Abstract
Glucagon-like peptide-1 (GLP-1) has been shown to potentiate glucose-stimulated insulin secretion binding GLP-1 receptor on pancreatic β cells. β-arrestin 1 (βARR1) is known to regulate the desensitization of GLP-1 receptor. Mounting evidence indicates that microRNAs (miRNAs, miRs) are fundamental in the regulation of β cell function and insulin release. However, the regulation of GLP-1/βARR1 pathways by miRs has never been explored. Our hypothesis is that specific miRs can modulate the GLP-1/βARR1 axis in β cells. To test this hypothesis, we applied a bioinformatic approach to detect miRs that could target βARR1; we identified hsa-miR-7-5p (miR-7) and we validated the specific interaction of this miR with βARR1. Then, we verified that GLP-1 was indeed able to regulate the transcription of miR-7 and βARR1, and that miR-7 significantly regulated GLP-1-induced insulin release and cyclic AMP (cAMP) production in β cells. Taken together, our findings indicate, for the first time, that miR-7 plays a functional role in the regulation of GLP-1-mediated insulin release by targeting βARR1. These results have a decisive clinical impact given the importance of drugs modulating GLP-1 signaling in the treatment of patients with type 2 diabetes mellitus.
1. Introduction
The incretin glucagon-like peptide-1 (GLP-1) is a polypeptide hormone produced mainly in entero-endocrine L cells of the gut known to potentiate glucose-stimulated insulin secretion (GSIS) in pancreatic β cells [1,2]. GLP-1 action is mediated by GLP-1 receptors, a member of the seven-transmembrane family of G protein-coupled receptors (GPCRs) [3,4,5,6].
MicroRNAs (miRNAs, miRs) are small non-coding single-stranded ribonucleic acids (RNAs), highly conserved from plants to mammals, which are able to enhance messenger RNA (mRNA) degradation and/or inhibit protein translation by binding to the 3′-untranslated regions (UTRs) of target mRNAs [7,8,9,10,11,12,13]. They play crucial regulatory roles in a variety of biological processes, including regulation of differentiation, development, and function of β cells [14,15,16]. Specifically, recent studies have identified a number of miRs involved in the regulation of insulin release [17,18,19]. In terms of therapeutic potential, miRs represent a novel and very appealing strategy to manipulate metabolic processes as their activity can be efficiently modulated with RNA-based technologies [20,21,22].
Experimental evidence has recently shown that β-arrestin 1 (βARR1), a protein known to be involved in the regulation of signal transduction of GPCRs [23,24,25,26,27,28,29], plays a key role in the desensitization of GLP-1 receptor in β cells [30]. Therefore, targeting βARR1 by miR intervention could be a promising strategy for the treatment of diabetes mellitus. The aim of the present study was to identify a miR that targets βARR1 to modulate β cell function. A bioinformatic screen resulted in the identification of hsa-miR-7-5p (briefly noted as miR-7 in the rest of the paper) as potentially capable of repressing βARR1 mRNA expression. In our biological validation of this screen, miR-7 was found to actually target βARR1 and to be upregulated in β cells after GLP-1 stimulation. The mechanistic role of miR-7 was further confirmed by assessing the regulation of insulin release in β cells.
2. Materials and Methods
2.1. Cell Culture and Reagents
INS-1 β cells were cultured in a humidified atmosphere (37 °C) containing 5% CO2 in RPMI-1640 medium, and insulin secretion was assessed as we previously described and validated [31,32,33,34]. All experiments were performed using INS-1 β cells between the 20th and 40th passage. All reagents were from Millipore-Sigma (Burlington, MA, USA), unless otherwise stated.
2.2. Insulin Secretion
Insulin release in response to different stimuli was assessed using a commercially available enzyme-linked immunosorbent assay, following the manufacturer’s instructions (Mercodia, Uppsala, Sweden).
2.3. Cyclic AMP (cAMP) Assay
Intracellular cAMP content was measured by using the cAMP enzyme immunoassay kit (Enzo Life Sciences, Farmingdale, NY, USA), according to the manufacturer’s instructions; final cAMP concentrations per well were normalized by total protein as described.
2.4. Identification of miR-7 as a Regulator of βARR1
To identify miRs targeting the 3′-UTR of βARR1, we used online target prediction tools, including miRWalk-3 and Targetscan version 7.2, as we previously described [35,36]. These programs predict biological targets of miRs by searching for the presence of conserved 8mer and 7mer sites that match the seed region of miRs.
2.5. Biological Validation of miR-7 as a Regulator of βARR1
To assess the actual effects of miR-7 on βARR1 gene transcription, we used a luciferase reporter containing the 3’-UTR segment of the predicted miR interaction sites, both wild-type and mutated, in INS-1 cells. The mutant construct of βARR1 3′-UTR, carrying a substitution of two nucleotides within the predicted miR-7 binding sites of βARR1 3′-UTR (see Figure 1A) was obtained using a commercially available site-directed mutagenesis kit (New England Biolabs, Ipswich, MA, USA), as we described [35]. Using Lipofectamine RNAiMAX (ThermoFisher Scientific, Waltham, MA, USA), cells were transfected with the 3′-UTR reporter plasmid (0.05 μg) and miR-7 mirVanaTM (50 nM) mimics or inhibitors (ThermoFisher Scientific) as well as a non-targeting negative control (scramble), according to the manufacturer’s instructions, as described [35]. Forty-eight hours after transfection, Firefly and Renilla luciferase activities were assessed using a commercially available Luciferase Reporter Assay System (Promega, Madison, WI, USA). Firefly luciferase was normalized to Renilla luciferase activity. Levels of miR-7 were measured using individual TaqMan miRNA assays, according to the manufacturer’s instructions; miR expression was normalized to the level of U6; standard TaqMan gene expression assays from Applied Biosystem were used, as we previously described and validated [35,36,37]. Cellular expression of βARR1 was determined by RT-qPCR, as we previously described [32,33,37], normalizing to endogenous glyceraldehyde 3-phosphate dehydrogenase (GAPDH). Sequences of oligonucleotide primers (Merck KGaA, Darmstadt, Germany) are presented in Supplementary Table S1.

Figure 1.
