ZLN005, a PGC-1α Activator, Protects the Liver against Ischemia–Reperfusion Injury and the Progression of Hepatic Metastases
Abstract
:1. Introduction
2. Materials and Methods
2.1. Animals
2.2. ZLN005 Treatment
2.3. Liver I/R Model
2.4. Metastasis Models
2.5. Liver Damage Assessment in Mouse Liver Samples
2.6. Tumor Burden Analysis
2.7. Immunofluorescence
2.8. Mouse Cytokine/Chemokine 44-Plex Discovery Assay® Array (MD44)
2.9. NET Quantification Using MPO-DNA ELISA
2.10. Primary Hepatocyte Isolation and Treatment
2.11. Flow Cytometry
2.12. Quantitative Real-Time PCR
2.13. mtDNA Count Quantification
2.14. Cytochrome c ELISA
2.15. Statistical Analyses
3. Results
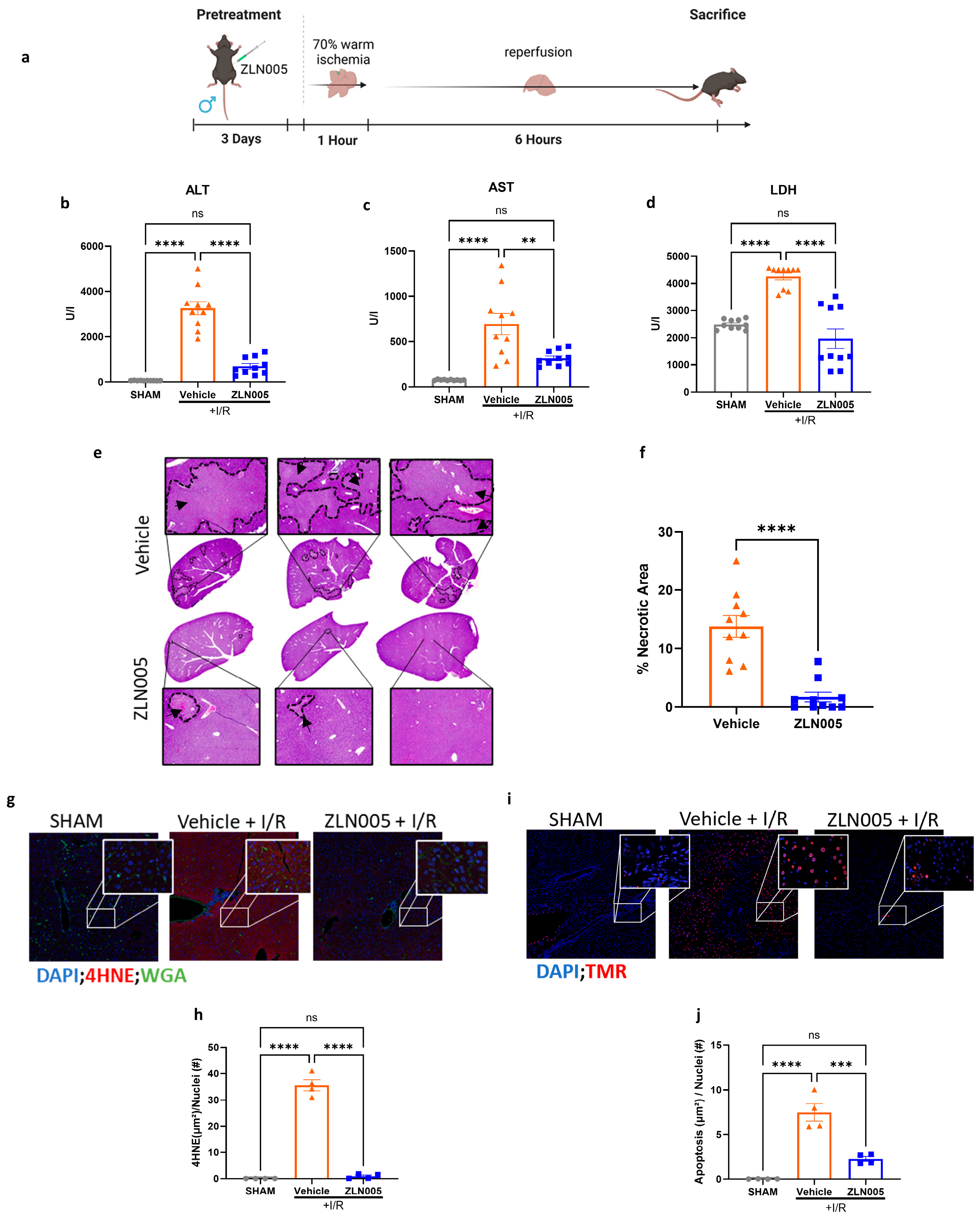
3.1. ZLN005 Pretreatment Decreases Liver Ischemia–Reperfusion Injury
3.2. ZLN005 Decreases Acute Systemic and Intrahepatic Inflammatory Response to Liver I/R
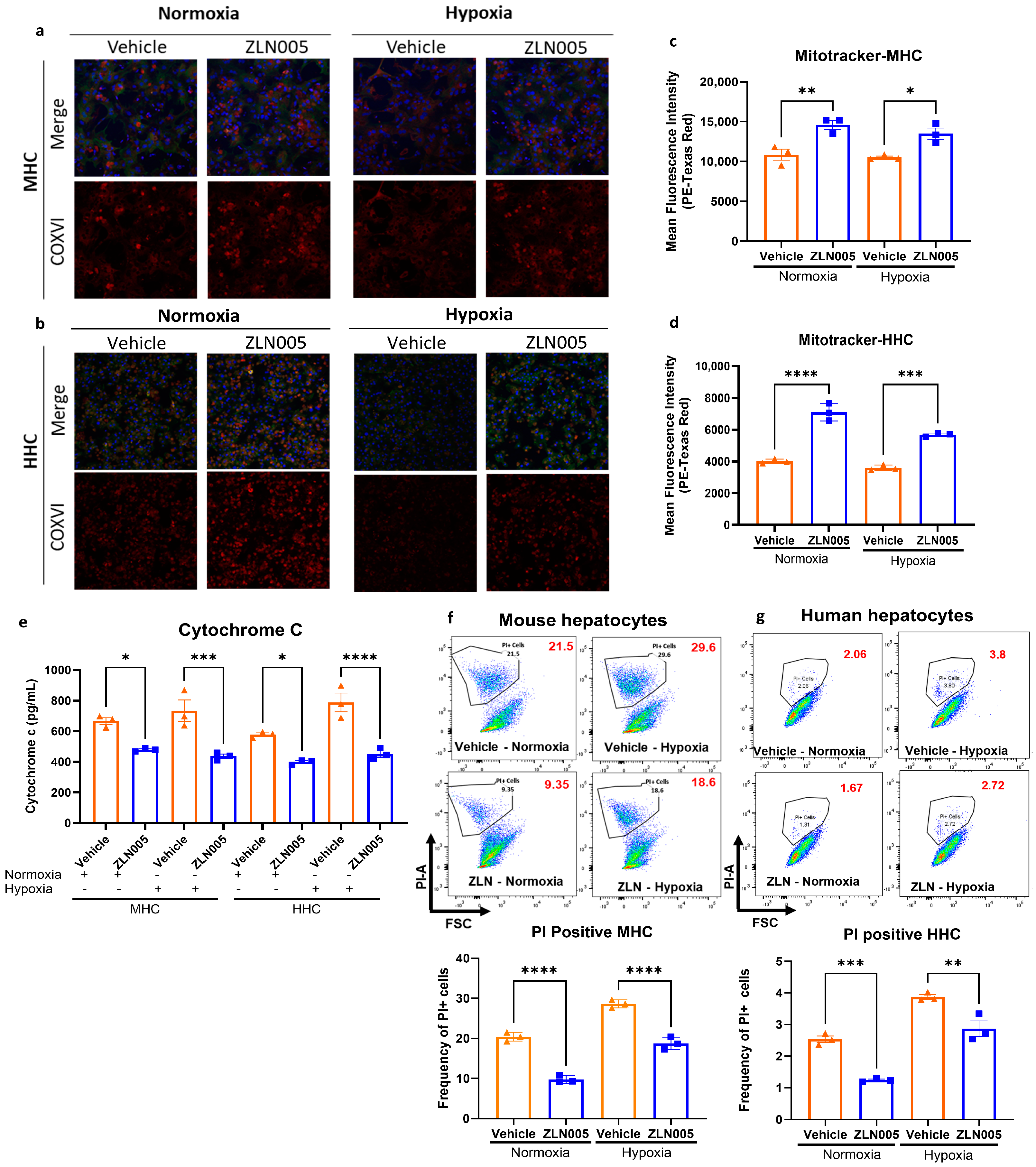
3.3. ZLN005 Pretreatment Upregulates the Hepatic Mitochondrial Biogenesis Pathway
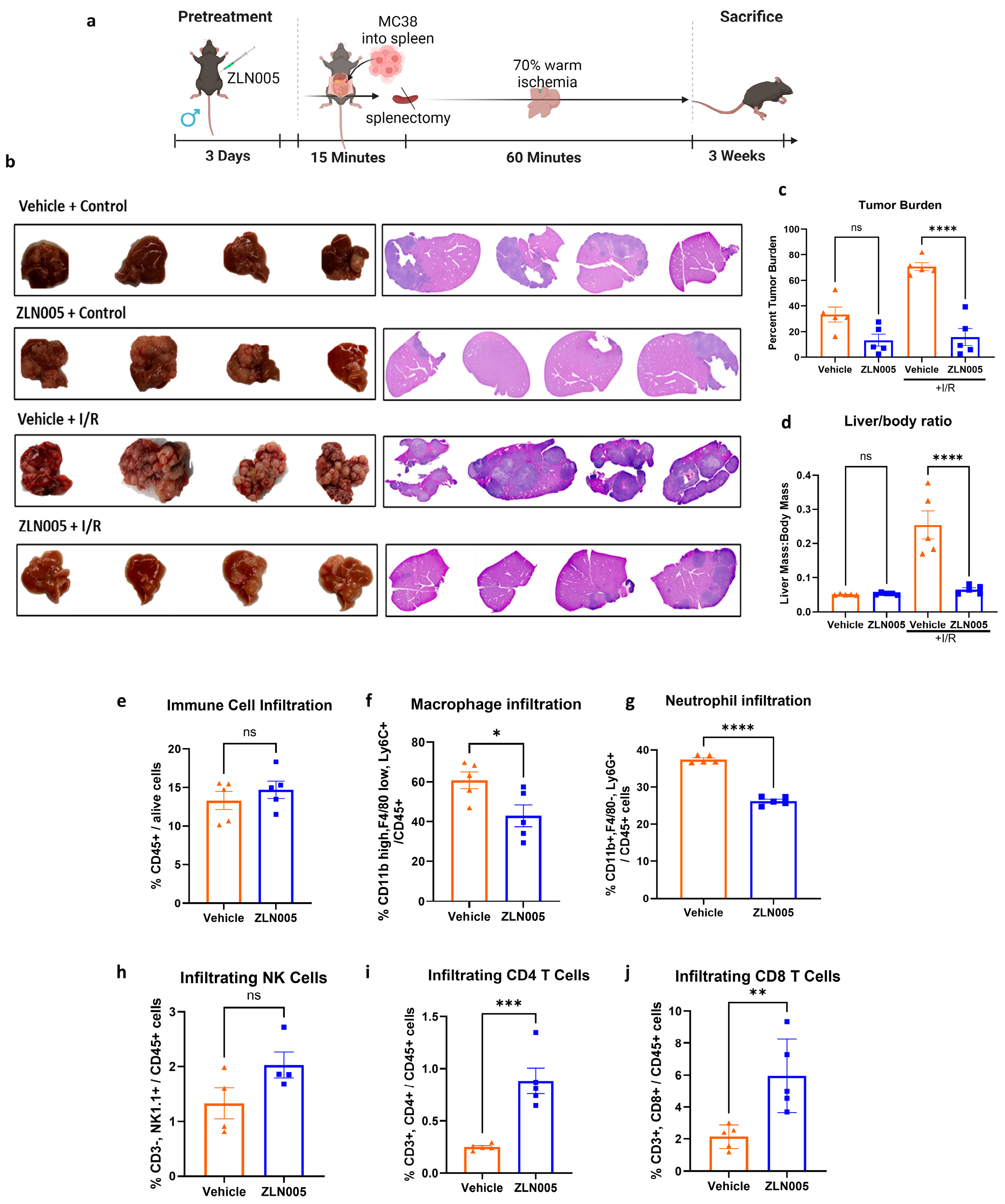
3.4. ZLN005 Protects the Liver against Liver IRI-Induced Tumor Metastasis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Martin, J.; Petrillo, A.; Smyth, E.C.; Shaida, N.; Khwaja, S.; Cheow, H.; Duckworth, A.; Heister, P.; Praseedom, R.; Jah, A.; et al. Colorectal Liver Metastases: Current Management and Future Perspectives. World J. Clin. Oncol. 2020, 11, 761–808. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.-C.; Yeh, D.-C.; Ho, W.-M.; Yu, C.-L.; Cheng, S.-B.; Liu, T.-J.; P’eng, F.-K. Occlusion of Hepatic Blood Inflow for Complex Central Liver Resections in Cirrhotic Patients: A Randomized Comparison of Hemihepatic and Total Hepatic Occlusion Techniques. Arch. Surg. 2002, 137, 1369–1376. [Google Scholar] [CrossRef]
