Effects of Claroideoglomus etunicatum Fungus on the Growth Parameters of Maize (Zea mays L.) Plants under Boron Toxicity and Salt Stress
Abstract
:1. Introduction
2. Materials and Methods
2.1. Soil Preparation
2.2. Experimental Setup
2.3. Sampling and Analysis
2.4. Statistical Analysis
3. Results
3.1. Plant Growth
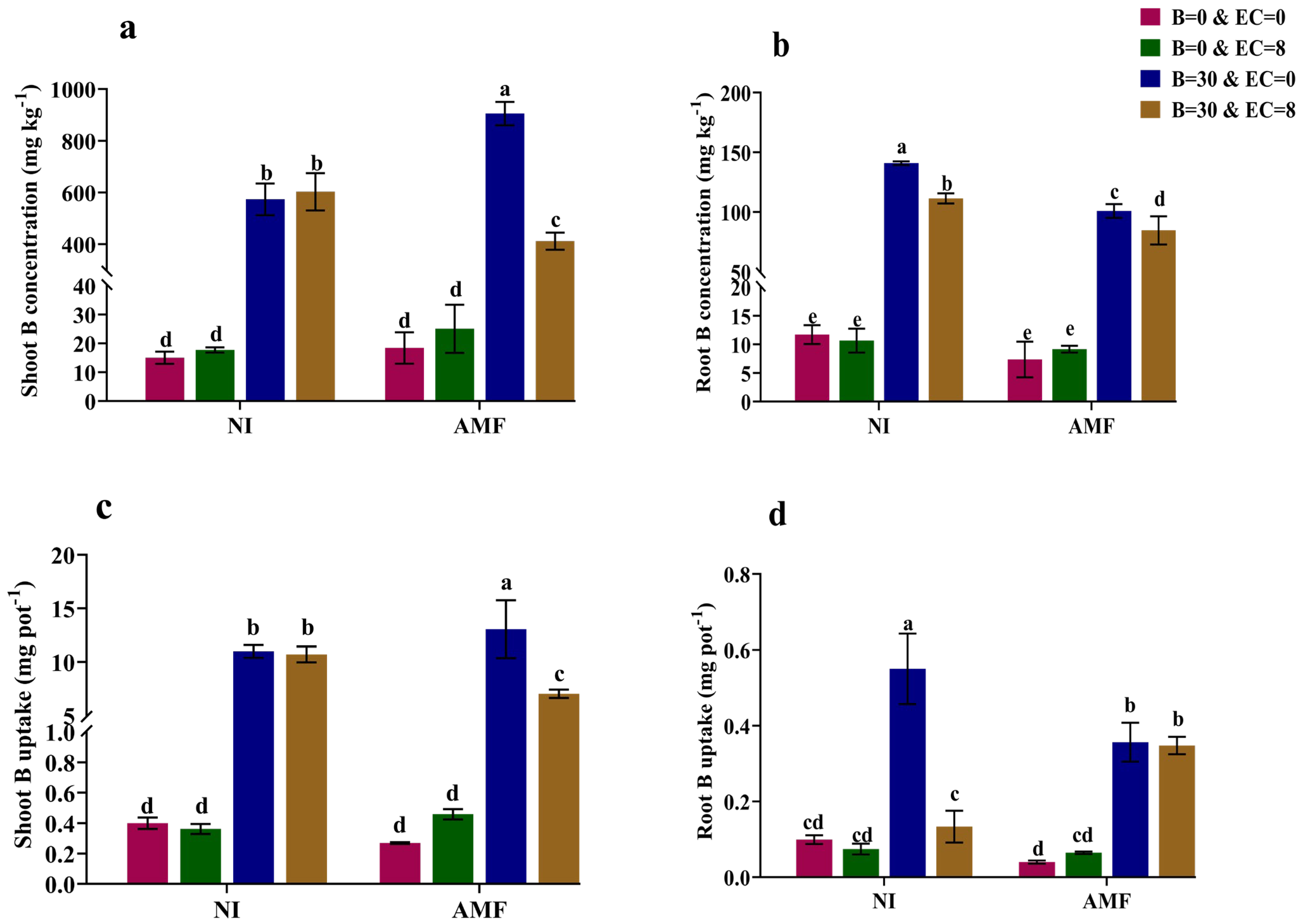
3.2. Plant B Concentrations and Uptake
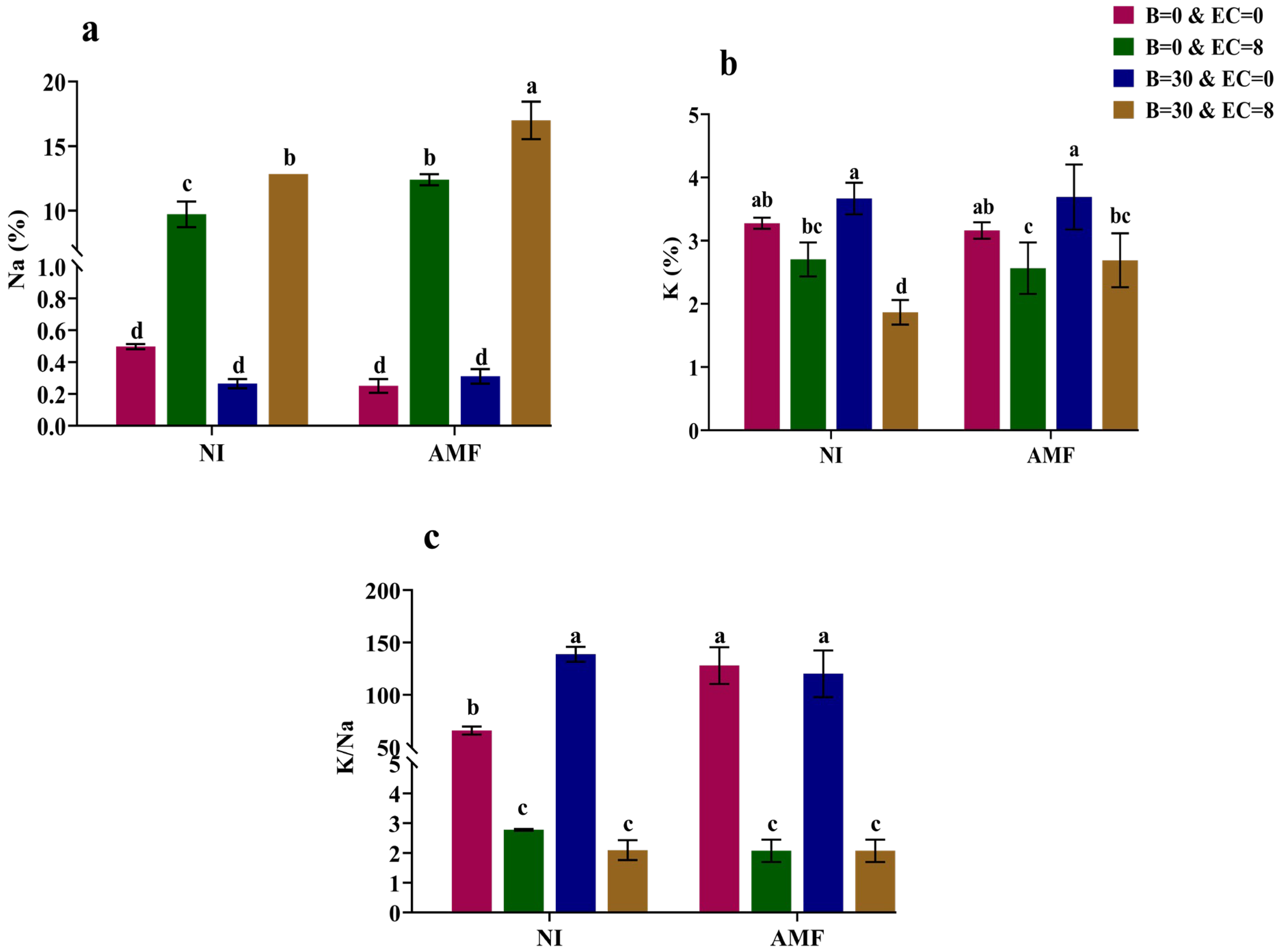
3.3. Shoot Na and K Concentrations and Uptake
3.4. Shoot Zn, Cu, Fe, and Mn Concentrations and Uptake
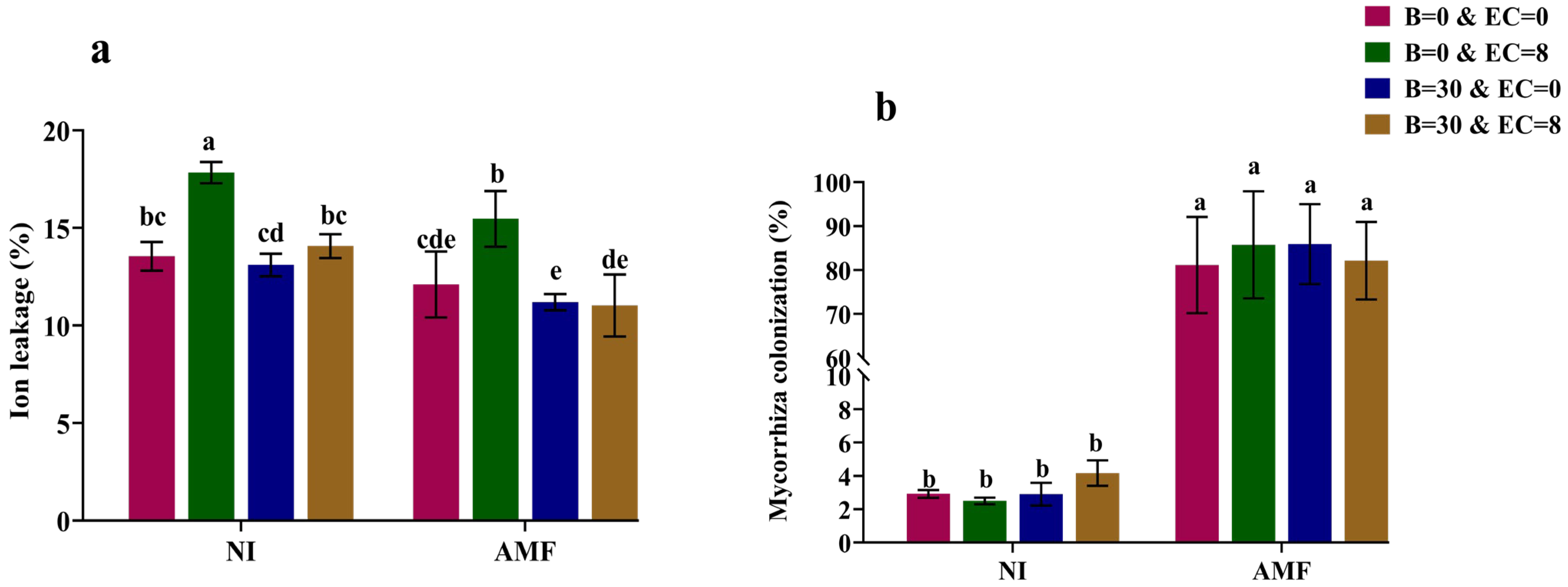
3.5. Ion Leakage
3.6. Mycorrhizal Colonization
4. Discussion
4.1. Detrimental Effects of Combined B and Salt Stress on Maize Plant Growth and the Influence of AMF Inoculation
4.2. Role of AMF Inoculation and Determination of Plant Nutrient Concentrations
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Wang, G.; DiTusa, S.F.; Oh, D.H.; Herrmann, A.D.; Mendoza-Cozatl, D.G.; O’Neill, M.A.; Dassanayake, M. Cross species multi-omics reveals cell wall sequestration and elevated global transcript abundance as mechanisms of boron tolerance in plants. New Phytol. 2021, 230, 1985–2000. [Google Scholar] [CrossRef] [PubMed]
- Pandey, A.; Khan, M.K.; Hakki, E.E.; Gezgin, S.; Hamurcu, M. Combined boron toxicity and salinity stress—An insight into its interaction in plants. Plants 2019, 8, 364. [Google Scholar] [CrossRef]
- Turhan, A. Interactive effects of boron stress and mycorrhizal (AMF) treatments on tomato growth, yield, leaf chlorophyll and boron accumulation, and fruit characteristics. Arch. Agron. Soil Sci. 2021, 67, 1974–1985. [Google Scholar] [CrossRef]
- Ozturk, M.; Sakcali, S.; Gucel, S.; Tombuloglu, H. Boron and Plants. In Plant Adaptation and Phytoremediation; Ashraf, M., Ozturk, M., Ahmad, M.S.A., Eds.; Springer: Dordrecht, The Netherlands, 2010; pp. 275–311. [Google Scholar]
