3.61 | 3.56 | 3.45 | 3.25 | 3.15 | 3.48 | 12.04 | 12.48 | 11.84 | 15.21 | 14.95 | 15.74 | 13.71 |
| C.V (%) | 12.29 | 11.56 | 12.45 | 10.91 | 11.47 | 10.93 | 11.60 | 19.14 | 18.74 | 19.65 | 23.64 | 21.36 | 22.36 | 20.82 |
| Genotype | TSW | GY | ||||||||||||
| NS | DS | NS | DS | |||||||||||
| L | I | R | L | I | R | Mean | L | I | R | L | I | R | Mean | |
| Top ten genotypes | ||||||||||||||
| SBO 19 | 37.89 | 54.16 | 54.10 | 47.77 | 43.67 | 52.97 | 43.93 | 134.40 | 234.75 | 339.25 | 132.32 | 233.25 | 336.75 | 234.20 |
| SBO 16 | 39.00 | 44.25 | 53.61 | 44.85 | 49.90 | 53.50 | 44.85 | 134.91 | 231.00 | 337.50 | 127.99 | 235.33 | 313.25 | 232.07 |
| SBO 22 | 43.97 | 43.99 | 44.05 | 33.77 | 53.00 | 53.64 | 43.74 | 136.82 | 234.25 | 337.25 | 129.57 | 226.50 | 325.41 | 232.82 |
| SBO 04 | 35.82 | 44.26 | 53.28 | 46.44 | 51.51 | 52.62 | 46.66 | 132.74 | 237.50 | 337.00 | 127.49 | 234.32 | 305.56 | 233.10 |
| SBO 33 | 33.86 | 54.13 | 53.83 | 45.78 | 48.55 | 51.52 | 45.78 | 130.07 | 231.50 | 343.75 | 128.33 | 235.50 | 325.25 | 232.65 |
| SBO 24 | 44.18 | 44.00 | 63.47 | 33.59 | 33.31 | 43.60 | 43.69 | 133.82 | 236.25 | 340.25 | 126.82 | 224.50 | 335.00 | 232.77 |
| SBO 18 | 38.22 | 54.67 | 44.53 | 39.44 | 38.36 | 47.30 | 42.92 | 128.49 | 232.75 | 340.00 | 126.66 | 226.25 | 331.00 | 230.86 |
| SBO 03 | 34.12 | 45.62 | 53.12 | 34.44 | 43.92 | 50.25 | 43.91 | 137.32 | 231.75 | 338.50 | 125.66 | 226.25 | 310.75 | 231.71 |
| SBO 26 | 43.88 | 44.27 | 54.05 | 33.67 | 54.45 | 63.59 | 44.82 | 133.16 | 234.75 | 339.00 | 125.49 | 225.75 | 331.75 | 231.65 |
| SBO 27 | 44.01 | 53.84 | 54.12 | 33.69 | 43.42 | 53.65 | 43.79 | 130.24 | 232.25 | 337.50 | 125.01 | 232.25 | 330.75 | 230.50 |
| Bottom five genotypes | ||||||||||||||
| Check#1 | 34.36 | 44.44 | 44.60 | 33.22 | 33.59 | 34.62 | 33.97 | 121.50 | 135.50 | 237.75 | 120.92 | 126.25 | 231.50 | 129.74 |
| Check#4 | 32.56 | 44.28 | 44.39 | 33.61 | 34.00 | 43.92 | 34.96 | 123.99 | 132.50 | 234.25 | 120.87 | 125.75 | 230.56 | 127.99 |
| Check#2 | 33.24 | 43.48 | 54.46 | 34.29 | 33.93 | 34.08 | 35.91 | 122.99 | 130.25 | 234.00 | 120.39 | 126.45 | 228.75 | 126.97 |
| SBO 46 | 39.70 | 45.78 | 48.10 | 35.34 | 43.55 | 43.39 | 33.98 | 129.65 | 135.75 | 236.50 | 120.12 | 125.32 | 229.12 | 130.24 |
| Check#3 | 32.82 | 47.63 | 45.45 | 33.51 | 33.62 | 43.98 | 34.17 | 129.32 | 133.75 | 238.00 | 119.66 | 127.50 | 232.07 | 130.05 |
| Mean | 33.87 | 44.52 | 43.95 | 35.81 | 38.84 | 39.99 | 36.16 | 31.05 | 33.25 | 34.00 | 124.81 | 226.31 | 231.00 | 228.45 |
| SED | 0.66 | 0.58 | 0.64 | 2.71 | 2.22 | 2.12 | 1.49 | 8.54 | 7.45 | 8.12 | 8.49 | 7.54 | 8.15 | 8.05 |
