Structure and Function of Human Matrix Metalloproteinases
Abstract
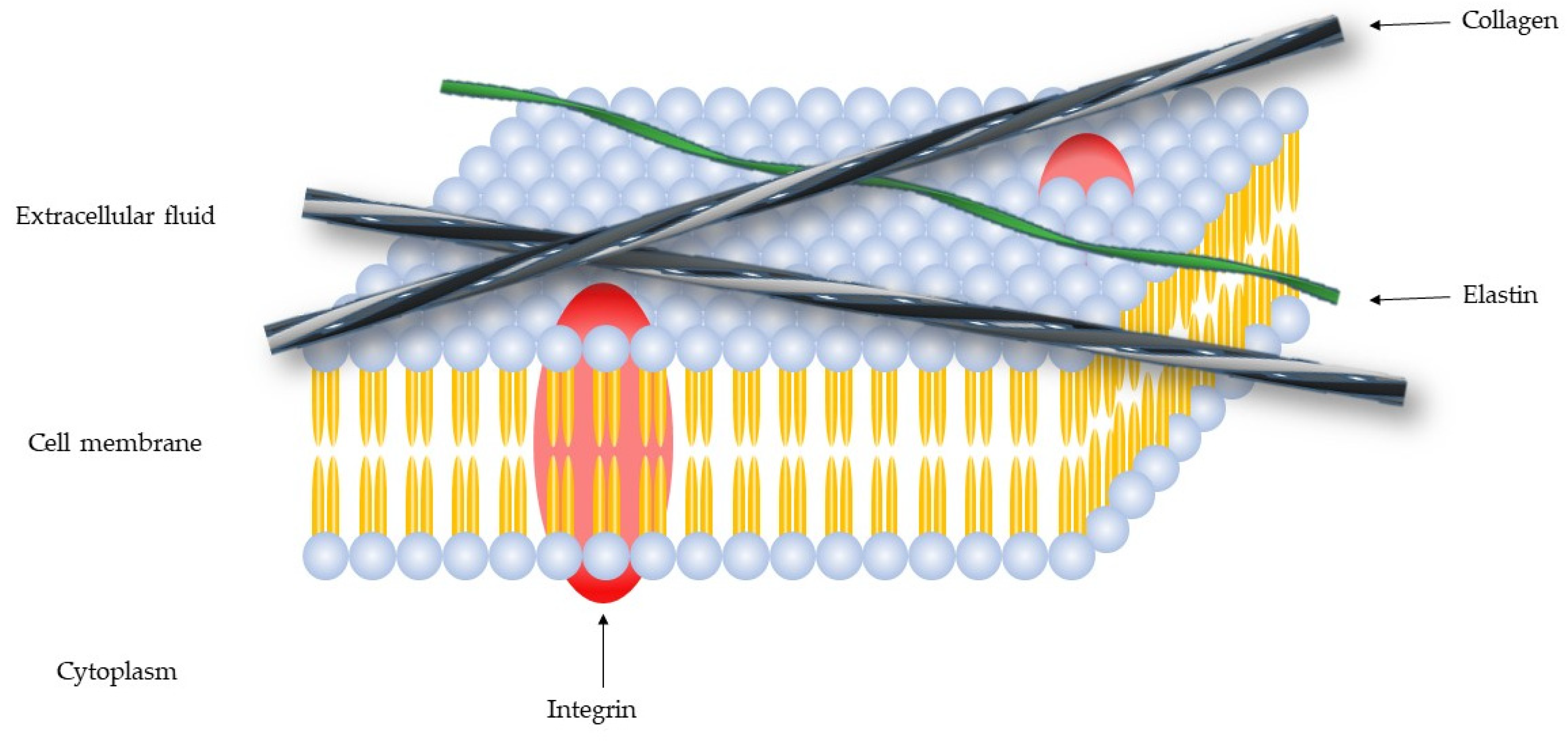
:1. Extracellular Matrix—Collagen
2. Metzincs Superfamily—Matrix Metalloproteinases (MMPs)
3. MMPs Functions
4. Types of MMPs
- Non-furin regulated MMPs (MMP-1, -3, -7, -8, -10, -12, -13, -20, and -27);
- MMPs bearing three fibronectin-like inserts in the catalytic domain (MMP-2 and -9);
- MMPs anchored to the cellular membrane by a C-terminal glycosylphosphatidylinositol (GPI) moiety (MMP-11, -17 and -25);
- MMPs bearing a transmembrane domain (MMP-14, -15, -16, and -24) and
- All the other MMPs (MMP-19, -21, -23, -26 and -28).
