Reproductive Biology of Solanum orbiculatum ssp. orbiculatum, an Australian Endemic Bush Tomato
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material
2.2. Floral Structure Study
2.3. Pollen Collection and Storage Conditions
2.4. Pollen Culture in Artificial Media
- A.
- Media composition: Culture media with different components were screened for optimal pollen germination. The components of the pollen germination medium (PGM) were optimised using BK basal medium [9] enriched with different concentrations of sucrose and polyethylene glycol 4000 (PEG). The experiment has been carried out using 6 different sucrose concentrations (0, 5, 10, 15, 20, and 25% w/v) and 3 different concentrations of PEG (0, 2.5, and 5% w/v). The basic BK medium contains 100 mg/L H3BO3, 300 mg/L Ca (NO3)2·4H2O), 200 mg/L MgSO4·7H2O, and 100 mg/L KNO3.
- B.
- Slide preparation: Pollen was collected from the flowers of a single plant the morning after anthesis and distributed on glass slides with one drop of medium placed onto each slide. The slides were inverted and maintained on Petri plates lined with moist filter paper (RH > 95%) for 2 h at 22.5 °C.
- C.
- Scoring of pollen cultures: The responses of cultured pollen grains are assessed as a percentage for pollen germination. A pollen grain is considered germinated when the length of its tube is equal to or more than the diameter of the pollen grain. The pollen grains were scored for germination using arbitrarily selected microscope fields in a completely randomised design, with a factorial treatment structure. Each treatment contained 3 replicates, and 3 fields of view were assessed in each replicate. Pollen grains in at least three arbitrarily selected microscopic fields of each slide were observed after 2 h incubation at 22.5 °C. It was convenient to move the preparation under the microscope in consecutive rows and score one or two arbitrary fields in each row. This would eliminate the probability of scoring the same group of pollen grains again. For each field, the total number of pollen grains and the number of germinated grains were recorded.
- D.
- Pollen hydration: The pollen grains were hydrated by incubation at 22.5 °C inside the brass ring over moist filter paper in a covered container for 15 min.
