A Broad-Spectrum Chemokine Inhibitor Drives M2 Macrophage Polarization Through Modulation of the Myometrial Secretome
Abstract
:1. Introduction
2. Materials and Methods
2.1. Ethics
2.2. Myometrial Cell Isolation and Culture
2.3. Vacuum-Driven Static Stretch of Myocytes
2.4. Monocyte Harvesting and Differentiation
2.5. Flow Cytometry
2.6. Cell Imaging
2.7. Protein Extraction and Immunoblotting
2.8. Collagen Contractility Assay
2.9. Cell Viability Assay
2.10. Phagocytosis Assay
2.11. Mass Spectrometry Sample Preparation
2.12. Mass Spectrometry Analysis
2.13. Mass Spectrometry Data Analysis
2.14. Bioinformatics Analysis
2.15. Statistical Analysis
3. Results
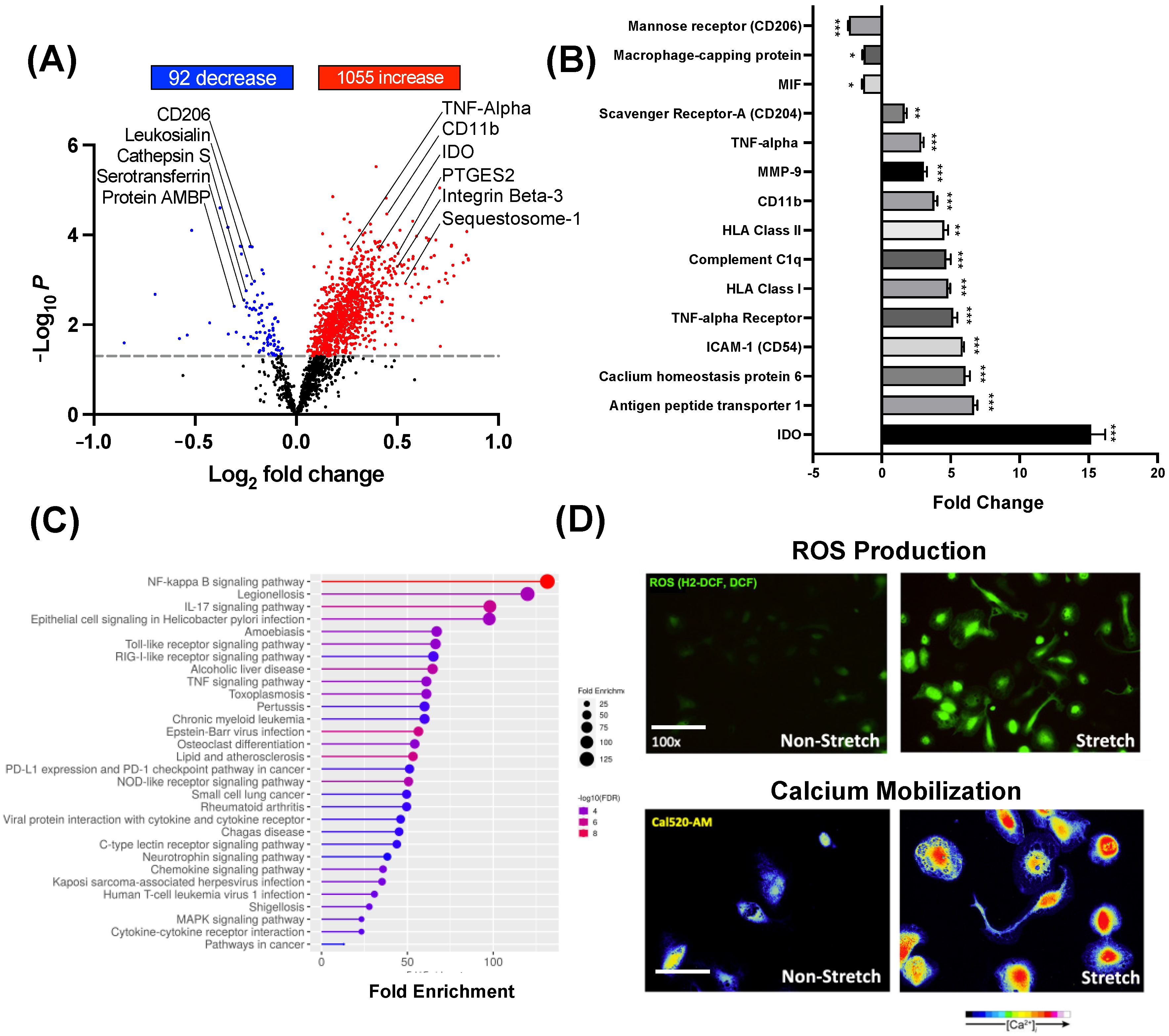
3.1. Mass Spectrometry Analysis of Proteins Secreted by Stretched Myometrial Cells
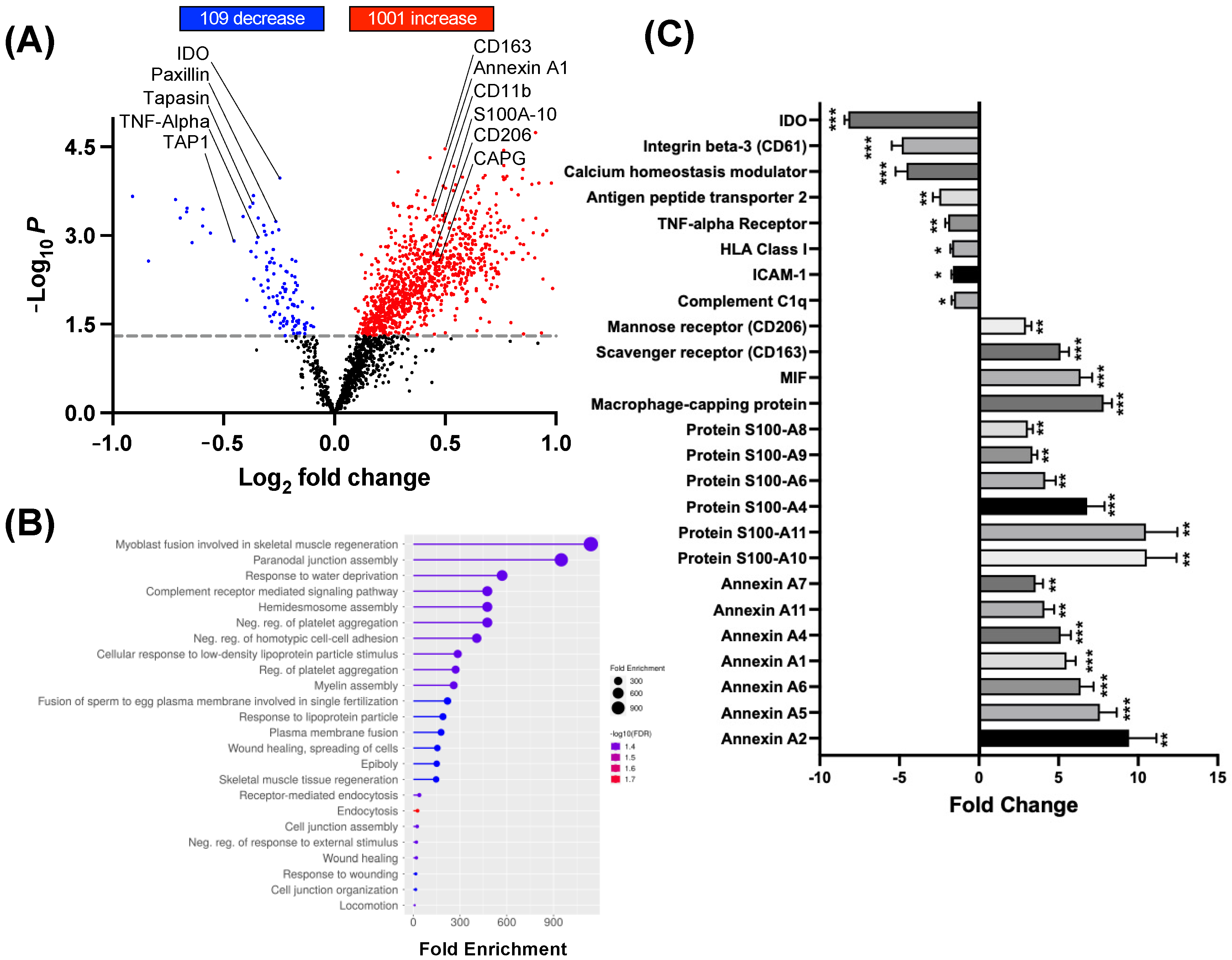
3.2. Monocytes Treated with Myometrial S-CM Are Differentiated and Polarized into M1 Macrophages
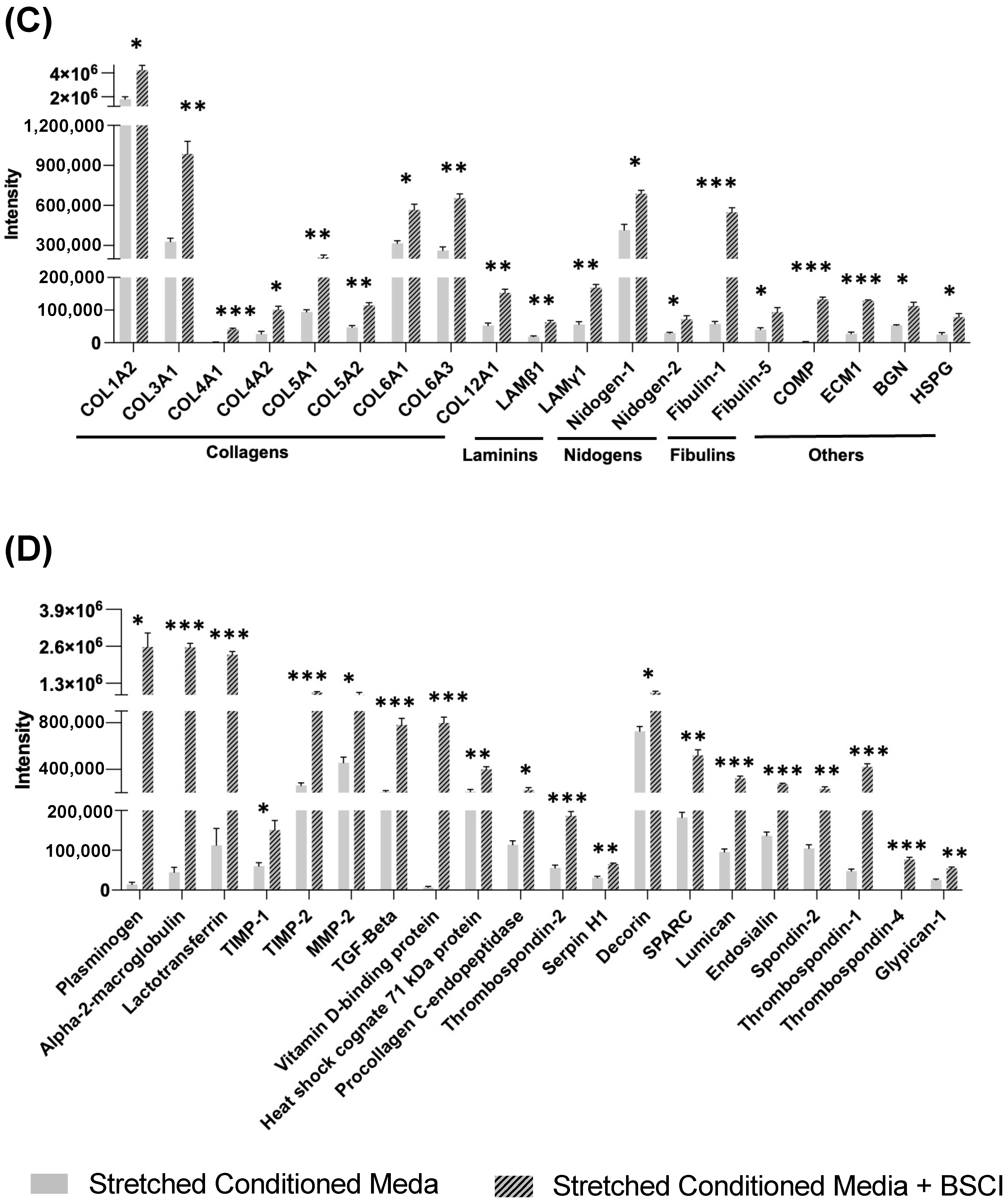
3.3. BSCI-Treated Human Myocytes Secrete Tissue-Remodeling Proteins
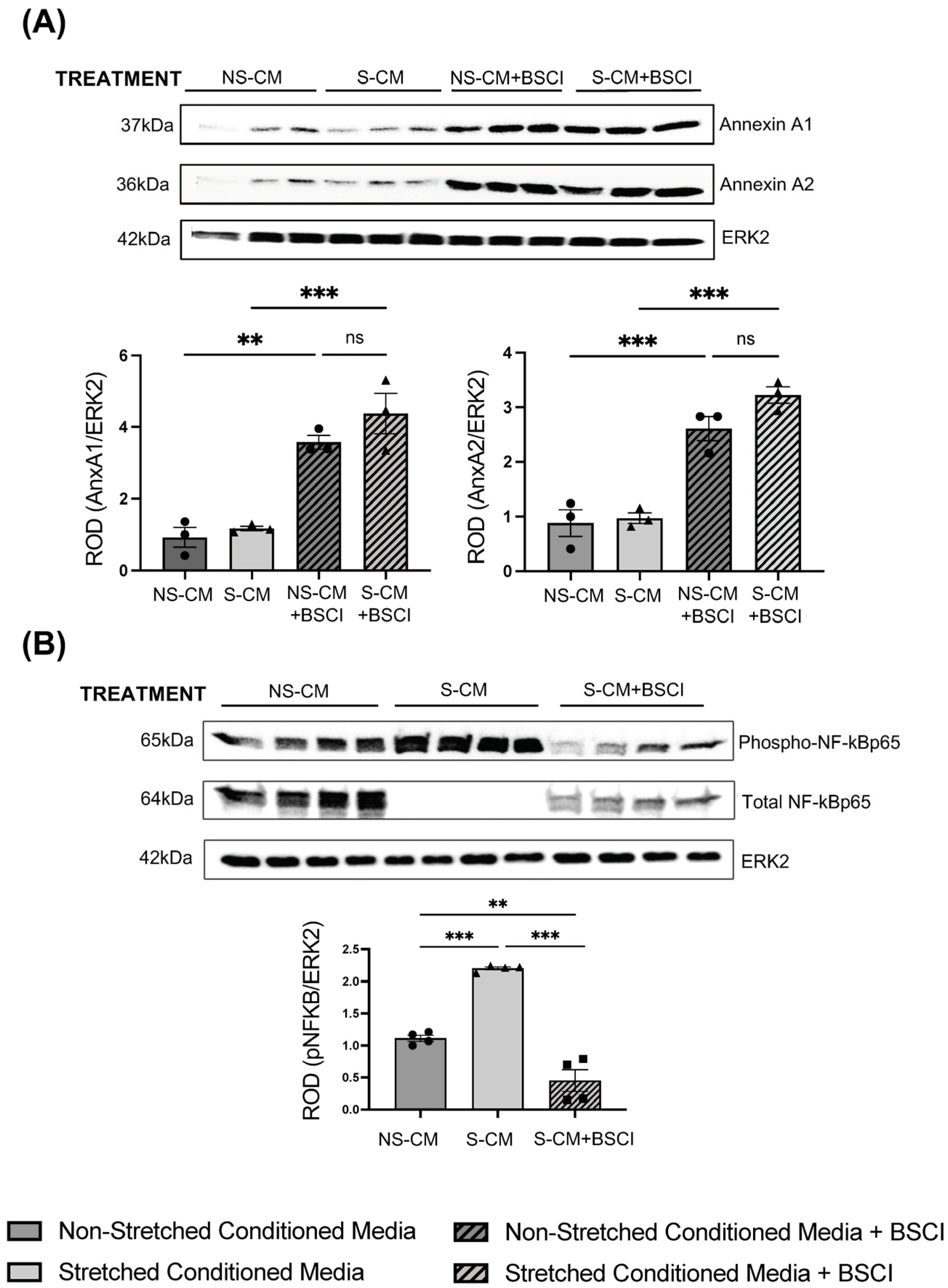
3.4. Myocyte BSCI Treatment Drives Homeostatic M2 Macrophage Polarization
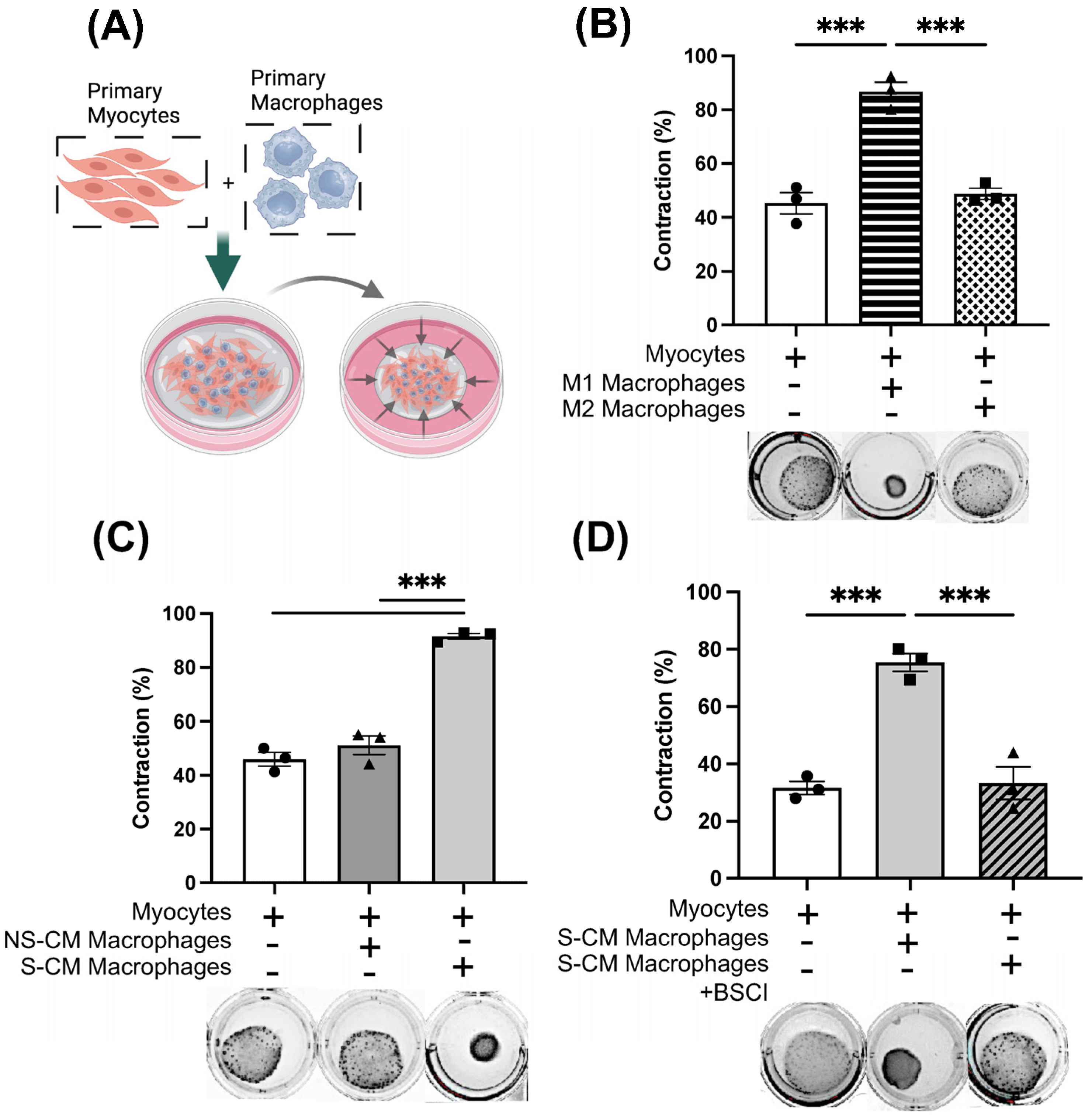
3.5. The BSCI Prevents the Contraction of Collagen Gel Myocyte–Macrophage Co-Cultures
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Quinn, J.-A.; Munoz, F.M.; Gonik, B.; Frau, L.; Cutland, C.; Mallett-Moore, T.; Kissou, A.; Wittke, F.; Das, M.; Nunes, T.; et al. Preterm Birth: Case Definition & Guidelines for Data Collection, Analysis, and Presentation of Immunisation Safety Data. Vaccine 2016, 34, 6047–6056. [Google Scholar] [CrossRef]
- Lakshmanan, A.; Song, A.Y.; Belfort, M.B.; Yieh, L.; Dukhovny, D.; Friedlich, P.S.; Gong, C.L. The Financial Burden Experienced by Families of Preterm Infants after NICU Discharge. J. Perinatol. 2022, 42, 223–230. [Google Scholar] [CrossRef]
- March of Dimes Report Card. Available online: https://www.marchofdimes.org/report-card (accessed on 14 January 2025).