5. Structure
- 1-
- A signal N-terminal peptide with variable length, that targets the peptide for secretion;
- 2-
- A pro-domain (with about 80 aa), which keeps MMP inactive and is removed when the enzyme is proteolytically activated;
- 3-
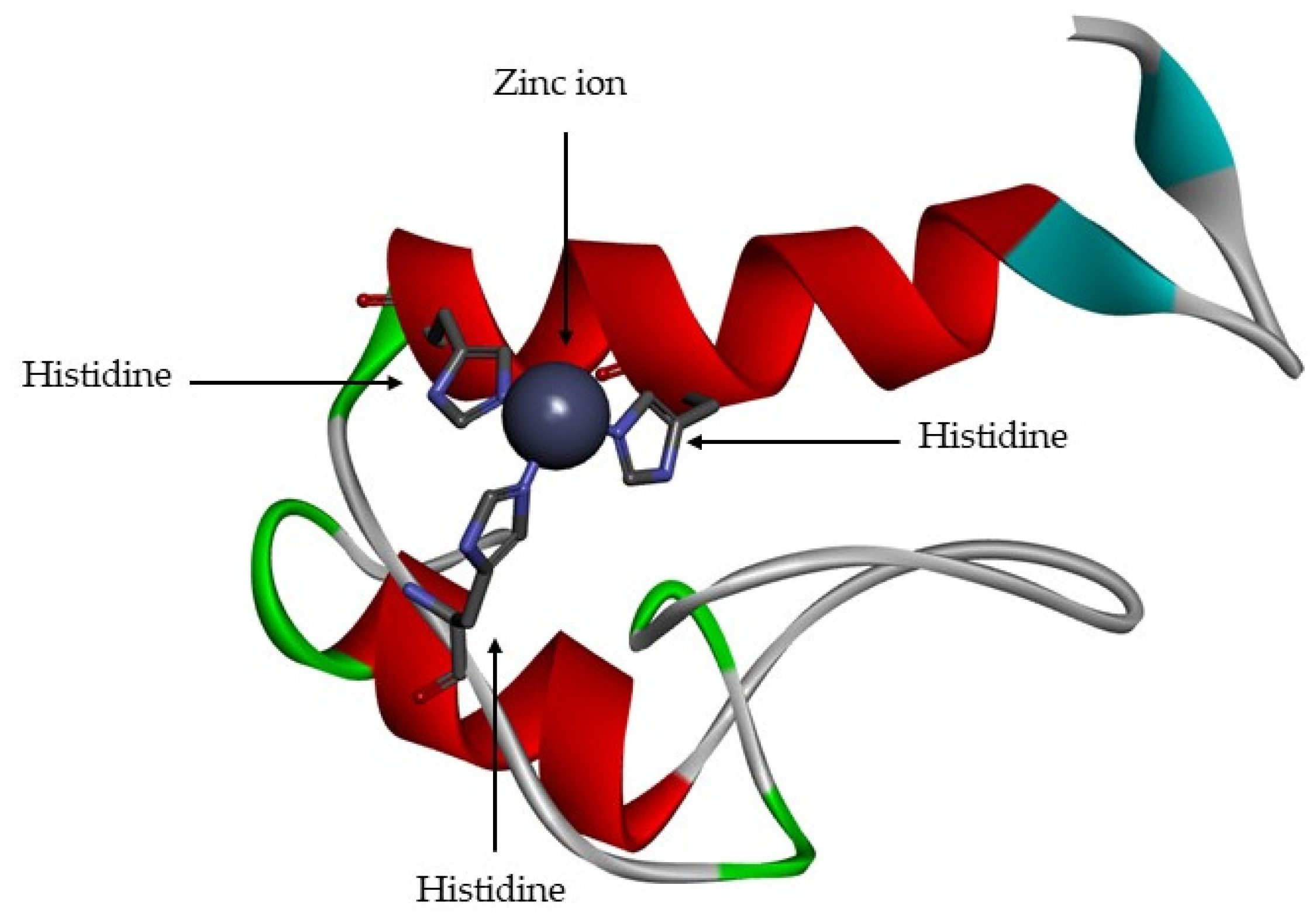
- A catalytic domain (with about 160 aa), with a zinc ion, that consists of five β-sheets, three α-helixes and three calcium ions;
- 4-
- A linker of variable length (14–69 aa), which links the catalytic domain to hemopexin-like domain—“hinge region”;
- 5-
- A hemopexin-like domain (with about 210 aa) that is characterized by four β-propeller and
- 6-
- An additional transmembrane domain with the small cytoplasmatic C-terminal domain, only present in MMPs-14, -15, -16 and -24.