- Choi, E.K.; Lim, D.G. Hepatic Ischemia-Reperfusion Injury with Respect to Oxidative Stress and Inflammatory Response: A Narrative Review. J. Yeungnam Med. Sci. 2022, 40, 115–122. [Google Scholar] [CrossRef] [PubMed]
- Creasy, J.M.; Sadot, E.; Koerkamp, B.G.; Chou, J.F.; Gonen, M.; Kemeny, N.E.; Balachandran, V.P.; Kingham, T.P.; DeMatteo, R.P.; Allen, P.J.; et al. Actual 10-Year Survival after Hepatic Resection of Colorectal Liver Metastases: What Factors Preclude Cure? Surgery 2018, 163, 1238–1244. [Google Scholar] [CrossRef] [PubMed]
- Peralta, C.; Jiménez-Castro, M.B.; Gracia-Sancho, J. Hepatic Ischemia and Reperfusion Injury: Effects on the Liver Sinusoidal Milieu. J. Hepatol. 2013, 59, 1094–1106. [Google Scholar] [CrossRef]
- Kaltenmeier, C.; Wang, R.; Popp, B.; Geller, D.; Tohme, S.; Yazdani, H.O. Role of Immuno-Inflammatory Signals in Liver Ischemia-Reperfusion Injury. Cells 2022, 11, 2222. [Google Scholar] [CrossRef]
- Huang, H.; Tohme, S.; Al-Khafaji, A.B.; Tai, S.; Loughran, P.; Chen, L.; Wang, S.; Kim, J.; Billiar, T.; Wang, Y.; et al. DAMPs-Activated Neutrophil Extracellular Trap Exacerbates Sterile Inflammatory Liver Injury. Hepatology 2015, 62, 600–614. [Google Scholar] [CrossRef]
- Kaltenmeier, C.; Yazdani, H.O.; Handu, S.; Popp, B.; Geller, D.; Tohme, S. The Role of Neutrophils as a Driver in Hepatic Ischemia-Reperfusion Injury and Cancer Growth. Front. Immunol. 2022, 13, 887565. [Google Scholar] [CrossRef]
- Tohme, S.; Kameneva, M.V.; Yazdani, H.O.; Sud, V.; Goswami, J.; Loughran, P.; Huang, H.; Simmons, R.L.; Tsung, A. Drag Reducing Polymers Decrease Hepatic Injury and Metastases after Liver Ischemia-Reperfusion. Oncotarget 2017, 8, 59854–59866. [Google Scholar] [CrossRef]
