Human Adipose Mesenchymal Stromal/Stem Cells Improve Fat Transplantation Performance
Abstract
:1. Introduction
2. Materials and Methods
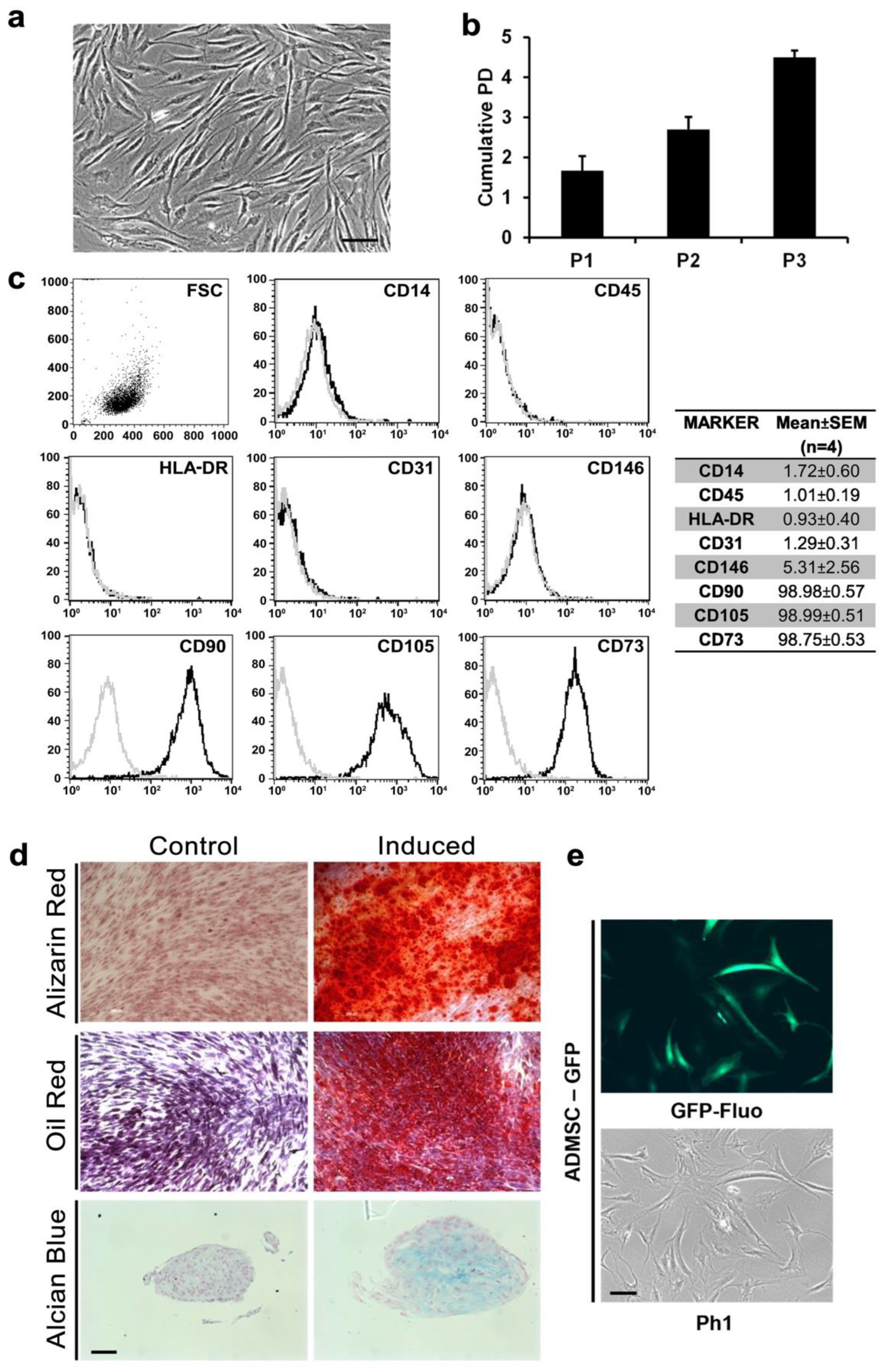
2.1. Isolation and Expansion of Human AD-MSCs
2.2. Fluorescence-Activated Cell Sorting Analyses (FACS)
2.3. Differentiation Assays
2.4. Prostaglandin E2 (PGE2) Evaluation in AD-MSCs
2.5. Retroviral Transduction of Human AD-MSCs to Express GFP
2.6. Microbiology Test
2.7. Human AD-MSC-Assisted Autologous Fat Transplantation in a Xenogeneic Model
2.8. Hematology and Biochemistry
2.9. Tissue Handling and Histology
2.10. Statistical Analysis
3. Results
3.1. AD-MSCs Could Be Isolated and Genetically Modified from Small Amounts of Adipose Tissue
3.2. Xenotransplantation Was Well Tolerated by NOD/SCID Mice
3.3. AD-MSC-GFP Differentiated into Adipocytes within 60 Days after Transplantation
3.4. Proangiogenic and Pro-Inflammatory Characterization of AFT-AD-MSC-GFP
3.5. Wild-Type AD-MSCs Preserved Their Proangiogenic and Anti-Inflammatory Properties
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Krastev, T.K.; Schop, S.J.; Hommes, J.; Piatkowski, A.; van der Hulst, R.R.W.J. Autologous fat transfer to treat fibrosis and scar-related conditions: A systematic review and meta-analysis. J. Plast. Reconstr. Aesthetic Surg. 2020, 73, 2033–2048. [Google Scholar] [CrossRef] [PubMed]
- Raj, S.; Abu-Ghname, A.; Davis, M.J.; Izaddoost, S.A.; Winocour, S.J. Safety and Regulation of Fat Grafting. In Seminars in Plastic Surgery; Thieme Medical Publishers: Leipzig, Germany, 2020; Volume 34, pp. 59–64. [Google Scholar]
- Bauer-Kreisel, P.; Goepferich, A.; Blunk, T. Cell-Delivery Therapeutics for Adipose Tissue Regeneration. Adv. Drug Deliv. Rev. 2010, 62, 798–813. [Google Scholar] [CrossRef]
- Kim, H.Y.; Jung, B.K.; Lew, D.H.; Lee, D.W. Autologous Fat Graft in the Reconstructed Breast: Fat Absorption Rate and Safety based on Sonographic Identification. Arch. Plast. Surg. 2014, 41, 740–747. [Google Scholar] [CrossRef]