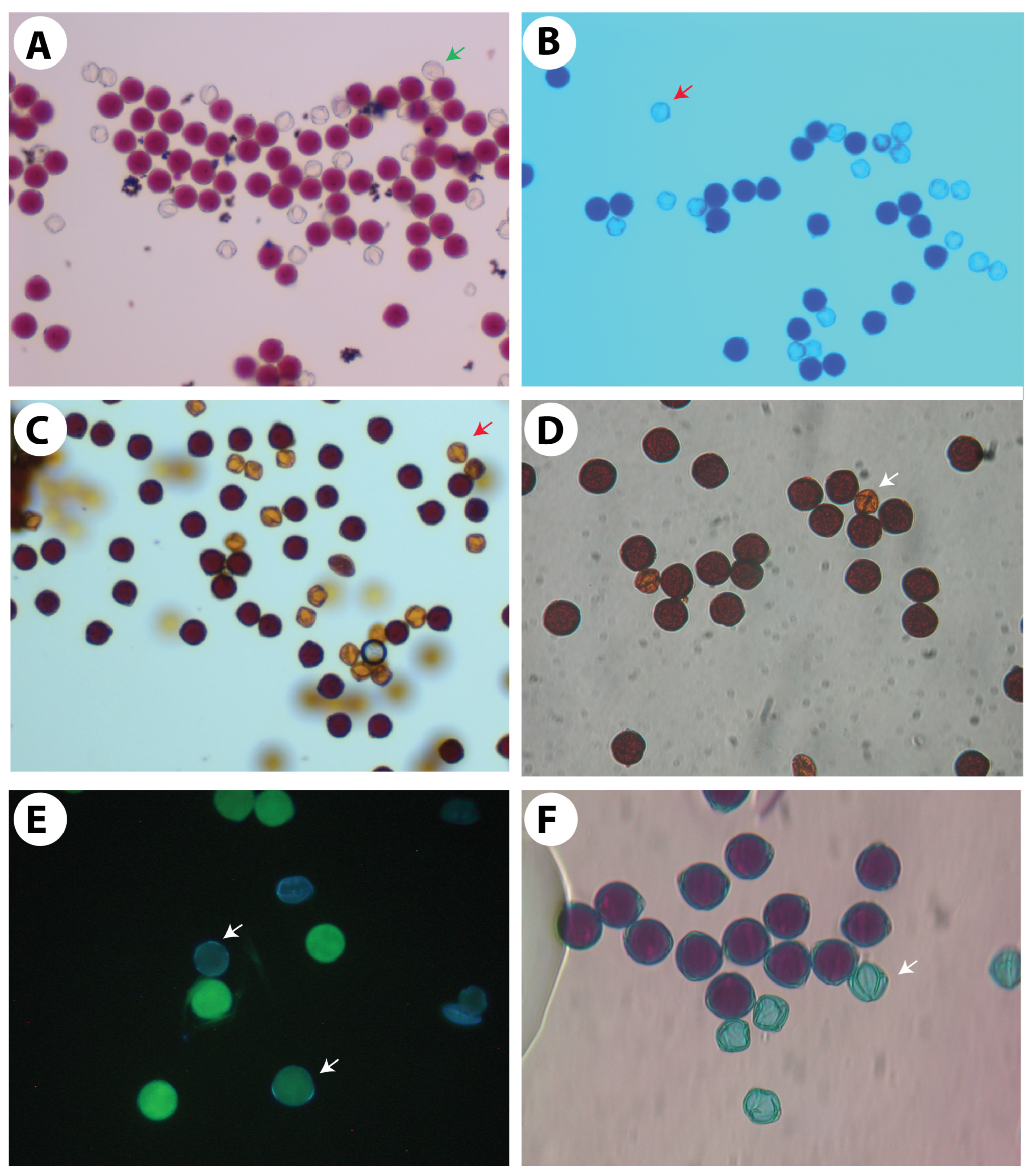
2.5. Pollen Histo-Chemical Tests
- A.
- Cotton blue-lactophenol (0.5%): Hydrated fresh and mature pollen grains were dusted onto a drop of stain on a slide, a cover glass was placed on the smear, and the preparation was warmed for 15 min. The pollen grains were then examined under the high power of a microscope and classified as follows: (1) deeply stained (dark blue, uniform intensity), (2) moderately stained (light blue), and (3) unstained (colourless, shrivelled, often without cytoplasm).
- B.
- Acetocarmine (1%): Pollen grains were dispersed in a drop of stain on a microscope slide, covered with a coverslip, and examined under a light microscope to be scored for stainability. Red-stained pollen grains were considered viable, while light and unstained pollen grains were considered non-viable.
- C.
- I-KI (1% and 2%) solution: The same procedure was carried out as that for acetocarmine. Darkly stained pollen grains were considered viable, whereas light-brown-coloured and unstained pollen grains were considered non-viable.
- D.
- Fluorochromatic reaction test (FCR): The test media were prepared using the rapid semi-empirical method described by Heslop-Harrison et al. [10]. Fluorescein diacetate was made up as a stock solution in acetone at 2 mg/mL. Immediately before use, dilutions of 1% were prepared in 25% sucrose solution. The pollen samples were dispersed in a drop of this medium on a microscope slide, and the fluorochromatic reaction was allowed to proceed for 10 min at room temperature under a covered Petri dish lined with moistened filter paper before viewing with fluorescence microscopy using a Leica DMIL compound microscope. The viable pollen grains fluoresced bright yellow-green under ultraviolet light.
- E.
- Alexander’s stain: Alexander’s stain [11] has been used as a differential stain to differentiate between aborted and non-aborted pollen grains. Pollen grains have been incubated on a slide in a drop of the stain and incubated for at least one hour in the dark at 22.5 °C before viewing under the compound microscope, as mentioned above.
