The Interplay among Subunit Composition, Cardiolipin Content, and Aggregation State of Bovine Heart Cytochrome c Oxidase
Abstract
:1. Introduction
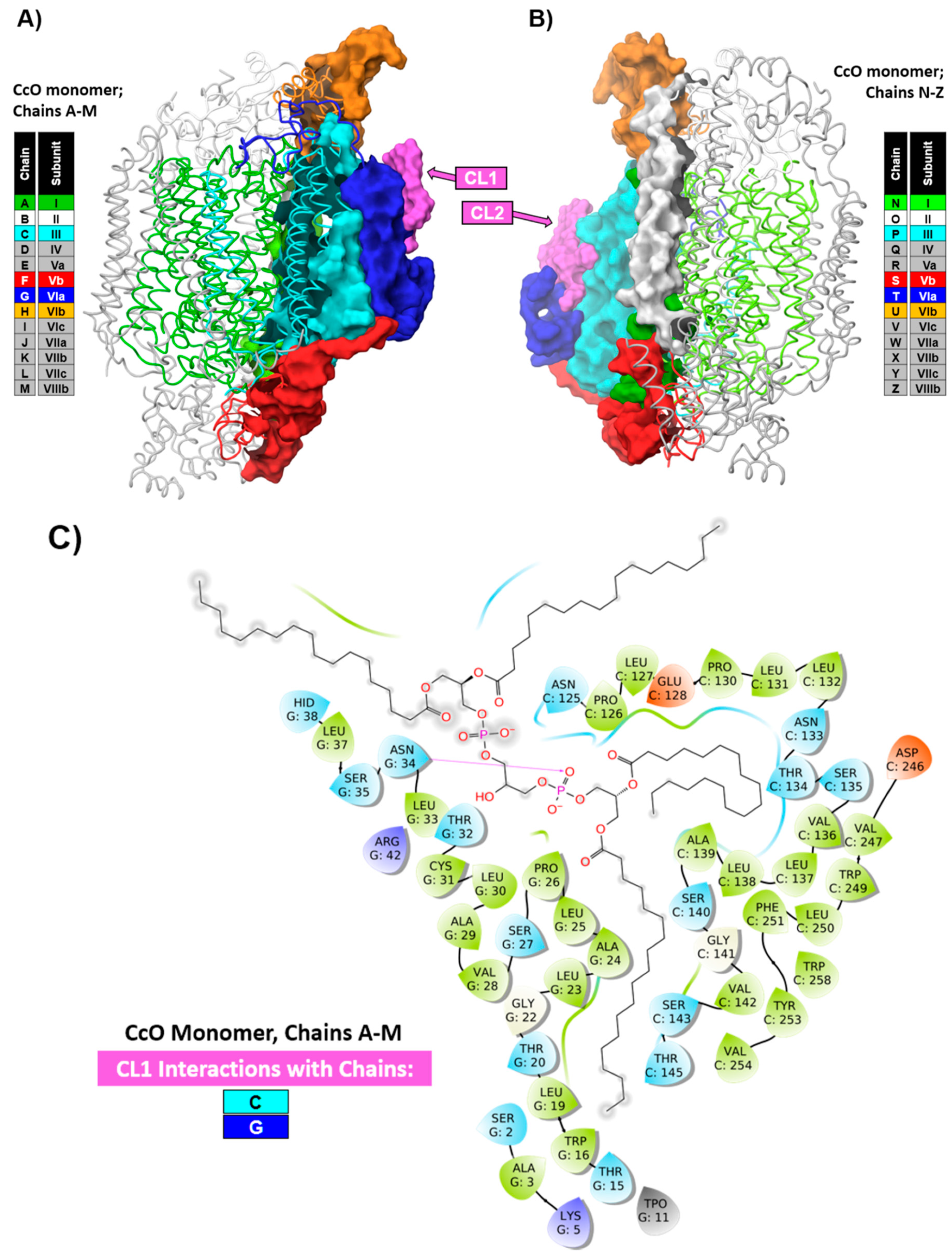
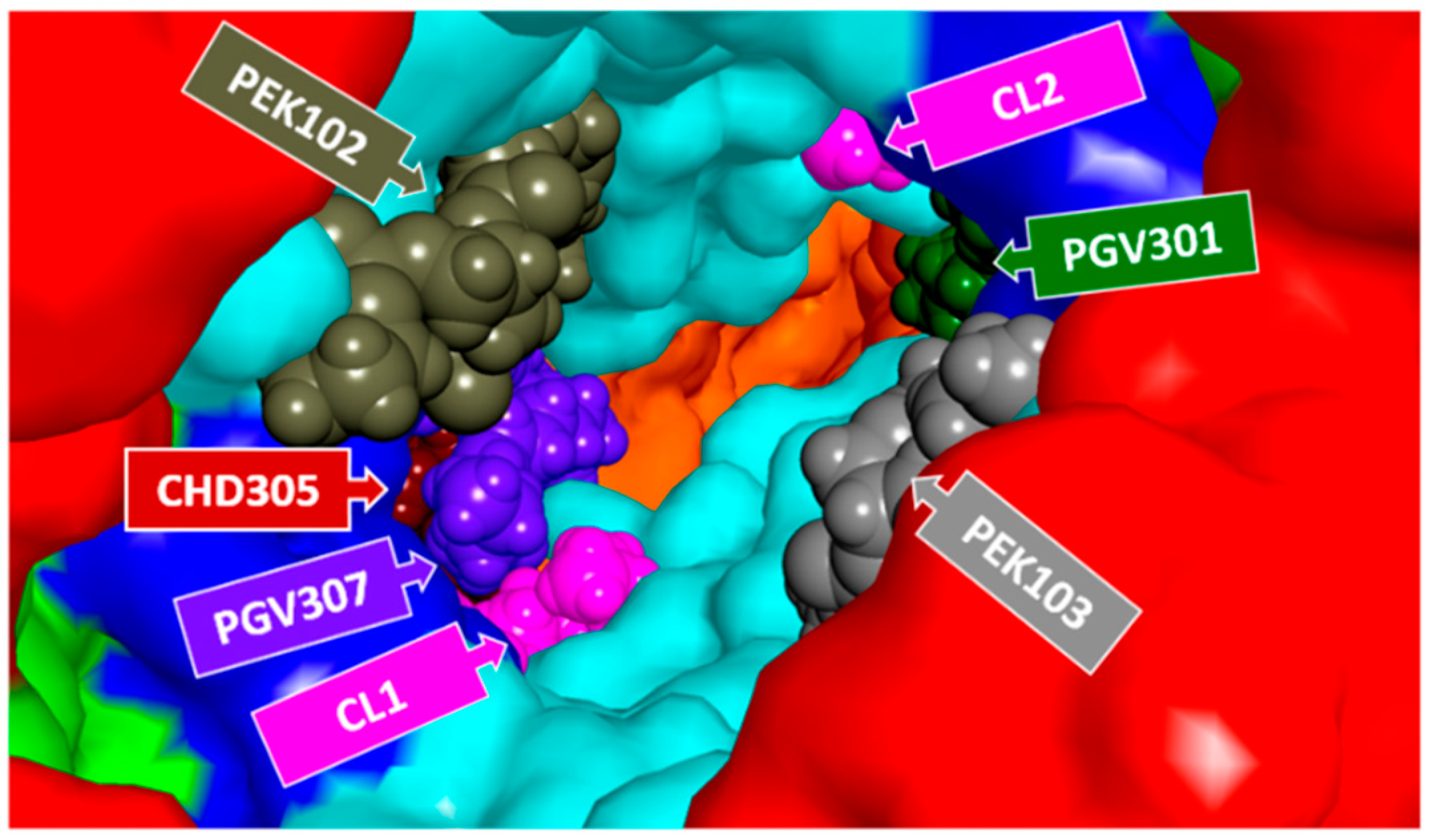
1.1. Specific Cardiolipin Interactions with CcO Surface
1.2. Nonspecific Entropic Effect of Phospholipids on Dimerization of Membrane Proteins
1.3. Specific Protein–Lipid and Protein–Protein Interactions in Dimerization of Membrane Proteins
2. Role of Amphiphilic Environment in Aggregation State of Cytochrome c Oxidase
2.1. Evidence for Existence of Dimeric Structure of CcO
2.2. Modulation of the Aggregation State of CcO by “Linear” Detergents
2.3. Dimerization of CcO Induced by “Planar” Bile Salt Detergents
3. Role of Interface Subunits and Cardiolipin in Function and Stability of Cytochrome c Oxidase Dimeric Form
3.1. Role of Unspecific Phospholipids in Stabilization of Dimeric CcO
3.2. Relationship between Specific CL Binding/Dimerization and Proton Pumping of CcO
3.3. Evidences for a Monomeric Electron-Transfer Competing Form of CcO
3.4. Fast Labile Monomer versus Tunable Stable Dimer of CcO
4. Posttranslational Modifications in Cytochrome c Oxidase
5. Cytochrome c Oxidase in Supercomplexes of the Respiratory Chain
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Liko, I.; Degiacomi, M.T.; Mohammed, S.; Yoshikawa, S.; Schmidt, C.; Robinson, C.V. Dimer interface of bovine cytochrome c oxidase is influenced by local posttranslational modifications and lipid binding. Proc. Natl. Acad. Sci. USA 2016, 113, 8230–8235. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kadenbach, B.; Jarausch, J.; Hartmann, R.; Merle, P. Separation of mammalian cytochrome c oxidase into 13 polypeptides by a sodium dodecyl sulfate-gel electrophoretic procedure. Anal. Biochem. 1983, 129, 517–521. [Google Scholar] [CrossRef]
- Kadenbach, B.; Huttemann, M. The subunit composition and function of mammalian cytochrome c oxidase. Mitochondrion 2015, 24, 64–76. [Google Scholar] [CrossRef] [PubMed]
- Shinzawa-Itoh, K.; Sugimura, T.; Misaki, T.; Tadehara, Y.; Yamamoto, S.; Hanada, M.; Yano, N.; Nakagawa, T.; Uene, S.; Yamada, T.; et al. Monomeric structure of an active form of bovine cytochrome c oxidase. Proc. Natl. Acad. Sci. USA 2019, 116, 19945–19951. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hendler, R.W.; Pardhasaradhi, K.; Reynafarje, B.; Ludwig, B. Comparison of energy-transducing capabilities of the two- and three-subunit cytochromes aa3 from Paracoccus denitrificans and the 13-subunit beef heart enzyme. Biophys. J. 1991, 60, 415–423. [Google Scholar] [CrossRef] [Green Version]
- Zong, S.; Wu, M.; Gu, J.; Liu, T.; Guo, R.; Yang, M. Structure of the intact 14-subunit human cytochrome c oxidase. Cell. Res. 2018, 28, 1026–1034. [Google Scholar] [CrossRef] [Green Version]
- Kadenbach, B. Regulation of Mammalian 13-Subunit Cytochrome c Oxidase and Binding of other Proteins: Role of NDUFA4. Trends. Endocrinol. Metab. 2017, 28, 761–770. [Google Scholar] [CrossRef]