- Smith, P.; Adams, W.P.; Lipschitz, A.H.; Chau, B.; Sorokin, E.; Rohrich, R.J.; Brown, S.A. Autologous Human Fat Grafting: Effect of Harvesting and Preparation Techniques on Adipocyte Graft Survival. Plast. Reconstr. Surg. 2006, 117, 1836–1844. [Google Scholar] [CrossRef]
- Zuk, P.A.; Zhu, M.I.; Mizuno, H.; Huang, J.; Futrell, J.W.; Katz, A.J.; Benhaim, P.; Lorenz, H.P.; Hedrick, M.H. Multilineage Cells from Human Adipose Tissue: Implications for Cell-Based Therapies. Tissue Eng. 2001, 7, 211–228. [Google Scholar] [CrossRef]
- Kesten, S.; Fraser, J.K. Autologous Adipose Derived Regenerative Cells: A Platform for Therapeutic Applications. Surg. Technol. Int. 2016, 29, 38–44. [Google Scholar]
- Yoshimura, K.; Aoi, N.; Suga, H.; Inoue, K.; Eto, H.; Sato, K.; Kurita, M.; Harii, K.; Hirohi, T. Ectopic Fibrogenesis Induced by Transplantation of Adipose-Derived Progenitor Cell Suspension Immediately after Lipoinjection. Transplantation 2008, 85, 1868–1869. [Google Scholar] [CrossRef]
- Matsumoto, D.; Sato, K.; Gonda, K.; Takaki, Y.; Shigeura, T.; Sato, T.; Aiba-Kojima, E.; Iizuka, F.; Inoue, K.; Suga, H.; et al. Cell-Assisted Lipotransfer: Supportive Use of Human Adipose-Derived Cells for Soft Tissue Augmentation with Lipoinjection. Tissue Eng. 2006, 12, 3375–3382. [Google Scholar] [CrossRef]
- Toyserkani, N.M.; Quaade, M.L.; Sørensen, J.A. Cell-Assisted Lipotransfer: A Systematic Review of Its Efficacy. Aesthetic Plast. Surg. 2016, 40, 309–318. [Google Scholar] [CrossRef]
- Prantl, L.; Brix, E.; Kempa, S.; Felthaus, O.; Eigenberger, A.; Brébant, V.; Anker, A.; Strauss, C. Facial Rejuvenation with Concentrated Lipograft—A 12 Month Follow-Up Study. Cells 2021, 10, 594. [Google Scholar] [CrossRef] [PubMed]
- Piccinno, M.S.; Veronesi, E.; Loschi, P.; Pignatti, M.; Murgia, A.; Grisendi, G.; Castelli, I.; Bernabei, D.; Candini, O.; Conte, P.; et al. Adipose stromal/stem cells assist fat transplantation reducing necrosis and increasing graft performance. Apoptosis 2013, 18, 1274–1289. [Google Scholar] [CrossRef] [Green Version]
- Coleman, S.R. Structural Fat Grafting: More Than a Permanent Filler. Plast. Reconstr. Surg. 2006, 118, 108S–120S. [Google Scholar] [CrossRef]