Identification of miR-7 as modulator of βARR1. (A) Complementary nucleotides between the target region of βARR1 3′-UTR (in yellow) and hsa-miR-7-5p (miR-7) are conserved across different species. Luciferase activity was measured 48 h after transfection, using the vector without βARR1 3′-UTR (empty vector), the vector containing the wild-type βARR1 3′-UTR, and the vector containing a mutated βARR1 3′-UTR (βARR1 MUT); (B) A non-targeting miR (scramble) has been employed as further control. Means ± S.E.M. are shown alongside actual values; * p < 0.05.
2.6. Immunoblotting
Immunoblotting assays were performed, as previously described and validated by our group described [36,37,38,39,40] and developed with the Odyssey system (LI-COR Biosciences, Lincoln, NE, USA). The intensity of the bands was quantified by using the FIJI (Fiji Is Just ImageJ) software. Antibodies were purchased from Cell Signaling Technology (Danvers, MA, USA): CREB (catalog number: #4820), Phospho-CREB (pCREB Ser133; catalog number: #9198), ERK1/2 (catalog number: #9102); and from Santa Cruz Biotechnology (Dallas, TX, USA): p-ERK Antibody (catalog number: #sc-7383).
2.7. Statistical Analysis
Data are expressed as means ± standard error of means (SEM). Statistical analyses were performed in Prism (GraphPad Software, Version 8.0; Prism, San Diego, CA, USA). Statistical significance was tested using the nonparametric Mann–Whitney U test or two-way ANOVA followed by Tukey–Kramer multiple comparison test, as appropriate. Significant differences were established at a p-value < 0.05.
3. Results
3.1. βARR1 Is a Molecular Target of miR-7
Through bioinformatic analyses, we identified miR-7 as a potential regulator of βARR1. We selected miR-7 because it had been previously described as a key player in beta cell physiology. Specifically, miR-7 is considered to be a prototypical neuroendocrine miR, being highly expressed in neuroendocrine organs, including the endocrine pancreas and the pituitary and adrenal glands [41,42,43,44]. Moreover, the complementary nucleotides between the target region of βARR1 3’ untranslated region (3′-UTR) and miR-7 are evolutionarily highly conserved across different species, including humans, non-human primates, and rodents (Figure 1A). The proposed relationship was substantiated by an actual validation of seed complementarity, confirming the interaction between miR-7 and βARR1 3′-UTR in INS-1 β cells through a luciferase assay (Figure 1B).
3.2. GLP-1 Triggers miR-7 Transcription
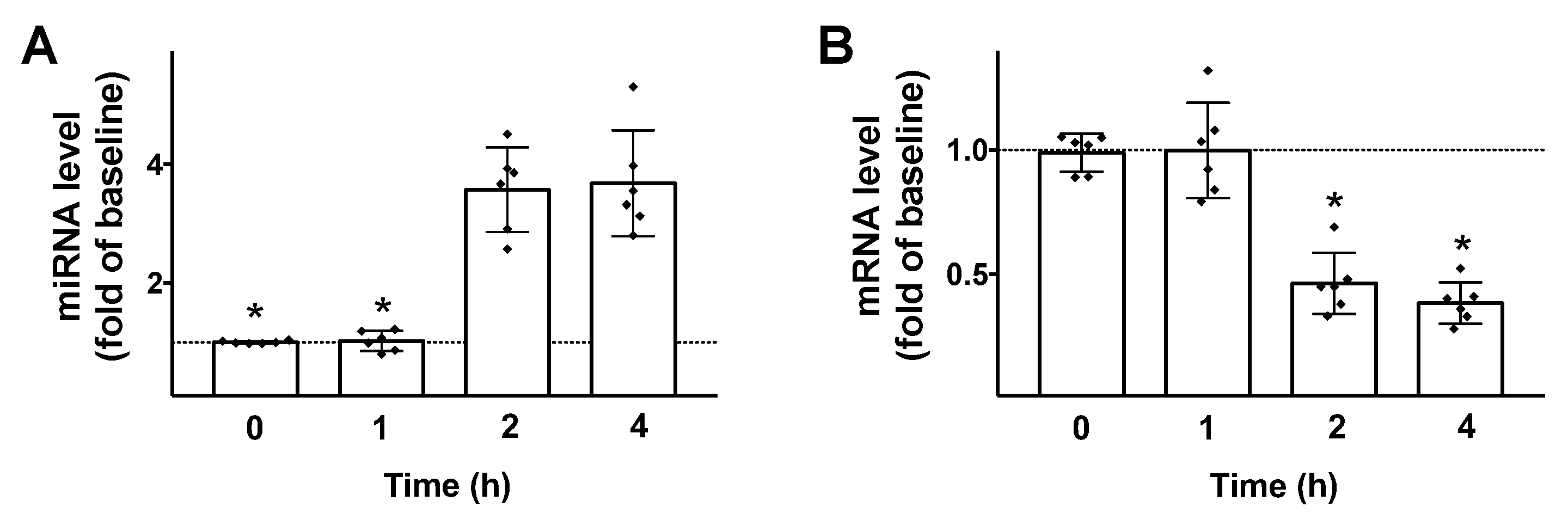
Then, we tested the effects of GLP-1 on the transcription of both miR-7 and βARR1 in β cells. We found that 100 nM GLP-1 (a dose previously verified to be effective to induce insulin release in INS1 cells [30]) induced a significant upregulation of miR-7 and downregulation of βARR1 (Figure 2), 2 h post stimulation.
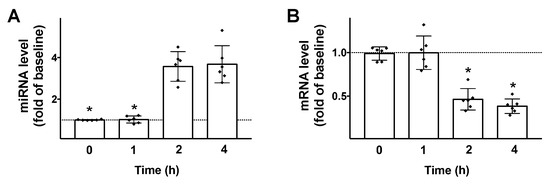
Figure 2.
GLP-1 regulates miR-7 and βARR1 transcription in pancreatic β cells. Stimulation of INS-1 β cells with GLP-1 (100 nM) induces an upregulation of miR-7 (A); and a downregulation of βARR1 (B). Means ± S.E.M. are shown alongside actual values. * p < 0.05.