- Yermiyahu, U.; Ben-Gal, A.; Keren, R.; Reid, R.J. Combined effect of salinity and excess boron on plant growth and yield. Plant Soil 2008, 304, 73–87. [Google Scholar] [CrossRef]
- Abdar, N.; Zarei, M.; Ronaghi, A.M. Mitigation effects of Rhizophagus intraradices and Micrococcus yunnanensis on boron toxicity in maize (Zea may L.) plant. J. Plant Nutr. 2023, 46, 3312–3324. [Google Scholar] [CrossRef]
- Coninx, L.; Martinova, V.; Rineau, F. Mycorrhiza-assisted phytoremediation. Adv. Bot. Res. 2017, 83, 127–188. [Google Scholar]
- Khan, A.; Zhao, X.Q.; Javed, M.T.; Khan, K.S.; Bano, A.; Shen, R.F.; Masood, S. Bacillus pumilus enhances tolerance in rice (Oryza sativa L.) to combined stresses of NaCl and high B due to limited uptake of Na+. Environ. Exp. Bot. 2016, 124, 120–129. [Google Scholar] [CrossRef]
- Sonmez, O.; Aydemir, S.; Kaya, C. Mitigation effects of mycorrhiza on boron toxicity in wheat (Triticum durum) plants. N. Z. J. Crop Hortic. Sci. 2009, 37, 99–104. [Google Scholar] [CrossRef]
- Sarafi, E.; Siomos, A.; Tsouvaltzis, P.; Therios, I.; Chatzissavvidis, C. Boron toxicity effects on the concentration of pigments, carbohydrates and nutrient elements in six nongrafted pepper cultivars (Capsicum annuum L.). Indian J. Plant Physiol. 2018, 23, 474–485. [Google Scholar] [CrossRef]
- Simón-Grao, S.; Nieves, M.; Martínez-Nicolás, J.J.; Alfosea-Simón, M.; Cámara-Zapata, J.M.; Fernández-Zapata, J.C.; García-Sánchez, F. Arbuscular mycorrhizal symbiosis improves tolerance of Carrizo citrange to excess boron supply by reducing leaf B concentration and toxicity in the leaves and roots. Ecotoxicol. Environ. Saf. 2019, 173, 322–330. [Google Scholar] [CrossRef]