- Moutquin, J.M. Classification and Heterogeneity of Preterm Birth. BJOG Int. J. Obstet. Gynaecol. 2003, 110 (Suppl. 20), 30–33. [Google Scholar] [CrossRef]
- Sheikh, I.A.; Ahmad, E.; Jamal, M.S.; Rehan, M.; Assidi, M.; Tayubi, I.A.; AlBasri, S.F.; Bajouh, O.S.; Turki, R.F.; Abuzenadah, A.M.; et al. Spontaneous Preterm Birth and Single Nucleotide Gene Polymorphisms: A Recent Update. BMC Genom. 2016, 17, 759. [Google Scholar] [CrossRef]
- Osman, I.; Young, A.; Ledingham, M.A.; Thomson, A.J.; Jordan, F.; Greer, I.A.; Norman, J.E. Leukocyte Density and Pro-Inflammatory Cytokine Expression in Human Fetal Membranes, Decidua, Cervix and Myometrium before and during Labour at Term. Mol. Hum. Reprod. 2003, 9, 41–45. [Google Scholar] [CrossRef] [PubMed]
- Shynlova, O.; Nedd-Roderique, T.; Li, Y.; Dorogin, A.; Lye, S.J. Myometrial Immune Cells Contribute to Term Parturition, Preterm Labour and Post-Partum Involution in Mice. J. Cell. Mol. Med. 2013, 17, 90–102. [Google Scholar] [CrossRef] [PubMed]
- Thomson, A.J.; Telfer, J.F.; Young, A.; Campbell, S.; Stewart, C.J.; Cameron, I.T.; Greer, I.A.; Norman, J.E. Leukocytes Infiltrate the Myometrium during Human Parturition: Further Evidence That Labour Is an Inflammatory Process. Hum. Reprod. 1999, 14, 229–236. [Google Scholar] [CrossRef]
- Srikhajon, K.; Shynlova, O.; Preechapornprasert, A.; Chanrachakul, B.; Lye, S. A New Role for Monocytes in Modulating Myometrial Inflammation during Human Labor. Biol. Reprod. 2014, 91, 10. [Google Scholar] [CrossRef]
- Lee, Y.-H.; Shynlova, O.; Lye, S.J. Stretch-Induced Human Myometrial Cytokines Enhance Immune Cell Recruitment via Endothelial Activation. Cell. Mol. Immunol. 2015, 12, 231–242. [Google Scholar] [CrossRef]
- Shynlova, O.; Lee, Y.-H.; Srikhajon, K.; Lye, S.J. Physiologic Uterine Inflammation and Labor Onset: Integration of Endocrine and Mechanical Signals. Reprod. Sci. 2013, 20, 154–167. [Google Scholar] [CrossRef]
- Waldorf, K.M.A.; Singh, N.; Mohan, A.R.; Young, R.C.; Ngo, L.; Das, A.; Tsai, J.; Bansal, A.; Paolella, L.; Herbert, B.R.; et al. Uterine Overdistention Induces Preterm Labor Mediated by Inflammation: Observations in Pregnant Women and Nonhuman Primates. Am. J. Obstet. Gynecol. 2015, 213, 830.e1–830.e19. [Google Scholar] [CrossRef]
- Arrowsmith, S. Multiple Pregnancies, the Myometrium and the Role of Mechanical Factors in the Timing of Labour. Curr. Res. Physiol. 2023, 6, 100105. [Google Scholar] [CrossRef] [PubMed]
- Hunt, J.S. Immunologically Relevant Cells in the Uterus. Biol. Reprod. 1994, 50, 461–466. [Google Scholar] [CrossRef]
- Hunt, J.S.; Manning, L.S.; Mitchell, D.; Selanders, J.R.; Wood, G.W. Localization and Characterization of Macrophages in Murine Uterus. J. Leukoc. Biol. 1985, 38, 255–265. [Google Scholar] [CrossRef]
- Hamilton, S.; Oomomian, Y.; Stephen, G.; Shynlova, O.; Tower, C.L.; Garrod, A.; Lye, S.J.; Jones, R.L. Macrophages Infiltrate the Human and Rat Decidua during Term and Preterm Labor: Evidence That Decidual Inflammation Precedes Labor. Biol. Reprod. 2012, 86, 39. [Google Scholar] [CrossRef]
- Shynlova, O.; Tsui, P.; Dorogin, A.; Lye, S.J. Monocyte Chemoattractant Protein-1 (CCL-2) Integrates Mechanical and Endocrine Signals That Mediate Term and Preterm Labor. J. Immunol. 2008, 181, 1470–1479. [Google Scholar] [CrossRef] [PubMed]
- Shan, Y.; Shen, S.; Long, J.; Tang, Z.; Wu, C.; Ni, X. Term and Preterm Birth Initiation Is Associated with the Macrophages Shifting to M1 Polarization in Gestational Tissues in Mice. Biology 2022, 11, 1759. [Google Scholar] [CrossRef]
- Strizova, Z.; Benesova, I.; Bartolini, R.; Novysedlak, R.; Cecrdlova, E.; Foley, L.K.; Striz, I. M1/M2 Macrophages and Their Overlaps—Myth or Reality? Clin. Sci. 2023, 137, 1067–1093. [Google Scholar] [CrossRef]
- Mizoguchi, M.; Ishida, Y.; Nosaka, M.; Kimura, A.; Kuninaka, Y.; Yahata, T.; Nanjo, S.; Toujima, S.; Minami, S.; Ino, K.; et al. Prevention of Lipopolysaccharide-Induced Preterm Labor by the Lack of CX3CL1-CX3CR1 Interaction in Mice. PLoS ONE 2018, 13, e0207085. [Google Scholar] [CrossRef]
- Gonzalez, J.M.; Franzke, C.-W.; Yang, F.; Romero, R.; Girardi, G. Complement Activation Triggers Metalloproteinases Release Inducing Cervical Remodeling and Preterm Birth in Mice. Am. J. Pathol. 2011, 179, 838–849. [Google Scholar] [CrossRef]
- Yellon, S.M.; Greaves, E.; Heuerman, A.C.; Dobyns, A.E.; Norman, J.E. Effects of Macrophage Depletion on Characteristics of Cervix Remodeling and Pregnancy in CD11b-Dtr Mice. Biol. Reprod. 2019, 100, 1386–1394. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Flores, V.; Liu, Z.; Romero, R.; Pique-Regi, R.; Xu, Y.; Miller, D.; Levenson, D.; Galaz, J.; Winters, A.D.; Farias-Jofre, M.; et al. Homeostatic Macrophages Prevent Preterm Birth and Improve Neonatal Outcomes by Mitigating In Utero Sterile Inflammation in Mice. J. Immunol. 2024, 213, 1620–1634. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Lopez, N.; Garcia-Flores, V.; Chin, P.Y.; Groome, H.M.; Bijland, M.T.; Diener, K.R.; Romero, R.; Robertson, S.A. Macrophages Exert Homeostatic Actions in Pregnancy to Protect against Preterm Birth and Fetal Inflammatory Injury. JCI Insight 2021, 6, e146089. [Google Scholar] [CrossRef]