- Ramzan, R.; Rhiel, A.; Weber, P.; Kadenbach, B.; Vogt, S. Reversible dimerization of cytochrome c oxidase regulates mitochondrial respiration. Mitochondrion 2019, 49, 149–155. [Google Scholar] [CrossRef]
- Sedlak, E.; Panda, M.; Dale, M.P.; Weintraub, S.T.; Robinson, N.C. Photolabeling of cardiolipin binding subunits within bovine heart cytochrome c oxidase. Biochemistry 2006, 45, 746–754. [Google Scholar] [CrossRef] [Green Version]
- Tsukihara, T.; Shimokata, K.; Katayama, Y.; Shimada, H.; Muramoto, K.; Aoyama, H.; Mochizuki, M.; Shinzawa-Itoh, K.; Yamashita, E.; Yao, M.; et al. The low-spin heme of cytochrome c oxidase as the driving element of the proton-pumping process. Proc. Natl. Acad. Sci. USA 2003, 100, 15304–15309. [Google Scholar] [CrossRef] [Green Version]
- Arnarez, C.; Marrink, S.J.; Periole, X. Erratum: Identification of cardiolipin binding sites on cytochrome c oxidase at the entrance of proton channels. Sci. Rep. 2013, 3. [Google Scholar] [CrossRef] [Green Version]
- Yano, N.; Muramoto, K.; Shimada, A.; Takemura, S.; Baba, J.; Fujisawa, H.; Mochizuki, M.; Shinzawa-Itoh, K.; Yamashita, E.; Tsukihara, T.; et al. The Mg2+-containing Water Cluster of Mammalian Cytochrome c Oxidase Collects Four Pumping Proton Equivalents in Each Catalytic Cycle. J. Biol. Chem. 2016, 291, 23882–23894. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Marianayagam, N.J.; Sunde, M.; Matthews, J.M. The power of two: Protein dimerization in biology. Trends. Biochemistry. Sci. 2004, 29, 618–625. [Google Scholar] [CrossRef] [PubMed]
- Mei, G.; Di Venere, A.; Rosato, N.; Finazzi-Agro, A. The importance of being dimeric. FEBS J. 2005, 272, 16–27. [Google Scholar] [CrossRef] [PubMed]
- Matthews, J.M.; Sunde, M. Dimers, Oligomers, Everywhere; Springer Science and Business Media LLC: Berlin, Germany, 2012; Volume 747, pp. 1–18. [Google Scholar]
- Helms, V. Attraction within the membrane. Forces behind transmembrane protein folding and supramolecular complex assembly. EMBO. Rep. 2002, 3, 1133–1138. [Google Scholar] [CrossRef]
- White, S.H.; Ladokhin, A.S.; Jayasinghe, S.; Hristova, K. How membranes shape protein structure. J. Biol. Chem. 2001, 276, 32395–32398. [Google Scholar] [CrossRef] [Green Version]
- Bowie, J.U. Helix packing in membrane proteins. J. Mol. Biol. 1997, 272, 780–789. [Google Scholar] [CrossRef]
- Langosch, D.; Heringa, J. Interaction of transmembrane helices by a knobs-into-holes packing characteristic of soluble coiled coils. Proteins 1998, 31, 150–159. [Google Scholar] [CrossRef]
- Senes, A.; Gerstein, M.; Engelman, D.M. Statistical analysis of amino acid patterns in transmembrane helices: The GxxxG motif occurs frequently and in association with beta-branched residues at neighboring positions. J. Mol. Biol. 2000, 296, 921–936. [Google Scholar] [CrossRef] [Green Version]
- Teese, M.G.; Langosch, D. Role of GxxxG Motifs in Transmembrane Domain Interactions. Biochemistry 2015, 54, 5125–5135. [Google Scholar] [CrossRef]