- Naz, T.; Alihtar, J.; Iqbal, M.M.; Anwar-ul-Haq, M.; Murtaza, G.; Niazi, N.K.; Dell, B. Assessment of gas exchange attributes, chlorophyll contents, ionic composition and antioxidant enzymes of bread wheat genotypes in boron toxic, saline and boron toxic-saline soils. Int. J. Agric. Biol. 2019, 21, 1271–1281. [Google Scholar]
- Lindsay, W.L.; Norvell, W.A. Development of a DTPA soil test for zinc, iron, manganese, and copper. Soil Sci. Soc. Am. J. 1978, 42, 421–428. [Google Scholar] [CrossRef]
- Berger, K.C.; Truog, E. Boron determination in soils and plants. Ind. Eng. Chem. Anal. Ed. 1939, 11, 540–545. [Google Scholar] [CrossRef]
- John, M.K.; Chuah, H.H.; Neufeld, J.H. Application of improved azomethine-H method to the determination of boron in soils and plants. Analy. Lett. 1975, 8, 559–568. [Google Scholar] [CrossRef]
- Thomas, G.W. Soil pH and soil acidity. In Methods of Soil Analysis: Part 3 Chemical Methods; Wiley: Hoboken, NJ, USA, 1996; Volume 5, pp. 475–490. [Google Scholar]
- Rhoades, J.; Sparks, D.; Page, A.; Helmke, P.; Loeppert, R.; Soltanpour, P.; Summer, M. Salinity: Electrical Methods of Soil Analysis Part 3-Chemical Methods Conductivity and Total Dissolved Solids; Wiley Online Library: Hoboken, NJ, USA, 1996; pp. 417–435. [Google Scholar]
- Nelson, D.W.; Sommers, L.E. Total carbon, organic carbon, and organic matter. In Methods of Soil Analysis: Part 2 Chemical and Microbiological Properties; Wiley Online Library: Hoboken, NJ, USA, 1996; Volume 5, pp. 961–1010. [Google Scholar]
- Bremner, J.M. Nitrogen-total. In Methods of Soil Analysis: Part 3 Chemical Methods; Wiley Online Library: Hoboken, NJ, USA, 1996; Volume 5, pp. 1085–1121. [Google Scholar]
- Olsen, S.R.; Sommers, L.E.; Phosphorus, P. Methods of Soil Analysis, Part 2; Page, A.L., Ed.; Soil Science Society of America: Madison, WI, USA, 1982; pp. 403–424. [Google Scholar]