S1′ Pocket Selectivity
6. MMP Activity Regulation
7. Catalytic Mechanism
8. Conclusions
Funding
Conflicts of Interest
Appendix A
| MMP | Name | Substrate | Production | Diseases | Other Information |
|---|---|---|---|---|---|
| 1 | Collagenase-1; Interstitial or Fibroblast collagenase | Collagen type I, II, III, VII, VIII, X and XI, gelatin, entactin, tenascin, aggrecan, fibronectin, vitronectin, myelin basic protein, ovostatin, casein | Sensitive to oxidative stress. Cells: fibroblasts, keratinocytes, endothelial cells, macrophages, hepatocytes, chondrocytes, platelets and osteoblasts. | Rheumatoid arthritis, atherosclerosis, pulmonary emphysema, fibrosis, autoimmune disease, wound healing and cancer | Identified in 1962. MMP-1 cleaves pro-MMP-2 and -9 into its active form. MMP-1 expression is increased by inflammatory cytokines (TNF-α and IL-1). |
| 8 | Collagenase-2; Neutrophil collagenase | Collagen type I, II and III, fibronectin, aggrecan and ovostatin. | Cells: chondrocytes, endothelial cells, activated macrophages and smooth muscle cells. | Rheumatoid arthritis, asthma, wound healing, periodontitis and cancer. | Identified in 1990 and it was discovered in cDNA library constructed from mRNA extracted from peripheral leukocytes of a patient with chronic granulocytic leukemia. MMP-8 is secreted by pro-MMP-8 form and it is activated by MMP-3 and -10 |
| 13 | Collagenase-3 | Collagen type I, II, III, IV, IX, X and XIV, tenascin C isoform, fibronectin, laminin, aggrecan core protein, gelatin, plasminogen, osteonectin, casein, fibrillin-1 and serine proteinases inhibitors | Connective tissue (cartilage and developing bone) Cells: epithelial and neuronal cells. | Osteoarthritis, lung diseases (lung injury, viral infection and chronic obstructive pulmonary disease), liver fibroses, cancer and metastasis | MMP-13 have gelatinolytic activity. MMP-13 active the pro-MMP-2 and -9. |
| 18 | Collagenase-4 | Collagen and gelatin | Organs: mammary glands, placenta, lung, pancreas, ovary, intestine, spleen, thymus, prostate, colon and heart | MMP-18 has not been directly linked to a specific pathological condition. | Identified in 1990s, in sequence similarity studies. Show closest identity with MMPs-1, -3, -10 and -11. MMP-18 differs from other MMPs in its amino acids sequence contains two cleavage sites for activation. |
| MMP | Name | Substrate | Production | Diseases | Other Information |
|---|---|---|---|---|---|
| 2 | Gelatinase A | Collagen typeI, III, IV, V, VII and X, gelatin, some glycoprotein of ECM, fibronectin, laminin, aggrecan, elastin, tenascin, myelin basic protein and vitronectin | Cells: dermal fibroblasts, keratinocytes, endothelial cells, chondrocytes, osteoblasts, leukocytes, platelets and monocytes | Promotion and inhibition of inflammation, asthma, fibrosis, cardiovascular diseases and cancer | MMP-2 expression is constitutive and TNF-α and -β stimulate its production, but IFN-τ suppresses its production. |
| 9 | Gelatinase B | Collagen type IV, V, and XI, cytokines, elastin, aggrecan, decorin, laminin, entactin, myelin basic protein, casein, chemokines, IL-8 and IL-1β | Cells: neutrophils, macrophages, polymorphonuclear leucocytes, osteoblasts, epithelial cells, fibroblasts, dendritic cells, granulocytes, T-cells and keratinocytes. | Cardiovascular diseases, inflammation and esophageal cancer | Identified such as neutrophil in 1974. MMP-9 has a strongly O-glycosylated collagen type V insert |
| MMP | Name | Substrate | Production | Diseases | Other Information |
|---|---|---|---|---|---|