- Cymer, F.; Veerappan, A.; Schneider, D. Transmembrane helix-helix interactions are modulated by the sequence context and by lipid bilayer properties. BBA 2012, 1818, 963–973. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McAuley, K.E.; Fyfe, P.K.; Ridge, J.P.; Isaacs, N.W.; Cogdell, R.J.; Jones, M.R. Structural details of an interaction between cardiolipin and an integral membrane protein. Proc. Natl. Acad. Sci. USA 1999, 96, 14706–14711. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Palsdottir, H.; Hunte, C. Lipids in membrane protein structures. BBA 2004, 1666, 2–18. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jones, M.R. Lipids in photosynthetic reaction centres: Structural roles and functional holes. Prog. Lipid. Res. 2007, 46, 56–87. [Google Scholar] [CrossRef]
- Shinzawa-Itoh, K.; Aoyama, H.; Muramoto, K.; Terada, H.; Kurauchi, T.; Tadehara, Y.; Yamasaki, A.; Sugimura, T.; Kurono, S.; Tsujimoto, K.; et al. Structures and physiological roles of 13 integral lipids of bovine heart cytochrome c oxidase. EMBO J. 2007, 26, 1713–1725. [Google Scholar] [CrossRef]
- Nałȩcz, K.A.; Bolli, R.; Azzi, A. Preparation of monomeric cytochrome C oxidase: Its kinetics differ from those of the dimeric enzyme. Biochem. Biophys. Res. Commun. 1983, 114, 822–828. [Google Scholar] [CrossRef]
- Bolli, R.; Nalecz, K.A.; Azzi, A. The aggregation state of bovine heart cytochrome c oxidase and its kinetics in monomeric and dimeric form. Arch. Biochem. Biophys. 1985, 240, 102–116. [Google Scholar] [CrossRef]
- Bolli, R.; Nałecz, K.A.; Azzi, A. The interconversion between monomeric and dimeric bovine heart cytochrome c oxidase. Biochimie 1985, 67, 119–128. [Google Scholar] [CrossRef]
- Kwee, S.; Møller, J.V.; Le Maire, M. Binding of Detergents by Membrane Proteins. In Surfactants in Solution; Mittal, K.L., Bothorel, P., Eds.; Springer: Boston, MA, USA, 1986; pp. 853–860. [Google Scholar]
- Robinson, N.C.; Capaldi, R.A. Interaction of detergents with cytochrome c oxidase. Biochemistry 1977, 16, 375–381. [Google Scholar] [CrossRef]
- Bisson, R.; Jacobs, B.; Capaldi, R.A. Binding of arylazidocytochrome c derivatives to beef heart cytochrome c oxidase: Cross-linking in the high- and low-affinity binding sites. Biochemistry 1980, 19, 4173–4178. [Google Scholar] [CrossRef]
- Henderson, R.; Capaldi, R.A.; Leigh, J.S. Arrangement of cytochrome oxidase molecules in two-dimensional vesicle crystals. J. Mol. Biol. 1977, 112, 631–648. [Google Scholar] [CrossRef]
- Yoshikawa, S.; Tera, T.; Takahashi, Y.; Tsukihara, T.; Caughey, W.S. Crystalline cytochrome c oxidase of bovine heart mitochondrial membrane: Composition and X-ray diffraction studies. Proc. Natl. Acad. Sci. USA 1988, 85, 1354–1358. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tsukihara, T.; Aoyama, H.; Yamashita, E.; Tomizaki, T.; Yamaguchi, H.; Shinzawa-Itoh, K.; Nakashima, R.; Yaono, R.; Yoshikawa, S. The whole structure of the 13-subunit oxidized cytochrome c oxidase at 2.8 A. Science 1996, 272, 1136–1144. [Google Scholar] [CrossRef] [PubMed]
