Abstract
Coffee berry disease (CBD) is not present in the Americas and presents a potential risk for growing coffee. Therefore, Colombia, which has been in scientific cooperation with the Centro de Investigação de Ferrugens do Cafeeiro (CIFC) of Portugal for more than 30 years, has been evaluating the genetic resistance of nine populations of C. arabica to 13 isolates of Colletotrichum kahawae JM Waller and PD Bridge, which are diverse in terms of aggressiveness and geographical origin. The phenotypes observed in the interaction between C. arabica and C. kahawae were used to develop a statistically reliable scale (p-value ≥ 0.001) to categorize resistance in C. arabica into five classes, and this scale was used to classify the nine populations of C. arabica evaluated. The results allowed us to corroborate the potential of Timor Hybrid CIFC 1343 (TH CIFC 1343) as a source of genetic resistance to CBD and to identify new genetic sources not yet explored for the development of varieties in Colombia that may eventually mitigate the effects of CBD in the face of increasing rainfall events and minimum temperatures due to climate change, which can favor disease development. Additionally, the results suggest that the existence of races in the C. arabica–C. kahawae complex is probable, and a selection of genotypes was identified as a possible differential series of races in C. kahawae.
1. Introduction
Coffee berry disease (CBD) is caused by Colletotrichum kahawae JM Waller and PD Bridge [1,2]. This fungus is classified among the ten main genera that cause diseases in crops and is currently restricted only to the African continent [3,4,5]. C. kahawae affects Coffea arabica crops, causing production losses of more than 80% when weather conditions favor pathogen development and preventive measures are not taken to control it [6,7,8]. The effects of C. kahawae on C. arabica cultivation are far superior to those caused by Hemileia vastatrix Berk and Broome [9]. Its aggressiveness is highly variable across isolates and geographic origins; it presents a clearly defined population structure and is composed of three geographic groups, namely Angola, Cameroon, and East Africa [10,11,12].
CBD is dispersed across altitudes, affecting coffee plantations established at altitudes as low as 1000 m above sea level (masl) [13,14]. However, the most severe effects occur in coffee plantations established at altitudes above 1400 masl [4,15] in combination with climate conditions that favor epidemics, including relative humidity close to the saturation point (>95%) [13,16,17,18] and temperatures ranging between 15 °C and 25 °C [18].
In Africa, in countries where varieties with genetic resistance to CBD are not cultivated, the disease must be controlled with the application of chemically synthesized products [19]. In Kenya, no fewer than eight applications are recommended during the year, distributed in the rainy season, between five and six applications in the first half and between two and three in the second half (personal communication, Dr. Elijah Gichuru, Coffee Research Institute—Kenya). Additionally, fungicide application should be complemented with cultural crop management practices, including pruning and the regulation of conditions that produce favorable microclimates [16,18,20].
From an economic and environmental conservation perspective, the development of varieties with genetic resistance to diseases is the most suitable strategy for ensuring the production and profitability of crops. Proof of this are the unprecedented impacts achieved in Colombia, where the adoption of highly productive varieties of C. arabica that are resistant to H. vastatrix has had positive impacts on the economy for coffee growers and on environmental sustainability [21,22]. It has also been demonstrated that CBD can be controlled through the development of varieties resistant to C. kahawae [16,19,23,24]. This resistance can be found in different sources of C. arabica, some of which are derived from interspecific hybridization, such as Timor Hybrid (TH) [25].
TH has been noted as the main source of genetic resistance to diseases that limit C. arabica production, and for this reason, it has been used extensively in genetic improvement programs around the world [26]. In Africa, progeny derived from the TH have been shown to be resistant to CBD, providing effective and durable genetic control of epidemics caused by C. kahawae [19,27,28,29].
In the Americas, specifically in Colombia, the Coffee Research Center, Cenicafé, has developed varieties with resistance to CBD from TH CIFC 1343, and the processes of selecting genotypes with resistance to this disease have been based on the following three strategies:
- Indirect selection or assisted selection by molecular markers (MAS). In Colombia, SSR markers associated with the Ck-1 gene of resistance to CBD are currently used [30,31,32]. This biotechnological tool has made it possible to identify the Ck-1 gene in rust-resistant varieties developed in Colombia [33].
- Direct selection through field tests. Advanced progeny evaluations of TH CIFC 1343 derivatives were carried out from scientific cooperation agreements with the Coffee Research Institute, formerly the Coffee Research Foundation (CRI), Ruiru, in Kenya, between 1975 and 1977 [34,35]. Later, in Kenya, they developed the varieties Ruiru 11 and Batian from derivatives of TH CIFC 1343 [24,36].
- Direct selection via the hypocotyl inoculation method. The advantage of this method is that it allows the identification of resistance to CBD in advanced nonclonal genotypes [37] and presents the highest correlation (>0.73) with the behavior of the disease under natural crop or epidemic conditions [38]. The first tests using this method were carried out 50 years ago at the CRI in Kenya [39]. The results made it possible to identify the resistance of TH CIFC 1343 to C. kahawae, and evaluation is currently being carried out at the CIFC.
Historically, the method of evaluating resistance to CBD via the hypocotyl inoculation method uses severity scales [40,41]. However, when scales of this type are used, their interpretation can become controversial [42]. Using already developed scales of evaluation of CBD resistance [40,41] allows for the identification of highly resistant and completely susceptible genotypes with great ease. However, the classification of individuals with intermediate levels of resistance is not clear [40,41], and its complexity lies mainly in the very nature of the resistance character and how it is inherited, aspects of which there has been no clear consensus on to date [9,30,43,44,45].
In 1997, the Cenicafé genetic improvement program and the CIFC began evaluating nine populations of C. arabica for their resistance to different C. kahawae isolates, which are diverse in aggressiveness and geographic origin. The purpose of this research was (1) to develop a scale for the determination of classes of resistance to CBD in improved genotypes and (2) to identify resistance to this disease in unexplored genotypes of the Colombian coffee collection (CCC), which have potential as progenitors of new varieties of coffee.
2. Materials and Methods
2.1. Location
The evaluated populations were established at the Naranjal Experimental Station (04°58′ N, 75°39′ W), which is located in the municipality of Chinchiná (Caldas, Colombia) at 1381 m above sea level; the average temperature is 21.4 °C, the annual precipitation is 2782 mm, and the average relative humidity is 77.5%. Evaluations of resistance to C. kahawae were carried out in the laboratories of the CIFC in Portugal for 25 years (1997–2022).
2.2. Genotypes Evaluated
In total, 512 genotypes derived from nine populations were evaluated (Table 1). The Caturra variety, which is susceptible to C. kahawae, was used as a control genotype.

Table 1.
Populations evaluated and number of genotypes for each population.
2.3. Isolates of C. kahawae
The evaluated populations were inoculated with 13 isolates of C. kahawae, which are diverse in aggressiveness and belong to the biological collection of the CIFC (Table 2).

Table 2.
Geographical origins of C. kahawae isolates and their classification by aggressiveness.
2.4. Evaluation of Resistance via Hypocotyl Testing
In each of the evaluations (genotype isolate), between 100 and 120 hypocotyls were tested using the methodology developed by [38,40] and modifications made by the CIFC. Hypocotyls 3 to 6 cm long (5–6 weeks of development) were arranged in a humid chamber on nylon sponges moistened with water and subsequently inoculated with a solution of C. kahawae conidia in suspension at an approximate concentration of 2 × 106 conidia per milliliter of water.
Conidia were obtained from in vitro cultures of C. kahawae in malt extract agar medium (GEM) [Oxoid, 3.4%] and were grown for eight days at 22 °C. The culture was subsequently resuspended in 5 mL of sterile distilled water. The suspension was filtered through layers of sterile muslin cloth to remove the mycelia. The solution was sprayed on each hypocotyl with an atomizer coupled to an air pressure pump (Pumpentyp Siehe unterseite Pumpe VDE 0530). The inoculated hypocotyls were kept in a humid chamber for 48 h, and a second inoculation was subsequently carried out under the same conditions.
During the first four days of incubation, the humid chambers remained at an average temperature of 22 °C, then subsequently at 19 °C; the relative humidity was close to the saturation point and the photoperiod was 12 h. The infection process was monitored for 4 weeks; during this time, the hypocotyls were classified according to the severity scale [41] (Table 3).

Table 3.
CBD phenotypic severity scale in C. arabica hypocotyls and description of the development of symptoms caused by C. kahawae.
2.5. Data Analysis
Each hypocotyl evaluated (observation unit) was classified as resistant if it developed a degree of infection of 1, 2, or 3 and as susceptible if it was Grade 4 according to the severity scale (Table 3) [41]. From each of the populations and isolates (treatments), groups of 10 observation units (sampling units) were randomly formed, with the percentage of resistant hypocotyls as a variable of interest, i.e., of the 10 observation units per sampling unit, the percentage of those that were not classified as Grade 4. For each combination (genotype isolate), the average percentage of resistant hypocotyls was estimated, with their respective intervals. For a confidence coefficient of 95% and with the percentile distribution of the averages, a classification by resistance classes was established.
For each resistance class, the average percentage of resistant hypocotyls was estimated, and the Duncan test at 5% was used to compare the averages. Once the significant differences between classification groups were obtained (categorization of resistance classes), the classification criterion was applied to the populations derived from the Cat. × TH CIFC 832/1, TH CIFC 2252, TH CIFC 1343, Cat. × TH CIFC 1343, and CCC populations.
3. Results
3.1. Resistance Class Scale
With the described methodology, a scale was obtained for the classification of C. arabica genotypes on the basis of their CBD resistance. The scale is made up of four resistance classes, namely high resistance (HR), moderate resistance (MR), low resistance (LR), and very low resistance (VLR), as well as a susceptible class (S). Each resistance class was established according to the percentage of hypocotyls with phenotypic reactions of severity between 1 and 3, that is, classified as resistant (Table 3 and Table 4).

Table 4.
Scale for the classification of C. kahawae resistance in C. arabica and the percentage of hypocotyls with phenotypic reactions of severity 1, 2, and 3.
The classification did not include a class that considers the absence of the disease because in the populations evaluated, no immunity to C. kahawae was found and to date, there are no reports of immunity in C. arabica.
Duncan’s comparison test at 5% revealed differences between the averages of the five classes, statistically separating them (Table 5) with a reliability greater than 95%. Therefore, the statistically obtained scale is a reliable tool for phenotypic categorization by classes of C. kahawae resistance in C. arabica.

Table 5.
Average percentage of hypocotyls resistant to C. kahawae by class and their respective confidence intervals.
The classification of a genotype into one of the classes with the highest level of resistance to CBD, HR (Figure 1A) or MR (Figure 1B), is mainly due to the percentage of hypocotyls that were less affected by the disease, that is, Grades 1 and 2 (Table 3). In these cases, the advancement and development of C. kahawae in the hypocotyls was restricted to the formation of small lesions, which may eventually coalesce without the disease progressing to the most advanced stages (Grades 3 and 4).

Figure 1.