- Pique-Regi, R.; Romero, R.; Garcia-Flores, V.; Peyvandipour, A.; Tarca, A.L.; Pusod, E.; Galaz, J.; Miller, D.; Bhatti, G.; Para, R.; et al. A Single-Cell Atlas of the Myometrium in Human Parturition. JCI Insight 2022, 7, e153921. [Google Scholar] [CrossRef]
- Xu, Y.; Romero, R.; Miller, D.; Kadam, L.; Mial, T.N.; Plazyo, O.; Garcia-Flores, V.; Hassan, S.S.; Xu, Z.; Tarca, A.L.; et al. An M1-like Macrophage Polarization in Decidual Tissue during Spontaneous Preterm Labor That Is Attenuated by Rosiglitazone Treatment. J. Immunol. 2016, 196, 2476–2491. [Google Scholar] [CrossRef]
- Wendremaire, M.; Hadi, T.; Pezze, M.; Barrichon, M.; Lopez, T.; Neiers, F.; Sagot, P.; Garrido, C.; Lirussi, F. Macrophage-Induced Reactive Oxygen Species Promote Myometrial Contraction and Labor-Associated Mechanisms. Biol. Reprod. 2020, 102, 1326–1339. [Google Scholar] [CrossRef] [PubMed]
- Rajagopal, S.P.; Hutchinson, J.L.; Dorward, D.A.; Rossi, A.G.; Norman, J.E. Crosstalk between Monocytes and Myometrial Smooth Muscle in Culture Generates Synergistic Pro-Inflammatory Cytokine Production and Enhances Myocyte Contraction, with Effects Opposed by Progesterone. Mol. Hum. Reprod. 2015, 21, 672–686. [Google Scholar] [CrossRef]
- Boros-Rausch, A.; Shynlova, O.; Lye, S.J. A Broad-Spectrum Chemokine Inhibitor Blocks Inflammation-Induced Myometrial Myocyte–Macrophage Crosstalk and Myometrial Contraction. Cells 2022, 11, 128. [Google Scholar] [CrossRef]
- Nadeem, L.; Sharma, A.; Pelletier, L.; Shynlova, O.; Lye, S. Macrophage-myocyte crosstalk: Evidence of tunneling nanotube mediated physical connection and cargo transfer. Reprod. Sci. 2023, 30, 47A–48A. [Google Scholar] [CrossRef]
- Olson, D.M.; Ammann, C. Role of the Prostaglandins in Labour and Prostaglandin Receptor Inhibitors in the Prevention of Preterm Labour. Front. Biosci. 2007, 12, 1329–1343. [Google Scholar] [CrossRef]
- Chioss, G.; Costantine, M.M.; Bytautiene, E.; Betancourt, A.; Hankins, G.D.; Saade, G.R.; Longo, M. In Vitro Myometrial Contractility Profiles of Different Pharmacological Agents Used for Induction of Labor. Am. J. Perinatol. 2012, 29, 699–704. [Google Scholar] [CrossRef] [PubMed]
- Shynlova, O.; Dorogin, A.; Li, Y.; Lye, S. Inhibition of Infection-Mediated Preterm Birth by Administration of Broad Spectrum Chemokine Inhibitor in Mice. J. Cell Mol. Med. 2014, 18, 1816–1829. [Google Scholar] [CrossRef] [PubMed]
- Coleman, M.; Orvis, A.; Wu, T.-Y.; Dacanay, M.; Merillat, S.; Ogle, J.; Baldessari, A.; Kretzer, N.M.; Munson, J.; Boros-Rausch, A.J.; et al. A Broad Spectrum Chemokine Inhibitor Prevents Preterm Labor but Not Microbial Invasion of the Amniotic Cavity or Neonatal Morbidity in a Non-Human Primate Model. Front. Immunol. 2020, 11, 770. [Google Scholar] [CrossRef]
- Shynlova, O.; Boros-Rausch, A.; Farine, T.; Waldorf, K.M.A.; Dunk, C.; Lye, S.J. Decidual Inflammation Drives Chemokine-Mediated Immune Infiltration Contributing to Term Labor. J. Immunol. 2021, 207, 2015–2026. [Google Scholar] [PubMed]
- Chimal-Ramírez, G.K.; Espinoza-Sánchez, N.A.; Chávez-Sánchez, L.; Arriaga-Pizano, L.; Fuentes-Pananá, E.M. Monocyte Differentiation towards Protumor Activity Does Not Correlate with M1 or M2 Phenotypes. J. Immunol. Res. 2016, 2016, 6031486. [Google Scholar] [CrossRef]
- Soares-Schanoski, A.; Jurado, T.; Córdoba, R.; Siliceo, M.; Fresno, C.; Gómez-Piña, V.; Toledano, V.; Vallejo-Cremades, M.T.; Alfonso-Iñiguez, S.; Carballo-Palos, A.; et al. Impaired Antigen Presentation and Potent Phagocytic Activity Identifying Tumor-Tolerant Human Monocytes. Biochem. Biophys. Res. Commun. 2012, 423, 331–337. [Google Scholar] [CrossRef]
- Cappelletti, M.; Doll, J.R.; Stankiewicz, T.E.; Lawson, M.J.; Sauer, V.; Wen, B.; Kalinichenko, V.V.; Sun, X.; Tilburgs, T.; Divanovic, S. Maternal Regulation of Inflammatory Cues Is Required for Induction of Preterm Birth. JCI Insight 2020, 5, e138812. [Google Scholar] [CrossRef]
- Garcia-Flores, V.; Romero, R.; Peyvandipour, A.; Galaz, J.; Pusod, E.; Panaitescu, B.; Miller, D.; Xu, Y.; Tao, L.; Liu, Z.; et al. A Single-Cell Atlas of Murine Reproductive Tissues during Preterm Labor. Cell Rep. 2023, 42, 111846. [Google Scholar] [CrossRef]
- Saliba, J.; Coutaud, B.; Solovieva, V.; Lu, F.; Blank, V. Regulation of CXCL1 Chemokine and CSF3 Cytokine Levels in Myometrial Cells by the MAFF Transcription Factor. J. Cell. Mol. Med. 2019, 23, 2517–2525. [Google Scholar] [CrossRef]
- Hertelendy, F.; Romero, R.; Molnár, M.; Todd, H.; Baldassare, J.J. Cytokine-Initiated Signal Transduction in Human Myometrial Cells. Am. J. Reprod. Immunol. 1993, 30, 49–57. [Google Scholar] [CrossRef]
- Korbecki, J.; Barczak, K.; Gutowska, I.; Chlubek, D.; Baranowska-Bosiacka, I. CXCL1: Gene, Promoter, Regulation of Expression, mRNA Stability, Regulation of Activity in the Intercellular Space. Int. J. Mol. Sci. 2022, 23, 792. [Google Scholar] [CrossRef]