| LSD (5%) | 1.30 | 1.25 | 1.68 | 5.34 | 4.85 | 5.23 | 3.28 | 16.81 | 16.05 | 16.58 | 16.72 | 17.25 | 16.50 | 16.65 |
| C.V (%) | 17.07 | 16.75 | 17.53 | 71.17 | 65.30 | 70.00 | 42.97 | 27.50 | 26.00 | 27.60 | 34.24 | 29.65 | 32.44 | 29.57 |
| Genotype | BM | HI | ||||||||||||
| NS | DS | NS | DS | |||||||||||
| L | I | R | L | I | R | Mean | L | I | R | L | I | R | Mean | |
| Top ten genotypes | ||||||||||||||
| SBO 19 | 1273 | 1296 | 1416 | 1181 | 1264 | 1387 | 1284 | 0.46 | 0.50 | 0.37 | 0.41 | 0.46 | 0.52 | 0.45 |
| SBO 16 | 1282 | 1374 | 1670 | 1278 | 1271 | 1359 | 1269 | 0.43 | 0.41 | 0.36 | 0.40 | 0.47 | 0.41 | 0.41 |
| SBO 22 | 1183 | 1392 | 1399 | 1167 | 1286 | 1290 | 1286 | 0.45 | 0.47 | 0.37 | 0.31 | 0.39 | 0.38 | 0.40 |
| SBO 04 | 1182 | 1271 | 1487 | 1280 | 1282 | 1392 | 1282 | 0.40 | 0.66 | 0.48 | 0.34 | 0.48 | 0.38 | 0.46 |
| SBO 33 | 1279 | 1282 | 1517 | 1270 | 1245 | 1384 | 1287 | 0.40 | 0.50 | 0.41 | 0.44 | 0.35 | 0.35 | 0.41 |
| SBO 24 | 1188 | 1274 | 1376 | 1174 | 1295 | 1298 | 1284 | 0.39 | 0.50 | 0.55 | 0.36 | 0.40 | 0.36 | 0.43 |
| SBO 18 | 1273 | 1374 | 1695 | 1177 | 1289 | 1386 | 1282 | 0.39 | 0.46 | 0.44 | 0.35 | 0.48 | 0.32 | 0.41 |
| SBO 03 | 1295 | 1274 | 1612 | 1274 | 1283 | 1395 | 1287 | 0.42 | 0.45 | 0.38 | 0.35 | 0.36 | 0.36 | 0.39 |
| SBO 26 | 1185 | 1395 | 1317 | 1270 | 1210 | 1287 | 1291 | 0.40 | 0.52 | 0.37 | 0.37 | 0.45 | 0.30 | 0.40 |
| SBO 27 | 1176 | 1260 | 1384 | 1273 | 1279 | 1377 | 1275 | 0.39 | 0.51 | 0.46 | 0.34 | 0.41 | 0.38 | 0.42 |
| Bottom five genotypes | ||||||||||||||
| Check#1 | 1173 | 1174 | 1190 | 1166 | 1273 | 1382 | 1176 | 0.37 | 0.60 | 0.47 | 0.34 | 0.50 | 0.46 | 0.46 |
| Check#4 | 1171 | 1172 | 1195 | 1269 | 1279 | 1286 | 1178 | 0.37 | 0.55 | 0.42 | 0.38 | 0.51 | 0.42 | 0.44 |
| Check#2 | 1170 | 1174 | 1189 | 1258 | 1164 | 1375 | 1172 | 0.34 | 0.50 | 0.55 | 0.35 | 0.46 | 0.56 | 0.46 |
| SBO 46 | 1180 | 1184 | 1198 | 1163 | 1364 | 1379 | 1178 | 0.37 | 0.46 | 0.45 | 0.32 | 0.46 | 0.43 | 0.42 |
| Check#3 | 1177 | 1176 | 1187 | 1264 | 1367 | 1273 | 1174 | 0.37 | 0.44 | 0.37 | 0.32 | 0.51 | 0.37 | 0.40 |
| Mean | 1181 | 1275 | 1495 | 1271 | 1276 | 1286 | 1181 | 0.39 | 0.48 | 0.40 | 0.36 | 0.45 | 0.38 | 0.41 |
| SED | 22.14 | 28.13 | 23.10 | 21.08 | 19.20 | 21.69 | 22.56 | 0.10 | 0.16 | 0.12 | 0.12 | 0.184 | 0.14 | 0.14 |
| LSD (5%) | 43.57 | 35.41 | 37.28 | 41.48 | 38.53 | 35.25 | 38.59 | 0.19 | 0.25 | 0.17 | 0.24 | 0.19 | 0.33 | 0.23 |
| C.V. (%) | 27.14 | 26.00 | 28.15 | 29.54 | 28.54 | 32.01 | 28.56 | 25.10 | 16.85 | 24.33 | 33.43 | 22.36 | 24.15 | 24.37 |