- Yazdani, H.O.; Kaltenmeier, C.; Morder, K.; Moon, J.; Traczek, M.; Loughran, P.; Zamora, R.; Vodovotz, Y.; Li, F.; Wang, J.H.-C.; et al. Exercise Training Decreases Hepatic Injury via Changes in Immune Response to Liver Ischemia/Reperfusion in Mice. Hepatology 2021, 73, 2494. [Google Scholar] [CrossRef]
- van der Windt, D.J.; Sud, V.; Zhang, H.; Tsung, A.; Huang, H. The Effects of Physical Exercise on Fatty Liver Disease. Gene Expr. 2018, 18, 89–101. [Google Scholar] [CrossRef] [PubMed]
- Sheinboim, D.; Parikh, S.; Manich, P.; Markus, I.; Dahan, S.; Parikh, R.; Stubbs, E.; Cohen, G.; Zemser-Werner, V.; Bell, R.E.; et al. An Exercise-Induced Metabolic Shield in Distant Organs Blocks Cancer Progression and Metastatic Dissemination. Cancer Res. 2022, 82, 4164–4178. [Google Scholar] [CrossRef]
- Warren, J.L.; Hunter, G.R.; Gower, B.A.; Bamman, M.M.; Windham, S.T.; Moellering, D.R.; Fisher, G. Exercise Effects on Mitochondrial Function and Lipid Metabolism during Energy Balance. Med. Sci. Sports Exerc. 2020, 52, 827–834. [Google Scholar] [CrossRef]
- Lima, F.D.; Stamm, D.N.; Della-Pace, I.D.; Dobrachinski, F.; de Carvalho, N.R.; Royes, L.F.F.; Soares, F.A.; Rocha, J.B.; González-Gallego, J.; Bresciani, G. Swimming Training Induces Liver Mitochondrial Adaptations to Oxidative Stress in Rats Submitted to Repeated Exhaustive Swimming Bouts. PLoS ONE 2013, 8, e55668. [Google Scholar] [CrossRef] [PubMed]
- Stevanović, J.; Beleza, J.; Coxito, P.; Ascensão, A.; Magalhães, J. Physical Exercise and Liver “Fitness”: Role of Mitochondrial Function and Epigenetics-Related Mechanisms in Non-Alcoholic Fatty Liver Disease. Mol. Metab. 2019, 32, 1–14. [Google Scholar] [CrossRef]