| 3 | Stromelysin-1 | Collagen type I, II, III, IV, V, X and IX, fibronectin, gelatin, laminin, aggrecan, vitronectin, entactin, tenascin, decorin, myelin basic protein, ovostatin, casein, osteonectin elastin and proteoglycans. | Cells: fibroblasts and platelets MMP-3 has been detected in the nucleus, and it may function as a trans-regulator of connective tissue growth factor. | Arthritis, osteoarthritis, asthma, aneurism, atherosclerosis, coronary artery diseases, periodontitis, wound healing, Alzheimer diseases and cancer. | Identified in 1985. MMP-3 is secreted as inactive enzyme and actives pro-MMP-1, pro-MMP-13 and gelatinases. MMP-3 retains protease capability regardless of metal center, but this replace leads to sensitivity changes for different substrates. MMP-3 has a unique deep active site that transverses the length of the enzyme. |
| 10 | Stromelysin-2 | Collagen type III, IV, V, IX and X, proteoglycans, gelatin, fibronectin, laminin, elastin, aggrecan, cassein and fibrilin-10 | Cells: keratinocytes, macrophages and epithelium | Wound healing, arthritis, fibrosis, idiopathic pulmonary fibrosis, peripheral arterial disease and cancer. | MMP-10 has 82% MMP-3 homologous sequence. MMP-10 is secreted as pro-MMP-10. Actives others pro-MMPs, such as pro-collagenases. MMP-10 plays a role in liver regeneration. |
| 11 | - | No protein of major relevance to ECM can be degraded by MMP-11 but it degrades laminin receptor and serine proteinases inhibitors, α1-proteinases and α1-antitrypsin inhibitors. | Cells: fibroblasts Organs: uterus, placenta and mammary glands | Would healing, progression of epithelial malignancies and cancer | Identified in 1990, in stromal cells surrounding invasive breast carcinoma.MMP-11 is activated intracellularly by furin, since it has a furin recognition sequence (RXRXKR) and secreted in active form. |
| MMP | Name | Substrate | Production | Diseases | Other Information |
|---|---|---|---|---|---|
| 7 | - | Faz-ligand, pro-TNF-α, E-cadherin, syndecan-1, fibronectin, laminin, elastin, casein, gelatin type I, II, IV and V, collagen type I and IVm vitronectin, entactin, tenascin, aggrecan, myelin, and proteoglycans | Cells: epithelia cells, mammalian glands, liver, pancreas, prostate and skin | Idiopathic pulmonary fibrosis, cancer, metastasis and inflammatory processes. | MMP-7 was described such as putative uterine metalloprotease- 1 in 1988. MMP-7 acts intracellularly in the intestine to process procryptidins to bactericidal forms. |
| 26 | Matrilysin-2 or endometase | (in vitro): collagen type IV, fibronectin, fibrinogen, gelatin, vitronectin, α1-antipripsin, β-casein, α2-macroglobulin and IGFBP-1 | Cancer cells of epithelial origin | Carcinomas of the lung, prostate and breast, angiogenesis and tumor progression. | MMP-26 contains a signal sequence for secretion and a prodomain with an unusual cysteine switch for latency preservation. Active pro-MMP-9. MMP-26 is negatively regulated by TIMP-2 and -4, with TIMP-4 being more potent inhibitor. |
| MMP | Name | Substrate | Production | Diseases | Other Information |
|---|---|---|---|---|---|
| 14 | MT1-MMP (Membrane-type) | Collagen type I, II and III; gelatin, fibronectin, laminin-1, vitronectin, cartilage proteoglycans, fibrilin-1, tenascin, entactin, aggrecan, α1-proteinase inhibitor and α2-macroglobulin. | Cells: fibroblasts, platelets and osteoblasts | Cancer | Identified in 1994. MMP-14 actives the MMP-2, -8 and -13 latent forms. MMP-14 activates pro-MMP-13 on the cell surface. |