- Grisendi, G.; Bussolari, R.; Cafarelli, L.; Petak, I.; Rasini, V.; Veronesi, E.; De Santis, G.; Spano, C.; Tagliazzucchi, M.; Barti-Juhasz, H.; et al. Adipose-Derived Mesenchymal Stem Cells as Stable Source of Tumor Necrosis Factor–Related Apoptosis-Inducing Ligand Delivery for Cancer Therapy. Cancer Res. 2010, 70, 3718–3729. [Google Scholar] [CrossRef]
- Grisendi, G.; Annerén, C.; Cafarelli, L.; Sternieri, R.; Veronesi, E.; Cervo, G.L.; Luminari, S.; Maur, M.; Frassoldati, A.; Palazzi, G.; et al. GMP-manufactured density gradient media for optimized mesenchymal stromal/stem cell isolation and expansion. Cytotherapy 2010, 12, 466–477. [Google Scholar] [CrossRef] [PubMed]
- Lin, T.; Pajarinen, J.; Nabeshima, A.; Lu, L.; Nathan, K.; Jämsen, E.; Yao, Z.; Goodman, S.B. Preconditioning of murine mesenchymal stem cells synergistically enhanced immunomodulation and osteogenesis. Stem Cell Res. Ther. 2017, 8, 277. [Google Scholar] [CrossRef]
- Zhu, M.; Zhou, Z.; Chen, Y.; Schreiber, R.; Ransom, J.T.; Fraser, J.K.; Hedrick, M.H.; Pinkernell, K.; Kuo, H.-C. Supplementation of Fat Grafts with Adipose-Derived Regenerative Cells Improves Long-Term Graft Retention. Ann. Plast. Surg. 2010, 64, 222–228. [Google Scholar] [CrossRef] [PubMed]
- Dominici, M.; Rasini, V.; Bussolari, R.; Chen, X.; Hofmann, T.J.; Spano, C.; Bernabei, D.; Veronesi, E.; Bertoni, F.; Paolucci, P.; et al. Restoration and reversible expansion of the osteoblastic hematopoietic stem cell niche after marrow radioablation. Blood 2009, 114, 2333–2343. [Google Scholar] [CrossRef]
- Debels, H.; Galea, L.; Han, X.-L.; Palmer, J.; Van Rooijen, N.; Morrison, W.; Abberton, K. Macrophages Play a Key Role in Angiogenesis and Adipogenesis in a Mouse Tissue Engineering Model. Tissue Eng. Part A 2013, 19, 2615–2625. [Google Scholar] [CrossRef]
- Charles River Research Model, Technical Shit about NOD SCID Mouse Hematology. Available online: https://larc.ucsf.edu/sites/larc.ucsf.edu/files/wysiwyg/RM-catalog-2019-US-full-version.pdf (accessed on 19 June 2022).