Phenotypic response of C. arabica to C. kahawae and its classification on the scale of resistance. (A) Phenotypic response of the genotypes of the Cat. × TH CIFC 1343 population classified in the high resistance (HR) class to the Zim12 (Zimbabwe) isolate. (B) Phenotypic response of the genotypes of the Cat. × TH CIFC 1343 population classified in the moderate resistance (MR) class to the Zim12 (Zimbabwe) isolate. (C) Phenotypic response of the genotypes of the Cat. × TH CIFC 1343 population classified into the low resistance (LR) class to the Cam1 (Cameroon) isolate. (D) Phenotypic response of the genotypes of the Cat. × TH CIFC 1343 population classified in the very low resistance (VLR) class to the Ang29 (Angola) isolate. (E) Phenotypic response of the genotypes of the Cat. × TH CIFC 1343 population classified in the susceptible class to the A (Angola) isolate.
For the HR class, the hypocotyls least affected by CBD (Grade 1) contributed the most to categorization into this group, since they represented greater than 70% of the tested hypocotyls, with 28% developing Grade 2 symptoms and 2% developing Grade 3 symptoms. In Grade 3, lesions that affect the circumference of the hypocotyl were present, and although the disease progressed, the hypocotyl survived until the end of the evaluation.
The MR class presented a lower percentage of Grade 1 hypocotyls (55%) than the HR class and greater percentages of hypocotyls with Grade 2 and Grade 3 symptoms. At least 45% of the hypocotyls presented Grades 2 and 3 symptoms in the MR class, corresponding to a 15% decrease in the number of hypocotyls with Grade 1 symptoms compared with the HR class.
The genotypes in the LR (Figure 1C) and VLR (Figure 1D) classes mainly presented Grade 1 resistance (Table 4), corresponding to 54% and 72% of the hypocotyls, respectively. The remaining hypocotyls were Grades 2 and 3. As in the other resistance classes, the hypocotyls that reached Grade 3 represented the lowest proportion.
The S class (Table 3 and Table 4) consisted completely of Grade 4 phenotypes (Figure 1D). This level of CBD development is characterized by the presence of lesions that completely necrotize the stem, followed by the complete death of the hypocotyl tissues.
In the genotypes least affected by C. kahawae (Grades 1 and 2), more than 90% of the hypocotyls presented resistance, thus defining the resistance class in which a genotype can be classified. However, the percentage of resistant hypocotyls taken as a reference for categorizing a genotype into one of the four resistance classes included those in the first three degrees of phenotypic qualification (Grades 1, 2, and 3). That is, the resistance class of a genotype is not exclusively due to the concentration of symptoms to a defined degree.
In the hypocotyls cataloged within the HR class, 70% were Grade 1, among which the selection limit of the class (≥79%) was present in 58.8% of the cases, and 10.9% were Grade 2. In other words, it is highly probable (≥95%) to find HR genotypes, and their resistance classification (class) is exclusive to Grade 1.
For the MR class, the selection range corresponded to >50% and ≤79% of the hypocotyls as a result of the sum of Grade 1 + Grade 2 + Grade 3 hypocotyls (Table 3 and Table 4). However, unlike those in the HR class, the hypocotyls within the selection limit were not concentrated within a specific severity degree. Therefore, the sum of the number of hypocotyls in the two lowest severity grades (Grades 1 and 2) ultimately defines the resistance class (MR).
Among the genotypes of the LR class (>20% and ≤50%), the resistant hypocotyls were equally distributed among the three degrees of severity, similar to MR. However, for a genotype to be considered LR, a greater number of hypocotyls with Grade 2 phenotypic reactions are needed. The VLR class is the product of a higher frequency of Grade 2 phenotypic responses, with fewer Grade 1 and Grade 3 responses. Finally, the S genotypes present exclusively Grade 4 phenotypic reactions.
Of the 512 genotypes evaluated, 98.2% were evaluated with isolates from two of the three documented population origins, Cameroon and East Africa. The East Africa isolates were obtained from five countries (Malawi, Kenya, Rwanda, Uganda, and Zimbabwe) (Table 6). The remaining percentage was evaluated with isolates from the third geographical origin (Angola).

Table 6.
Percentage of genotypes evaluated by isolate and the C. kahawae resistance classes in which they were grouped.
A total of 38.73% of the evaluated progeny developed HR and MR phenotypes to the Cam1, M, M2, Que2, R, Z12, Z9, Zim1, and Zim12 isolates, and more severe reactions to C. kahawae (LR and VLR) occurred in 40.5% of the progeny when evaluated with the same isolates. Reactions of complete susceptibility (class S) developed in 20.77% of the progeny (Table 6).
In the evaluated populations, no high or medium resistance was observed for isolates originating from Angola and Uganda. The highest proportion of the evaluated genotypes presented high/medium resistance to isolates from Kenya (Que2), Zimbabwe (Zim1, Z9), and Malawi (M) (Table 6).
3.2. Resistance to CBD in TH from the Class Scale
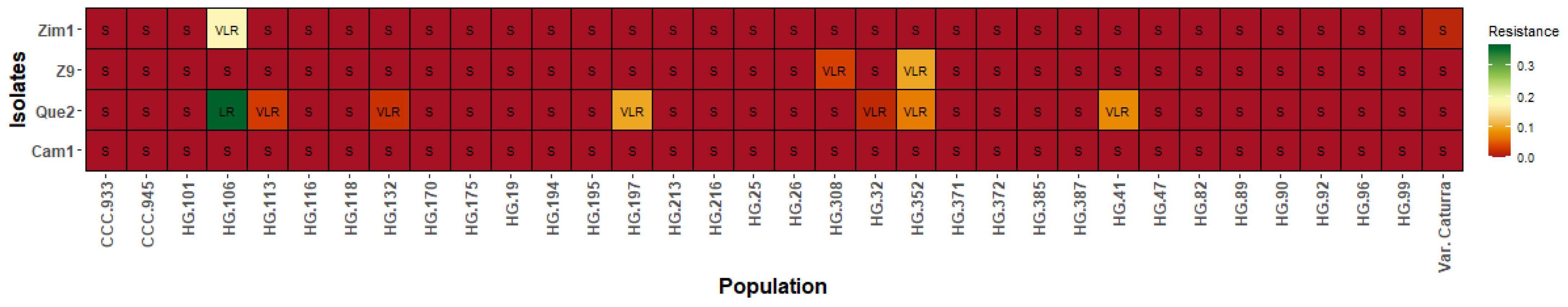
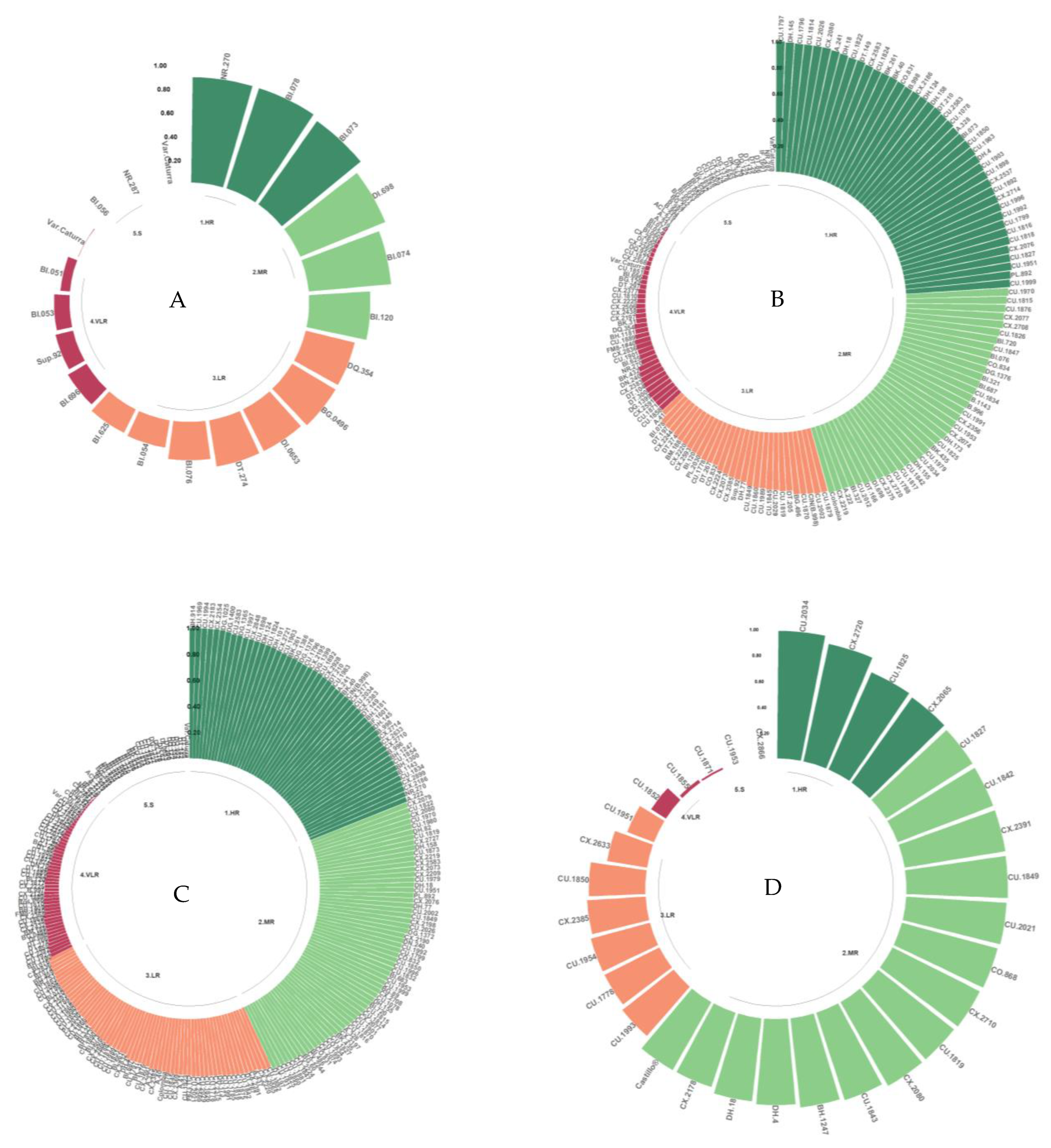
In the populations derived from TH CIFC 832/1, only the LR class was identified for the isolate Que2 in one of the progeny (HG.106), which also presented VLR to the isolate Zim1. The other progeny were classified as VLR and S for the four isolates (Cam1, Que2, Z9, Zim1). The S class predominated in the population derived from TH CIFC 832/1, similar to the results obtained for the Caturra variety (Figure 2).
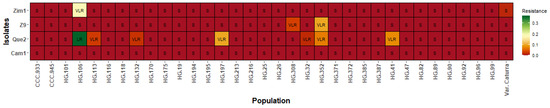
Figure 2.
Classification of resistance to the Cam1, Que2, Z9, and Zim1 isolates of C. kahawae in the genotypes of the Cat. × TH CIFC 832/1 population. HR: high resistance; MR: moderate resistance; LR: low resistance; VLR: very low resistance; S: susceptible.