- Knudsen, D.; Peterson, G.A.; Pratt, P.F. Lithium, sodium, and potassium. In Methods of Soil Analysis: Part 2 Chemical and Microbiological Properties; Wiley Online Library: Hoboken, NJ, USA, 1983; Volume 9, pp. 225–246. [Google Scholar]
- Bouyoucos, G.J. A recalibration of the hydrometer method for making mechanical analysis of soils. Agro. J. 1962, 43, 434–438. [Google Scholar] [CrossRef]
- Zarei, M.; König, S.; Hempel, S.; Nekouei, M.K.; Savaghebi, G.; Buscot, F. Community structure of arbuscular mycorrhizal fungi associated to Veronica rechingeri at the Anguran zinc and lead mining region. Environ. Pollut. 2008, 156, 1277–1283. [Google Scholar] [CrossRef] [PubMed]
- Kormanik, P.P.; McGraw, A.C. Qualification of Vesicular Arbuscular Mycorrhizae in Plant Root. In Method and Principles of Mycorrhizal Research; Scheneck, N.C., Ed.; American Phytopathological Society: St. Paul, MN, USA, 1982; pp. 37–45. [Google Scholar]
- Bañuelos, G.S.; LeDuc, D.; Johnson, J. Evaluating the tolerance of young hybrid poplar trees to recycled waters high in salinity and boron. Int. J. Phytoremediat. 2010, 12, 419–439. [Google Scholar] [CrossRef] [PubMed]
- Lutts, S.; Kinet, J.M.; Bouharmont, J. Effects of salt stress on growth, mineral nutrition and proline accumulation in relation to osmotic adjustment in rice (Oryza sativa L.) cultivars differing in salinity resistance. Plant Growth Regul. 1996, 19, 207–218. [Google Scholar] [CrossRef]
- Chapman, H.D.; Pratt, P.F. Methods of analysis for soils, plants and waters. Soil Sci. 1962, 93, 68. [Google Scholar] [CrossRef]
- Page, A.L.; Miller, R.H.; Keeny, D.R. Methods of Soil and Plant Analysis; American Society of Agronomy: Madison, WI, USA, 1982. [Google Scholar]