3.3. miR-7 Regulates GLP-1-Induced Insulin Secretion in β Cells
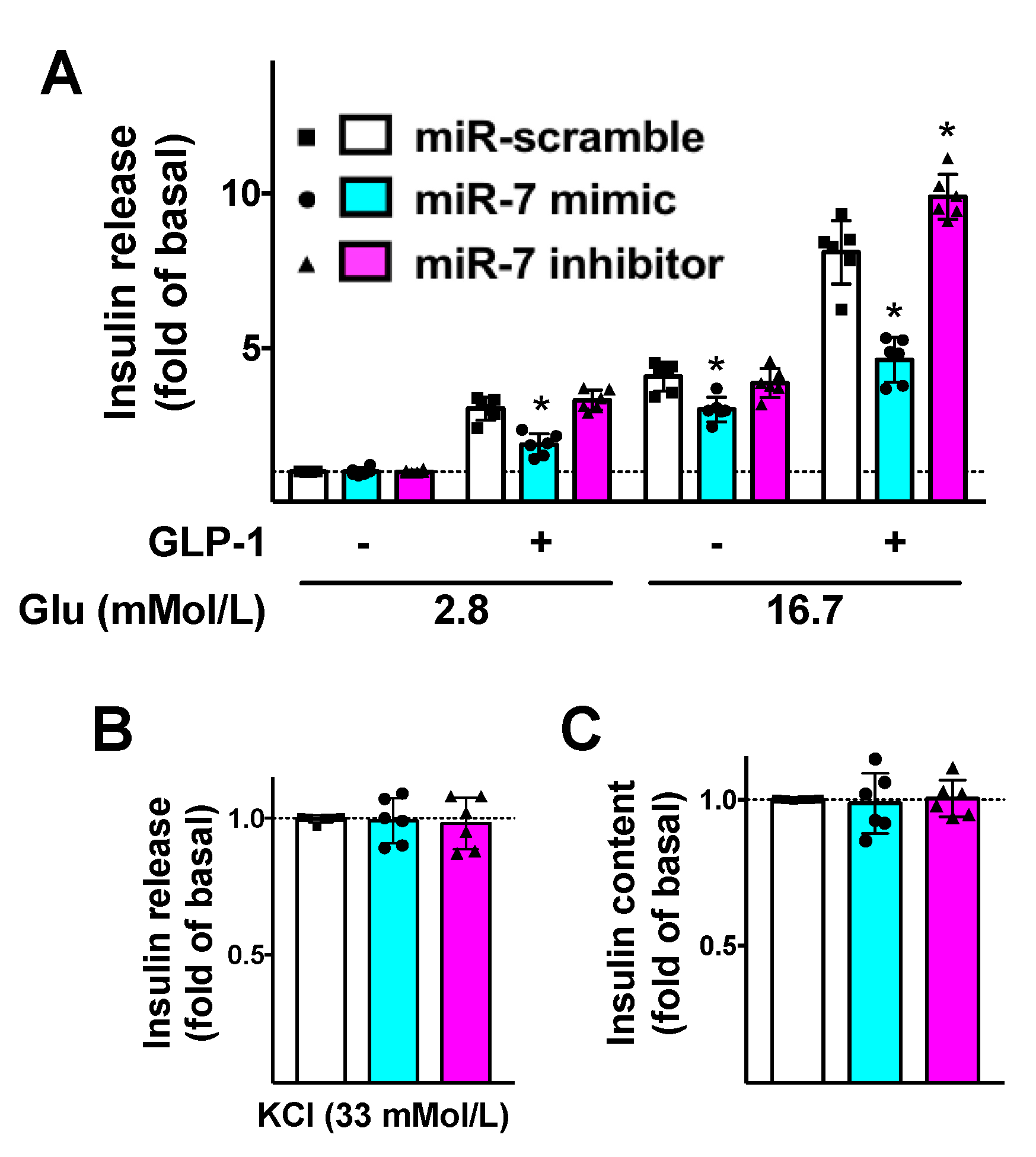
To further substantiate the functional aspects of our results, we evaluated the effects of miR-7 on GLP-1-mediated insulin release. We demonstrated that miR-7 significantly reduced GLP-1-induced GSIS in β cells (Figure 3A), whereas no significant effects were noted on KCl-induced insulin secretion (Figure 3B) or insulin content (Figure 3C).
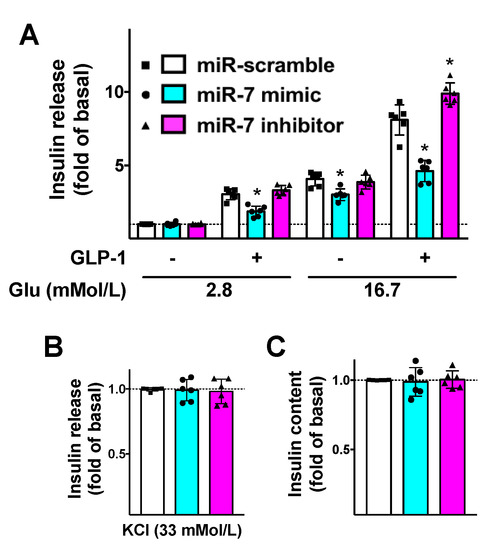
Figure 3.
Mechanistic role of miR-7 in GLP-1-mediated insulin secretion. INS-1 cells treated with miR-7 mimic, inhibitor, or scramble (negative control) were stimulated for 2 h with GLP-1 (100 nM) or vehicle and insulin release was measured (A); No significant differences were observed in terms of insulin release in response to KCl (B) or insulin content (C). Means ± S.E.M. are shown alongside actual values. * p < 0.05 vs. miR-scramble.
Strikingly, these alterations were rescued when β cells were treated with a specific inhibitor of miR-7 (Figure 3).
We also verified that miR-7 mimic significantly decreased the transcription levels of βARR1 in β cells (Supplementary Figure S1).