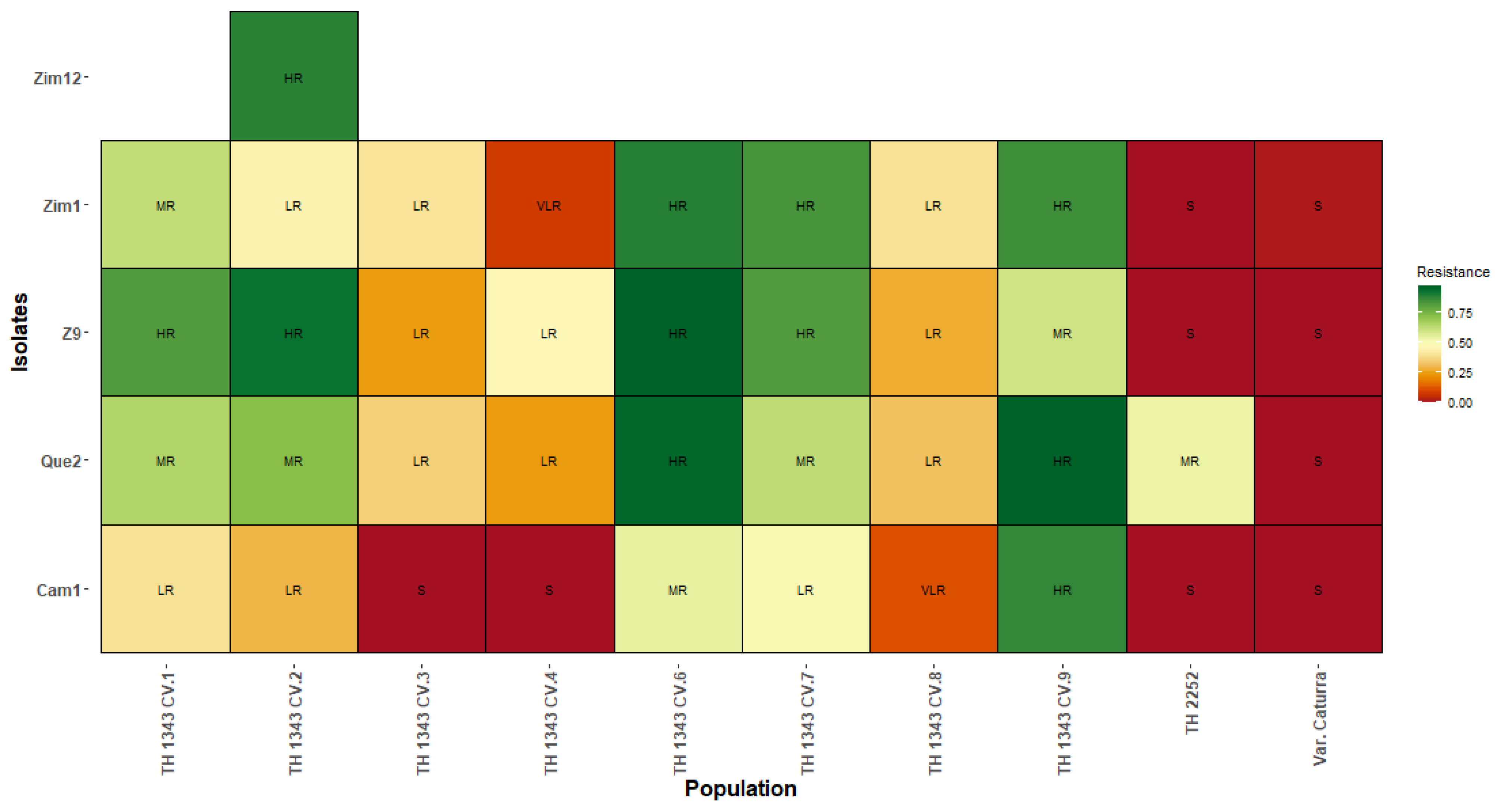
In the results obtained from the classification of resistance to C. kahawae in TH CIFC 1343, the CV.9 genotype presented an HR phenotype for the Cam1 isolates, which have been reported as being highly aggressive, and for the Que2 and Zim1 isolates, which are of medium–low aggressiveness. TH CIFC 2252 presented an MR phenotype for the Que2 isolate and susceptibility to the other isolates (Figure 3).
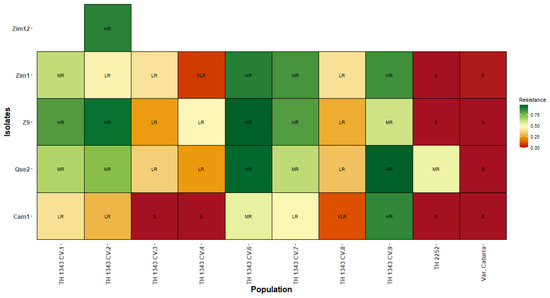
Figure 3.
Classification of resistance to the Cam1, Que2, Z9, Zim1, and Zim12 isolates of C. kahawae in TH CIFC 1343 and TH CIFC 2252. HR: high resistance; MR: moderate resistance; LR: low resistance; VLR: very low resistance; S: susceptible.
3.3. CBD Resistance Classes in Cat. × TH CIFC 1343
The populations derived from Cat. × TH CIFC 1343 consisted of 275 genotypes of advanced generations (F3–F6), which were analyzed and classified according to the phenotypic classes of resistance determined in this study (Table 4). This population was evaluated with 11 isolates of different levels of aggressiveness and diverse geographic origins. All the phenotypic classes of resistance to C. kahawae were identified in the genotypes evaluated, ranging from genotypes of the HR class, through to the intermediate classes of resistance, to the VLR and susceptible classes (Table 7).

Table 7.
Total percentage of genotypes evaluated by isolate and the C. kahawae resistance classes in which the Cat. × TH CIFC 1343 population was grouped.
3.4. Resistance to C. kahawae Isolates in Cat. × TH CIFC 1343
3.4.1. Resistance to the Ang29 Isolate
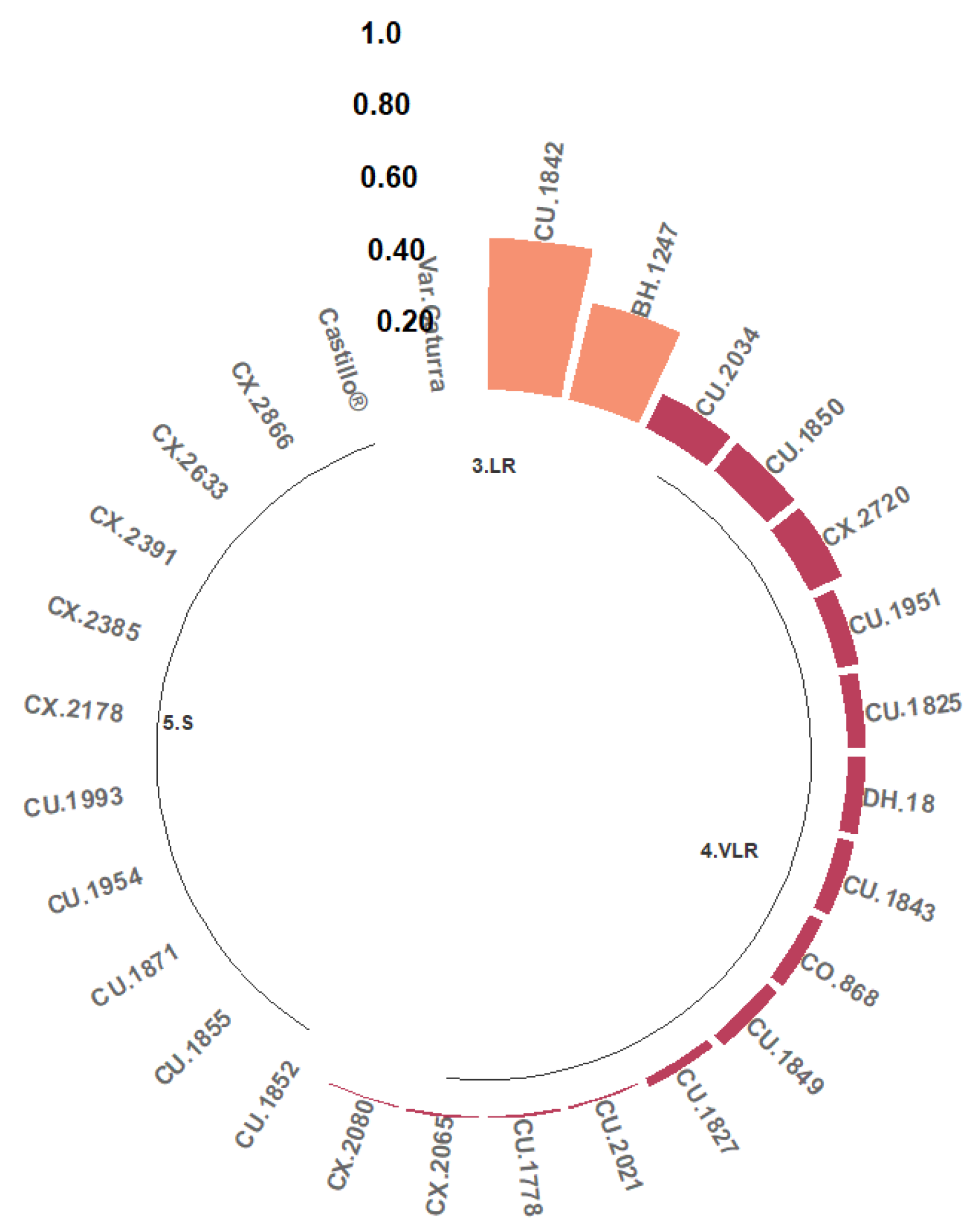
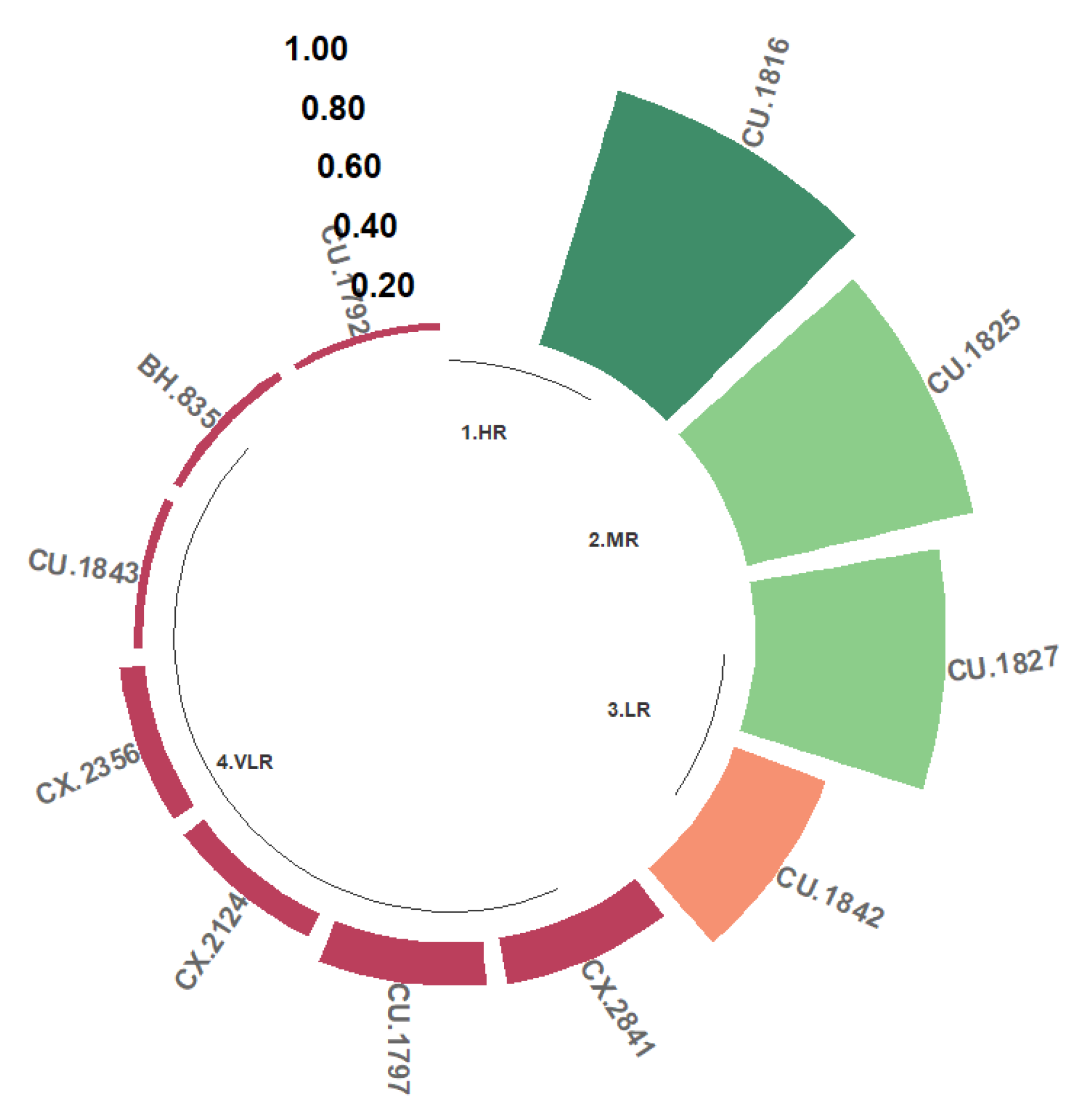
Two classes of resistance to the Ang29 isolate of Angolan origin were found, namely LR and VLR. A total of 7.41% of the progeny were grouped in the LR class; that is, between 20% and 50% of their hypocotyls presented Grade 1, 2, or 3 reactions. The progeny identified in this class are CU.1842 and BH.1247 (Figure 4). The remaining hypocotyls developed phenotypic reactions of the VLR class (51.85%) or were completely susceptible (40.74%).