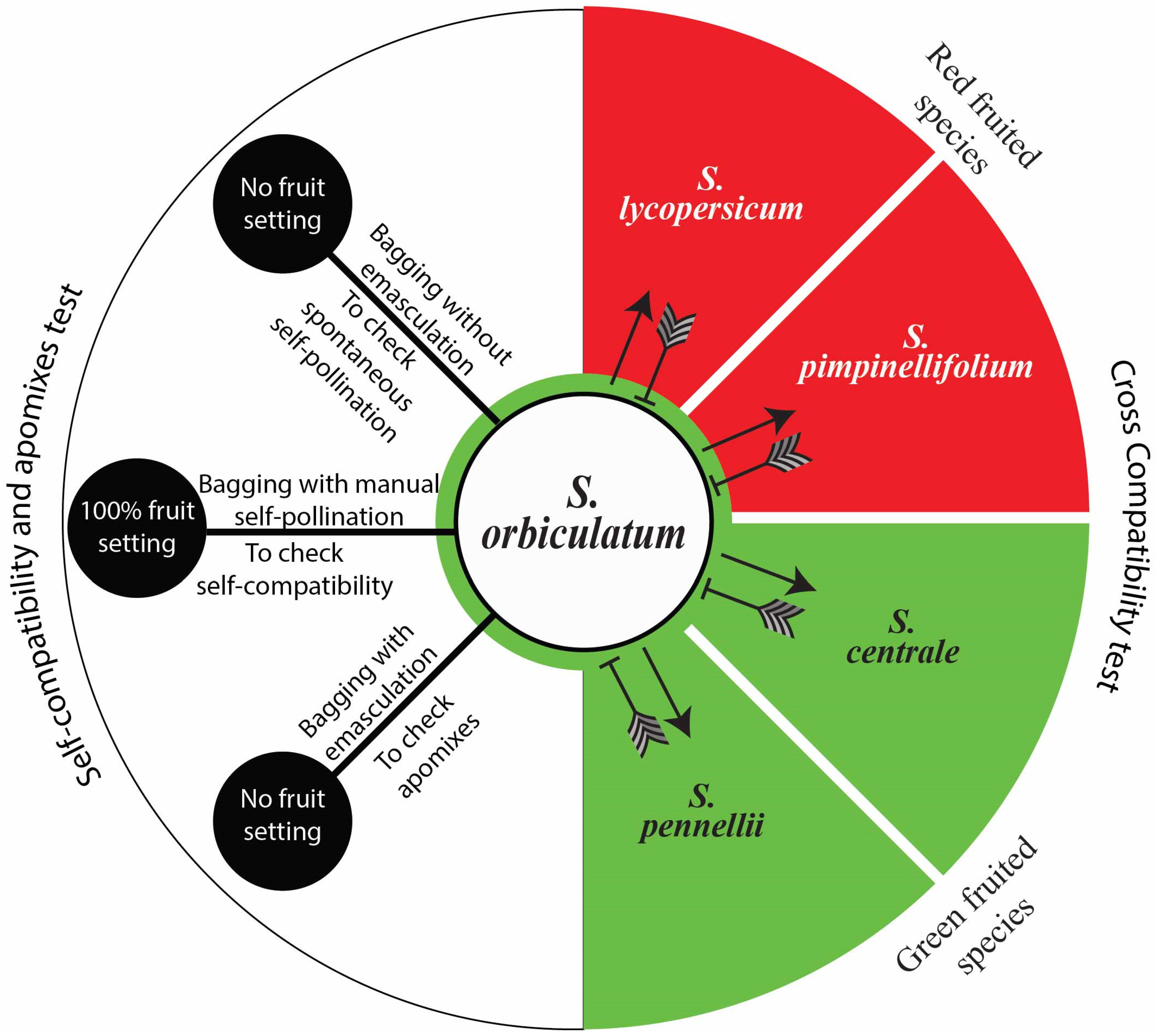
2.6. Controlled Pollination for Compatibility Studies
2.7. Data Collection and Analysis
3. Results and Discussion
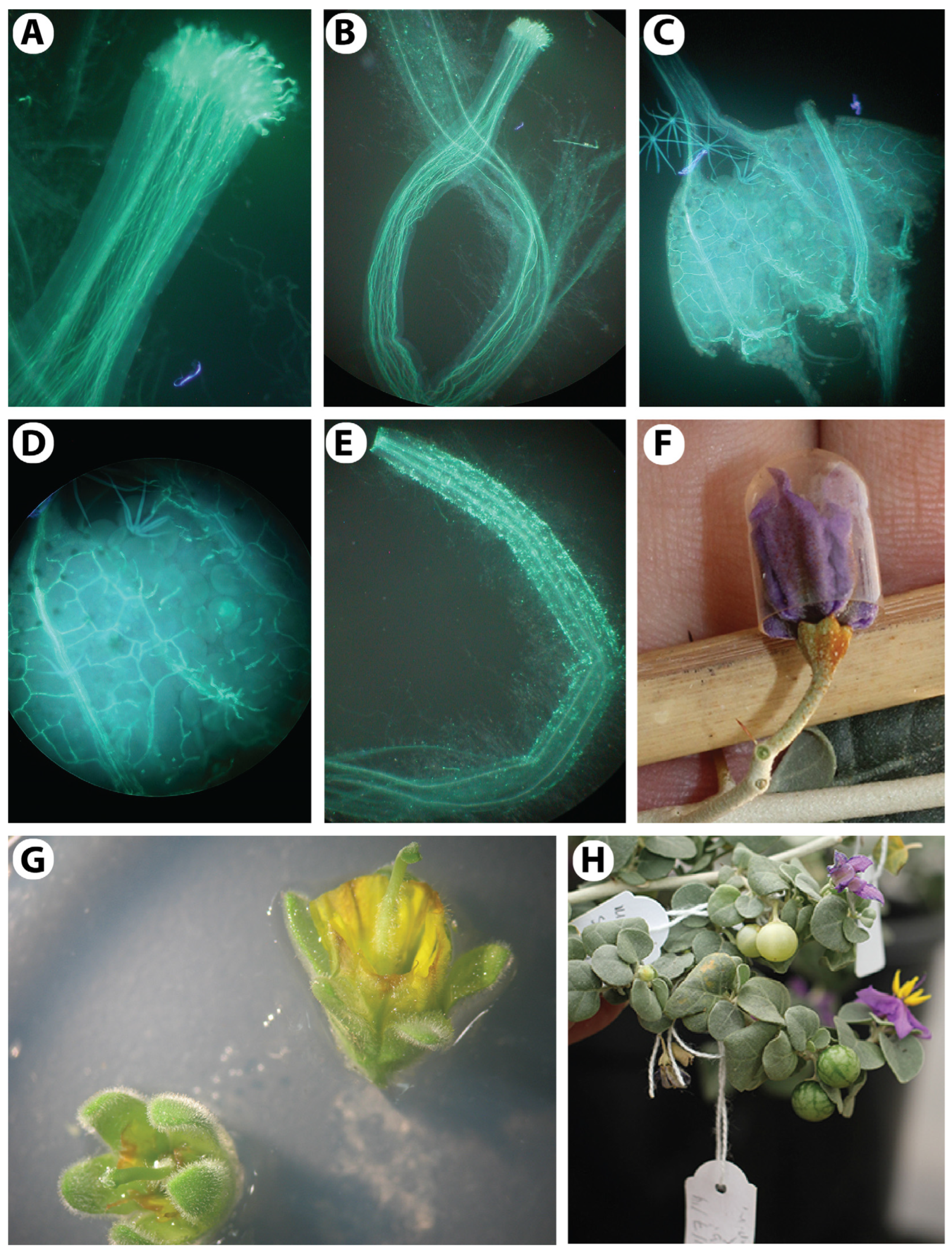
3.1. Floral Structure and Morphological Variation
3.2. Relationship between Pollen Size and Style Length
3.3. Optimising the In Vitro Pollen Germination Medium
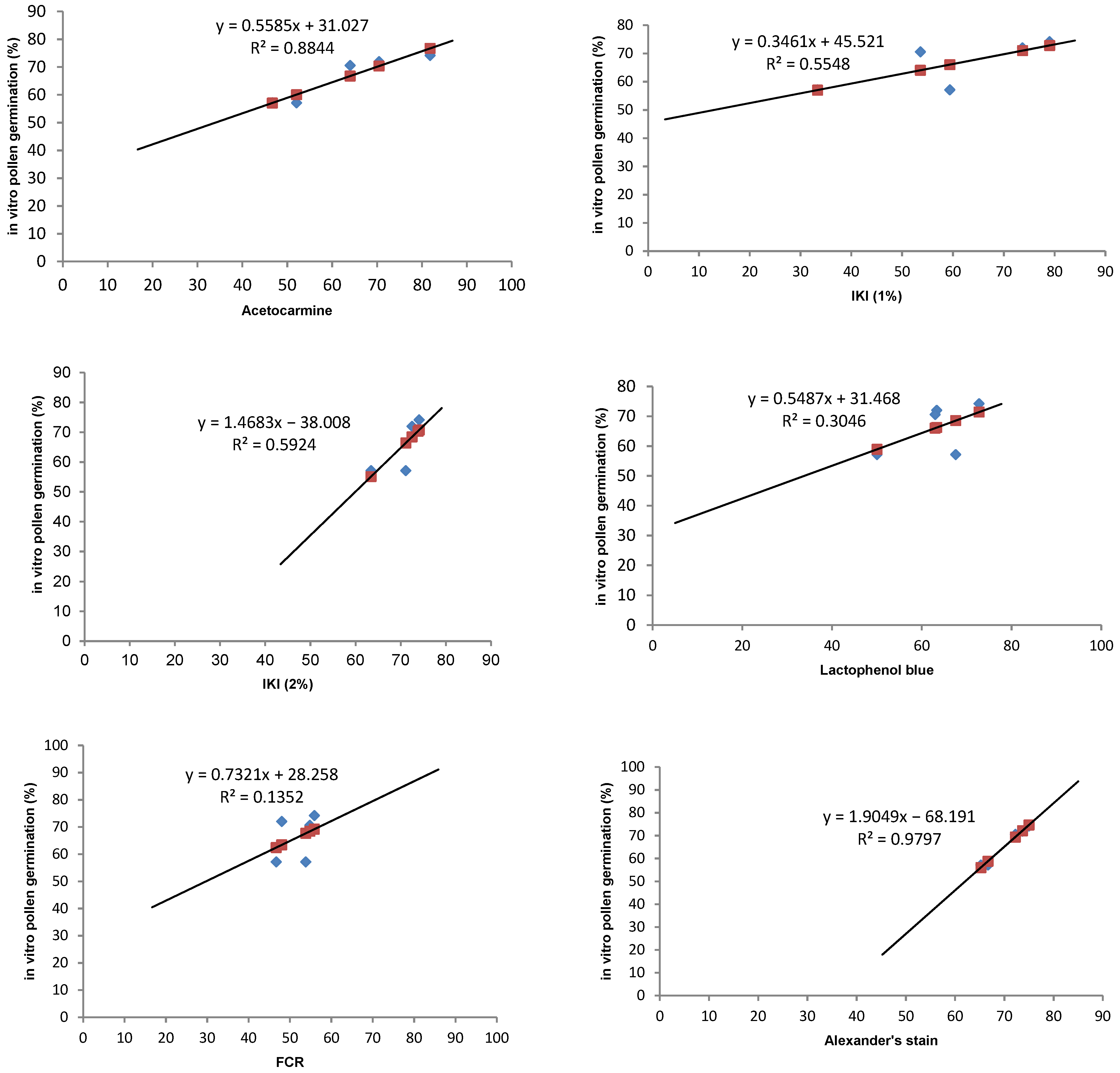
3.4. Pollen Stainability Test
3.5. Pollen Storage and Viability
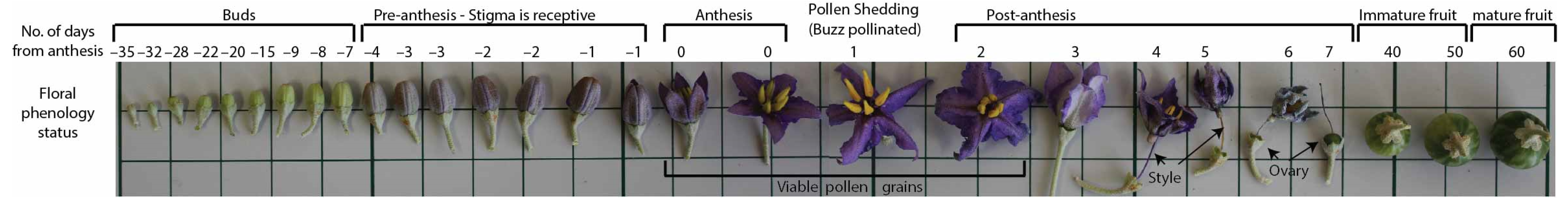
3.6. Timing of Pollen Viability and Stigma Receptivity
3.7. Sequence of Floral Development
3.8. Developing an Efficient Pollination Technique
3.9. Self- and Cross-Compatibility
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Moore, P.A. Guide to Plants of Inland Australia; Reed New Holland: Wahroonga, Australia, 2005. [Google Scholar]
- Commander, L.E.; Merritt, D.J.; Rokich, D.P.; Dixon, K.W. Seed biology of Australian arid zone species: Germination of 18 species used for rehabilitation. J. Arid Environ. 2009, 73, 617–625. [Google Scholar] [CrossRef]
- Commander, L.E.; Merritt, D.J.; Rokich, D.P.; Flematti, G.R.; Dixon, K.W. Seed germination of Solanum spp. (Solanaceae) for use in rehabilitation and commercial industries. Aust. J. Bot. 2008, 56, 333–341. [Google Scholar] [CrossRef]