3.4. miR-7 Regulates GLP-1-Mediated cAMP Production in β Cells
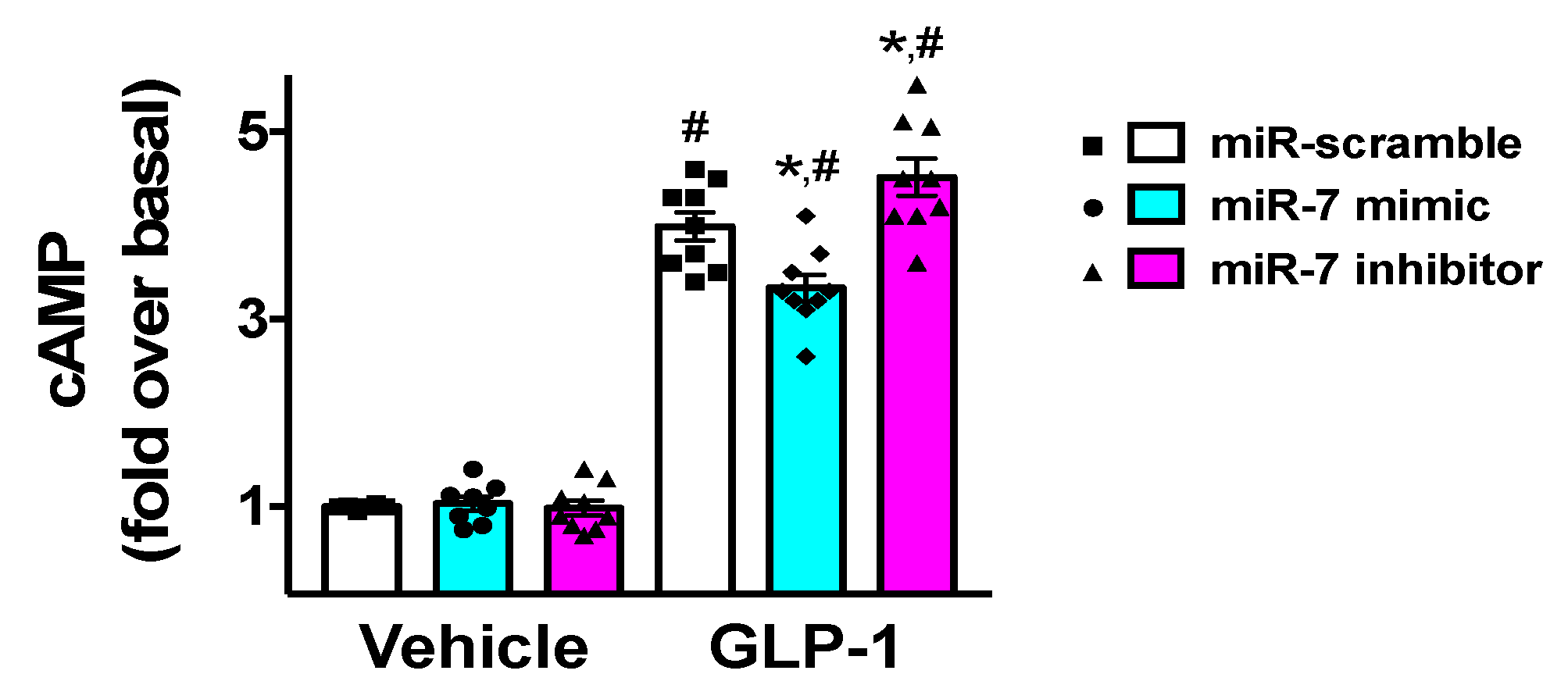
Since GLP-1 has been shown to induce insulin release via an increased production of cyclic AMP (cAMP) [30,45], we verified the effects of miR-7 on the generation of cAMP in β cells. We found that miR-7 significantly decreased GLP-1-induced cAMP levels (Figure 4), whereas incubation with a specific miR-7 inhibitor markedly increased cAMP production (Figure 4).
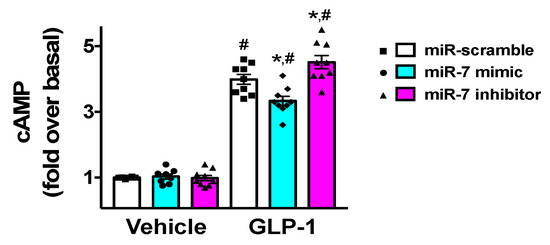
Figure 4.
Effects of miR-7 on GLP-1-mediated cAMP production. INS-1 cells treated with miR-7 mimic, inhibitor, or scramble (negative control) were stimulated for 2 h with GLP-1 (100 nM) or vehicle and cAMP was measured. Means ± S.E.M. are shown alongside actual values. Basal, vehicle + miR-scramble; * p < 0.05 vs. miR-scramble, # p < 0.05 vs. vehicle.
3.5. miR-7 Modulates the GLP-1-Mediated Activation of ERK and CREB
Finally, since βARR1 knockdown has been shown [30] to reduce the phosphorylation of ERK and CREB, two factors downstream of GLP-1 signaling, we tested the effects of miR-7 on both these signaling pathways. We found that the GLP-1-induced activation of ERK and CREB was significantly attenuated by miR-7 (Supplementary Figure S2).
4. Discussion
In the present study, we demonstrate, for the first time, that miR-7 plays a pivotal role in the regulation of GLP-1-mediated insulin release in β cells via a mechanism that involves its direct targeting of βARR1.
Our results are consistent with the evidence of a functional connection between βARR1 and GLP-1 receptor, which had been previously established by Sonoda and collaborators in INS-1 β cells [30]. Another study, fully in line with our observations, has demonstrated that transgenic mice overexpressing miR-7a in pancreatic β cells developed diabetes mellitus due to impaired insulin release and β cell dedifferentiation [44]. Similarly, the specific knock-out of miR-7 in β cells increased GSIS and improved glucose homeostasis in vivo [46,47].
The key importance of miR-7 in β cell physiology is also corroborated by the fact that this miR has been shown to be one of the most abundant miRs in both human and murine islets [19], with a ratio >150 as compared with its expression between islet and surrounding acinar tissue [48].
Since βARR1 is known to play a crucial role in the desensitization of diverse GPCRs [49], our results could open the field to new research in order to verify the role of miR-7 in other tissues and cell types. βARRs can also mediate non-canonical signaling via ERK1/2 and other kinases, which are linked to β cell apoptosis [50], as well as be involved in biased agonism [51,52]. These mechanisms could be regulated by miR-7 by its direct targeting of βARR1. Intriguingly, we observed that miR-7 mimic led to a decreased GSIS as compared with scramble, even in the absence of GLP-1 stimulation (Figure 3A), suggesting that other targets of miR-7 could be involved in insulin secretion. Similarly, we cannot exclude the fact that the miR-7/βARR1 axis regulates other GPCRs in β cells. In this sense, β adrenergic receptors are known to be regulated by βARR1 [53,54,55,56]; consistent with the present findings, our group has previously demonstrated that β2 adrenergic receptors stimulated GSIS [57].