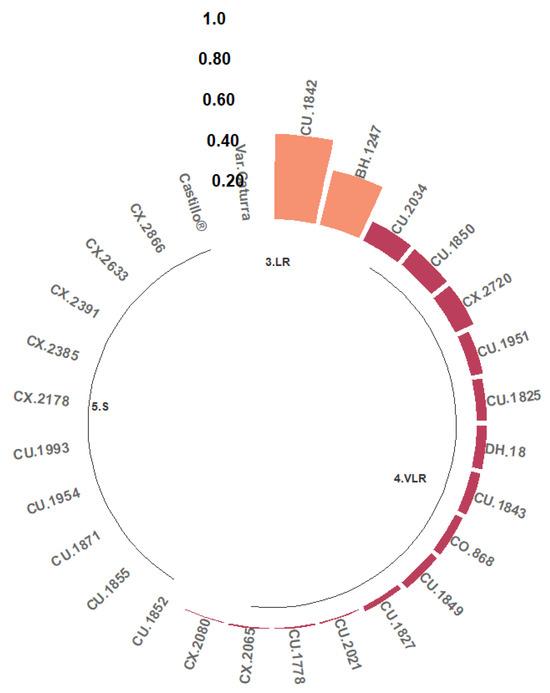
Figure 4.
Distribution of resistance classes to the C. kahawae Ang29 isolate in progeny derived from the Cat. × TH CIFC 1343 population. LR: low resistance; VLR: very low resistance; S: susceptible. The amount of resistance to CBD as determined by the resistance scale is represented by the numbers on the Y-axis.
3.4.2. Resistance to the Cam1 and CC Isolates
The resistance classes for these isolates were more diverse than those for Ang29. For the Cam1 isolate, 0.37% of the genotypes were categorized as HR (CU.1997) and 4.4% were categorized as MR (A.323, BK.261, B.998, DH.155, DH.145, BI. 486, A.437, DG.1365, DT.210, DG.1376, CX.2171, and BH.914) or LR (DH.124, DH.77, DG.1025, CX.2714, DG.1386, DG.1399, CX.2073, A.41, B.996, CU.1994, A.222, BG.89, CU.1796, A.328, CU.1826, CIN (B.998), BH.1247, BK.40, CU.1903, CX.2369, DH.82, CX.2354, BI.720, NR.270, and CX.2224). The remaining percentage was classified as VLR or susceptible (Figure 5A). For the CC isolate, all genotypes were classified into the VLR or S classes (Figure 5B).
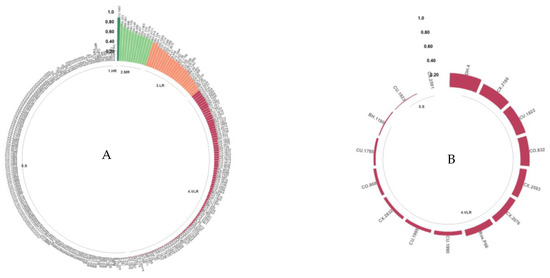
Figure 5.
Distribution of resistance to C. kahawae isolates Cam1 (A) and CC (B) in the progeny derived from the Cat. × TH CIFC 1343 population. HR: high resistance; MR: moderate resistance; LR: low resistance; VLR: very low resistance; S: susceptible. The amount of resistance to CBD as determined by the resistance scale is represented by the numbers on the Y-axis.
3.4.3. Resistance to the M and M2 Isolates
For the isolates from Malawi (M and M2), more than 75% of the population evaluated was classified as HR or MR. Very few genotypes were grouped into the least resistant classes (Figure 6A,B). In the resistance tests with M2, no S class genotypes were found.
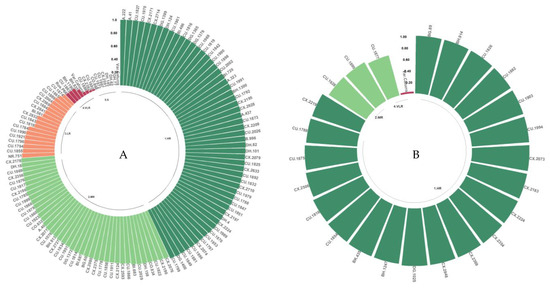
Figure 6.
Distribution of resistance to C. kahawae isolates M (A) and M2 (B) in the progeny derived from the Cat. × TH CIFC 1343 population. High resistance; MR: moderate resistance; LR: low resistance; VLR: very low resistance; S: susceptible. The amount of resistance to CBD as determined by the resistance scale is represented by the numbers on the Y-axis.
3.4.4. Resistance to the Que2 Isolate
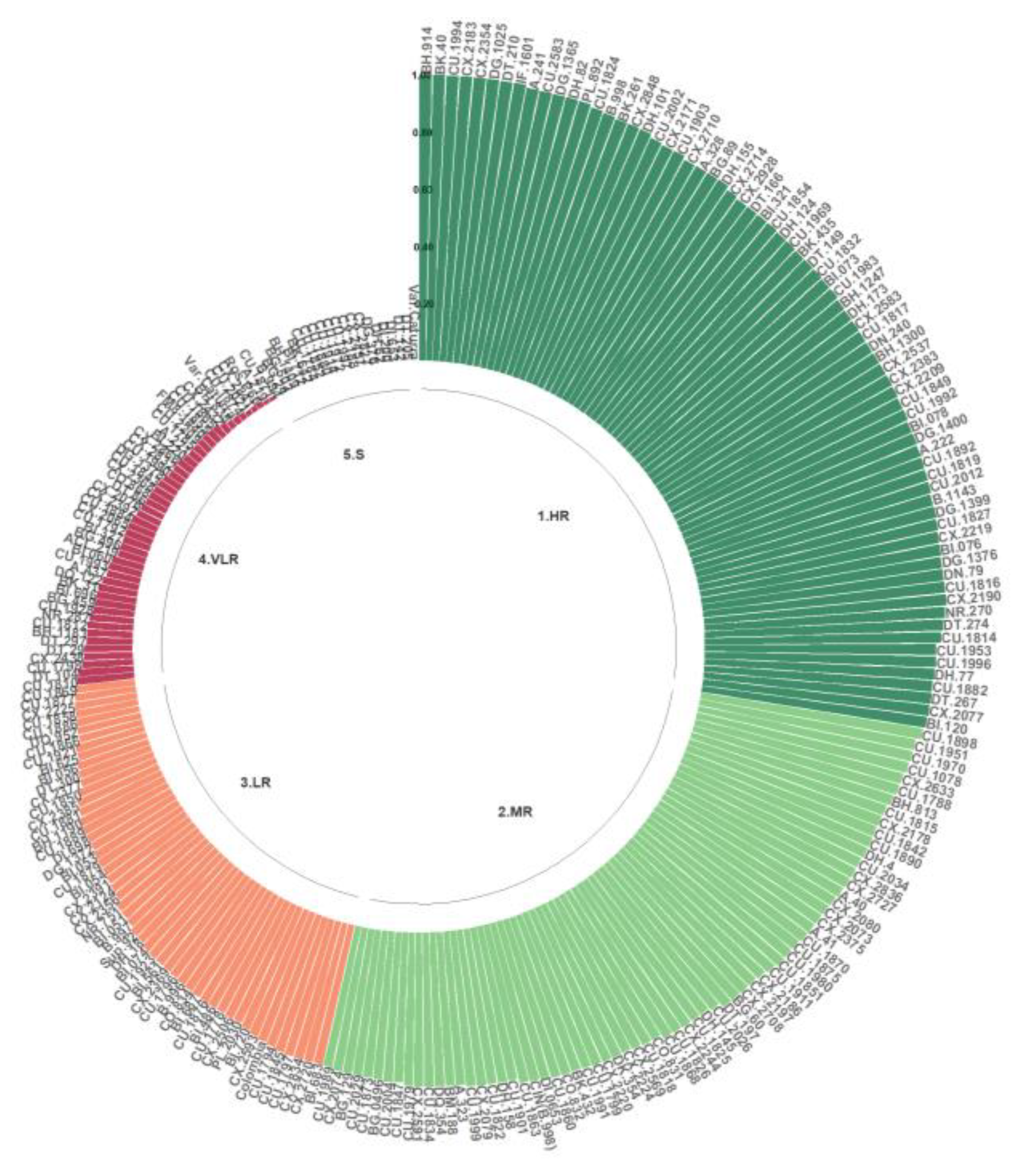
For the Kenyan isolate, the HR, MR, and LR classes were observed. The genotypes with the highest resistance corresponded to 73.76% of the population evaluated with Que2, most notably BH.914, BK.40, CU.1994, CX.2183, CX.2354, DG.1025, DT.210, and IF.1601, along with A.241, CU.2583, DG.1365, DH.82, PL.892, CU.1824, B.998, BK.261, CX.2848, DH.101, CU.2002, CX.2171, CU. 1903, CX.2710, A.328, BG.89, DH.155, CX.2714, CX.2928, DT.166, BI.321, CU.1854, DH.124, CU.1969, BK.435, DT.149, CU.1832, BI.073, CU.1983, BH.1247, DH.173, CX.2583, CU.1817, DN.240, BH.1300, CX.2537, CX.2383, CX. 2209, CU.1849, CU.1992, BI.078, DG.1400, A.222, CU.1892, CU.1819, CU.2012, B.1143, DG.1399, CU.1827, CX.2219, BI.076, DG.1376, DN.79, CU.1816, CX.2190, NR.270, DT.274, CU.1814, CU.1953, CU.1996, DH.77, CU.1882, DT.267, CX.2077, and BI.120. Only 18.25% of the genotypes were categorized in the VLR class, and only 7.98% were grouped in the S class (Figure 7).