- Symon, D.E. A revision of the genus Solanum in Australia. J. Adel. Bot. Gard. 1981, 4, 1–367. [Google Scholar]
- Milner, K.V.; French, K.; Krix, D.W.; Valenzuela, S.M.; Leigh, A. The effects of spring versus summer heat events on two arid zone plant species under field conditions. Funct. Plant Biol. 2023, 50, 455–469. [Google Scholar] [CrossRef] [PubMed]
- Anderson, G.J.; Symon, D.E. Functional dioecy and andromonoecy in Solanum. Evolution 1989, 43, 204–219. [Google Scholar] [CrossRef] [PubMed]
- Martine, C.T.; Symon, D.E.; Evans, E.C. A new cryptically dioecious species of bush tomato (Solanum) from the Northern Territory, Australia. PhytoKeys 2013, 30, 23–31. [Google Scholar] [CrossRef] [PubMed]
- Stanley, R.G.; Linskens, H.F. Pollen: Biology, Biochemistry, Management; Springer: Berlin/Heidelberg, Germany, 1974; p. ix + 307. [Google Scholar]
- Brewbake, J.L.; Kwack, B.H. Essential role of calcium ion in pollen germination and pollen tube growth. Am. J. Bot. 1963, 50, 859. [Google Scholar] [CrossRef]
- Heslop-Harrison, J.; Heslop-Harrison, Y.; Shivanna, K.R. The evaluation of pollen quality, and a further appraisal of the fluorochromatic (FCR) test procedure. Theor. Appl. Genet. 1984, 67, 367–375. [Google Scholar] [CrossRef] [PubMed]