| 15 | MT2-MMP (Membrane-type) | Laminin, fibronectin, entactin, aggrecan, gelatin, vibronectin and tenascin. | Organs: placenta, heart and brain | Cancer (glioblastoma, ovarian and breast carcinoma) | Identified in 1995. MMP-15 can active MMP-2 and -13 latent forms. |
| 16 | MT3-MMP (Membrane-type) | Gelatin, casein, collagen type III, laminin and fibronectin. | Organs: lungs, placenta, kidney, ovaries, intestine, prostate, spleen, heart and skeletal muscle Cells: cardiomyocytes progenitor cells | Tumor invasion | Identified in 1997. MMP-16 can activate MMP-2 and -9. |
| 17 | MT4-MMP (GPI-anchored) | Gelatin, fibrinogen and fibrin | Cells: leucocytes Organs: brain, colon, ovaries and testicles | Inflammatory processes, cancer and tumor progression. | In the mid-1990s, MMP-17 was cloned from a human breast carcinoma cDNA library. ADAMTS-4 activator. MMP-17 cannot active pro-MMP-2. |
| 24 | MT5-MMP (Membrane-type) | Fibronectin, gelatin and proteoglycans | Organs: brain, kidney, pancreas and lung | Brain tumor (astrocytomas and glioblastomas) and tumor progression and angiogenesis. | Identified in 1999 and cloned from a human brain cDNA library. MMP-24 can activate MMP-2 latent form. MMP-24 is neuro-specific and contribute to neuronal circuit formation and plasticity. It has a role in the development of dermal neuro-immune synapses. |
| 25 | MT6-MMP (GPI-anchored) | Collagen type IV, fibronectin, gelatin and proteoglycans. | Cells: leucocytes and cancer tissue Organs: testicles, kidney and skeletal muscle | Cancer | The stem region contains three cysteine residues which may contribute to dimerization by inter- and intramolecular disulfide bond. MMP-25 cannot degrade laminin-1. MMP-25 actives pro-MMP-2, but differently than the other MT-MMPS. |
| MMP | Name | Substrate | Production | Diseases | Other Information |
|---|---|---|---|---|---|
| 12 | Macrophage metalloelastase | Gelatin type I, elastin, fibronectin, laminin, vitronectin, proteoglycans, elastin, collagen type I, IV and V, entactin, osteonectin, aggrecan, myelin, fibrinogen and α1-antitripsin | Cells: chondrocytes, macrophages and other stromal cells, osteoblasts. Organs: placenta | Chronic pulmonary disease, atherosclerosis, emphysema and lung cancer. | MMP-12 may affect the blood-brain barrier after cerebral ischemia. |
| 19 | RASI-1 or stromelysin-4 | Collagen type I and IV, laminin and nidogen, tenascin-C isoform, entactin, aggrecan, fibronectin and gelatin type I, in vitro | Cells: leucocytes Organs: colon, intestine, ovary, testis, prostate, thymus, spleen, pancreas, kidney, skeletal muscle, liver, lung, placenta, brain and heart | Wound healing and arthritic disease. | The MMP-19 can activate pro-MMP-9, but cannot activate other latent forms (MMP-1, -2, -3, -13 and -14, in vitro) |
| 20 | Enamelysin | Ameloblasts, aggrecan, odontoblasts and amelogenin | Organs: dental tissue (enamel) | Tooth development | MMP-20 is a tooth-specific MMP expressed in newly formed tooth enamel. MMP-20 contains a very basic hinge region compared to the hinge region of stromelysins (hydrophobic) or MMP-19 (acidic) |
| 21 | Xenopus-MMP | - | Cells: leucocytes, macrophages, fibroblasts, basal and squamous cell Organs: ovary, kidney, lung, placenta, intestine, neuroectoderm, skin and brain. | Embryogenesis, pancreatic cancer and tumor progression | - |
| 22 | Chicken-MMP | - | - | - | MMP-22 catalytic domain is closely related to stromelysin-3. |
| 23 | Cysteine array (CA)-MMP | Gelatin | Organs: ovary, testicles and prostate | - | MMP-23 lacks a signal sequence, it has a short pro-domain and the C-terminal domain is considerable shortened and shows no sequence similarity to hemopexin.MMP-23 is the only one that lacks the hemopexin domain, having a cysteine rich immunoglobulin-like domain.MMP-23 lacks the cysteine switch motif in propeptide. |