- Estey, L.A.; Prochaska, L.J. Detection of bovine heart mitochondrial cytochrome c oxidase dimers in Triton X-100 and phospholipid vesicles by chemical cross-linking. Biochemistry 1993, 32, 13270–13276. [Google Scholar] [CrossRef]
- Rubinson, K.A.; Pokalsky, C.; Krueger, S.; Prochaska, L.J. Structure determination of functional membrane proteins using small-angle neutron scattering (sans) with small, mixed-lipid liposomes: Native beef heart mitochondrial cytochrome c oxidase forms dimers. Protein J. 2013, 32, 27–38. [Google Scholar] [CrossRef]
- Gahbauer, S.; Bockmann, R.A. Comprehensive Characterization of Lipid-Guided G Protein-Coupled Receptor Dimerization. J. Phys. Chem. B. 2020, 124, 2823–2834. [Google Scholar] [CrossRef]
- Fisher, L.E.; Engelman, D.M.; Sturgis, J.N. Detergents modulate dimerization, but not helicity, of the glycophorin A transmembrane domain. J. Mol. Biol. 1999, 293, 639–651. [Google Scholar] [CrossRef] [Green Version]
- Capaldi, R.A.; Malatesta, F.; Darley-Usmar, V.M. Structure of cytochrome c oxidase. Biochim Biophys Acta Rev. Bioenrg. 1983, 726, 135–148. [Google Scholar] [CrossRef]
- Saraste, M.; Penttila, T.; Wikstrom, M. Quaternary structure of bovine cytochrome oxidase. Eur. J. Biochemistry. 1981, 115, 261–268. [Google Scholar] [CrossRef]
- Georgevich, G.; Darley-Usmar, V.M.; Malatesta, F.; Capaldi, R.A. Electron transfer in monomeric forms of beef and shark heart cytochrome c oxidase. Biochemistry 1983, 22, 1317–1322. [Google Scholar] [CrossRef]
- Darley-Usmar, V.M.; Alizai, N.; Al-Ayash, A.I.; Jones, G.D.; Sharpe, A.; Wilson, M.T. A comparison of the structural and functional properties of cytochrome c oxidase isolated from beef (Bos tauros), camel (Camelus dromedarius), chicken (Gallus domesticus) and rat (Rattus norvegicus). Comp. Biochem. Physiol. B: Comp. Biochem. 1981, 68, 445–456. [Google Scholar] [CrossRef]
- Robinson, N.C.; Talbert, L. Triton X-100 induced dissociation of beef heart cytochrome c oxidase into monomers. Biochemistry 1986, 25, 2328–2335. [Google Scholar] [CrossRef] [PubMed]
- Musatov, A.; Ortega-Lopez, J.; Robinson, N.C. Detergent-solubilized bovine cytochrome c oxidase: Dimerization depends on the amphiphilic environment. Biochemistry 2000, 39, 12996–13004. [Google Scholar] [CrossRef] [PubMed]
- Musatov, A.; Ortega-Lopez, J.; Demeler, B.; Osborne, J.P.; Gennis, R.B.; Robinson, N.C. Detergent-solubilized Escherichia coli cytochrome bo 3 ubiquinol oxidase: A monomeric, not a dimeric complex. FEBS Lett. 1999, 457, 153–156. [Google Scholar] [CrossRef] [Green Version]
- Musatov, A.; Robinson, N.C. Cholate-induced dimerization of detergent- or phospholipid-solubilized bovine cytochrome C oxidase. Biochemistry 2002, 41, 4371–4376. [Google Scholar] [CrossRef]