- Zhang, L.-N.; Zhou, H.-Y.; Fu, Y.-Y.; Li, Y.-Y.; Wu, F.; Gu, M.; Wu, L.-Y.; Xia, C.-M.; Dong, T.-C.; Li, J.-Y.; et al. Novel Small-Molecule PGC-1α Transcriptional Regulator With Beneficial Effects on Diabetic Db/Db Mice. Diabetes 2013, 62, 1297–1307. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Kabba, J.A.; Ruan, W.; Wang, Y.; Zhao, S.; Song, X.; Zhang, L.; Li, J.; Pang, T. The PGC-1α Activator ZLN005 Ameliorates Ischemia-Induced Neuronal Injury In Vitro and In Vivo. Cell. Mol. Neurobiol. 2018, 38, 929–939. [Google Scholar] [CrossRef]
- Wang, Z.; Fu, Z.; Wang, C.; Xu, J.; Ma, H.; Jiang, M.; Xu, T.; Feng, X.; Zhang, W. ZLN005 Protects against Ischemia-Reperfusion-Induced Kidney Injury by Mitigating Oxidative Stress through the Restoration of Mitochondrial Fatty Acid Oxidation. Am. J. Transl. Res. 2021, 13, 10014–10037. [Google Scholar]
- Yazdani, H.O.; Tohme, S. Murine Model of Metastatic Liver Tumors in the Setting of Ischemia Reperfusion Injury. J. Vis. Exp. JoVE 2019, 150, e59748. [Google Scholar] [CrossRef]
- Tsung, A.; Hoffman, R.A.; Izuishi, K.; Critchlow, N.D.; Nakao, A.; Chan, M.H.; Lotze, M.T.; Geller, D.A.; Billiar, T.R. Hepatic Ischemia/Reperfusion Injury Involves Functional TLR4 Signaling in Nonparenchymal Cells1. J. Immunol. 2005, 175, 7661–7668. [Google Scholar] [CrossRef]
- Tohme, S.; Yazdani, H.O.; Sud, V.; Loughran, P.; Huang, H.; Zamora, R.; Simmons, R.L.; Vodovotz, Y.; Tsung, A. Computational Analysis Supports IL-17A as a Central Driver of Neutrophil Extracellular Trap-Mediated Injury in Liver Ischemia Reperfusion. J. Immunol. 2019, 202, 268–277. [Google Scholar] [CrossRef] [PubMed]
- Charni-Natan, M.; Goldstein, I. Protocol for Primary Mouse Hepatocyte Isolation. STAR Protoc. 2020, 1, 100086. [Google Scholar] [CrossRef] [PubMed]