Our data are significant also in the clinical scenario; indeed, several drugs targeting the GLP-1 pathway are available for diabetic patients. The first group of drugs is represented by GLP-1 receptor agonists which are a group of peptides that display structural similarities to native GLP-1 and activate the GLP-1 receptor; they include exenatide, lixisenatide, albiglutide, dulaglutide, liraglutide, CJC 1134, and CJC 1131 [58]. The main adverse effects of GLP-1 receptor agonists include nausea, vomiting and diarrhea, injection-site reactions, antibody formation, and increased heart rate. The gliptins are another class of drugs indirectly acting on GLP-1, via the inhibition of GLP-1 degrading enzyme dipeptidyl peptidase 4 (DPP4). These drugs, which include sitagliptin, saxagliptin, linagliptin, alogliptin, and vildagliptin, raise the plasma levels of native GLP-1 by preventing its proteolytic degradation; some concerns have been raised regarding the metabolic and cardiovascular adverse effects of these drugs [59,60].
Notably, timing seems to be essential in the cellular responses observed by us and other investigators [3,30]. Indeed, whereas acute responses to GLP-1 (5–10 min) appear to be potentiated by knockdown of βARR1, longer-term responses (16 h) suggest a more “classical” effect of βARR1 on trafficking of the receptor with internalization suppressing downstream signaling and GSIS [3,61]. Equally important, the effects of miR-7 on the βARR1-GLP-1 axis could be different when testing lower doses of GLP-1, or when stimulating GLP-1 receptor with pharmacologic agonists.
Although miRs mainly exert their biological function to bind mRNA transcript and inhibit their translation in the cytosol [22], they have also been implicated in transcriptional gene regulation and alternative splicing, which are restricted to the cell nucleus [62,63]. Since βARR1 is known to translocate from the cytosol to the nucleus, where it regulates gene transcription [64], it would be interesting to determine the exact localization of the action of miR-7 on βARR1 within the cell.
Our study does have some limitations. First, we only conducted in vitro experiments which need to be verified in vivo in animal models. Second, we focused on βARR1, without investigating the potential contribution of other βARRs [65]. Moreover, we only used a clonal β cell line (INS-1) and we did not confirm our results in other cell lines or in human islets. Nevertheless, the GLP-1/βARR1 signaling pathway has been confirmed in murine MIN6 β cells and in human β cells [3] and we have shown that the interaction between miR-7, which is one of the most highly conserved miRs during evolution [66], and the 3′-UTR of βARR1 is conserved among species (Figure 1A). Since most of the results shown are with exogenously expressed targets or miRs, further studies are necessary to appraise the translational potential of our findings.
Supplementary Materials
The following are available online at https://www.mdpi.com/2073-4409/9/7/1621/s1: Figure S1. βARR1 expression in pancreatic β cells is reduced by miR-7, Figure S2. GLP-1-induced phosphorylation of ERK and CREB is significantly attenuated by miR-7. Table S1. Sequences of oligonucleotide primers and product sizes.
Author Contributions
G.S. designed the study and wrote the manuscript; A.M., J.G., A.L., and X.W. carried out and analyzed experiments. All authors have read and agreed to the published version of the manuscript.
Funding
This study was supported in part by the NIH (R01-DK123259, R00-DK107895, R01-HL146691, R01-DK033823 to G.S.).
Conflicts of Interest
The authors declare no conflict of interest.
References
- Liang, Y.L.; Khoshouei, M.; Glukhova, A.; Furness, S.G.B.; Zhao, P.; Clydesdale, L.; Koole, C.; Truong, T.T.; Thal, D.M.; Lei, S.; et al. Phase-plate cryo-EM structure of a biased agonist-bound human GLP-1 receptor-Gs complex. Nature 2018, 555, 121–125. [Google Scholar] [CrossRef] [PubMed]
- Drucker, D.J. Mechanisms of Action and Therapeutic Application of Glucagon-like Peptide-1. Cell Metab. 2018, 27, 740–756. [Google Scholar] [CrossRef] [PubMed]
- Jones, B.; Buenaventura, T.; Kanda, N.; Chabosseau, P.; Owen, B.M.; Scott, R.; Goldin, R.; Angkathunyakul, N.; Correa, I.R., Jr.; Bosco, D.; et al. Targeting GLP-1 receptor trafficking to improve agonist efficacy. Nat. Commun. 2018, 9, 1602. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Sun, B.; Feng, D.; Hu, H.; Chu, M.; Qu, Q.; Tarrasch, J.T.; Li, S.; Sun Kobilka, T.; Kobilka, B.K.; et al. Cryo-EM structure of the activated GLP-1 receptor in complex with a G protein. Nature 2017, 546, 248–253. [Google Scholar] [CrossRef] [PubMed]
- Jazayeri, A.; Rappas, M.; Brown, A.J.H.; Kean, J.; Errey, J.C.; Robertson, N.J.; Fiez-Vandal, C.; Andrews, S.P.; Congreve, M.; Bortolato, A.; et al. Crystal structure of the GLP-1 receptor bound to a peptide agonist. Nature 2017, 546, 254–258. [Google Scholar] [CrossRef] [PubMed]