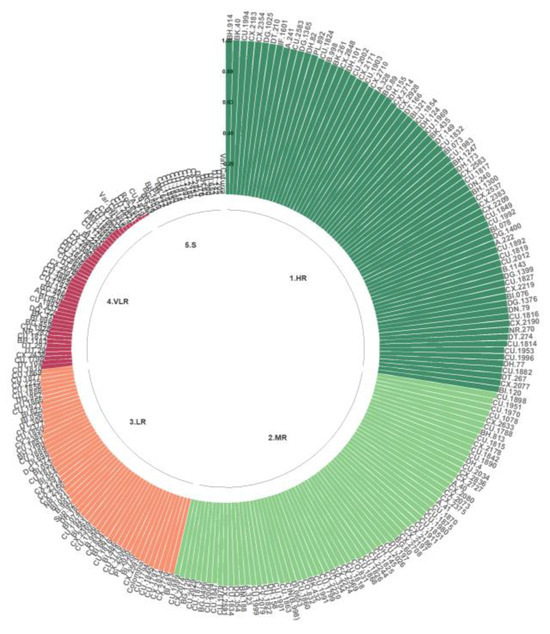
Figure 7.
Distribution of resistance to C. kahawae isolate Que2 in the progeny derived from the Cat. × TH CIFC 1343 population. HR: high resistance; MR: moderate resistance; LR: low resistance; VLR: very low resistance; S: susceptible. The amount of resistance to CBD as determined by the resistance scale is represented by the numbers on the Y-axis.
3.4.5. Resistance to the R Isolate
Four different classes of resistance to C. kahawae of Rwandan origin were observed. In this sense, 36.36% of the progenies (CU.1816, CU.1825, CU.1827, and CU.1842) were grouped into the HR, MR, and LR classes, and 63.64% were in the VLR class (Figure 8). No genotypes were identified that were classified in class S for the R isolate.
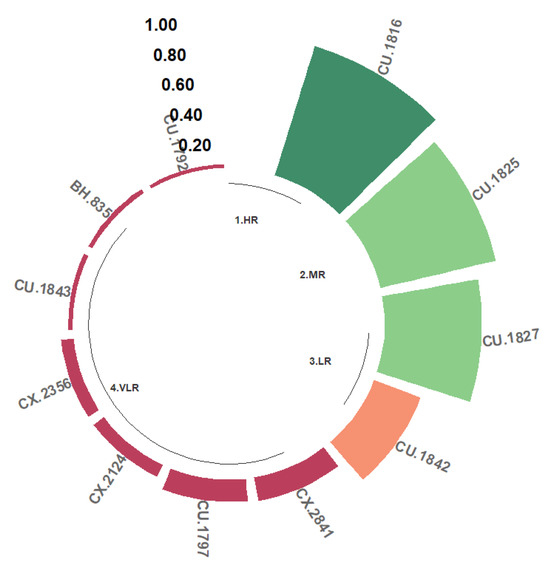
Figure 8.
Distribution of resistance to C. kahawae isolate R in the progeny derived from the Cat. × TH CIFC 1343 population. HR: high resistance; MR: moderate resistance; LR: low resistance; VLR: very low resistance; S: susceptible. The amount of resistance to CBD as determined by the resistance scale is represented by the numbers on the Y-axis.
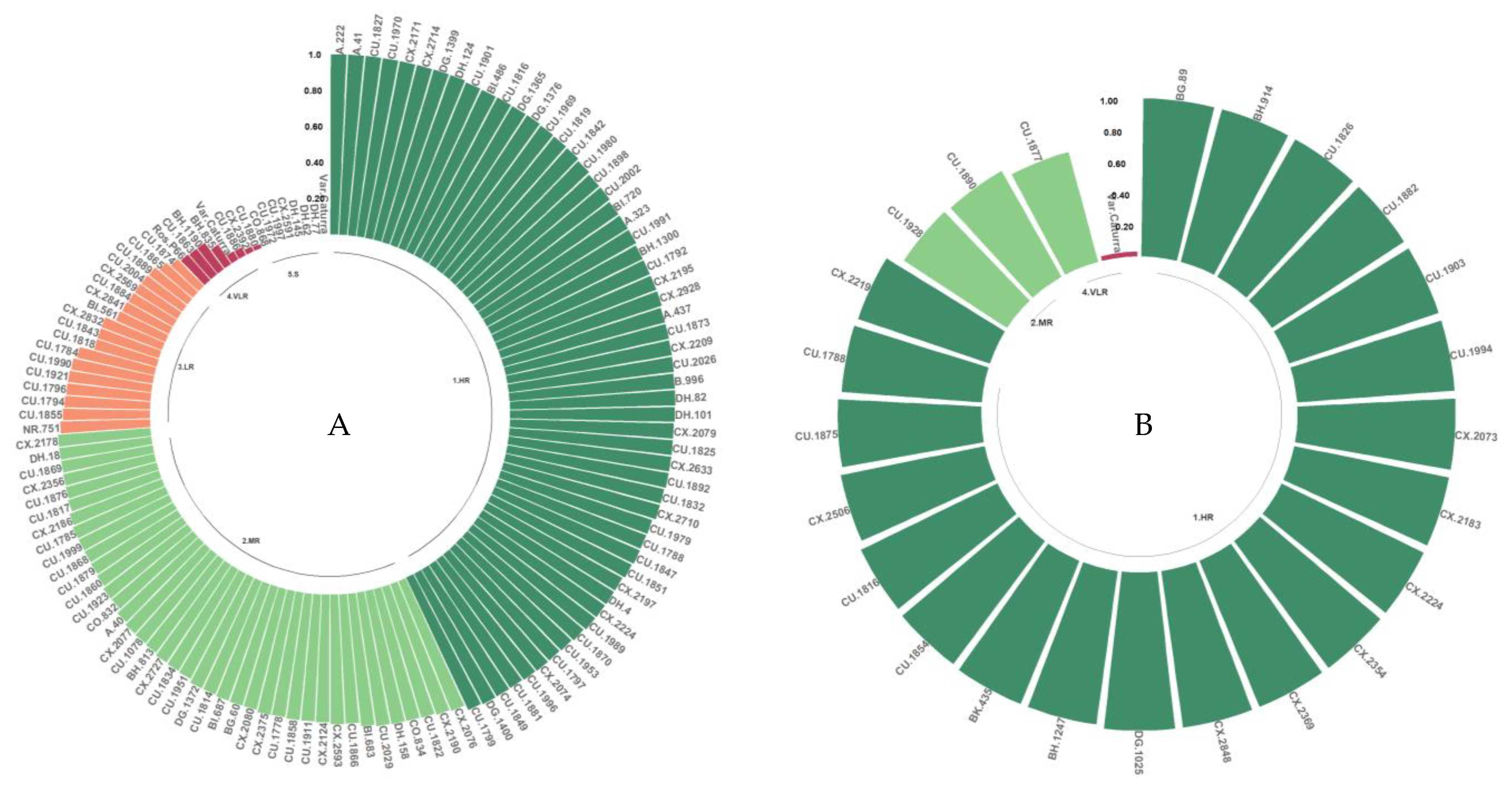
3.4.6. Resistance to the Z12, Z9, Zim1, and Zim12 Isolates
The resistance classes observed for isolates Z12, Z9, Zim1, and Zim12, which originated in Zimbabwe, were mainly HR, MR, and LR (Figure 9A–D). Of the evaluated genotypes, 87.4% were classified as HR, MR, or LR, of which 20.4% were HR. The S class constituted 12.6% of the genotypes evaluated with these isolates.
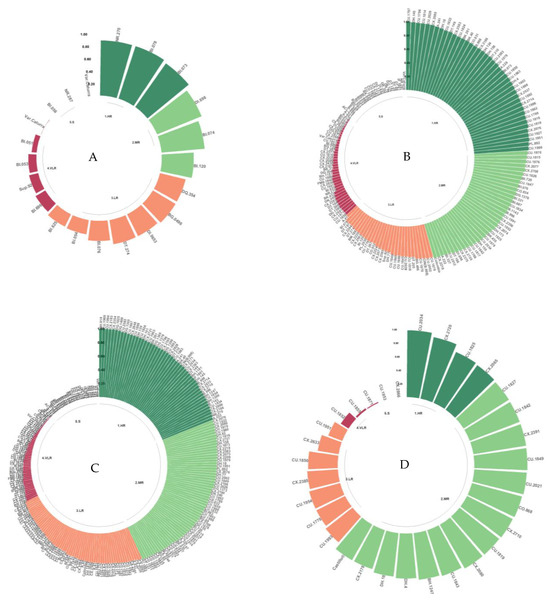
Figure 9.
Distribution of resistance to C. kahawae isolates Z12 (A), Z9 (B), Zim1 (C), and Zim12 (D) in the progeny derived from the Cat. × TH CIFC 1343 population. HR: high resistance; MR: moderate resistance; LR: low resistance; VLR: very low resistance; S: susceptible. The amount of resistance to CBD as determined by the resistance scale is represented by the numbers on the Y-axis.
3.5. Resistance in Other Populations
When accessions of Ethiopian origin from the CCC were evaluated, all four resistance classes and the susceptibility class were observed, with high and medium resistance (HR and MR) to isolates from Cameroon and Angola, which are reported to be highly aggressive (Cam1, Ang29), which is particularly noteworthy (Table 2). Similarly, for isolates of high/medium and medium/low aggressiveness from Kenya and Zimbabwe, between 4.0% and 10.8% of genotypes were classified in the HR class and between 3.7% and 17.8% in the MR class. The remaining genotypes were grouped into the least resistant classes (Table 8). In the Cat. × Ethiopian population, the evaluated genotypes were grouped into the least resistant classes, LR and VLR, with no genotypes classified into the HR and MR classes.

Table 8.