- Rooney, J.; Ryde, I.; Sanders, L.; Howlett, E.; Colton, M.; Germ, K.; Mayer, G.; Greenamyre, J.; Meyer, J. PCR Based Determination of Mitochondrial DNA Copy Number in Multiple Species. Methods Mol. Biol. 2015, 1241, 23–38. [Google Scholar] [CrossRef]
- Kaltenmeier, C.; Yazdani, H.; Molinari, M.; Simmons, R.; Geller, D.; Tohme, S. The Role of Exercise Training in Preconditioning the Liver against Ischemia/Reperfusion Injury in an Orthotopic Liver Murine Transplant Model. HPB 2021, 23, S452. [Google Scholar] [CrossRef]
- Arhin, N.D.; Shen, C.; Bailey, C.E.; Matsuoka, L.K.; Hawkins, A.T.; Holowatyj, A.N.; Ciombor, K.K.; Hopkins, M.B.; Geiger, T.M.; Kam, A.E.; et al. Surgical Resection and Survival Outcomes in Metastatic Young Adult Colorectal Cancer Patients. Cancer Med. 2021, 10, 4269–4281. [Google Scholar] [CrossRef] [PubMed]
- Machado, I.F.; Palmeira, C.M.; Rolo, A.P. Preservation of Mitochondrial Health in Liver Ischemia/Reperfusion Injury. Biomedicines 2023, 11, 948. [Google Scholar] [CrossRef]
- Rossmann, M.P.; Dubois, S.M.; Agarwal, S.; Zon, L.I. Mitochondrial Function in Development and Disease. Dis. Model. Mech. 2021, 14, dmm048912. [Google Scholar] [CrossRef]
- Adzigbli, L.; Sokolov, E.P.; Wimmers, K.; Sokolova, I.M.; Ponsuksili, S. Effects of Hypoxia and Reoxygenation on Mitochondrial Functions and Transcriptional Profiles of Isolated Brain and Muscle Porcine Cells. Sci. Rep. 2022, 12, 19881. [Google Scholar] [CrossRef]
- Azimifar, S.B.; Nagaraj, N.; Cox, J.; Mann, M. Cell-Type-Resolved Quantitative Proteomics of Murine Liver. Cell Metab. 2014, 20, 1076–1087. [Google Scholar] [CrossRef]
- Martins, R.M.; Teodoro, J.S.; Furtado, E.; Rolo, A.P.; Palmeira, C.M.; Tralhão, J.G. Evaluation of Bioenergetic and Mitochondrial Function in Liver Transplantation. Clin. Mol. Hepatol. 2019, 25, 190–198. [Google Scholar] [CrossRef]