- Alexander, M.P. Differential staining of aborted and nonaborted pollen. Stain Technol. 1969, 44, 117. [Google Scholar] [CrossRef] [PubMed]
- Lundqvist, A.A. rapid method for the analysis of incompatibilities in grasses. Hereditas 1961, 47, 705–707. [Google Scholar] [CrossRef]
- Martin, F.W. Staining and observing pollen tubes in the style by means of fluorescence. Stain Technol. 1959, 34, 125–128. [Google Scholar] [CrossRef] [PubMed]
- Torres, C. Pollen size evolution: Correlation between pollen volume and pistil length in Asteraceae. Sex. Plant Reprod. 2000, 12, 365–370. [Google Scholar] [CrossRef]
- Aguilar, R.; Bernardello, G.; Galetto, L. Pollen-pistil relationships and pollen size-number trade-off in species of the tribe Lycieae (Solanaceae). J. Plant Res. 2002, 115, 335–340. [Google Scholar] [CrossRef] [PubMed]
- Proctor, M.; Yeo, P.; Lack, A. The Natural History of Pollination; Timber Press: Portland, OR, USA, 1996; p. 479. [Google Scholar]
- Routley, M.B.; Bertin, R.I.; Husband, B.C. Correlated evolution of dichogamy and self incompatibility: A phylogenetic perspective. Int. J. Plant Sci. 2004, 165, 983–993. [Google Scholar] [CrossRef]




| Flower No. | No. of Petals | Petal Width (mm) | Petal Length (mm) | No. of Anthers | Anther Length (mm) | Style Length (mm) | Comments (Petals, Pedicel) |
