Comprehensive Analysis of HMCN1 Somatic Mutation in Clear Cell Renal Cell Carcinoma
Abstract
1. Introduction
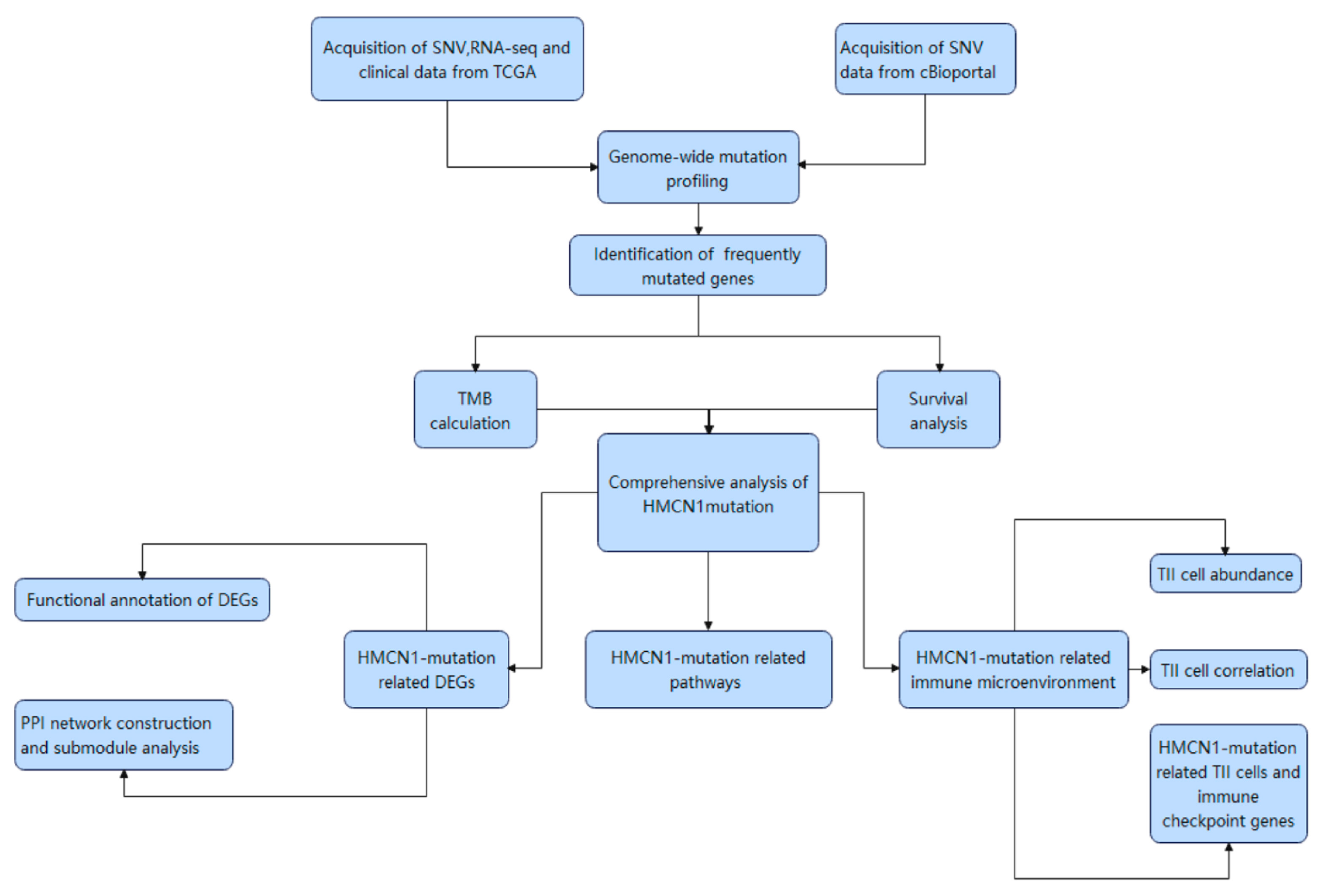
2. Materials and Methods
2.1. Data Acquisition
2.2. Genome-Wide Mutation Profiling