- Kolios, G.; Valatas, V.; Kouroumalis, E. Role of Kupffer Cells in the Pathogenesis of Liver Disease. World J. Gastroenterol. WJG 2006, 12, 7413–7420. [Google Scholar] [CrossRef]
- Deshmane, S.L.; Kremlev, S.; Amini, S.; Sawaya, B.E. Monocyte Chemoattractant Protein-1 (MCP-1): An Overview. J. Interferon Cytokine Res. 2009, 29, 313–326. [Google Scholar] [CrossRef] [PubMed]
- Nairz, M.; Sonnweber, T.; Schroll, A.; Theurl, I.; Weiss, G. The Pleiotropic Effects of Erythropoietin in Infection and Inflammation. Microbes Infect. Inst. Pasteur 2012, 14, 238–246. [Google Scholar] [CrossRef] [PubMed]
- Jiao, Q.; Xiang, L.; Chen, Y. Mitochondrial Transplantation: A Promising Therapy for Mitochondrial Disorders. Int. J. Pharm. 2024, 658, 124194. [Google Scholar] [CrossRef]
- Cruz-Gregorio, A.; Aranda-Rivera, A.K.; Amador-Martinez, I.; Maycotte, P. Mitochondrial Transplantation Strategies in Multifaceted Induction of Cancer Cell Death. Life Sci. 2023, 332, 122098. [Google Scholar] [CrossRef]
- Ulger, O.; Kubat, G.B. Therapeutic Applications of Mitochondrial Transplantation. Biochimie 2022, 195, 1–15. [Google Scholar] [CrossRef]
- Wang, C.; Li, Z.; Zhao, B.; Wu, Y.; Fu, Y.; Kang, K.; Li, Y.; Dong, L.; Li, X.; Zhang, B.; et al. PGC-1α Protects against Hepatic Ischemia Reperfusion Injury by Activating PPARα and PPARγ and Regulating ROS Production. Oxid. Med. Cell. Longev. 2021, 2021, 6677955. [Google Scholar] [CrossRef]
- Yuan, Y.; Tian, Y.; Jiang, H.; Cai, L.; Song, J.; Peng, R.; Zhang, X. Mechanism of PGC-1α-Mediated Mitochondrial Biogenesis in Cerebral Ischemia–Reperfusion Injury. Front. Mol. Neurosci. 2023, 16, 1224964. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Jiao, Y.; Liu, Y.; Fu, J.; Sun, L.; Su, J. PGC-1α Protects from Myocardial Ischaemia-reperfusion Injury by Regulating Mitonuclear Communication. J. Cell. Mol. Med. 2022, 26, 593–600. [Google Scholar] [CrossRef]