- Stanicova, J.; Sedlak, E.; Musatov, A.; Robinson, N.C. Differential stability of dimeric and monomeric cytochrome c oxidase exposed to elevated hydrostatic pressure. Biochemistry 2007, 46, 7146–7152. [Google Scholar] [CrossRef] [Green Version]
- Musatov, A.; Robinson, N.C. Bound cardiolipin is essential for cytochrome c oxidase proton translocation. Biochimie 2014, 105, 159–164. [Google Scholar] [CrossRef] [Green Version]
- Van Holde, K.E.; Weischet, W.O. Boundary analysis of sedimentation-velocity experiments with monodisperse and paucidisperse solutes. Biopolymers 1978, 17, 1387–1403. [Google Scholar] [CrossRef]
- Stafford, W.F. Boundary analysis in sedimentation transport experiments: A procedure for obtaining sedimentation coefficient distributions using the time derivative of the concentration profile. Anal. Biochem. 1992, 203, 295–301. [Google Scholar] [CrossRef]
- Tanford, C.; Nozaki, Y.; Reynolds, J.A.; Makino, S. Molecular characterization of proteins in detergent solutions. Biochemistry 1974, 13, 2369–2376. [Google Scholar] [CrossRef]
- Demeler, B.; Saber, H. Determination of molecular parameters by fitting sedimentation data to finite-element solutions of the Lamm equation. Biophys. J. 1998, 74, 444–454. [Google Scholar] [CrossRef] [Green Version]
- Reynolds, J.A.; Tanford, C. Determination of molecular weight of the protein moiety in protein-detergent complexes without direct knowledge of detergent binding. Proc. Natl. Acad. Sci. USA 1976, 73, 4467–4470. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Musatov, A.; Robinson, N.C. Detergent-solubilized monomeric and dimeric cytochrome bc1 isolated from bovine heart. Biochemistry 1994, 33, 13005–13012. [Google Scholar] [CrossRef] [PubMed]
- Napiwotzki, J.; Shinzawa-Itoh, K.; Yoshikawa, S.; Kadenbach, B. ATP and ADP bind to cytochrome c oxidase and regulate its activity. Biol. Chem. 1997, 378, 1013–1021. [Google Scholar] [CrossRef] [PubMed]
- Dainese, E.; Oddi, S.; Maccarrone, M. Lipid-mediated dimerization of beta2-adrenergic receptor reveals important clues for cannabinoid receptors. Cell. Mol. Life Sci. 2008, 65, 2277–2279. [Google Scholar] [CrossRef] [PubMed]
- Mondal, S.; Khelashvili, G.; Johner, N.; Weinstein, H. How the dynamic properties and functional mechanisms of GPCRs are modulated by their coupling to the membrane environment. Adv. Exp. Med. Biol. 2014, 796, 55–74. [Google Scholar] [CrossRef] [PubMed]
- Gahbauer, S.; Bockmann, R.A. Membrane-Mediated Oligomerization of G Protein Coupled Receptors and Its Implications for GPCR Function. Front. Physiol. 2016, 7, 494. [Google Scholar] [CrossRef] [Green Version]
- Wittig, I.; Braun, H.P.; Schagger, H. Blue native PAGE. Nat. Protoc. 2006, 1, 418–428. [Google Scholar] [CrossRef]
- Bazan, S.; Mileykovskaya, E.; Mallampalli, V.K.; Heacock, P.; Sparagna, G.C.; Dowhan, W. Cardiolipin-dependent reconstitution of respiratory supercomplexes from purified Saccharomyces cerevisiae complexes III and IV. J. Biol. Chem. 2013, 288, 401–411. [Google Scholar] [CrossRef] [Green Version]