| 27 | - | Gelatin | Cells: B-lymphocytes Organs: testicles, intestine, lung and skin. | Ovarian or peritoneal endometriotic lesions, breast cancer development and tumor progression. | MMP-27 is classified as stromelysin and holds 51.6% structural homology with MMP-10. |
| 28 | Epilysin | Casein | Cells: basal keratinocytes Organs: epidermis. High levels- testis. Low levels-lungs heart, intestine, colon, placenta and brain. | Tissue homeostasis and repair, osteoarthritis and rheumatoid arthritis. | - |
References
- Cui, N.; Hu, M.; Khalil, R.A. Biochemical and Biological Attributes of Matrix Metalloproteinases. Prog. Mol. Biol. Transl. Sci. 2017, 147, 1–73. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, J.; Khalil, R.A. Matrix Metalloproteinase Inhibitors as Investigational and Therapeutic Tools in Unrestrained Tissue Remodeling and Pathological Disorders. Prog. Mol. Biol. Transl. Sci. 2017, 148, 355–420. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhu, J.; Hoop, C.L.; Case, D.A.; Baum, J. Cryptic binding sites become accessible through surface reconstruction of the type I collagen fibril. Sci. Rep. 2018, 8, 16646. [Google Scholar] [CrossRef] [PubMed]
- Chung, L.; Dinakarpandian, D.; Yoshida, N.; Lauer-Fields, J.L.; Fields, G.B.; Visse, R.; Nagase, H. Collagenase unwinds triple-helical collagen prior to peptide bond hydrolysis. EMBO J. 2004, 23, 3020–3030. [Google Scholar] [CrossRef]
- Ricard-Blum, S. The collagen family. Cold Spring Harb. Perspect. Biol. 2011, 3, a004978. [Google Scholar] [CrossRef] [Green Version]
- Fischer, T.; Senn, N.; Riedl, R. Design and Structural Evolution of Matrix Metalloproteinase Inhibitors. Chemistry 2019, 25, 7960–7980. [Google Scholar] [CrossRef]
- Yu, Z.; Visse, R.; Inouye, M.; Nagase, H.; Brodsky, B. Defining requirements for collagenase cleavage in collagen type III using a bacterial collagen system. J. Biol. Chem. 2012, 287, 22988–22997. [Google Scholar] [CrossRef] [Green Version]
- Nagase, H.; Visse, R.; Murphy, G. Structure and function of matrix metalloproteinases and TIMPs. Cardiovasc. Res. 2006, 69, 562–573. [Google Scholar] [CrossRef] [Green Version]
- Amălinei, C.; Căruntu, I.D.; Bălan, R.A. Biology of metalloproteinases. Rom. J. Morphol. Embryol. 2007, 48, 323–334. [Google Scholar]
- Goodwin, L. A Closer Look At Metalloproteinases; Nova Science Publisher Inc.: Hauppauge, NY, USA, 2019; p. 310. [Google Scholar]
- Klein, T.; Bischoff, R. Physiology and pathophysiology of matrix metalloproteases. Amino Acids 2011, 41, 271–290. [Google Scholar] [CrossRef] [Green Version]
- Visse, R.; Nagase, H. Matrix metalloproteinases and tissue inhibitors of metalloproteinases: Structure, function, and biochemistry. Circ. Res. 2003, 92, 827–839. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maskos, K. Crystal structures of MMPs in complex with physiological and pharmacological inhibitors. Biochimie 2005, 87, 249–263. [Google Scholar] [CrossRef] [PubMed]
- Tallant, C.; Marrero, A.; Gomis-Rüth, F.X. Matrix metalloproteinases: Fold and function of their catalytic domains. Biochim. Biophys. Acta 2010, 1803, 20–28. [Google Scholar] [CrossRef] [PubMed]
- Jacobsen, J.A.; Major Jourden, J.L.; Miller, M.T.; Cohen, S.M. To bind zinc or not to bind zinc: An examination of innovative approaches to improved metalloproteinase inhibition. Biochim. Biophys. Acta 2010, 1803, 72–94. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Verma, R.P.; Hansch, C. Matrix metalloproteinases (MMPs): Chemical-biological functions and (Q)SARs. Bioorg. Med. Chem. 2007, 15, 2223–2268. [Google Scholar] [CrossRef] [PubMed]