- Wang, D.; Cao, L.; Zhou, X.; Wang, G.; Ma, Y.; Hao, X.; Fan, H. Mitigation of Honokiol on Fluoride-Induced Mitochondrial Oxidative Stress, Mitochondrial Dysfunction, and Cognitive Deficits through Activating AMPK/PGC-1α/Sirt3. J. Hazard. Mater. 2022, 437, 129381. [Google Scholar] [CrossRef]
- Abu Shelbayeh, O.; Arroum, T.; Morris, S.; Busch, K.B. PGC-1α Is a Master Regulator of Mitochondrial Lifecycle and ROS Stress Response. Antioxidants 2023, 12, 1075. [Google Scholar] [CrossRef] [PubMed]
- Li, C.X.; Ling, C.C.; Shao, Y.; Xu, A.; Li, X.C.; Ng, K.T.-P.; Liu, X.B.; Ma, Y.Y.; Qi, X.; Liu, H.; et al. CXCL10/CXCR3 Signaling Mobilized-Regulatory T Cells Promote Liver Tumor Recurrence after Transplantation. J. Hepatol. 2016, 65, 944–952. [Google Scholar] [CrossRef]
- Maspero, M.; Yilmaz, S.; Cazzaniga, B.; Raj, R.; Ali, K.; Mazzaferro, V.; Schlegel, A. The Role of Ischaemia-Reperfusion Injury and Liver Regeneration in Hepatic Tumour Recurrence. JHEP Rep. 2023, 5, 100846. [Google Scholar] [CrossRef] [PubMed]
- Fnu, G.; Weber, G.F. Osteopontin Induces Mitochondrial Biogenesis in Deadherent Cancer Cells. Oncotarget 2023, 14, 957–969. [Google Scholar] [CrossRef]
- Bai, R.; Cui, J. Mitochondrial Immune Regulation and Anti-Tumor Immunotherapy Strategies Targeting Mitochondria. Cancer Lett. 2023, 564, 216223. [Google Scholar] [CrossRef]
- Li, W.; Li, X.; Wang, B.; Chen, Y.; Xiao, A.; Zeng, D.; Ou, D.; Yan, S.; Li, W.; Zheng, Q. ZLN005 Protects Cardiomyocytes against High Glucose-Induced Cytotoxicity by Promoting SIRT1 Expression and Autophagy. Exp. Cell Res. 2016, 345, 25–36. [Google Scholar] [CrossRef] [PubMed]