- Shen, Y.; Duan, T. The Interaction between Arbuscular Mycorrhizal Fungi (AMF) and Grass Endophyte (Epichloë) on Host Plants: A Review. J. Fungi 2024, 10, 174. [Google Scholar] [CrossRef]
- Jongen, M.; Albadran, B.; Beyschlag, W.; Unger, S. Can arbuscular mycorrhizal fungi mitigate drought stress in annual pasture legumes? Plant Soil 2022, 472, 295–310. [Google Scholar] [CrossRef]
- Liu, C.; Dai, Z.; Xia, J.; Chang, C.; Sun, H. Combined effect of salt and drought on boron toxicity in Puccinellia tenuiflora. Ecotoxicol. Environ. Saf. 2018, 157, 395–402. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Dai, Z.; Cui, M.; Lu, W.; Sun, H. Arbuscular mycorrhizal fungi alleviate boron toxicity in Puccinellia tenuiflora under the combined stresses of salt and drought. Environ. Pollut. 2018, 240, 557–565. [Google Scholar] [CrossRef] [PubMed]
- Porcel, R.; Aroca, R.; Ruiz-Lozano, J.M. Salinity stress alleviation using arbuscular mycorrhizal fungi. A review. Agron. Sustain. Dev. 2012, 32, 181–200. [Google Scholar] [CrossRef]
- Abdar, N.; Zarei, M.; Shahriari, A.G.; Mirmazloum, I. The effect of arbuscular mycorrhizal inoculation and plant growth-promoting rhizobacteria on maize (Zea mays L.) under boron toxicity stress. Not. Bot. Horti. Agrobot. 2023, 51, 13473. [Google Scholar] [CrossRef]
- Pan, G.; Wei, Y.; Zhao, N.; Gu, M.; He, B.; Wang, X. Effects of Claroideoglomus etunicatum Fungi Inoculation on Arsenic Uptake by Maize and Pteris vittata L. Toxics 2022, 10, 574. [Google Scholar] [CrossRef] [PubMed]
- Alharby, H.F.; Nahar, K.; Al-Zahrani, H.S.; Hakeem, K.R.; Hasanuzzaman, M. Enhancing salt tolerance in soybean by exogenous boron: Intrinsic study of the ascorbate-glutathione and glyoxalase pathways. Plants 2021, 10, 2085. [Google Scholar] [CrossRef]