- Ansari, A.M.; Ahmed, A.K.; Matsangos, A.E.; Lay, F.; Born, L.J.; Marti, G.; Harmon, J.W.; Sun, Z. Cellular GFP Toxicity and Immunogenicity: Potential Confounders in in Vivo Cell Tracking Experiments. Stem Cell Rev. Rep. 2016, 12, 553–559. [Google Scholar] [CrossRef]
- Moustaki, M.; Papadopoulos, O.; Verikokos, C.; Karypidis, D.; Masud, D.; Kostakis, A.; Papastefanaki, F.; Roubelakis, M.G.; Perrea, D. Application of adipose-derived stromal cells in fat grafting: Basic science and literature review. Exp. Ther. Med. 2017, 14, 2415–2423. [Google Scholar] [CrossRef]
- Fisher, C.; Grahovac, T.L.; Schafer, M.E.; Shippert, R.D.; Marra, K.; Rubin, J.P. Comparison of Harvest and Processing Techniques for Fat Grafting and Adipose Stem Cell Isolation. Plast. Reconstr. Surg. 2013, 132, 351–361. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zielins, E.R.; Brett, E.A.; Longaker, M.T.; Wan, D.C. Autologous Fat Grafting: The Science Behind the Surgery. Aesthetic Surg. J. 2016, 36, 488–496. [Google Scholar] [CrossRef]
- Mantha, S.; Pillai, S.; Khayambashi, P.; Upadhyay, A.; Zhang, Y.; Tao, O.; Pham, H.M.; Tran, S.D. Smart Hydrogels in Tissue Engineering and Regenerative Medicine. Materials 2019, 12, 3323. [Google Scholar] [CrossRef]
- Skelton, D.; Satake, N.; Kohn, D. The enhanced green fluorescent protein (eGFP) is minimally immunogenic in C57BL/6 mice. Gene Ther. 2001, 8, 1813–1814. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Lee, H.J.; Song, Y.S. Tracking Transplanted Stem Cells Using Magnetic Resonance Imaging and the Nanoparticle Labeling Method in Urology. BioMed Res. Int. 2015, 2015, 231805. [Google Scholar] [CrossRef]
- Guo, Y.; Su, L.; Wu, J.; Zhang, N.; Zhang, X.; Zhang, G.; Li, T.; Wang, J.; Liu, C. Assessment of the green florescence protein labeling method for tracking implanted mesenchymal stem cells. Cytotechnology 2012, 64, 391–401. [Google Scholar] [CrossRef] [PubMed]
- Wei, N.; Sun, Z.; Yu, J.; Jia, Y.; Zheng, P.; Tang, H.; Chen, J. Immunological Responses to Transgene-Modified Neural Stem Cells After Transplantation. Front. Immunol. 2021, 12, 697203. [Google Scholar] [CrossRef] [PubMed]
- Stillaert, F.; Findlay, M.; Palmer, J.; Idrizi, R.; Cheang, S.; Messina, A.; Abberton, K.; Morrison, W.; Thompson, E.W. Host Rather than Graft Origin of Matrigel-Induced Adipose Tissue in the Murine Tissue-Engineering Chamber. Tissue Eng. 2007, 13, 2291–2300. [Google Scholar] [CrossRef]
- Bowles, A.C.; Wise, R.M.; Gerstein, B.Y.; Thomas, R.C.; Ogelman, R.; Febbo, I.; Bunnell, B.A. Immunomodulatory Effects of Adipose Stromal Vascular Fraction Cells Promote Alternative Activation Macrophages to Repair Tissue Damage. Stem Cells 2017, 35, 2198–2207. [Google Scholar] [CrossRef]