- Zhu, P.; Ma, H.; Cui, S.; Zhou, X.; Xu, W.; Yu, J.; Li, J. ZLN005 Alleviates In Vivo and In Vitro Renal Fibrosis via PGC-1α-Mediated Mitochondrial Homeostasis. Pharmaceuticals 2022, 15, 434. [Google Scholar] [CrossRef]
- Suzuki, Y.; Kami, D.; Taya, T.; Sano, A.; Ogata, T.; Matoba, S.; Gojo, S. ZLN005 Improves the Survival of Polymicrobial Sepsis by Increasing the Bacterial Killing via Inducing Lysosomal Acidification and Biogenesis in Phagocytes. Front. Immunol. 2023, 14, 1089905. [Google Scholar] [CrossRef]
- Maurice, N.M.; Bedi, B.; Yuan, Z.; Lin, K.-C.; Goldberg, J.B.; Hart, C.M.; Bailey, K.L.; Sadikot, R.T. The Effect of PGC-1alpha-SIRT3 Pathway Activation on Pseudomonas Aeruginosa Infection. Pathogens 2022, 11, 116. [Google Scholar] [CrossRef]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tohme, C.; Haykal, T.; Yang, R.; Austin, T.J.; Loughran, P.; Geller, D.A.; Simmons, R.L.; Tohme, S.; Yazdani, H.O. ZLN005, a PGC-1α Activator, Protects the Liver against Ischemia–Reperfusion Injury and the Progression of Hepatic Metastases. Cells 2024, 13, 1448. https://doi.org/10.3390/cells13171448
Tohme C, Haykal T, Yang R, Austin TJ, Loughran P, Geller DA, Simmons RL, Tohme S, Yazdani HO. ZLN005, a PGC-1α Activator, Protects the Liver against Ischemia–Reperfusion Injury and the Progression of Hepatic Metastases. Cells. 2024; 13(17):1448. https://doi.org/10.3390/cells13171448
Chicago/Turabian StyleTohme, Celine, Tony Haykal, Ruiqi Yang, Taylor J. Austin, Patricia Loughran, David A. Geller, Richard L. Simmons, Samer Tohme, and Hamza O. Yazdani. 2024. "ZLN005, a PGC-1α Activator, Protects the Liver against Ischemia–Reperfusion Injury and the Progression of Hepatic Metastases" Cells 13, no. 17: 1448. https://doi.org/10.3390/cells13171448