- Song, G.; Yang, D.; Wang, Y.; de Graaf, C.; Zhou, Q.; Jiang, S.; Liu, K.; Cai, X.; Dai, A.; Lin, G.; et al. Human GLP-1 receptor transmembrane domain structure in complex with allosteric modulators. Nature 2017, 546, 312–315. [Google Scholar] [CrossRef]
- Rupaimoole, R.; Slack, F.J. MicroRNA therapeutics: Towards a new era for the management of cancer and other diseases. Nat. Rev. Drug Discov. 2017, 16, 203–222. [Google Scholar] [CrossRef]
- Hess, A.L.; Larsen, L.H.; Udesen, P.B.; Sanz, Y.; Larsen, T.M.; Dalgaard, L.T. Levels of Circulating miR-122 are Associated with Weight Loss and Metabolic Syndrome. Obesity (Silver Spring) 2020, 28, 493–501. [Google Scholar] [CrossRef]
- Santulli, G. microRNAs Distinctively Regulate Vascular Smooth Muscle and Endothelial Cells: Functional Implications in Angiogenesis, Atherosclerosis, and In-Stent Restenosis. Adv. Exp. Med. Biol. 2015, 887, 53–77. [Google Scholar]
- Mendell, J.T.; Olson, E.N. MicroRNAs in stress signaling and human disease. Cell 2012, 148, 1172–1187. [Google Scholar] [CrossRef]
- Vonhogen, I.G.C.; El Azzouzi, H.; Olieslagers, S.; Vasilevich, A.; de Boer, J.; Tinahones, F.J.; da Costa Martins, P.A.; de Windt, L.J.; Murri, M. MiR-337-3p Promotes Adipocyte Browning by Inhibiting TWIST1. Cells 2020, 9, 1056. [Google Scholar] [CrossRef] [PubMed]
- Santulli, G. Exosomal microRNA: The revolutionary endogenous Innerspace nanotechnology. Sci. Transl. Med. 2018, 10. [Google Scholar] [CrossRef] [PubMed]
- Sorensen, A.E.; Wissing, M.L.; Salo, S.; Englund, A.L.; Dalgaard, L.T. MicroRNAs Related to Polycystic Ovary Syndrome (PCOS). Genes 2014, 5, 684–708. [Google Scholar] [CrossRef] [PubMed]
- Eliasson, L.; Esguerra, J.L. Role of non-coding RNAs in pancreatic beta-cell development and physiology. Acta Physiol. (Oxf.) 2014, 211, 273–284. [Google Scholar] [CrossRef]
- Michell, D.L.; Zhao, S.; Allen, R.M.; Sheng, Q.; Vickers, K.C. Pervasive Small RNAs in Cardiometabolic Research: Great Potential Accompanied by Biological and Technical Barriers. Diabetes 2020, 69, 813–822. [Google Scholar] [CrossRef] [PubMed]
- Wong, W.K.M.; Sorensen, A.E.; Joglekar, M.V.; Hardikar, A.A.; Dalgaard, L.T. Non-Coding RNA in Pancreas and beta-Cell Development. Noncoding RNA 2018, 4, 41. [Google Scholar]
- Ozcan, S. microRNAs in Pancreatic beta-Cell Physiology. Adv. Exp. Med. Biol. 2015, 887, 101–117. [Google Scholar] [PubMed]
- Joglekar, M.V.; Wong, W.K.M.; Maynard, C.L.; Umrani, M.R.; Martin, D.; Loudovaris, T.; Thomas, H.E.; Dalgaard, L.T.; Hardikar, A.A. Expression of miR-206 in human islets and its role in glucokinase regulation. Am. J. Physiol. Endocrinol. Metab. 2018, 315, E634–E637. [Google Scholar] [CrossRef]
- Eliasson, L.; Esguerra, J.L.S. MicroRNA Networks in Pancreatic Islet Cells: Normal Function and Type 2 Diabetes. Diabetes 2020, 69, 804–812. [Google Scholar] [CrossRef]
- Christopher, A.F.; Kaur, R.P.; Kaur, G.; Kaur, A.; Gupta, V.; Bansal, P. MicroRNA therapeutics: Discovering novel targets and developing specific therapy. Perspect. Clin. Res. 2016, 7, 68–74. [Google Scholar]
- Barraclough, J.Y.; Joan, M.; Joglekar, M.V.; Hardikar, A.A.; Patel, S. MicroRNAs as Prognostic Markers in Acute Coronary Syndrome Patients-A Systematic Review. Cells 2019, 8, 1572. [Google Scholar] [CrossRef] [PubMed]
- Santulli, G. MicroRNA: From Molecular Biology to Clinical Practice; Springer Nature: New York, NY, USA, 2016. [Google Scholar]
- Cahill, T.J., 3rd; Thomsen, A.R.; Tarrasch, J.T.; Plouffe, B.; Nguyen, A.H.; Yang, F.; Huang, L.Y.; Kahsai, A.W.; Bassoni, D.L.; Gavino, B.J.; et al. Distinct conformations of GPCR-beta-arrestin complexes mediate desensitization, signaling, and endocytosis. Proc. Natl. Acad. Sci. USA 2017, 114, 2562–2567. [Google Scholar] [CrossRef] [PubMed]
- Baidya, M.; Kumari, P.; Dwivedi-Agnihotri, H.; Pandey, S.; Sokrat, B.; Sposini, S.; Chaturvedi, M.; Srivastava, A.; Roy, D.; Hanyaloglu, A.C.; et al. Genetically encoded intrabody sensors report the interaction and trafficking of beta-arrestin 1 upon activation of G protein-coupled receptors. J. Biol. Chem. 2020. [Google Scholar] [CrossRef] [PubMed]
- Dabul, S.; Bathgate-Siryk, A.; Valero, T.R.; Jafferjee, M.; Sturchler, E.; McDonald, P.; Koch, W.J.; Lymperopoulos, A. Suppression of adrenal betaarrestin1-dependent aldosterone production by ARBs: Head-to-head comparison. Sci. Rep. 2015, 5, 8116. [Google Scholar] [CrossRef]
- Slosky, L.M.; Bai, Y.; Toth, K.; Ray, C.; Rochelle, L.K.; Badea, A.; Chandrasekhar, R.; Pogorelov, V.M.; Abraham, D.M.; Atluri, N.; et al. beta-Arrestin-Biased Allosteric Modulator of NTSR1 Selectively Attenuates Addictive Behaviors. Cell 2020, 181, 1364–1379.e14. [Google Scholar] [CrossRef]