- Lu, X.; Han, J.; Xu, X.-P.; Xu, J.; Liu, L.; Huang, Y.; Yang, Y.; Qiu, H. PGE2 Promotes the Migration of Mesenchymal Stem Cells through the Activation of FAK and ERK1/2 Pathway. Stem Cells Int. 2017, 2017, 8178643. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Daaka, Y. PGE2 promotes angiogenesis through EP4 and PKA Cγ pathway. Blood 2011, 118, 5355–5364. [Google Scholar] [CrossRef] [Green Version]
- Lee, B.-C.; Kim, H.-S.; Shin, T.-H.; Kang, I.; Lee, J.Y.; Kim, J.-J.; Kang, H.K.; Seo, Y.; Lee, S.; Yu, K.-R.; et al. PGE2 maintains self-renewal of human adult stem cells via EP2-mediated autocrine signaling and its production is regulated by cell-to-cell contact. Sci. Rep. 2016, 6, 26298. [Google Scholar] [CrossRef]
- Fraser, J.K.; Wulur, I.; Alfonso, Z.; Hedrick, M.H. Fat Tissue: An Underappreciated Source of Stem Cells for Biotechnology. Trends Biotechnol. 2006, 24, 150–154. [Google Scholar] [CrossRef]
- Dong, Z.; Peng, Z.; Chang, Q.; Zhan, W.; Zeng, Z.; Zhang, S.; Lu, F. The Angiogenic and Adipogenic Modes of Adipose Tissue after Free Fat Grafting. Plast. Reconstr. Surg. 2015, 135, 556e–567e. [Google Scholar] [CrossRef] [PubMed]
- Kølle, S.-F.T.; Fischer-Nielsen, A.; Mathiasen, A.B.; Elberg, J.J.; Oliveri, R.S.; Glovinski, P.V.; Kastrup, J.; Kirchhoff, M.; Rasmussen, B.S.; Talman, M.-L.M.; et al. Enrichment of autologous fat grafts with ex-vivo expanded adipose tissue-derived stem cells for graft survival: A randomised placebo-controlled trial. Lancet 2013, 382, 1113–1120. [Google Scholar] [CrossRef]





Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Piccinno, M.S.; Petrachi, T.; Pignatti, M.; Murgia, A.; Grisendi, G.; Candini, O.; Resca, E.; Bergamini, V.; Ganzerli, F.; Portone, A.; et al. Human Adipose Mesenchymal Stromal/Stem Cells Improve Fat Transplantation Performance. Cells 2022, 11, 2799. https://doi.org/10.3390/cells11182799
Piccinno MS, Petrachi T, Pignatti M, Murgia A, Grisendi G, Candini O, Resca E, Bergamini V, Ganzerli F, Portone A, et al. Human Adipose Mesenchymal Stromal/Stem Cells Improve Fat Transplantation Performance. Cells. 2022; 11(18):2799. https://doi.org/10.3390/cells11182799
Chicago/Turabian StylePiccinno, Maria Serena, Tiziana Petrachi, Marco Pignatti, Alba Murgia, Giulia Grisendi, Olivia Candini, Elisa Resca, Valentina Bergamini, Francesco Ganzerli, Alberto Portone, and et al. 2022. "Human Adipose Mesenchymal Stromal/Stem Cells Improve Fat Transplantation Performance" Cells 11, no. 18: 2799. https://doi.org/10.3390/cells11182799