- Lee, S.J.; Yamashita, E.; Abe, T.; Fukumoto, Y.; Tsukihara, T.; Shinzawa-Itoh, K.; Ueda, H.; Yoshikawa, S. Intermonomer interactions in dimer of bovine heart cytochrome c oxidase. Acta Cryst. 2001, 57, 941–947. [Google Scholar] [CrossRef]
- Sedlak, E.; Robinson, N.C. Phospholipase A(2) digestion of cardiolipin bound to bovine cytochrome c oxidase alters both activity and quaternary structure. Biochemistry 1999, 38, 14966–14972. [Google Scholar] [CrossRef] [PubMed]
- Sedlak, E.; Robinson, N.C. Destabilization of the Quaternary Structure of Bovine Heart Cytochrome c Oxidase upon Removal of Tightly Bound Cardiolipin. Biochemistry 2015, 54, 5569–5577. [Google Scholar] [CrossRef] [PubMed]
- Daum, G. Lipids of mitochondria. BBA 1985, 822, 1–42. [Google Scholar] [CrossRef]
- Musatov, A.; Sedlak, E. Role of cardiolipin in stability of integral membrane proteins. Biochimie 2017, 142, 102–111. [Google Scholar] [CrossRef] [PubMed]
- Berg, J.; Liu, J.; Svahn, E.; Ferguson-Miller, S.; Brzezinski, P. Structural changes at the surface of cytochrome c oxidase alter the proton-pumping stoichiometry. BBA Bioenerg. 2020, 1861, 148116. [Google Scholar] [CrossRef] [PubMed]
- Musatov, A.; Siposova, K.; Kubovcikova, M.; Lysakova, V.; Varhac, R. Functional and structural evaluation of bovine heart cytochrome c oxidase incorporated into bicelles. Biochimie 2016, 121, 21–28. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Siposova, K.; Petrenko, V.I.; Ivankov, O.I.; Bulavin, L.A.; Musatov, A. Small-Angle Neutron Scattering Study of Bicelles and Proteobicelles with Incorporated Mitochondrial Cytochrome c Oxidase. Ukrainian. J. Phys. 2020, 65, 662. [Google Scholar] [CrossRef]
- Sedlak, E.; Varhac, R.; Musatov, A.; Robinson, N.C. The kinetic stability of cytochrome C oxidase: Effect of bound phospholipid and dimerization. Biophys. J. 2014, 107, 2941–2949. [Google Scholar] [CrossRef] [Green Version]
- Sedlak, E.; Robinson, N.C. Sequential dissociation of subunits from bovine heart cytochrome C oxidase by urea. Biochemistry 2009, 48, 8143–8150. [Google Scholar] [CrossRef] [Green Version]
- Musatov, A.; Hebert, E.; Carroll, C.A.; Weintraub, S.T.; Robinson, N.C. Specific modification of two tryptophans within the nuclear-encoded subunits of bovine cytochrome c oxidase by hydrogen peroxide. Biochemistry 2004, 43, 1003–1009. [Google Scholar] [CrossRef]
- Musatov, A. Contribution of peroxidized cardiolipin to inactivation of bovine heart cytochrome c oxidase. Free Radic. Biol. Med. 2006, 41, 238–246. [Google Scholar] [CrossRef]
- Musatov, A.; Carroll, C.A.; Liu, Y.C.; Henderson, G.I.; Weintraub, S.T.; Robinson, N.C. Identification of bovine heart cytochrome c oxidase subunits modified by the lipid peroxidation product 4-hydroxy-2-nonenal. Biochemistry 2002, 41, 8212–8220. [Google Scholar] [CrossRef] [PubMed]