|---|---|---|---|---|---|---|---|
| 1 | 5 | 6.77 | 12.07 | 5 | 6.76 | 10.69 | Long pedicel |
| 2 | 5 | 8.03 | 16.69 | 5 | 7.38 | 12.33 | |
| 3 | 5 | 7.51 | 15.14 | 5 | 7.45 | 12.65 | Long pedicel |
| 4 | 5 | 6.73 | 12.4 | 5 | 7.24 | 9.62 | |
| 5 | 4 | 7.45 | 14.27 | 4 | 6.93 | 11.33 | Light purple petals |
| 6 | 5 | 9.84 | 17.77 | 5 | 7.31 | 12.18 | Very light purple petals |
| 7 | 5 | 7.56 | 15.84 | 5 | 6.37 | 10.9 | Very light purple petals |
| 8 | 5 | 6.31 | 15.74 | 5 | 8.09 | 9.25 | - |
| 9 | 3 | 9.17 | 12.74 | 3 | 6.9 | 9.74 | - |
| 10 | 3 | 7.84 | 11.05 | 3 | 6.6 | 9.69 | - |
| 11 | 4 | 7.27 | 12.59 | 4 | 7.3 | 11.6 | - |
| 12 | 4 | 8.24 | 13.78 | 4 | 7.16 | 11.62 | - |
| 13 | 5 | 5.31 | 10.78 | 5 | 5.91 | 10.47 | - |
| 14 | 3 | 8.15 | 12.05 | 3 | 6.71 | 9.7 | - |
| 15 | 5 | 6.67 | 10.87 | 5 | 5.99 | 10.3 | - |
| 16 | 5 | 5.82 | 10.31 | 5 | 5.84 | 10.35 | - |
| 17 | 5 | 6.42 | 14.42 | 5 | 6.63 | 10.64 | Light purple petals |
| 18 | 5 | 8.95 | 16.57 | 5 | 7.2 | 9.74 | Light purple/long pedicel |
| 19 | 5 | 9.3 | 15.14 | 5 | 6.65 | 10.67 | - |
| 20 | 5 | 7.19 | 12.15 | 5 | 7.24 | 9.9 | - |
| Mean ± s.e. | 7.53 ± 0.27 | 13.62 ± 0.50 | 6.88 ± 0.13 | 10.67 ± 0.22 |