- Murphy, G.; Nagase, H. Progress in matrix metalloproteinase research. Mol. Aspects Med. 2008, 29, 290–308. [Google Scholar] [CrossRef] [Green Version]
- Mannello, F.; Medda, V. Nuclear localization of matrix metalloproteinases. Prog. Histochem. Cytochem. 2012, 47, 27–58. [Google Scholar] [CrossRef]
- Rangasamy, L.; Geronimo, B.D.; Ortín, I.; Coderch, C.; Zapico, J.M.; Ramos, A.; de Pascual-Teresa, B. Molecular Imaging Probes Based on Matrix Metalloproteinase Inhibitors (MMPIs). Molecules 2019, 24, E2982. [Google Scholar] [CrossRef] [Green Version]
- Gersh, I.; Catchpole, H.R. The organization of ground substance and basement membrane and its significance in tissue injury disease and growth. Am. J. Anat. 1949, 85, 457–521. [Google Scholar] [CrossRef]
- Whittaker, M.; Floyd, C.D.; Brown, P.; Gearing, A.J. Design and therapeutic application of matrix metalloproteinase inhibitors. Chem. Rev. 1999, 99, 2735–2776. [Google Scholar] [CrossRef]
- Gross, J.; Lapiere, C.M. Collagenolytic activity in amphibian tissues: A tissue culture assay. Proc. Natl. Acad. Sci. USA 1962, 48, 1014–1022. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cerofolini, L.; Fragai, M.; Luchinat, C. Mechanism and Inhibition of Matrix Metalloproteinases. Curr. Med. Chem. 2019, 26, 2609–2633. [Google Scholar] [CrossRef] [PubMed]
- Tokuhara, C.K.; Santesso, M.R.; Oliveira, G.S.N.; Ventura, T.M.D.S.; Doyama, J.T.; Zambuzzi, W.F.; Oliveira, R.C. Updating the role of matrix metalloproteinases in mineralized tissue and related diseases. J. Appl. Oral Sci. 2019, 27, e20180596. [Google Scholar] [CrossRef] [PubMed]
- Vandenbroucke, R.E.; Dejonckheere, E.; Libert, C. A therapeutic role for matrix metalloproteinase inhibitors in lung diseases? Eur. Respir. J. 2011, 38, 1200–1214. [Google Scholar] [CrossRef]
- Huang, H. Matrix Metalloproteinase-9 (MMP-9) as a Cancer Biomarker and MMP-9 Biosensors: Recent Advances. Sensors 2018, 18, E3249. [Google Scholar] [CrossRef] [Green Version]
- Zajkowska, M.; Zbucka-Krętowska, M.; Sidorkiewicz, I.; Lubowicka, E.; Będkowska, G.E.; Gacuta, E.; Szmitkowski, M.; Ławicki, S. Human Plasma Levels of Vascular Endothelial Growth Factor, Matrix Metalloproteinase 9, and Tissue Inhibitor of Matrix Metalloproteinase 1 and Their Applicability as Tumor Markers in Diagnoses of Cervical Cancer Based on ROC Analysis. Cancer Control 2018, 25, 1073274818789357. [Google Scholar] [CrossRef] [Green Version]
- Blanco-Prieto, S.; Barcia-Castro, L.; Páez de la Cadena, M.; Rodríguez-Berrocal, F.J.; Vázquez-Iglesias, L.; Botana-Rial, M.I.; Fernández-Villar, A.; De Chiara, L. Relevance of matrix metalloproteases in non-small cell lung cancer diagnosis. BMC Cancer 2017, 17, 823. [Google Scholar] [CrossRef] [Green Version]
- Silvello, D.; Narvaes, L.B.; Albuquerque, L.C.; Forgiarini, L.F.; Meurer, L.; Martinelli, N.C.; Andrades, M.E.; Clausell, N.; dos Santos, K.G.; Rohde, L.E. Serum levels and polymorphisms of matrix metalloproteinases (MMPs) in carotid artery atherosclerosis: Higher MMP-9 levels are associated with plaque vulnerability. Biomarkers 2014, 19, 49–55. [Google Scholar] [CrossRef]
- Goncalves, I.; Bengtsson, E.; Colhoun, H.M.; Shore, A.C.; Palombo, C.; Natali, A.; Edsfeldt, A.; Dunér, P.; Fredrikson, G.N.; Björkbacka, H.; et al. Elevated Plasma Levels of MMP-12 Are Associated With Atherosclerotic Burden and Symptomatic Cardiovascular Disease in Subjects With Type 2 Diabetes. Arterioscler. Thromb. Vasc. Biol. 2015, 35, 1723–1731. [Google Scholar] [CrossRef] [Green Version]