- Srinivasan, S.; Avadhani, N.G. Cytochrome c oxidase dysfunction in oxidative stress. Free Radic. Biol. Med. 2012, 53, 1252–1263. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Musatov, A.; Robinson, N.C. Susceptibility of mitochondrial electron-transport complexes to oxidative damage. Focus on cytochrome c oxidase. Free Radic. Res. 2012, 46, 1313–1326. [Google Scholar] [CrossRef] [PubMed]
- Arnold, S.; Kadenbach, B. Cell respiration is controlled by ATP, an allosteric inhibitor of cytochrome-c oxidase. Eur. J. Biochem. 1997, 249, 350–354. [Google Scholar] [CrossRef]
- Arnold, S.; Kadenbach, B. The intramitochondrial ATP/ADP-ratio controls cytochrome c oxidase activity allosterically. FEBS Lett. 1999, 443, 105–108. [Google Scholar] [CrossRef] [Green Version]
- Bender, E.; Kadenbach, B. The allosteric ATP-inhibition of cytochrome c oxidase activity is reversibly switched on by cAMP-dependent phosphorylation. FEBS Lett. 2000, 466, 130–134. [Google Scholar] [CrossRef] [Green Version]
- Kadenbach, B.; Arnold, S. A second mechanism of respiratory control. FEBS Lett. 1999, 447, 131–134. [Google Scholar] [CrossRef] [Green Version]
- Kadenbach, B.; Ramzan, R.; Vogt, S. High efficiency versus maximal performance--the cause of oxidative stress in eukaryotes: A hypothesis. Mitochondrion 2013, 13, 1–6. [Google Scholar] [CrossRef]
- Ramzan, R.; Vogt, S.; Kadenbach, B. Stress-mediated generation of deleterious ROS in healthy individuals - role of cytochrome c oxidase. J. Mol. Med. (Berl). 2020, 98, 651–657. [Google Scholar] [CrossRef] [Green Version]
- Pearson, R.B.; Kemp, B.E. Protein kinase phosphorylation site sequences and consensus specificity motifs: Tabulations. Methods Enzymol. 1991. [Google Scholar] [CrossRef]
- Ramzan, R.; Weber, P.; Kadenbach, B.; Vogt, S. Individual Biochemistryical Behaviour Versus Biological Robustness: Spotlight on the Regulation of Cytochrome c Oxidase. Adv. Exp. Med. Biol. 2012, 265–281. [Google Scholar] [CrossRef]
- Lenaz, G.; Baracca, A.; Barbero, G.; Bergamini, C.; Dalmonte, M.E.; Del Sole, M.; Faccioli, M.; Falasca, A.; Fato, R.; Genova, M.L.; et al. Mitochondrial respiratory chain super-complex I-III in physiology and pathology. BBA 2010, 1797, 633–640. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schagger, H.; Pfeiffer, K. Supercomplexes in the respiratory chains of yeast and mammalian mitochondria. EMBO J. 2000, 19, 1777–1783. [Google Scholar] [CrossRef] [Green Version]
- Dudkina, N.V.; Eubel, H.; Keegstra, W.; Boekema, E.J.; Braun, H.P. Structure of a mitochondrial supercomplex formed by respiratory-chain complexes I and III. Proc. Natl. Acad. Sci. USA 2005, 102, 3225–3229. [Google Scholar] [CrossRef] [Green Version]
- Shinzawa-Itoh, K.; Shimomura, H.; Yanagisawa, S.; Shimada, S.; Takahashi, R.; Oosaki, M.; Ogura, T.; Tsukihara, T. Purification of Active Respiratory Supercomplex from Bovine Heart Mitochondria Enables Functional Studies. J. Biol. Chem. 2016, 291, 4178–4184. [Google Scholar] [CrossRef] [Green Version]
- Althoff, T.; Mills, D.J.; Popot, J.L.; Kuhlbrandt, W. Arrangement of electron transport chain components in bovine mitochondrial supercomplex I1III2IV1. EMBO J. 2011, 30, 4652–4664. [Google Scholar] [CrossRef] [Green Version]