- Chaturvedi, M.; Maharana, J.; Shukla, A.K. Terminating G-Protein Coupling: Structural Snapshots of GPCR-beta-Arrestin Complexes. Cell 2020, 180, 1041–1043. [Google Scholar] [CrossRef]
- Santulli, G. Adrenal signaling in heart failure: Something more than a distant ship’s smoke on the horizon. Hypertension 2014, 63, 215–216. [Google Scholar] [CrossRef]
- Alexander, R.A.; Lot, I.; Saha, K.; Abadie, G.; Lambert, M.; Decosta, E.; Kobayashi, H.; Beautrait, A.; Borrull, A.; Asnacios, A.; et al. Beta-arrestins operate an on/off control switch for focal adhesion kinase activity. Cell. Mol. Life Sci. 2020. [Google Scholar] [CrossRef]
- Sonoda, N.; Imamura, T.; Yoshizaki, T.; Babendure, J.L.; Lu, J.C.; Olefsky, J.M. Beta-Arrestin-1 mediates glucagon-like peptide-1 signaling to insulin secretion in cultured pancreatic beta cells. Proc. Natl. Acad. Sci. USA 2008, 105, 6614–6619. [Google Scholar] [CrossRef]
- Maier, C.; Truong, A.; Auld, S.; Polly, D.; Tanksley, C.; Duncan, A. COVID-19-associated hyperviscosity: A link between inflammation and thrombophilia? Lancet 2020. [Google Scholar] [CrossRef]
- Lombardi, A.; Trimarco, B.; Iaccarino, G.; Santulli, G. Impaired mitochondrial calcium uptake caused by tacrolimus underlies beta-cell failure. Cell Commun. Signal. 2017, 15, 47. [Google Scholar] [CrossRef] [PubMed]
- Lombardi, A.; Gambardella, J.; Du, X.L.; Sorriento, D.; Mauro, M.; Iaccarino, G.; Trimarco, B.; Santulli, G. Sirolimus induces depletion of intracellular calcium stores and mitochondrial dysfunction in pancreatic beta cells. Sci. Rep. 2017, 7, 15823. [Google Scholar] [CrossRef] [PubMed]
- Santulli, G.; Pagano, G.; Sardu, C.; Xie, W.; Reiken, S.; D’Ascia, S.L.; Cannone, M.; Marziliano, N.; Trimarco, B.; Guise, T.A.; et al. Calcium release channel RyR2 regulates insulin release and glucose homeostasis. J. Clin. Investig. 2015, 125, 1968–1978. [Google Scholar] [CrossRef] [PubMed]
- Morelli, M.B.; Shu, J.; Sardu, C.; Matarese, A.; Santulli, G. Cardiosomal microRNAs Are Essential in Post-Infarction Myofibroblast Phenoconversion. Int. J. Mol. Sci. 2019, 21, 201. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Morelli, M.B.; Matarese, A.; Sardu, C.; Santulli, G. Cardiomyocyte-derived exosomal microRNA-92a mediates post-ischemic myofibroblast activation both in vitro and ex vivo. ESC Heart Fail. 2020, 7, 284–288. [Google Scholar] [CrossRef]
- Santulli, G.; Wronska, A.; Uryu, K.; Diacovo, T.G.; Gao, M.; Marx, S.O.; Kitajewski, J.; Chilton, J.M.; Akat, K.M.; Tuschl, T.; et al. A selective microRNA-based strategy inhibits restenosis while preserving endothelial function. J. Clin. Investig. 2014, 124, 4102–4114. [Google Scholar] [CrossRef]
- De Vitis, S.; Sonia Treglia, A.; Ulianich, L.; Turco, S.; Terrazzano, G.; Lombardi, A.; Miele, C.; Garbi, C.; Beguinot, F.; Di Jeso, B. Tyr phosphatase-mediated P-ERK inhibition suppresses senescence in EIA + v-raf transformed cells, which, paradoxically, are apoptosis-protected in a MEK-dependent manner. Neoplasia 2011, 13, 120–130. [Google Scholar] [CrossRef]
- Xie, W.; Santulli, G.; Reiken, S.R.; Yuan, Q.; Osborne, B.W.; Chen, B.X.; Marks, A.R. Mitochondrial oxidative stress promotes atrial fibrillation. Sci. Rep. 2015, 5, 11427. [Google Scholar] [CrossRef]
- Santulli, G.; Xie, W.; Reiken, S.R.; Marks, A.R. Mitochondrial calcium overload is a key determinant in heart failure. Proc. Natl. Acad. Sci. USA 2015, 112, 11389–11394. [Google Scholar] [CrossRef]
- Landgraf, P.; Rusu, M.; Sheridan, R.; Sewer, A.; Iovino, N.; Aravin, A.; Pfeffer, S.; Rice, A.; Kamphorst, A.O.; Landthaler, M.; et al. A mammalian microRNA expression atlas based on small RNA library sequencing. Cell 2007, 129, 1401–1414. [Google Scholar] [CrossRef]
- Kredo-Russo, S.; Ness, A.; Mandelbaum, A.D.; Walker, M.D.; Hornstein, E. Regulation of pancreatic microRNA-7 expression. Exp. Diabetes Res. 2012, 2012, 695214. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Vogel, G.; Yu, Z.; Richard, S. The QKI-5 and QKI-6 RNA binding proteins regulate the expression of microRNA 7 in glial cells. Mol. Cell Biol. 2013, 33, 1233–1243. [Google Scholar] [CrossRef] [PubMed]
- Latreille, M.; Hausser, J.; Stutzer, I.; Zhang, Q.; Hastoy, B.; Gargani, S.; Kerr-Conte, J.; Pattou, F.; Zavolan, M.; Esguerra, J.L.; et al. MicroRNA-7a regulates pancreatic beta cell function. J. Clin. Investig. 2014, 124, 2722–2735. [Google Scholar] [CrossRef]
- Lamont, B.J.; Li, Y.; Kwan, E.; Brown, T.J.; Gaisano, H.; Drucker, D.J. Pancreatic GLP-1 receptor activation is sufficient for incretin control of glucose metabolism in mice. J. Clin. Investig. 2012, 122, 388–402. [Google Scholar] [CrossRef] [PubMed]
- Latreille, M.; Herrmanns, K.; Renwick, N.; Tuschl, T.; Malecki, M.T.; McCarthy, M.I.; Owen, K.R.; Rulicke, T.; Stoffel, M. miR-375 gene dosage in pancreatic beta-cells: Implications for regulation of beta-cell mass and biomarker development. J. Mol. Med. 2015, 93, 1159–1169. [Google Scholar] [CrossRef]