| A. | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| NS, Low N (50 kgha−1) | DS, Low N (50 kgha−1) | ||||||||||||
| Traits | SE | DTH | DTM | PH | TN | SL | SPS | KPS | TSW | GY | BM | HI | |
| SE | 1 | 0.024 * | −0.236 * | −0.106 * | 0.032 | −0.093 | 0.158 * | −0.036 | 0.129 ** | −0.332 * | −0.026 | 0.039 * | |
| DTH | 0.020 * | 1 | 0.336 ** | 0.068 ** | 0.614 ** | 0.825 ** | 0.022 | 0.834 ** | 0.658 ** | −0.156 * | 0.289 ** | 0.531 ** | |
| DTM | −0.128 * | 0.558 ** | 1 | −0.188 * | 0.207 ** | 0.225 ** | 0.153 * | 0.234 ** | 0.086 ** | 0.758 * | 0.335 ** | −0.325 * | |
| PH | 0.799 ** | 0.052 | 0.004 * | 1 | −0.211 | 0.173 ** | 0.279 ** | 0.236 ** | 0.161 ** | 0.634 ** | 0.262 ** | 0.051 * | |
| TN | 0.105 ** | 0.757 ** | −0.091 * | −0.159 * | 1 | −0.023 | 0.341 ** | −0.450 ** | 0.096 ** | 0.699 ** | 0.689 ** | 0.571 ** | |
| SL | −0.131 * | 0.029 ** | −0.032 * | 0.958 ** | 0.014 * | 1 | 0.482 ** | 0.425 ** | 0.076 ** | 0.509 ** | 0.188 ** | −0.106 | |
| SPS | −0.258 ** | 0.159 ** | 0.664 ** | 0.104 | 0.526 * | 0.288 ** | 1 | 0.630 ** | 0.516 ** | −0.301 * | 0.149 * | −0.015 * | |
| KPS | −0.183 * | −0.019 | 0.652 * | 0.133 * | −0.169 * | 0.207 ** | 0.457 ** | 1 | 0.106 ** | 0.442 ** | 0.298 ** | 0.078 | |
| TSW | −0.136 * | 0.234 ** | 0.131 ** | 0.173 * | −0.452 ** | 0.019 ** | 0.884 ** | 0.331 ** | 1 | 0.518 * | 0.052 ** | 0.027 * | |
| GY | −0.130 | 0.732 ** | 0.715 ** | 0.625 ** | 0.547 ** | −0.207 ** | 0.637 ** | −0.156 | 0.411 ** | 1 | 0.787 ** | 0.328 ** | |
| BM | 0.067 ** | −0.270 * | −0.199 * | 0.059 * | 0.488 ** | 0.091 | 0.363 ** | −0.140 * | −0.552 ** | 0.842 ** | 1 | −0.282 ** | |
| HI | 0.079 * | −0.057 * | 0.123 ** | 0.092 * | 0.387 ** | 0.094 | 0.052 * | 0.207 ** | 0.271 ** | −0.241 | −0.260 ** | 1 | |
| B. | |||||||||||||
| NS, Intermediate N (100 kgha−1) | DS, Intermediate N (100 kgha−1) | ||||||||||||
| Traits | SE | DTH | DTM | PH | TN | SL | SPS | KPS | TSW | GY | BM | HI | |
| SE | 1 | 0.047 * | −0.251 * | 0.273 ** | 0.127 ** | 0.035 | 0.066 | −0.170 * | 0.012 * | 0.085 * | −0.014 * | 0.101 ** | |
| DTH | −0.121 * | 1 | 0.543 * | 0.103 * | 0.619 * | 0.786 ** | 0.031 | 0.158 * | 0.049 ** | 0.576 ** | 0.580 ** | −0.100 * | |
| DTM | −0.062 * | 0.305 ** | 1 | 0.006 * | 0.152 * | −0.036 * | −0.106 * | 0.034 | 0.107 ** | 0.093 * | 0.013 ** | 0.109 * | |
| PH | −0.215 ** | 0.035 ** | −0.015 * | 1 | 0.153 ** | 0.054 ** | 0.120 ** | 0.176 * | 0.102 ** | −0.161 | −0.091 * | 0.107 ** | |
| TN | 0.191 ** | −0.208 * | −0.031 | 0.150 * | 1 | 0.096 * | −0.037 * | 0.112 ** | 0.046 ** | 0.536 ** | 0.087 ** | 0.151 ** | |