- Schafer, E.; Dencher, N.A.; Vonck, J.; Parcej, D.N. Three-dimensional structure of the respiratory chain supercomplex I1III2IV1 from bovine heart mitochondria. Biochemistry 2007, 46, 12579–12585. [Google Scholar] [CrossRef]
- Gu, J.; Wu, M.; Guo, R.; Yan, K.; Lei, J.; Gao, N.; Yang, M. The architecture of the mammalian respirasome. Nature 2016, 537, 639–643. [Google Scholar] [CrossRef]
- Sousa, J.S.; Mills, D.J.; Vonck, J.; Kuhlbrandt, W. Functional asymmetry and electron flow in the bovine respirasome. Elife 2016, 5, e21290. [Google Scholar] [CrossRef]
- Stuchebrukhov, A.; Schafer, J.; Berg, J.; Brzezinski, P. Kinetic advantage of forming respiratory supercomplexes. BBA Bioenerg. 2020, 1861, 148193. [Google Scholar] [CrossRef] [PubMed]
- Wittig, I.; Schagger, H. Supramolecular organization of ATP synthase and respiratory chain in mitochondrial membranes. BBA 2009, 1787, 672–680. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Strecker, V.; Wumaier, Z.; Wittig, I.; Schagger, H. Large pore gels to separate mega protein complexes larger than 10 MDa by blue native electrophoresis: Isolation of putative respiratory strings or patches. Proteomics 2010, 10, 3379–3387. [Google Scholar] [CrossRef] [PubMed]
- Letts, J.A.; Fiedorczuk, K.; Sazanov, L.A. The architecture of respiratory supercomplexes. Nature 2016, 537, 644–648. [Google Scholar] [CrossRef]
- Guo, R.; Zong, S.; Wu, M.; Gu, J.; Yang, M. Architecture of Human Mitochondrial Respiratory Megacomplex I2III2IV2. Cell 2017, 170, 1247–1257.e1212. [Google Scholar] [CrossRef] [Green Version]
- Mileykovskaya, E.; Penczek, P.A.; Fang, J.; Mallampalli, V.K.; Sparagna, G.C.; Dowhan, W. Arrangement of the respiratory chain complexes in Saccharomyces cerevisiae supercomplex III2IV2 revealed by single particle cryo-electron microscopy. J. Biol. Chem. 2012, 287, 23095–23103. [Google Scholar] [CrossRef] [Green Version]
- Dudkina, N.V.; Kudryashev, M.; Stahlberg, H.; Boekema, E.J. Interaction of complexes I, III, and IV within the bovine respirasome by single particle cryoelectron tomography. Proc. Natl. Acad. Sci. USA 2011, 108, 15196–15200. [Google Scholar] [CrossRef] [Green Version]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sedlák, E.; Kožár, T.; Musatov, A. The Interplay among Subunit Composition, Cardiolipin Content, and Aggregation State of Bovine Heart Cytochrome c Oxidase. Cells 2020, 9, 2588. https://doi.org/10.3390/cells9122588
Sedlák E, Kožár T, Musatov A. The Interplay among Subunit Composition, Cardiolipin Content, and Aggregation State of Bovine Heart Cytochrome c Oxidase. Cells. 2020; 9(12):2588. https://doi.org/10.3390/cells9122588
Chicago/Turabian StyleSedlák, Erik, Tibor Kožár, and Andrey Musatov. 2020. "The Interplay among Subunit Composition, Cardiolipin Content, and Aggregation State of Bovine Heart Cytochrome c Oxidase" Cells 9, no. 12: 2588. https://doi.org/10.3390/cells9122588