Total percentage of genotypes evaluated by isolate and the C. kahawae resistance classes in which the populations of the Colombian coffee collection were grouped.
Genotypes derived from TH CIFC 1343 and the Typica/Bourbon group were classified as HR and MR for the isolates of Kenyan and Zimbabwean origin and MR for the Cam1 isolate. Other genotypes were classified into the VLR class. In the population derived from hybridization between C. arabica and a diploid Coffea species, C. canephora, a higher percentage of genotypes were grouped into the HR class than in the other populations. Similarly, a higher percentage of genotypes were classified as MR for the Cam1 isolate, which is considered highly aggressive (Table 8).
In the Typica/Bourbon group, resistance was distributed across all the resistance classes and the susceptibility class. Notably, some genotypes were entirely classified as HR to isolates of Zimbabwean origin (Table 8).
3.6. Evidence for the Existence of Races in C. kahawae
Although there is no conclusive evidence for the existence of races in C. kahawae, our results suggest that they likely occur in the C. arabica–C. kahawae complex. This behavior in pathogen specificity on the host (C. arabica) was observed in genotypes derived from Cat. × TH CIFC 1343, wild genotypes, and interspecific hybrids (C. arabica × C. canephora) evaluated with isolates of high and medium/low aggressiveness (Table 9). While some genotypes were classified in class S, some isolates were also grouped in the HR class or others.

Table 9.
Percentage of hypocotyls affected by CBD with high resistance (Grade 1) and complete susceptibility (Grade 4) in the C. arabica–C. kahawae complex in genotypes from four populations.
These genotypes can eventually be considered a differentiating series of possible races in C. kahawae.
4. Discussion
4.1. CBD Resistance Class Scale
In countries where CBD is absent, direct [40,47,48] and indirect [30,31,49] laboratory techniques are the tools that support quick decision-making, identifying genotypes with genetic resistance to the disease. However, direct techniques such as those used to identify resistance to CBD [40,47,48] and the information obtained can represent a challenge for interpretation when the method of qualification and registration of the information is not standard [42].
In this sense, the direct evaluation technique using hypocotyls [38,40,41] qualifies the phenotypic reaction against C. kahawae by registering the degree of disease development [40,41]. However, the C. arabica genetic improvement program in Colombia has shown the difficulty of interpreting these scales; while they unequivocally identify completely resistant and susceptible genotypes, classifying the intermediate categories is more complex. This difficulty has represented a challenge for plant breeders in Colombia; however, it has not been an impediment to advancing the development of varieties resistant to CBD. On the other hand, from the very moment of the conception of the hypocotyl methodology to evaluate CBD resistance in the 1970s, it was evident that identifying intermediate categories of resistance to CBD represented greater complexity, as suggested by Cook [40], who developed the method of evaluating resistance to CBD via the hypocotyl method.
These categories of intermediate resistance to CBD, which are highly complex with respect to classification, suggest that resistance to the disease is controlled by genes with an additive dominant effect and in some cases, with a recessive effect [43,49], but there is no current consensus in the scientific community on the type of resistance mechanisms present [9]. Therefore, having a standard protocol for objective data interpretation or the evaluation of resistance via hypocotyl tests is highly beneficial for genetic improvement programs, since this method presents a greater correlation (>0.73) with CBD incidence values under field conditions [38].
The classification scale of genetic resistance to CBD for C. arabica presented in this study was obtained from the rigorous evaluation of 331,386 hypocotyls using the standard evaluation method, corresponding to four times as many hypocotyls than that used when the method was first developed [48,49]. The scale groups resistance to CBD into five classes, HR, MR, LR, VLR, and S (Table 4), on the basis of the assessment of the degree of disease progression, with greater than 95% reliability [41]. Before this report, there was no scale that would allow this classification to be carried out with statistical support.
This advance in the categorization for the objective classification of resistance to CBD from a rapid, direct technique and with high correlation with the expression of the disease under epidemic conditions contributes to making decisions in the selection of genotypes resistant to C. kahawae. This contribution to the classification of the phenotypic expression of resistance to CBD complements the advances achieved in Kenya and Ethiopia in the 1970s and 1980s [38,40,48], which to date have supported the processes of genetic improvement for resistance to CBD in C. arabica.
The scale proposed here classifies resistance in C. arabica, regardless of the isolate, because it was obtained from different sources of genetic resistance; isolates with different levels of aggressiveness, high, medium, and low [10,12,46], and three geographical origins, Angola, Cameroon, and Kenya [4,11] were selected. Additionally, the adoption of the scale by genetic improvement programs for resistance to CBD will complement the classifications obtained from the average degree of infection [41,50] and may become a single and standard language, which can easily be interpreted by plant breeders and phytopathologists from countries seeking to advance the development of varieties of C. arabica with resistance to C. kahawae using the hypocotyl test.
On the other hand, this is the first time that a country without the disease has developed a statistically reliable tool to perform the phenotypic preselection of genotypes with resistance to CBD, which will doubtlessly benefit other countries in their selection processes.
4.2. Aggressiveness of C. kahawae Isolates in TH
No immunity to CBD was identified in the populations evaluated. The resistance expressed by the evaluated genotypes was distributed among the different resistance classes (Table 4). This behavior can be attributed to the very nature of the inheritance of the genes responsible for resistance [43,51], complemented by the evident genetic diversity in resistance to different isolates of C. kahawae. In this manner, what has been suggested by other researchers is confirmed [52].
Indirectly, the phenotypic response of the genotypes to specific isolates can be an indicator of the aggressiveness of each of the isolates. This result is in agreement with that reported for isolates originating from Angola and Cameroon, which have been classified as highly aggressive [10,12,46]. On the basis of the percentages of hypocotyls in the least resistant classes (VLR and S in the populations evaluated), C. kahawae from Angola (Ang29) and Cameroon (Cam1, CC) can be considered the most aggressive isolates among the isolates referenced in this research. Among the hypocotyls evaluated with these isolates, more than 86% developed lesions forming a ring around the entire hypocotyl or completely died. These symptoms are equivalent to the maximum expression of the disease [41].
Isolates of Rwandan origin have also been identified as highly aggressive [10,12]. The Cat. × TH CIFC 1343 population studied here was evaluated with the R isolate, which is different from the isolate used in studies where its level of aggressiveness was reported [10,12]. However, our results, which are based on the distribution of resistance classes, suggest that its pathogenicity does correspond to a highly aggressive isolate.
Aggressiveness was observed in isolates Zim12, Zim1, and Z9 from Zimbabwe and Que2 from Kenya, consistent with previous reports [12,46]. The advanced populations derived from Cat. × TH CIFC 1343 presented a greater level of resistance to these isolates compared to the isolates from Angola, Cameroon, and Rwanda.
The lowest aggressiveness was associated with isolates from Malawi and Zimbabwe (M2, M, and Z12). The phenotypic response of the hypocotyls was characterized by more than 84% of the hypocotyls having small lesions without them coalescing. In the population derived from TH CIFC 1343, these isolates had the least effect, as has been previously reported in evaluations of varieties considered susceptible [12].
In general terms, diverse degrees of aggressiveness in isolates from the same geographic origin was observed, as has been previously documented [12,43]. This same behavior was observed in progeny derived from Cat. × TH CIFC 1343, in which different levels of resistance were found in different isolates from the same geographical origin, such as isolates of Zimbabwean origin.
4.3. Diversity of Resistance to CBD in the CCC Genotypes
TH is the main source of resistance to limiting and potential diseases for growing coffee, and its genetic value is so important that it has been used as a source of resistance by different genetic improvement programs around the world [25]. TH is diverse [26], and in Colombia, varieties resistant to rust and CBD have been developed from TH CIFC 1343 [35,53,54].
Although the inheritance of resistance to CBD is complex [43,50], the results of evaluations in progenies derived from TH CIFC 1343 clearly show that it is possible to obtain lasting resistance to C. kahawae, as described previously with the hybrid variety Ruiru 11 in Africa [19,24,28,29]. This antecedent highlights the importance of this genetic resource (TH CIFC 1343) as a source of resistance to cherry disease [30,50,53,55]. On the other hand, it is important to note that this same TH is the source of resistance to rust in the coffee varieties developed in Colombia.
However, the universally documented resistance of TH, the variety from which it developed, is not synonymous with immunity to C. kahawae [39,52,55,56]. Therefore, knowledge of resistance in derived varieties is beneficial for strengthening crop protection strategies in the event of the eventual arrival of the pathogen in countries where the disease has not yet been reported. One of those strategies is the knowledge and use of various sources of resistance. In the genotypes of Ethiopian origin used in this study, as with those that constitute the other populations evaluated here, resistance was found to be distributed across the different resistance classes (Table 4), including the HR class (ET.37C9, ET.56, E.537, E.538, E.139, Rume Sudan, E.53, E.264, E.288, and E.417), and the resistance of some of these genotypes, such as ET.56 and Rume Sudan, has been previously reported [57,58,59].
Within the Typica/Bourbon group, Local Bronce.8, Local Bronce.12, Jackson.2 and Villalobos were noteworthy and were grouped in the highest resistance classes. Genotypes of the Local Bronce population have already been reported to present some degree of resistance to CBD [57,58,59]. We found no previous reports documenting resistance to Jackson and genotypes of Ethiopian origin. From the group of interspecific hybrids, the genotypes with the highest degree of resistance were obtained from C. arabica × C. canephora, and our findings agree with previous studies [52].
Although the possible existence of physiological races has been suggested in C. kahawae [52], to date there is no conclusive evidence to support this hypothesis. However, the results obtained from the Cat. × TH CIFC 1343 population suggest that the existence of races in the C. arabica–C. kahawae complex is probable (Table 9). This is how genotypes completely susceptible (Grade 4) to isolates such as Ang29 and Cam1, and at the same time, highly resistant to other isolates from, for example, Kenya, Malawi, and Zimbabwe, were identified (Table 3), indicating contrasting resistance reactions to different isolates. Similar results have been reported with isolates from Angola, Malawi, and Kenya [52].
From the perspective of our findings, the evaluation of C. arabica varieties for resistance to CBD is highly valuable, and none of the research centers other than those on the African continent have evaluated their gene banks for resistance to a potential disease such as CBD (or if so, their research data have not been published), except for the data previously reported by Cook and van der Vossen [47,48]. Therefore, contrary to expectations, countries that produce C. arabica, such as Colombia, have been working for more than 40 years to prepare their coffee growing industry in the event of the eventual arrival of C. kahawae. This is why we emphasize that management and control strategies have not been the sole responsibility of African countries, where the disease is currently present [9].
Cenicafé has advanced in the search for and incorporation of various sources of resistance to CBD (unpublished data). In the case of TH, the incorporation of disease resistance mechanisms in the varieties developed by Cenicafé has been validated since the 1970s [39]. More recently, varieties resistant to rust and currently planted by coffee growers in Colombia have been found to express Ck-1, which confers resistance to CBD [33] and displays a wide spectrum of resistance to different isolates with different levels of aggressiveness and geographical origins [32].