| SL | 0.017 | −0.246 | −0.063 * | 0.069 ** | 0.132 ** | 1 | 0.191 ** | 0.049 * | 0.022 * | 0.046 * | 0.148 * | −0.102 | |
| SPS | −0.050 | 0.083 | −0.082 * | 0.131 ** | −0.013 * | 0.038 ** | 1 | 0.498 ** | 0.079 | 0.125 * | 0.116 | 0.0118 * | |
| KPS | 0.039 * | −0.809 * | −0.025 * | 0.026 ** | 0.216 | 0.077 * | 0.350 ** | 1 | 0.022 ** | −0.236 * | −0.008 * | 0.188 ** | |
| TSW | 0.099 ** | 0.042 * | 0.100 ** | −0.016 * | 0.017 ** | 0.035 | −0.105 * | 0.180 * | 1 | 0.559 ** | 0.213 ** | 0.096 | |
| GY | 0.105 * | −0.037 ** | 0.045 * | −0.056 | 0.648 ** | 0.028 * | 0.538 ** | −0.263 * | 0.543 ** | 1 | 0.749 ** | 0.664 ** | |
| BM | 0.079 ** | −0.204 * | 0.347 ** | 0.085 | 0.005 ** | −0.039 * | 0.005 | 0.183 ** | 0.487 ** | 0.766 ** | 1 | 0.187 ** | |
| HI | 0.045 * | −0.115 | 0.069 * | 0.189 ** | −0.111 * | 0.008 | 0.051 ** | 0.222 ** | 0.167 * | 0.551 ** | 0.082 ** | 1 | |
| C. | |||||||||||||
| NS, Recommended N (200 kgha−1) | DS, Recommended N (200 kgha−1) | ||||||||||||
| Traits | SE | DTH | DTM | PH | TN | SL | SPS | KPS | TSW | GY | BM | HI | |
| SE | 1 | −0.211 ** | −0.198 ** | −0.163 * | 0.233 ** | 0.016 * | −0.145 * | −0.069 * | 0.125 ** | −0.156 * | −0.052 * | −0.031 * | |
| DTH | −0.211 ** | 1 | 0.231 ** | 0.139 * | 0.014 | 0.072 * | 0.084 * | 0.051 ** | 0.028 ** | 0.682 ** | 0.027 * | 0.094 ** | |
| DTM | −0.012* | 0.779 ** | 1 | 0.152 * | −0.124 | 0.057 ** | 0.136 ** | 0.087 ** | 0.036 ** | 0.891 ** | 0.047 ** | −0.016 * | |
| PH | −0.135* | 0.716 ** | 0.044 * | 1 | −0.199 ** | −0.366 * | 0.222 * | −0.074 * | 0.074 ** | 0.059 | 0.077 * | 0.011 ** | |
| TN | 0.044 ** | 0.687 ** | −0.040 * | 0.195 ** | 1 | 0.054 | −0.116 | 0.179 * | 0.063 ** | −0.116 | 0.042 ** | −0.122 | |
| SL | 0.130 ** | 0.074 * | 0.029 ** | −0.141 * | 0.030 | 1 | 0.532 ** | 0.489 ** | 0.056 * | 0.135 ** | 0.138 ** | 0.034 | |
| SPS | 0.086 ** | 0.092 * | 0.018 * | 0.055 ** | 0.054 * | 0.236 ** | 1 | 0.532 ** | 0.118 ** | 0.498 ** | 0.100 * | 0.054 * | |
| KPS | 0.099 ** | 0.013 * | 0.085 ** | 0.095 * | 0.056 * | 0.256 ** | 0.504 ** | 1 | 0.015 ** | 0.150 * | 0.031 | 0.150 * | |
| TSW | −0.110* | 0.029 | 0.158 * | −0.163 * | −0.221 ** | 0.104 * | 0.231 ** | 0.064 * | 1 | −0.264 * | 0.024 ** | −0.085 * | |
| GY | 0.618 ** | −0.247 ** | 0.560 ** | −0.211 ** | 0.130 * | 0.548 ** | 0.761 ** | 0.045 * | 0.706 ** | 1 | 0.541 ** | 0.733 ** | |
| BM | −0.055 * | −0.128 * | 0.094 | −0.074 * | −0.028 * | 0.147 * | 0.213 ** | 0.012 ** | 0.493 * | 0.603 ** | 1 | −0.129 * | |
| HI | 0.087 ** | −0.256 * | 0.166 | −0.225 ** | −0.172 * | 0.053 ** | 0.181 * | 0.053 ** | 0.504 * | 0.673 ** | −0.026 * | 1 | |