- Ciummo, S.L.; D’Antonio, L.; Sorrentino, C.; Fieni, C.; Lanuti, P.; Stassi, G.; Todaro, M.; Di Carlo, E. The C-X-C Motif Chemokine Ligand 1 Sustains Breast Cancer Stem Cell Self-Renewal and Promotes Tumor Progression and Immune Escape Programs. Front. Cell Dev. Biol. 2021, 9, 689286. [Google Scholar] [CrossRef] [PubMed]
- Acharyya, S.; Oskarsson, T.; Vanharanta, S.; Malladi, S.; Kim, J.; Morris, P.G.; Manova-Todorova, K.; Leversha, M.; Hogg, N.; Seshan, V.E.; et al. A CXCL1 Paracrine Network Links Cancer Chemoresistance and Metastasis. Cell 2012, 150, 165–178. [Google Scholar] [CrossRef]
- Boro, M.; Balaji, K.N. CXCL1 and CXCL2 Regulate NLRP3 Inflammasome Activation via G-Protein-Coupled Receptor CXCR2. J. Immunol. 2017, 199, 1660–1671. [Google Scholar] [CrossRef] [PubMed]
- Bacher, M.; Metz, C.N.; Calandra, T.; Mayer, K.; Chesney, J.; Lohoff, M.; Gemsa, D.; Donnelly, T.; Bucala, R. An Essential Regulatory Role for Macrophage Migration Inhibitory Factor in T-Cell Activation. Proc. Natl. Acad. Sci. USA 1996, 93, 7849–7854. [Google Scholar] [CrossRef] [PubMed]
- Calandra, T.; Roger, T. Macrophage Migration Inhibitory Factor: A Regulator of Innate Immunity. Nat. Rev. Immunol. 2003, 3, 791–800. [Google Scholar] [CrossRef]
- Javeed, A.; Zhao, Y.; Zhao, Y. Macrophage-Migration Inhibitory Factor: Role in Inflammatory Diseases and Graft Rejection. Inflamm. Res. 2008, 57, 45–50. [Google Scholar] [CrossRef]
- Hoi, A.Y.; Iskander, M.N.; Morand, E.F. Macrophage Migration Inhibitory Factor: A Therapeutic Target across Inflammatory Diseases. Inflamm. Allergy-Drug Targets-Inflamm. Allergy 2007, 6, 183–190. [Google Scholar] [CrossRef]
- Shynlova, O.; Chow, M.; Lye, S.J. Expression and Organization of Basement Membranes and Focal Adhesion Proteins in Pregnant Myometrium Is Regulated by Uterine Stretch. Reprod. Sci. 2009, 16, 960–969. [Google Scholar] [CrossRef]
- Nguyen, T.T.-T.N.; Shynlova, O.; Lye, S.J. Matrix Metalloproteinase Expression in the Rat Myometrium During Pregnancy, Term Labor, and Postpartum. Biol. Reprod. 2016, 95, 24. [Google Scholar] [CrossRef]
- Shynlova, O.; Tsui, P.; Jaffer, S.; Lye, S.J. Integration of Endocrine and Mechanical Signals in the Regulation of Myometrial Functions during Pregnancy and Labour. Eur. J. Obstet. Gynecol. Reprod. Biol. 2009, 144 (Suppl. 1), S2–S10. [Google Scholar] [CrossRef]
- Shynlova, O.; Tsui, P.; Dorogin, A.; Langille, B.L.; Lye, S.J. Insulin-like Growth Factors and Their Binding Proteins Define Specific Phases of Myometrial Differentiation during Pregnancy in the Rat. Biol. Reprod. 2007, 76, 571–578. [Google Scholar] [CrossRef] [PubMed]
- Shynlova, O.; Mitchell, J.A.; Tsampalieros, A.; Langille, B.L.; Lye, S.J.; Ruddock, N.T.; Wilson, K.J.; Cooney, M.A.; Korfiatis, N.A.; Tecirlioglu, R.T.; et al. Progesterone and Gravidity Differentially Regulate Expression of Extracellular Matrix Components in the Pregnant Rat Myometrium. Biol. Reprod. 2004, 70, 986–992. [Google Scholar] [CrossRef]
- Shynlova, O.; Oldenhof, A.; Dorogin, A.; Xu, Q.; Mu, J.; Nashman, N.; Lye, S.J. Myometrial Apoptosis: Activation of the Caspase Cascade in the Pregnant Rat Myometrium at Midgestation. Biol. Reprod. 2006, 74, 839–849. [Google Scholar] [CrossRef] [PubMed]
- Palmberg, L.; Sjölund, M.; Thyberg, J. Phenotype Modulation in Primary Cultures of Arterial Smooth-Muscle Cells: Reorganization of the Cytoskeleton and Activation of Synthetic Activities. Differentiation 1985, 29, 275–283. [Google Scholar] [CrossRef]
- Opitz, F.; Schenke-Layland, K.; Cohnert, T.U.; Stock, U.A. Phenotypical Plasticity of Vascular Smooth Muscle Cells-Effect of in Vitro and in Vivo Shear Stress for Tissue Engineering of Blood Vessels. Tissue Eng. 2007, 13, 2505–2514. [Google Scholar] [CrossRef]
- Timraz, S.B.; Farhat, I.A.; Alhussein, G.; Christoforou, N.; Teo, J.C. In-Depth Evaluation of Commercially Available Human Vascular Smooth Muscle Cells Phenotype: Implications for Vascular Tissue Engineering. Exp. Cell Res. 2016, 343, 168–176. [Google Scholar] [CrossRef] [PubMed]
- Metz, R.P.; Patterson, J.L.; Wilson, E. Vascular Smooth Muscle Cells: Isolation, Culture, and Characterization. Methods Mol. Biol. 2012, 843, 169–176. [Google Scholar] [CrossRef]
- Martino, M.M.; Briquez, P.S.; Güç, E.; Tortelli, F.; Kilarski, W.W.; Metzger, S.; Rice, J.J.; Kuhn, G.A.; Müller, R.; Swartz, M.A.; et al. Growth Factors Engineered for Super-Affinity to the Extracellular Matrix Enhance Tissue Healing. Science 2014, 343, 885–888. [Google Scholar] [CrossRef]
- Schultz, G.S.; Wysocki, A. Interactions between Extracellular Matrix and Growth Factors in Wound Healing. Wound Repair Regen. 2009, 17, 153–162. [Google Scholar] [CrossRef]
- Bi, Y.; Stuelten, C.H.; Kilts, T.; Wadhwa, S.; Iozzo, R.V.; Robey, P.G.; Chen, X.-D.; Young, M.F. Extracellular Matrix Proteoglycans Control the Fate of Bone Marrow Stromal Cells. J. Biol. Chem. 2005, 280, 30481–30489. [Google Scholar] [CrossRef] [PubMed]