In this research, improved progeny with the potential to be incorporated into a genetic diversity strategy and the central axis of rust-resistant varieties in Colombia were also evaluated. The results confirm that there is a diversity of resistance to C. kahawae; however, Colombia and the producing countries should continue working on strengthening their strategies for the incorporation of resistance mechanisms of different origins for the development of future varieties.
4.4. Potential of Genetic Resistance to CBD in a Climate Change Scenario
C. kahawae is a pathogen that is dispersed mainly by rain [60], and the disease is potentiated by specific weather conditions [18]. Therefore, the development of varieties resistant to CBD is an important challenge and requires a detailed understanding of the pathogen and local patterns of the real and probable climate conditions that can affect crop development [61].
The historical records of climate in Colombia since 1980 have made it possible to classify the hourly distribution of rainfall as light (0.1 to 10.0 mm h−1), light to moderate (10.1 to 20.0 mm h−1), moderately strong (20.1 to 40.0 mm h−1), strong (40.1 to 60.0 mm h−1), and torrential (>60.0 mm h−1) [62], and under a climate change scenario, a significant increase in the number of moderate and strong rainfall events in Colombia is projected [62,63]. Additionally, an increase is expected in the minimum and maximum historical temperatures in the Colombian coffee region [63].
These predicted changes in rain and temperature events could favor pathogen dispersion and disease development, with its most severe effects on susceptible varieties. Possible increases in rainfall and minimum temperatures warn of the need to work on strategic fronts that allow us to face potential diseases such as CBD. One of these strategies involves the search for sources of resistance for the development of varieties with genetic resistance to C. kahawae.
5. Conclusions
CBD represents a potential risk for coffee growing in the Americas, and the best way to prepare for the eventual arrival of the pathogen is the use of genetic diversity and resistance. The historical evaluations carried out by Cenicafé and the data published here confirm that genetic resistance to CBD is not synonymous with immunity. For this reason, all countries where coffee cultivation constitutes an important economic industry should continue to join efforts to evaluate their varieties and characterize their germplasm banks to identify possible sources of resistance to potential threats such as CBD.
The proposed CBD resistance classification tool unifies the language between research centers and their breeding programs for genetic resistance to CBD in such a way that it facilitates the evaluation, understanding, and interpretation of data to strengthen the knowledge network regarding CBD. On the other hand, the scale could contribute to the development of molecular markers linked to resistant genes to CBD, thereby allowing their validation under laboratory conditions.
Additionally, the results confirm the importance of TH CIFC 1343 as a source of resistance to CBD, the potential of CCC as a source of genes for resistance to this disease, and new evidence of the probable existence of races or pathotypes in C. kahawae.
Similarly, this is the first time that direct tests have shown the possible panorama of susceptibility to C. kahawae in coffee growing on the basis of TH CIFC 832/1. For this reason, the countries that support coffee growing using this source of resistance should move toward the use of new genetic sources and strategies to strengthen their coffee farms. Finally, the search for new sources of genes aimed at strengthening the genetic resistance already found in the varieties developed by Cenicafé in Colombia is recommended.
Author Contributions
Conceptualization, J.Q.-C., Á.L.G.-B. and C.P.F.-R.; investigation, J.Q.-C., C.P.F.-R. and V.M.P.V.; writing—original draft preparation, J.Q.-C.; writing—review and editing, J.Q.-C., C.P.F.-R., E.C.M.-R., Á.L.G.-B. and V.M.P.V. All authors have read and agreed to the published version of the manuscript.
Funding
This work was funded by the National Coffee Research Center (Cenicafé) (Crossref Funder ID 100019597).
Data Availability Statement
Restrictions apply to the dataset. The datasets presented in this article are not easily available because they were obtained with resources from the National Federation of Coffee Growers of Colombia and are part of the development program for C. arabica varieties with genetic resistance to CBD. Requests to access the datasets should be directed to the National Coffee Research Center (Cenicafé).
Acknowledgments
The authors thank the researchers Germán Moreno and María del Pilar Moncada for having initiated the work to explore resistance to CBD in the Colombian coffee collection (CCC). We would like to thank the National Federation of Coffee Growers of Colombia for funding this research and the General System of Royalties (SGR) of Colombia and the University of Caldas as the executing entity of project BPIN 2021000100132, Training of high-level human capital: doctorate master’s degree with emphasis on research and medical-surgical specialties to meet the demands in the territory of Caldas for the funding of academic training at the doctoral level in Agricultural Sciences in the form of a scholarship awarded to the main author of this article.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
References
- Waller, J.M.; Bridge, P.D.; Black, R.; Hakiza, G. Characterization of the coffee berry disease pathogen, Colletotrichum kahawae sp. nov. Mycol. Res. 1993, 97, 989–994. [Google Scholar] [CrossRef]
- Cabral, A.; Azinheira, H.G.; Talhinhas, P.; Batista, D.; Ramos, A.P.; do Céu Silva, M.; Oliveira, H.; Várzea, V. Pathological, Morphological, Cytogenomic, Biochemical and Molecular Data Support the Distinction between Colletotrichum cigarro comb. et stat. nov. and Colletotrichum kahawae. Plants 2020, 9, 502. [Google Scholar] [CrossRef] [PubMed]
- Dean, R.; Van Kan, J.L.; Pretorius, Z.; Hammond-Kosack, K.E.; Di Pietro, A.; Spanu, P.D.; Jason, J.; Dickman, M.; Kahmann, R.; Ellis, J.; et al. The Top 10 fungal pathogens in molecular plant pathology. Mol. Plant Pathol. 2012, 13, 414–430. [Google Scholar] [CrossRef] [PubMed]
- Vieira, A.; Nuno Silva, D.; Várzea, V.; Salgueiro Paulo, O.; Batista, D. Novel insights on colonization routes and evolutionary potential of Colletotrichum kahawae, a severe pathogen of Coffea arabica. Mol. Plant Pathol. 2019, 19, 2488–2501. [Google Scholar] [CrossRef]
- Liu, F.; Ma, Z.Y.; Hou, L.W.; Diao, Y.Z.; Wu, W.P.; Damm, U.; Song, S.; Cai, L. Updating species diversity of Colletotrichum, with a phylogenomic overview. Stud. Mycol. 2022, 101, 1–56. [Google Scholar] [CrossRef] [PubMed]
- Loureiro, A.; Nicole, M.R.; Várzea, V.; Moncada, M.P.; Bertrand, B.; do Céu Silva, M.C. Coffee resistance to Colletotrichum kahawae is associated with lignification, accumulation of phenols and cell death at infection sites. Physiol. Mol. Plant Pathol. 2012, 77, 23–32. [Google Scholar] [CrossRef]
- Garedew, W.; Lemessa, F.; Pinard, F. Assessment of berry drop due to coffee berry disease and non-CBD factors in Arabica coffee under farmers fields of Southwestern Ethiopia. Crop Prot. 2017, 98, 276–282. [Google Scholar] [CrossRef]
- Yemo Ngouegni, Y.; Gaston Noumbo, R.T.; Keuete Kamdoum, E.; Samuel, F.B.; Victor François, N. Morphocultural and aggressiveness variability of Colletotrichum kahawae isolates from different altitudes of the Western highlight zone of Cameroon. Acta Ecol. Sin. 2023, 43, 1058–1066. [Google Scholar] [CrossRef]
- Adugna, G. Coffee berry disease: A century-old anthracnose of green berries of Arabica coffee (Coffea arabica L.) in Africa. J. Plant Dis. Prot. 2023, 131, 315–328. [Google Scholar] [CrossRef]
- Loureiro, A.; Guerra-Guimarães, L.; Lidon, F.C.; Bertrand, B.; do Céu Silva, M.C.; Várzea, V. Isoenzymatic characterization of Colletotrichum kahawae isolates with different levels of aggressiveness. Trop. Plant Pathol. 2011, 36, 287–293. [Google Scholar] [CrossRef]
- Silva, D.N.; Talhinhas, P.; Luzolo Manuel, L.C.; Gichuru, E.K.; Loureiro, A.; Várzea, V.; Paulo Octávio, S.; Batista, D. Host-jump drives rapid and recent ecological speciation of the emergent fungal pathogen Colletotrichum kahawae. Mol. Ecol. 2012, 21, 2655–2670. [Google Scholar] [CrossRef] [PubMed]
- Vieira, A.; Diniz, I.; Pereira, A.P.; do Céu Silva, M.C.; Várzea, V.; Batista, D. Aggressiveness profiling of the coffee pathogen Colletotrichum kahawae. Plant Pathol. 2019, 68, 358–368. [Google Scholar] [CrossRef]
- Mouen Bedimo, J.A.; Bieysse, D.; Nyassé, S.; Nottéghem, J.L.; Cilas, C. Role of rainfall in the development of coffee berry disease in Coffea arabica caused by Colletotrichum kahawae, in Cameroon. Plant Pathol. 2010, 59, 324–329. [Google Scholar] [CrossRef]
- Yoganie, Y.N.; Gaston, T.N.; Elie, K.K.; Fru, B.S.; Nguetsop, V.F. Influence of altitudes on the cultural and morphological variation of Colletotrichum kahawae isolates, the causal agent of coffee berry disease in the West Region of Cameroon. Afr. J. Agric. Res. 2022, 18, 898–906. [Google Scholar] [CrossRef]
- Daba, G.; Helsen, K.; Berecha, G.; Lievens, B.; Debela, A.; Honnay, O. Seasonal and altitudinal differences in coffee leaf rust epidemics on coffee berry disease-resistant varieties in Southwest Ethiopia. Trop. Plant Pathol. 2019, 44, 244–250. [Google Scholar] [CrossRef]
- Rayner, R.W. Coffee Berry Disease—A Survey of Investigations Carried Out Up to 1950. East Afr. Agric. J. 1952, 17, 130–158. [Google Scholar] [CrossRef]
- Nutman, F.F.; Roberts, F.M.; Freeman, G.H. Seasonal variations in the sporulating capacity of the fungus causing coffee berry disease. Ann. Appl. Biol. 1969, 64, 85–99. [Google Scholar] [CrossRef]
- Motisi, N.; Ribeyre, F.; Poggi, S. Coffee tree architecture and its interactions with microclimates drive the dynamics of coffee berry disease in coffee trees. Sci. Rep. 2019, 9, 2544. [Google Scholar] [CrossRef]
- Uganda Coffee Development Authority (UCDA). Arabica Coffee Handbook: A Sustainable Coffee Industry with High Stakeholder Value for Social Economic Transformation; Uganda Coffee Development Authority: Kampala, Uganda, 2019; pp. 80–81.