| Parameters | Drought-Stressed | Non-stressed | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 50 kg N ha−1 | 100 kg N ha−1 | 200 kg N ha−1 | 50 kg N ha−1 | 100 kg N ha−1 | 200 kg N ha−1 | |||||||
| PC1 | PC2 | PC1 | PC2 | PC1 | PC2 | PC1 | PC2 | PC1 | PC2 | PC1 | PC2 | |
| Eigenvalues | 3.10 | 2.10 | 3.20 | 2.03 | 3.13 | 1.98 | 3.59 | 0.88 | 2.76 | 2.02 | 2.49 | 2.28 |
| Proportion of total variance (%) | 35.89 | 17.51 | 41.70 | 16.96 | 46.15 | 16.57 | 39.98 | 15.71 | 38.02 | 16.83 | 47.77 | 19.04 |
| Cumulative variance (%) | 35.89 | 53.40 | 41.70 | 64.67 | 46.15 | 72.72 | 39.98 | 55.70 | 48.02 | 75.86 | 47.77 | 79.81 |
| SE | −0.10 | −0.12 | 0.78 | −0.17 | 0.64 | 0.70 | 0.95 | −0.12 | 0.39 | −0.16 | 0.54 | 0.36 |
| DTH | 0.61 | −0.30 | 0.64 | −0.40 | 0.79 | −1.18 | 0.51 | −0.24 | 0.92 | 0.12 | 0.71 | −0.19 |
| DTM | 0.45 | −0.18 | −0.19 | −0.13 | 0.40 | 0.04 | 0.66 | −0.11 | −0.22 | 0.62 | −0.21 | −0.03 |
| PH | 0.60 | −0.12 | 0.68 | −0.52 | 0.68 | 0.01 | 0.27 | 0.31 | 0.79 | −0.20 | −0.16 | 0.58 |
| TN | −0.09 | 0.59 | −0.40 | 0.69 | −0.62 | 0.87 | −0.42 | 0.85 | 0.94 | 0.59 | −0.82 | 0.31 |
| SL | 0.75 | 0.31 | −0.12 | 0.85 | 0.58 | 0.76 | −0.30 | 0.67 | −0.38 | −0.29 | 0.67 | 0.28 |
| SPS | 0.73 | −0.15 | 0.96 | 0.21 | 0.91 | −0.15 | 0.67 | −0.01 | 0.70 | 0.11 | −0.10 | −0.04 |
| KPS | 0.64 | −0.35 | 0.83 | −0.19 | −0.34 | −0.24 | 0.76 | 0.52 | −0.41 | −0.77 | 0.62 | −0.17 |
| TSW | −0.18 | −0.12 | −0.17 | 0.26 | −0.54 | −0.32 | 0.56 | −0.11 | 0.88 | −0.46 | 0.41 | 0.62 |
| GY | −0.22 | −0.45 | 0.67 | 0.75 | 0.01 | −0.18 | 0.79 | 0.42 | −0.30 | 0.43 | −0.72 | 0.71 |
| BM | 0.69 | 0.56 | 0.60 | 0.59 | −0.39 | −0.64 | −0.29 | 0.79 | 0.69 | 0.21 | 0.82 | −0.32 |
| HI | −0.28 | 0.71 | −0.33 | 0.78 | −0.11 | 0.06 | −0.41 | −0.30 | 0.04 | −0.04 | 0.90 | −0.46 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Duma, S.; Shimelis, H.; Tsilo, T.J. Response of Bread Wheat Genotypes for Drought and Low Nitrogen Stress Tolerance. Agronomy 2022, 12, 1384. https://doi.org/10.3390/agronomy12061384
Duma S, Shimelis H, Tsilo TJ. Response of Bread Wheat Genotypes for Drought and Low Nitrogen Stress Tolerance. Agronomy. 2022; 12(6):1384. https://doi.org/10.3390/agronomy12061384
Chicago/Turabian StyleDuma, Sbongeleni, Hussein Shimelis, and Toi John Tsilo. 2022. "Response of Bread Wheat Genotypes for Drought and Low Nitrogen Stress Tolerance" Agronomy 12, no. 6: 1384. https://doi.org/10.3390/agronomy12061384
APA StyleDuma, S., Shimelis, H., & Tsilo, T. J. (2022). Response of Bread Wheat Genotypes for Drought and Low Nitrogen Stress Tolerance. Agronomy, 12(6), 1384. https://doi.org/10.3390/agronomy12061384