- Young, D.; Das, N.; Anowai, A.; Dufour, A. Matrix Metalloproteases as Influencers of the Cells’ Social Media. Int. J. Mol. Sci. 2019, 20, E3847. [Google Scholar] [CrossRef] [Green Version]
- Kwan, J.A.; Schulze, C.J.; Wang, W.; Leon, H.; Sariahmetoglu, M.; Sung, M.; Sawicka, J.; Sims, D.E.; Sawicki, G.; Schulz, R. Matrix metalloproteinase-2 (MMP-2) is present in the nucleus of cardiac myocytes and is capable of cleaving poly (ADP-ribose) polymerase (PARP) in vitro. FASEB J. 2004, 18, 690–692. [Google Scholar] [CrossRef] [PubMed]
- Lovejoy, B.; Cleasby, A.; Hassell, A.M.; Longley, K.; Luther, M.A.; Weigl, D.; McGeehan, G.; McElroy, A.B.; Drewry, D.; Lambert, M.H. Structure of the catalytic domain of fibroblast collagenase complexed with an inhibitor. Science 1994, 263, 375–377. [Google Scholar] [CrossRef] [PubMed]
- Tsukada, H.; Pourmotabbed, T. Unexpected crucial role of residue 272 in substrate specificity of fibroblast collagenase. J. Biol. Chem. 2002, 277, 27378–27384. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Knäuper, V.; Docherty, A.J.; Smith, B.; Tschesche, H.; Murphy, G. Analysis of the contribution of the hinge region of human neutrophil collagenase (HNC, MMP-8) to stability and collagenolytic activity by alanine scanning mutagenesis. FEBS Lett. 1997, 405, 60–64. [Google Scholar] [CrossRef] [Green Version]
- Gimeno, A.; Beltrán-Debón, R.; Mulero, M.; Pujadas, G.; Garcia-Vallvé, S. Understanding the variability of the S1’ pocket to improve matrix metalloproteinase inhibitor selectivity profiles. Drug Discov. Today 2019, 25, 38–57. [Google Scholar] [CrossRef] [PubMed]
- Bar-Or, A.; Nuttall, R.K.; Duddy, M.; Alter, A.; Kim, H.J.; Ifergan, I.; Pennington, C.J.; Bourgoin, P.; Edwards, D.R.; Yong, V.W. Analyses of all matrix metalloproteinase members in leukocytes emphasize monocytes as major inflammatory mediators in multiple sclerosis. Brain 2003, 126, 2738–2749. [Google Scholar] [CrossRef] [Green Version]










| Pathologies | Diseases |
|---|---|
| Tissue destruction | Cancer invasion and metastasis |
| Arthritis | |
| Ulcers | |
| Periodontal diseases | |
| Brain degenerative diseases | |
| Fibroses | Liver cirrhosis |
| Fibrotic lung disease | |
| Otosclerosis | |
| Atherosclerosis | |
| Multiple sclerosis | |
| Weakening of matrix | Dilated cardiomyopathy |
| Aortic aneurysm | |
| Varicose veins |
| Domain | Presence |
|---|---|
| Signal Peptide | All MMPs |
| Pro-domain | All MMPs |
| Catalytic | All MMPs |
| Hemopexin-like | All MMPs, except in MMP-7, -23, and -26 |
| Fibronectin | Only MMP-2 and -9 |
| Vitronectin insert | Only MMP-21 |
| Type I transmembrane | Only MMP-14, -15, -16, and -24 |
| Cytoplasmic | Only MMP-14, -15, -16, and -24 |
| GPI anchor | Only MMP-17 and -25 |
| Cysteine Array Region | Only MMP-23 |
| IgG-like domain | Only MMP-23 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Laronha, H.; Caldeira, J. Structure and Function of Human Matrix Metalloproteinases. Cells 2020, 9, 1076. https://doi.org/10.3390/cells9051076
Laronha H, Caldeira J. Structure and Function of Human Matrix Metalloproteinases. Cells. 2020; 9(5):1076. https://doi.org/10.3390/cells9051076
Chicago/Turabian StyleLaronha, Helena, and Jorge Caldeira. 2020. "Structure and Function of Human Matrix Metalloproteinases" Cells 9, no. 5: 1076. https://doi.org/10.3390/cells9051076
APA StyleLaronha, H., & Caldeira, J. (2020). Structure and Function of Human Matrix Metalloproteinases. Cells, 9(5), 1076. https://doi.org/10.3390/cells9051076