- Motisi, N.; Papaix, J.; Poggi, S. The dark side of shade: How microclimates drive the epidemiological mechanisms of coffee berry disease. Phytopathology 2022, 112, 1235–1243. [Google Scholar] [CrossRef]
- Duque Orrego, H. Adopción de Una Variedad de Café Resistente a la Roya: El Caso de la Variedad Colombia; Cenicafé: Manizales, Colombia, 2005; p. 76. [Google Scholar]
- Arias Suárez, J.C. Efecto de mezclas y unidad del área del genotipo sobre el incremento progresivo de Hemileia vastatrix Berk. y Br. Cenicafé 2019, 70, 30–44. [Google Scholar] [CrossRef]
- van der Graaff, N.A. Seleection of Arabica Coffee Types Resistance to Coffee Berry Disease in Ethiopia; Mededlingen Landbouwhogeschool: Wageningen, The Netherlands, 1981; p. 110. [Google Scholar]
- Nyoro, J.K.; Sprey, L.H. Introducing Ruiru 11 to the estates. Kenya Coffee 1986, 51, 7–14. [Google Scholar]
- Rodrigues, C.J., Jr.; Goncalves Mayer, M.; Várzea, V. Importância do Híbrido de Timor para o território e para o melhoramento da cafeicultura mundial. Rev. Ciênc. Agrár. 2001, 2, 203–216. [Google Scholar]
- Silva, R.A.; Zambolim, L.; Castro, S.L.; Rodrigues, H.S.; Cruz, C.D.; Caixeta, E.T. The Híbrido de Timor germplasm: Identification of molecular diversity and resistance sources to coffee berry disease and leaf rust. Euphytica 2018, 153, 214. [Google Scholar] [CrossRef]
- Vermeulen, H. Screening of Varieties for Resistance. In Annual Report Coffee Research Foundation Kenya 1965/1966; Coffee Research Foundation: Ruiru, Kenya, 1967; pp. 41–43. [Google Scholar]
- Omondi, C.O. Resistance to Coffee Berry Disease in Arabica Coffee Variety ‘Ruiru 11’. Plant Breed. 1994, 112, 256–259. [Google Scholar] [CrossRef]
- Gichimu, B.M.; Gichuru, E.K.; Mamati, G.E.; Nyende, A.B. Occurrence of Ck-1 gene conferring resistance to Coffee Berry Disease in Coffea arabica cv. Ruiru 11 and its parental genotypes. J. Agric. Crop Res. 2014, 2, 51–61. [Google Scholar]
- Gichuru, E.K.; Agwanda, C.O.; Combes, M.C.; Mutitu, E.W.K.; Ngugi, E.C.; Bertrand, B.; Lashermes, P. Identification of molecular markers linked to a gene conferring resistance to coffee berry disease (Colletotrichum kahawae) in Coffea arabica. Plant Pathol. 2008, 57, 1117–1124. [Google Scholar] [CrossRef]
- Guzmán, F.; Moncada-Botero, M.d.P. Evaluación de marcadores SSR ligados a resistencia a Colletotrichum kahawae en Coffea arabica. Cenicafé 2012, 63, 7–22. [Google Scholar]
- Centro Nacional de Investigaciones de Café-Cenicafé. Informe Anual 2023; Cenicafé: Chinchiná, Colombia, 2023; pp. 82–97. [Google Scholar] [CrossRef]
- Maldonado Londoño, C.E.; Ángel Giraldo, L. Resistencia genética a la Enfermedad de la Cereza del Café en variedades cultivadas en Colombia. Cenicafé 2020, 71, 68–90. [Google Scholar] [CrossRef]
- van der Vossen, H.A.; Walyaro, D.J. The coffee breeding programe in Kenya: A review of progress made since 1971 and plan of action for the coming years. Kenya Coffee 1981, 46, 113–130. [Google Scholar]
- Castillo Zapata, J.; Moreno Ruiz, G. La Variedad Colombia: Selección de un Cultivar Compuesto Resistente a la Roya del Cafeto; Cenicafé: Chinchiná, Colombia, 1986; p. 171. [Google Scholar]
- Gichuru, E.K.; Alwora, G.; Gimase, J.; Kathurima, C. Coffee Leaf Rust (Hemileia vastatrix) in Kenya—A Review. Agronomy 2021, 11, 2590. [Google Scholar] [CrossRef]
- van der Vossen, H.A. Screening for CBD Resistance. In Annual Report Coffee Research Foundation Kenya 1974/1975; Coffee Research Foundation: Ruiru, Kenya, 1976; pp. 84–103. [Google Scholar]
- van der Vossen, H.A.; Cook, R.T.; Murakaru, N.W. Breeding for resistance to coffee berry disease caused by Colletrotrichum coffeanum NOACK (Sensu HINDORF) in Coffea arabica L. I. Methods of preselection for resistance. Euphytica 1976, 25, 733–745. [Google Scholar] [CrossRef]
- van der Vossen, H.; Walyaro, D. Exchange Programme on Selection for CBD Resistance with Brazil and Colombia. In Annual Report Coffee Research Foundation Kenya 1977/1978; Coffee Research Foundation: Ruiru, Kenya, 1979; pp. 67–79. [Google Scholar]
- Cook, R.T. Screening Coffee Plants for CBD Resistance. In Annual Report Coffee Research Foundation, Kenya 1972/1973; Coffee Research Foundation: Ruiru, Kenya, 1973; pp. 66–68. [Google Scholar]
- van der Graaff, N.A. The principles of scaling and the inheritance of resistance to coffee berry disease in Coffea arabica. Euphytica 1982, 31, 735–740. [Google Scholar] [CrossRef]
- Postman, J.; Volk, G.; Aldwinckle, H. Standardized Plant Disease Evaluations Will Enhance Resistance Gene Discovery. HortScience 2010, 45, 1317–1320. [Google Scholar] [CrossRef]
- van der Vossen, H.A.; Walyaro, D.J. Breeding for resistance to coffee berry disease in Coffea arabica L. II. Inheritance of the resistance. Euphytica 1980, 29, 777–791. [Google Scholar] [CrossRef]
- Pinard, F.; Omondi, C.O.; Cilas, C. Detached berries inoculation for characterization of coffee resistance to coffee berry disease. J. Plant Pathol. 2012, 94, 517–523. [Google Scholar] [CrossRef]
- Alemu, K.; Adugna, G.; Lemessa, F.; Muleta, D. Induction of systemic resistance in Arabica coffee (Coffea arabica L.) against coffee berry disease (Colletotrichum kahawae Waller & Bridge) mediated through plant defense activator. Int. J. Pest Manag. 2018, 65, 313–323. [Google Scholar] [CrossRef]
- Pires, A.S.; Azinheira, H.G.; Cabral, A.; Tavares, S.; Tavares, D.; Castro, M.; Várzea, V.; do Céu Silva, M.C.; Abranches, R.; Loureiro, J.; et al. Cytogenomic characterization of Colletotrichum kahawae, the causal agent of coffee berry disease, reveals diversity in minichromosome profiles and genome size expansion. Plant Pathol. 2016, 65, 968–977. [Google Scholar] [CrossRef]
- Cook, R.T. Screening Coffee Plants for CBD Resistance. In Annual Report Coffee Research Foundation Kenya 1971/1972; Coffee Research Foundation: Ruiru, Kenya, 1972; pp. 66–74. [Google Scholar]
- van der Vossen, H.A. Screening for CBD and Leaf Rust Resistance. In Annual Report Coffee Research Foundation Kenya 1972/1973; Coffee Research Foundation: Ruiru, Kenya, 1973; pp. 54–57. [Google Scholar]
- Gimase, J.M.; Thagana, W.M.; Omondi, C.O.; Cheserek, J.J.; Gichimu, B.M.; Gichuru, E.K.; Cathrine, Z.; Sneller, C.H. Genome-Wide Association Study identify the genetic loci conferring resistance to Coffee Berry Disease (Colletotrichum kahawae) in Coffea arabica var. Rume Sudan. Euphytica 2020, 216, 86. [Google Scholar] [CrossRef]
- Mohammed, A.; Jambo, A. Importance and characterization of Coffee Berry Disease (Colletotrichum kahawae) in Borena and Guji zones, Sourthern Ethiopia. Plant Pathol. Microbiol. 2015, 6, 1000302. [Google Scholar] [CrossRef]
- van der Vossen, H.; Walyaro, D. Selection for CBD Resistance and Studies on its Inheritance. In Annual Report Coffee Research Foundation Kenya 1976/1977; Coffee Research Foundation: Ruiru, Kenya, 1978; pp. 76–95. [Google Scholar]
- Rodrigues, C.J., Jr.; Várzea, V.M. Evidence for the existence of physiological races of Colletotrichum coffeanum Noack sensu Hindorf. Kenya Coffee 1992, 57, 1417–1420. [Google Scholar]
- Flórez Ramos, C.P.; Maldonado, C.E.; Cortina Guerrero, H.A.; Moncada Botero, M.D.; Montoya, E.C.; Ibarra, R.L.N.; Unigarro, C.A.; Rendón, J.R.; Duque Orrego, H. Cenicafé 1: Nueva Variedad de Porte Bajo, Altamente Productiva, Resistente a la Roya y al CBD, con Mayor Calidad Física del Grano; Avance Técnico 469: Manizales, Colombia, 2016. [Google Scholar]
- Flórez Ramos, C.P.; Arias Suárez, J.C.; Maldonado, L.C.E.; Cortina, G.H.A.; Moncada Botero, M.D.P.; Quiroga-Cardona, J.; Molina-Vinasco, D.M.; García, J.C.; Duque, O.H. Variedades Castillo® Zonales Resistencia a la Roya con Mayor Productividad; Avance Técnico 489: Manizales, Colombia, 2018. [Google Scholar]
- Vermeulen, H. Screening of Varieties for Resistance. In Annual Report Coffee Research Foundation Kenya 1964/1965; Coffee Research Foundation: Ruiru, Kenya, 1966; pp. 57–60. [Google Scholar]
- Griffiths, E. Varietal resistance. In Annual Report Coffee Research Foundation Kenya 1967/1968; Coffee Research Foundation: Ruiru, Kenya, 1968; pp. 43–48. [Google Scholar]
- Coffee Research Foundation. Plant Pathology. In Annual Report Coffee Research Foundation Kenya 1962/1963; Coffee Research Foundation: Ruiru, Kenya, 1963; pp. 50–54. [Google Scholar]
- Firman, I.D. Screening of Coffee for Resistance to Coffee Berry Disease. East Afr. Agric. For. J. 1964, 29, 192–194. [Google Scholar] [CrossRef]
- Bouharmont, P. La Sélection du Caféier Arabica au Cameroun (1964–1991); CIRAD: Montpellier, France, 1995; p. 130.
- Griffiths, E.; Waller, J.M. Rainfall and cropping patterns in relation to coffee berry disease. Ann. Appl. Biol. 1971, 67, 75–91. [Google Scholar] [CrossRef]
- Wagner, S.; Jassogne, L.; Price, E.; Jones, M.; Preziosi, R. Impact of Climate Change on the Production of Coffea arabica at Mt. Kilimanjaro, Tanzania. Agriculture 2021, 11, 53. [Google Scholar] [CrossRef]
- Ramírez-Carabalí, C.; Sarmiento-Herrera, N.G.; García-López, J.C. Distribución y tendencias de las lluvias horarias en la región cafetera del Noreste de Sur América. Cenicafé 2024, 75, e75103. [Google Scholar] [CrossRef]
- Correa Ortiz, L.C.; Ocampo López, O.L.; Alba Castro, M.F. Análisis de tendencia de temperatura y precipitación para el departamento de Caldas (Colombia), mediante wavelets. Cienc. Ing. Neogranadina 2021, 31, 37–52. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).