- Pérez, S.; Rius-Pérez, S. Macrophage Polarization and Reprogramming in Acute Inflammation: A Redox Perspective. Antioxidants 2022, 11, 1394. [Google Scholar] [CrossRef] [PubMed]
- Chauhan, A.; Sun, Y.; Sukumaran, P.; Zangbede, F.O.Q.; Jondle, C.N.; Sharma, A.; Evans, D.L.; Chauhan, P.; Szlabick, R.E.; Aaland, M.O.; et al. M1 Macrophage Polarization Is Dependent on TRPC1-Mediated Calcium Entry. IScience 2018, 8, 85–102. [Google Scholar] [CrossRef]
- Sugimoto, M.A.; Ribeiro, A.L.C.; Costa, B.R.C.; Vago, J.P.; Lima, K.M.; Carneiro, F.S.; Ortiz, M.M.O.; Lima, G.L.N.; Carmo, A.A.F.; Rocha, R.M.; et al. Plasmin and Plasminogen Induce Macrophage Reprogramming and Regulate Key Steps of Inflammation Resolution via Annexin A1. Blood 2017, 129, 2896–2907. [Google Scholar] [CrossRef] [PubMed]
- Muhuitijiang, B.; Zhou, J.; Zhou, R.; Zhang, Z.; Yan, G.; Zheng, Z.; Zeng, X.; Zhu, Y.; Wu, H.; Gao, R.; et al. Development and Experimental Validation of an M2 Macrophage and Platelet-Associated Gene Signature to Predict Prognosis and Immunotherapy Sensitivity in Bladder Cancer. Cancer Sci. 2024, 115, 1417–1432. [Google Scholar] [CrossRef]
- Zhao, Y.; Yin, W.; Yang, Z.; Sun, J.; Chang, J.; Huang, L.; Xue, L.; Zhang, X.; Zhi, H.; Chen, S.; et al. Nanotechnology-Enabled M2 Macrophage Polarization and Ferroptosis Inhibition for Targeted Inflammatory Bowel Disease Treatment. J. Control. Release 2024, 367, 339–353. [Google Scholar] [CrossRef]
- Zhang, F.; Wang, H.; Wang, X.; Jiang, G.; Liu, H.; Zhang, G.; Wang, H.; Fang, R.; Bu, X.; Cai, S.; et al. TGF-β Induces M2-like Macrophage Polarization via SNAIL-Mediated Suppression of a pro-Inflammatory Phenotype. Oncotarget 2016, 7, 52294–52306. [Google Scholar] [CrossRef]
- Liang, S.; Cai, J.; Li, Y.; Yang, R. 1,25-Dihydroxy-Vitamin D3 Induces Macrophage Polarization to M2 by Upregulating T-cell Ig-mucin-3 Expression. Mol. Med. Rep. 2019, 19, 3707–3713. [Google Scholar] [CrossRef]
- Deng, S.-K.; Jin, Y.; Jin, Y.; Wang, J.-F. SPARC Induces M2 Polarization of Macrophages to Promote Proliferation, Migration, and Angiogenesis of Cholangiocarcinoma Cells. Neoplasma 2022, 69, 1101–1107. [Google Scholar] [CrossRef]
- Li, Q.; Fu, X.; Yuan, J.; Han, S. Contribution of Thrombospondin-1 and -2 to Lipopolysaccharide-Induced Acute Respiratory Distress Syndrome. Mediat. Inflamm. 2021, 2021, 8876484. [Google Scholar] [CrossRef]
- Perretti, M.; D’Acquisto, F. Annexin A1 and Glucocorticoids as Effectors of the Resolution of Inflammation. Nat. Rev. Immunol. 2009, 9, 62–70. [Google Scholar] [CrossRef] [PubMed]
- Perretti, M.; Dalli, J. Resolution Pharmacology: Focus on Pro-Resolving Annexin A1 and Lipid Mediators for Therapeutic Innovation in Inflammation. Annu. Rev. Pharmacol. Toxicol. 2023, 63, 449–469. [Google Scholar] [CrossRef] [PubMed]
- Ansari, J.; Kaur, G.; Gavins, F.N.E. Therapeutic Potential of Annexin A1 in Ischemia Reperfusion Injury. Int. J. Mol. Sci. 2018, 19, 1211. [Google Scholar] [CrossRef] [PubMed]
- Sheikh, M.H.; Solito, E. Annexin A1: Uncovering the Many Talents of an Old Protein. Int. J. Mol. Sci. 2018, 19, 1045. [Google Scholar] [CrossRef]
- Yang, Y.H.; Morand, E.; Leech, M. Annexin A1: Potential for Glucocorticoid Sparing in RA. Nat. Rev. Rheumatol. 2013, 9, 595–603. [Google Scholar] [CrossRef]
- Yan, Z.; Cheng, X.; Wang, T.; Hong, X.; Shao, G.; Fu, C. Therapeutic Potential for Targeting Annexin A1 in Fibrotic Diseases. Genes Dis. 2022, 9, 1493–1505. [Google Scholar] [CrossRef]
- Gobbetti, T.; Cooray, S.N. Annexin A1 and Resolution of Inflammation: Tissue Repairing Properties and Signalling Signature. Biol. Chem. 2016, 397, 981–993. [Google Scholar] [CrossRef]
- McArthur, S.; Juban, G.; Gobbetti, T.; Desgeorges, T.; Theret, M.; Gondin, J.; Toller-Kawahisa, J.E.; Reutelingsperger, C.P.; Chazaud, B.; Perretti, M.; et al. Annexin A1 Drives Macrophage Skewing to Accelerate Muscle Regeneration through AMPK Activation. J. Clin. Investig. 2020, 130, 1156–1167. [Google Scholar] [CrossRef]
- Häger, S.C.; Nylandsted, J. Annexins: Players of Single Cell Wound Healing and Regeneration. Commun. Integr. Biol. 2019, 12, 162–165. [Google Scholar] [CrossRef]
- Sugimoto, M.A.; Vago, J.P.; Teixeira, M.M.; Sousa, L.P. Annexin A1 and the Resolution of Inflammation: Modulation of Neutrophil Recruitment, Apoptosis, and Clearance. J. Immunol. Res. 2016, 2016, 8239258. [Google Scholar] [CrossRef]
- Drechsler, M.; de Jong, R.; Rossaint, J.; Viola, J.R.; Leoni, G.; Wang, J.M.; Grommes, J.; Hinkel, R.; Kupatt, C.; Weber, C.; et al. Annexin A1 Counteracts Chemokine-Induced Arterial Myeloid Cell Recruitment. Circ. Res. 2015, 116, 827–835. [Google Scholar] [CrossRef]
- Araújo, T.G.; Mota, S.T.S.; Ferreira, H.S.V.; Ribeiro, M.A.; Goulart, L.R.; Vecchi, L. Annexin A1 as a Regulator of Immune Response in Cancer. Cells 2021, 10, 2245. [Google Scholar] [CrossRef]
- Moraes, L.A.; Kar, S.; Foo, S.L.; Gu, T.; Toh, Y.Q.; Ampomah, P.B.; Sachaphibulkij, K.; Yap, G.; Zharkova, O.; Lukman, H.M.; et al. Annexin-A1 Enhances Breast Cancer Growth and Migration by Promoting Alternative Macrophage Polarization in the Tumour Microenvironment. Sci. Rep. 2017, 7, 17925. [Google Scholar] [CrossRef]