| Flower Type | Means ± s.e. | Significance |
|---|---|---|
| Flower with long style * | 345.73 ± 2.91 | T Stat = 3.887 and t Critical = 0.003, the two means are significantly different. |
| Flower with short style ** | 329.24 ± 3.09 |
| Flower Type | Means ± s.e. | Significance |
|---|---|---|
| Long style length * | 9588.02 ± 374.56 | T Stat = 15.310 and t Critical = 2.2235, the two means are significantly different. |
| Short style length ** | 2005.02 ± 143.89 |
| Medium | Sucrose (%) | PEG (%) | Germination (%) * |
|---|---|---|---|
| 1 | 0 | 0 | 11.61 hij |
| 2 | 0 | 2.5% | 16.68 ghi |
| 3 | 0 | 5% | 22.98 efg |
| 4 | 10% | 0 | 29.48 de |
| 5 | 10% | 2.5% | 28.60 de |
| 6 | 10% | 5% | 42.46 bc |
| 7 | 15% | 0 | 28.50 def |
| 8 | 15% | 2.5% | 37.96 cd |
| 9 | 15% | 5% | 52.04 ab |
| 10 | 20% | 0 | 31.70 de |
| 11 | 20% | 2.5% | 56.61 a |
| 12 | 20% | 5% | 50.31 ab |
| 13 | 25% | 0 | 11.36 hij |
| 14 | 25% | 2.5% | 13.30 ghij |
| 15 | 25% | 5% | 13.99 ghij |
| 16 | 30% | 0 | 19.11 fgh |
| 17 | 30% | 2.5% | 6.89 ij |
| 18 | 30% | 5% | 5.75 j |
| Stain | R2 | p Value * | Relationship Strength |
|---|---|---|---|
| Acetocarmine | 0.884 | 0.017 | Strong positive relationship |
| IKI (1%) | 0.555 | 0.149 | Weak relationship |
| IKI (2%) | 0.592 | 0.128 | Weak relationship |