- Xu, H.; Guo, S.; Li, W.; Yu, P. The circular RNA Cdr1as, via miR-7 and its targets, regulates insulin transcription and secretion in islet cells. Sci. Rep. 2015, 5, 12453. [Google Scholar] [CrossRef]
- Bravo-Egana, V.; Rosero, S.; Molano, R.D.; Pileggi, A.; Ricordi, C.; Dominguez-Bendala, J.; Pastori, R.L. Quantitative differential expression analysis reveals miR-7 as major islet microRNA. Biochem. Biophys. Res. Commun. 2008, 366, 922–926. [Google Scholar] [CrossRef]
- Goodman, O.B., Jr.; Krupnick, J.G.; Santini, F.; Gurevich, V.V.; Penn, R.B.; Gagnon, A.W.; Keen, J.H.; Benovic, J.L. Beta-arrestin acts as a clathrin adaptor in endocytosis of the beta2-adrenergic receptor. Nature 1996, 383, 447–450. [Google Scholar] [CrossRef]
- Quoyer, J.; Longuet, C.; Broca, C.; Linck, N.; Costes, S.; Varin, E.; Bockaert, J.; Bertrand, G.; Dalle, S. GLP-1 mediates antiapoptotic effect by phosphorylating Bad through a beta-arrestin 1-mediated ERK1/2 activation in pancreatic beta-cells. J. Biol. Chem. 2010, 285, 1989–2002. [Google Scholar] [CrossRef]
- Costa-Neto, C.M.; Parreiras, E.S.L.T.; Bouvier, M. A Pluridimensional View of Biased Agonism. Mol. Pharmacol. 2016, 90, 587–595. [Google Scholar] [CrossRef]
- Choi, M.; Staus, D.P.; Wingler, L.M.; Ahn, S.; Pani, B.; Capel, W.D.; Lefkowitz, R.J. G protein-coupled receptor kinases (GRKs) orchestrate biased agonism at the beta2-adrenergic receptor. Sci. Signal. 2018, 11. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Grotegut, C.A.; Wisler, J.W.; Li, T.; Mao, L.; Chen, M.; Chen, W.; Rosenberg, P.B.; Rockman, H.A.; Lefkowitz, R.J. beta-arrestin 1 regulates beta2-adrenergic receptor-mediated skeletal muscle hypertrophy and contractility. Skelet. Muscle 2018, 8, 39. [Google Scholar] [CrossRef] [PubMed]
- Luttrell, L.M.; Lefkowitz, R.J. The role of beta-arrestins in the termination and transduction of G-protein-coupled receptor signals. J. Cell Sci. 2002, 115 Pt 3, 455–465. [Google Scholar]
- Nguyen, A.H.; Thomsen, A.R.B.; Cahill, T.J., 3rd; Huang, R.; Huang, L.Y.; Marcink, T.; Clarke, O.B.; Heissel, S.; Masoudi, A.; Ben-Hail, D.; et al. Structure of an endosomal signaling GPCR-G protein-beta-arrestin megacomplex. Nat. Struct. Mol. Biol. 2019, 26, 1123–1131. [Google Scholar] [CrossRef] [PubMed]
- Luttrell, L.M.; Wang, J.; Plouffe, B.; Smith, J.S.; Yamani, L.; Kaur, S.; Jean-Charles, P.Y.; Gauthier, C.; Lee, M.H.; Pani, B.; et al. Manifold roles of beta-arrestins in GPCR signaling elucidated with siRNA and CRISPR/Cas9. Sci. Signal. 2018, 11, eaat7650. [Google Scholar] [CrossRef] [PubMed]
- Santulli, G.; Lombardi, A.; Sorriento, D.; Anastasio, A.; Del Giudice, C.; Formisano, P.; Beguinot, F.; Trimarco, B.; Miele, C.; Iaccarino, G. Age-related impairment in insulin release: The essential role of beta(2)-adrenergic receptor. Diabetes 2012, 61, 692–701. [Google Scholar] [CrossRef]
- Meier, J.J. GLP-1 receptor agonists for individualized treatment of type 2 diabetes mellitus. Nat. Rev. Endocrinol. 2012, 8, 728–742. [Google Scholar] [CrossRef]
- Tkac, I.; Raz, I. Combined Analysis of Three Large Interventional Trials With Gliptins Indicates Increased Incidence of Acute Pancreatitis in Patients with Type 2 Diabetes. Diabetes Care 2017, 40, 284–286. [Google Scholar] [CrossRef]
- Al-Mallah, M.; Hyafil, F.; Santulli, G. No pleotropic effects of linagliptin on atherosclerotic plaques: Case closed. Atherosclerosis 2020. [Google Scholar] [CrossRef]
- Carlessi, R.; Chen, Y.; Rowlands, J.; Cruzat, V.F.; Keane, K.N.; Egan, L.; Mamotte, C.; Stokes, R.; Gunton, J.E.; Bittencourt, P.I.H.; et al. GLP-1 receptor signalling promotes beta-cell glucose metabolism via mTOR-dependent HIF-1alpha activation. Sci. Rep. 2017, 7, 2661. [Google Scholar] [CrossRef]
- Turunen, T.A.; Roberts, T.C.; Laitinen, P.; Vaananen, M.A.; Korhonen, P.; Malm, T.; Yla-Herttuala, S.; Turunen, M.P. Changes in nuclear and cytoplasmic microRNA distribution in response to hypoxic stress. Sci. Rep. 2019, 9, 10332. [Google Scholar] [CrossRef] [PubMed]
- Boutz, P.L.; Chawla, G.; Stoilov, P.; Black, D.L. MicroRNAs regulate the expression of the alternative splicing factor nPTB during muscle development. Genes Dev. 2007, 21, 71–84. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Pei, G. Beta-arrestin signaling and regulation of transcription. J. Cell. Sci. 2007, 120 Pt 2, 213–218. [Google Scholar] [CrossRef]
- Srivastava, A.; Gupta, B.; Gupta, C.; Shukla, A.K. Emerging Functional Divergence of beta-Arrestin Isoforms in GPCR Function. Trends Endocrinol. Metab. 2015, 26, 628–642. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Sanchez, A.; Rutter, G.A.; Latreille, M. MiRNAs in beta-Cell Development, Identity, and Disease. Front. Genet. 2016, 7, 226. [Google Scholar] [PubMed]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).