- Novizio, N.; Belvedere, R.; Pessolano, E.; Morello, S.; Tosco, A.; Campiglia, P.; Filippelli, A.; Petrella, A. ANXA1 Contained in EVs Regulates Macrophage Polarization in Tumor Microenvironment and Promotes Pancreatic Cancer Progression and Metastasis. Int. J. Mol. Sci. 2021, 22, 11018. [Google Scholar] [CrossRef] [PubMed]
- de Graauw, M.; van Miltenburg, M.H.; Schmidt, M.K.; Pont, C.; Lalai, R.; Kartopawiro, J.; Pardali, E.; Le Dévédec, S.E.; Smit, V.T.; van der Wal, A.; et al. Annexin A1 Regulates TGF-β Signaling and Promotes Metastasis Formation of Basal-like Breast Cancer Cells. Proc. Natl. Acad. Sci. USA 2010, 107, 6340–6345. [Google Scholar] [CrossRef] [PubMed]
- Moraes, L.A.; Ampomah, P.B.; Lim, L.H.K. Annexin A1 in Inflammation and Breast Cancer: A New Axis in the Tumor Microenvironment. Cell Adhes. Migr. 2018, 12, 417–423. [Google Scholar] [CrossRef]
- Hatoum, D.; Yagoub, D.; Ahadi, A.; Nassif, N.T.; McGowan, E.M. Annexin/S100A Protein Family Regulation through p14ARF-P53 Activation: A Role in Cell Survival and Predicting Treatment Outcomes in Breast Cancer. PLoS ONE 2017, 12, e0169925. [Google Scholar] [CrossRef]
- Christensen, M.V.; Høgdall, C.; Jensen, S.G.; Lokman, N.; Ricciardelli, C.; Christensen, I.J.; Christiansen, P.; Brask, J.; Karlsen, M.A.; Nissen, T.K.; et al. Annexin A2 and S100A10 as Candidate Prognostic Markers in Epithelial Ovarian Cancer. Antican. Res. 2019, 39, 2475–2482. [Google Scholar] [CrossRef]
- Weisz, J.; Uversky, V.N. Zooming into the Dark Side of Human Annexin-S100 Complexes: Dynamic Alliance of Flexible Partners. Int. J. Mol. Sci. 2020, 21, 5879. [Google Scholar] [CrossRef]
- Rintala-Dempsey, A.C.; Rezvanpour, A.; Shaw, G.S. S100-Annexin Complexes—Structural Insights. FEBS J. 2008, 275, 4956–4966. [Google Scholar] [CrossRef]
- Liu, Y.; Myrvang, H.K.; Dekker, L.V. Annexin A2 Complexes with S100 Proteins: Structure, Function and Pharmacological Manipulation. Br. J. Pharmacol. 2015, 172, 1664–1676. [Google Scholar] [CrossRef]
- Zhang, Z.; Huang, L.; Zhao, W.; Rigas, B. Annexin 1 Induced by Anti-Inflammatory Drugs Binds to NF-κB and Inhibits Its Activation: Anticancer Effects In Vitro and In Vivo. Cancer Res. 2010, 70, 2379–2388. [Google Scholar] [CrossRef] [PubMed]
- Mussbacher, M.; Derler, M.; Basílio, J.; Schmid, J.A. NF-κB in Monocytes and Macrophages—An Inflammatory Master Regulator in Multitalented Immune Cells. Front. Immunol. 2023, 14, 1134661. [Google Scholar] [CrossRef]
- Alhasan, H.; Terkawi, M.A.; Matsumae, G.; Ebata, T.; Tian, Y.; Shimizu, T.; Nishida, Y.; Yokota, S.; Garcia-Martin, F.; Elwakil, M.M.A.; et al. Inhibitory Role of Annexin A1 in Pathological Bone Resorption and Therapeutic Implications in Periprosthetic Osteolysis. Nat. Commun. 2022, 13, 3919. [Google Scholar] [CrossRef] [PubMed]
- Juban, G.; Chazaud, B. Efferocytosis during Skeletal Muscle Regeneration. Cells 2021, 10, 3267. [Google Scholar] [CrossRef]
- Wang, X.; Zhou, L. The Many Roles of Macrophages in Skeletal Muscle Injury and Repair. Front. Cell Dev. Biol. 2022, 10, 952249. [Google Scholar] [CrossRef]
- Yu, Y.; Yue, Z.; Xu, M.; Zhang, M.; Shen, X.; Ma, Z.; Li, J.; Xie, X. Macrophages Play a Key Role in Tissue Repair and Regeneration. PeerJ 2022, 10, e14053. [Google Scholar] [CrossRef]
- Desgeorges, T.; Caratti, G.; Mounier, R.; Tuckermann, J.; Chazaud, B. Glucocorticoids Shape Macrophage Phenotype for Tissue Repair. Front. Immunol. 2019, 10, 1591. [Google Scholar] [CrossRef]
- Rőszer, T. Understanding the Mysterious M2 Macrophage through Activation Markers and Effector Mechanisms. Mediat. Inflamm. 2015, 2015, 816460. [Google Scholar] [CrossRef]
- Yao, Y.; Xu, X.-H.; Jin, L. Macrophage Polarization in Physiological and Pathological Pregnancy. Front. Immunol. 2019, 10, 792. [Google Scholar] [CrossRef]





Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Boros-Rausch, A.; Dorogin, A.; Nadeem, L.; Shynlova, O.; Lye, S.J. A Broad-Spectrum Chemokine Inhibitor Drives M2 Macrophage Polarization Through Modulation of the Myometrial Secretome. Cells 2025, 14, 514. https://doi.org/10.3390/cells14070514
Boros-Rausch A, Dorogin A, Nadeem L, Shynlova O, Lye SJ. A Broad-Spectrum Chemokine Inhibitor Drives M2 Macrophage Polarization Through Modulation of the Myometrial Secretome. Cells. 2025; 14(7):514. https://doi.org/10.3390/cells14070514
Chicago/Turabian StyleBoros-Rausch, Adam, Anna Dorogin, Lubna Nadeem, Oksana Shynlova, and Stephen James Lye. 2025. "A Broad-Spectrum Chemokine Inhibitor Drives M2 Macrophage Polarization Through Modulation of the Myometrial Secretome" Cells 14, no. 7: 514. https://doi.org/10.3390/cells14070514
APA StyleBoros-Rausch, A., Dorogin, A., Nadeem, L., Shynlova, O., & Lye, S. J. (2025). A Broad-Spectrum Chemokine Inhibitor Drives M2 Macrophage Polarization Through Modulation of the Myometrial Secretome. Cells, 14(7), 514. https://doi.org/10.3390/cells14070514