- Sharma, M.P.; Buyer, J.S. Comparison of biochemical and microscopic methods for quantification of arbuscular mycorrhizal fungi in soil and roots. Appl. Soil Ecol. 2015, 95, 86–89. [Google Scholar] [CrossRef]
- Lata, C.; Kumar, A.; Sharma, S.K.; Setter, T.L.; Singh, M.; Prasad, K.R.; Yaduvanshi, N.P. Combined effects of excess boron and salinity in wheat varieties differing in tolerance to these stresses. J. Soil Salin. Water Qual. 2015, 7, 107–114. [Google Scholar]
- Saxena, B.; Shukla, K.; Giri, B. Arbuscular Mycorrhizal Fungi and Tolerance of Salt Stress in Plants. In Arbuscular Mycorrhizas and Stress Tolerance of Plants; Springer: Berlin/Heidelberg, Germany, 2017; pp. 67–97. [Google Scholar]
- Borde, M.; Dudhane, M.; Kulkarni, M. Role of Arbuscular Mycorrhizal Fungi (AMF) in Salinity Tolerance and Growth Response in Plants under Salt Stress Conditions. In Mycorrhiza-Eco-Physiology, Secondary Metabolites, Nanomaterials; Springer: Berlin/Heidelberg, Germany, 2017; pp. 71–86. [Google Scholar]





| Soil Parameters | Measurements |
|---|---|
| pH in (H2O) | 7.68 |
| EC (dS m−1) | 0.6 |
| Organic matter (%) | 1.2 |
| Total N (%) | 0.05 |
| Available K (mg kg−1) | 293 |
| Available B (mg kg−1) | 0.25 |
| Available Fe (mg kg−1) | 2.1 |
| Available Mn (mg kg−1) | 4.7 |
| Available Zn (mg kg−1) | 0.6 |
| Available Cu (mg kg−1) | 0.5 |
| Clay (%) | 21.8 |
| Treatment | Shoot Fe Uptake (mg pot−1) | Shoot Zn Uptake (mg pot−1) | Shoot Cu Uptake (mg pot−1) | Shoot Mn Uptake (mg pot−1) | ||
|---|---|---|---|---|---|---|
| EC | B | Microbial | ||||
| 0 | 0 | No inoculation | 0.739 ± 0.07 ab | 0.35 ± 0.01 bc | 0.0809 ± 0.003 b | 0.984 ± 0.11 bc |
| 0 | 0 | Fungus | 0.513 ± 0.07 ed | 0.234 ± 0.03 de | 0.0242 ± 0.004 e | 0.802 ± 0.27 cd |
| EC | B | Microbial | ||||