| Lactophenol blue | 0.305 | 0.335 | Weak relationship |
| FDA | 0.135 | 0.543 | Very weak relationship |
| Alexander’s stain | 0.980 | 0.001 | Very strong positive relationship |
| Months after Storage | Pollen Germination (%) * |
|---|---|
| 0 | 59.26 ± 5.87 a |
| 1 | 56.92 ± 5.22 a |
| 2 | 39.31 ± 0.91 b |
| 3 | 35.05 ± 3.01 b |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ahmad, N.; Chamas, A.; Trethowan, R. Reproductive Biology of Solanum orbiculatum ssp. orbiculatum, an Australian Endemic Bush Tomato. Agronomy 2023, 13, 2701. https://doi.org/10.3390/agronomy13112701
Ahmad N, Chamas A, Trethowan R. Reproductive Biology of Solanum orbiculatum ssp. orbiculatum, an Australian Endemic Bush Tomato. Agronomy. 2023; 13(11):2701. https://doi.org/10.3390/agronomy13112701
Chicago/Turabian StyleAhmad, Nabil, Ali Chamas, and Richard Trethowan. 2023. "Reproductive Biology of Solanum orbiculatum ssp. orbiculatum, an Australian Endemic Bush Tomato" Agronomy 13, no. 11: 2701. https://doi.org/10.3390/agronomy13112701
APA StyleAhmad, N., Chamas, A., & Trethowan, R. (2023). Reproductive Biology of Solanum orbiculatum ssp. orbiculatum, an Australian Endemic Bush Tomato. Agronomy, 13(11), 2701. https://doi.org/10.3390/agronomy13112701