| 8 | 0 | No inoculation | 0.56 ± 0.07 cd | 0.405 ± 0.01 ab | 0.495 ± 0.005 d | 1.026 ± 0.04 b |
| 8 | 0 | Fungus | 0.642 ± 0.08 bcd | 0.454 ± 0.07 a | 0.0671 ± 0.005 c | 1.293 ± 0.003 a |
| EC | B | Microbial | ||||
| 0 | 30 | No inoculation | 0.597 ± 0.03 cd | 0.3 ± 0.05 cd | 0.0588 ± 0.01 cd | 0.673 ± 0.05 d |
| 0 | 30 | Fungus | 0.426 ± 0.09 e | 0.241 ± 0.03 de | 0.0355 ± 0.01 e | 0.389 ± 0.06 e |
| EC | B | Microbial | ||||
| 8 | 30 | No inoculation | 0.678 ± 0.08 bc | 0.337 ± 0.02 bc | 0.115 ± 0.01 a | 0.829 ± 0.07 bcd |
| 8 | 30 | Fungus | 0.838 ± 0.01 a | 0.202 ± 0.05 e | 0.0315 ± 0.01 e | 0.727 ± 0.02 d |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zarei, M.; Abdar, N.; Shahriari, A.G.; Mirmazloum, I.; Geösel, A. Effects of Claroideoglomus etunicatum Fungus on the Growth Parameters of Maize (Zea mays L.) Plants under Boron Toxicity and Salt Stress. Agronomy 2024, 14, 1013. https://doi.org/10.3390/agronomy14051013
Zarei M, Abdar N, Shahriari AG, Mirmazloum I, Geösel A. Effects of Claroideoglomus etunicatum Fungus on the Growth Parameters of Maize (Zea mays L.) Plants under Boron Toxicity and Salt Stress. Agronomy. 2024; 14(5):1013. https://doi.org/10.3390/agronomy14051013
Chicago/Turabian StyleZarei, Mehdi, Narges Abdar, Amir Ghaffar Shahriari, Iman Mirmazloum, and András Geösel. 2024. "Effects of Claroideoglomus etunicatum Fungus on the Growth Parameters of Maize (Zea mays L.) Plants under Boron Toxicity and Salt Stress" Agronomy 14, no. 5: 1013. https://doi.org/10.3390/agronomy14051013
APA StyleZarei, M., Abdar, N., Shahriari, A. G., Mirmazloum, I., & Geösel, A. (2024). Effects of Claroideoglomus etunicatum Fungus on the Growth Parameters of Maize (Zea mays L.) Plants under Boron Toxicity and Salt Stress. Agronomy, 14(5), 1013. https://doi.org/10.3390/agronomy14051013