2.3. TMB Calculation and Survival Analysis
2.4. Differentially Expressed Genes (DEGs) Analysis
2.5. Functional Annotation of DEGs
2.6. Protein–Protein Interaction (PPI) and Submodule Analysis
2.7. Estimation of Immune Cell Infiltrating
2.8. Statistical Analysis
3. Results
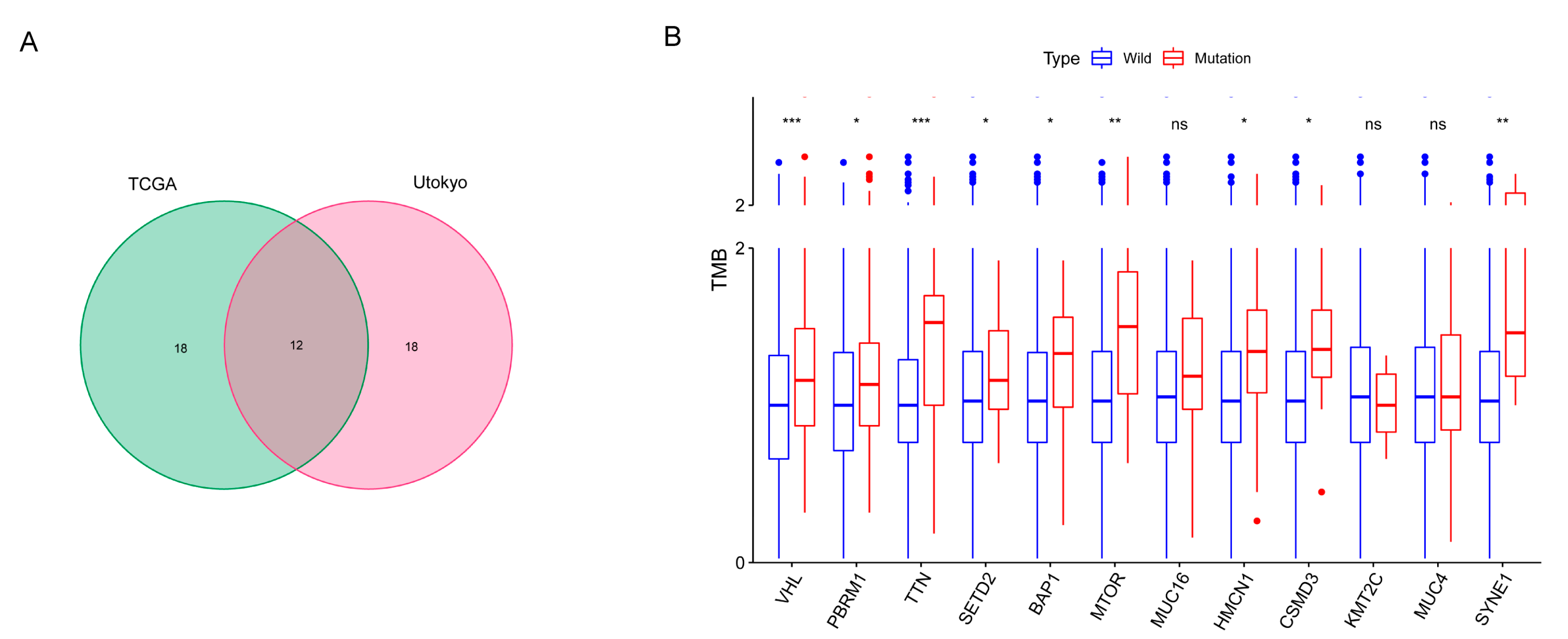
3.1. Somatic Mutation Landscape of ccRCC
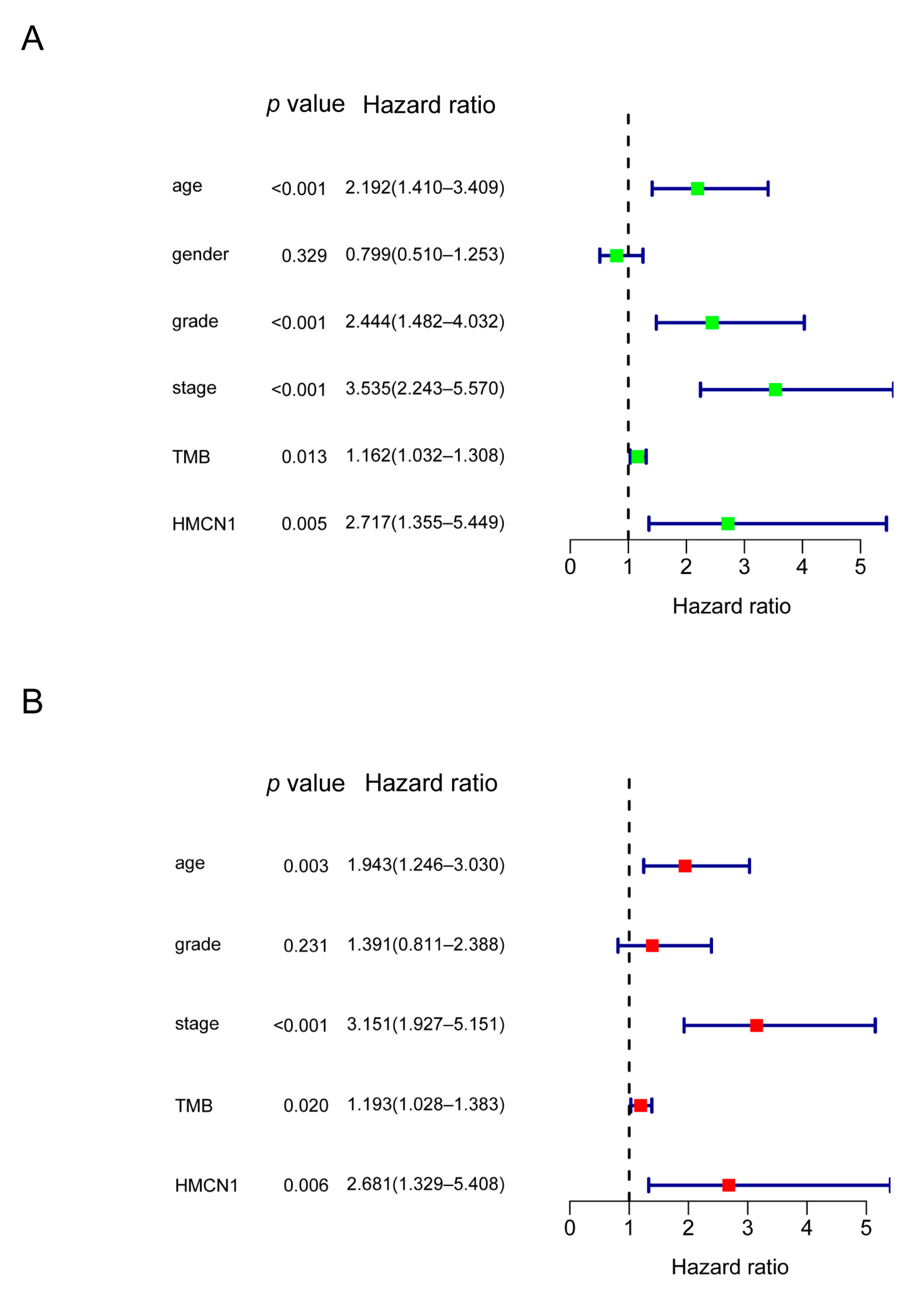
3.2. Gene Mutations Related to TMB and Prognosis
3.3. Identification of DEGs
3.4. Functional Annotations of DEGs
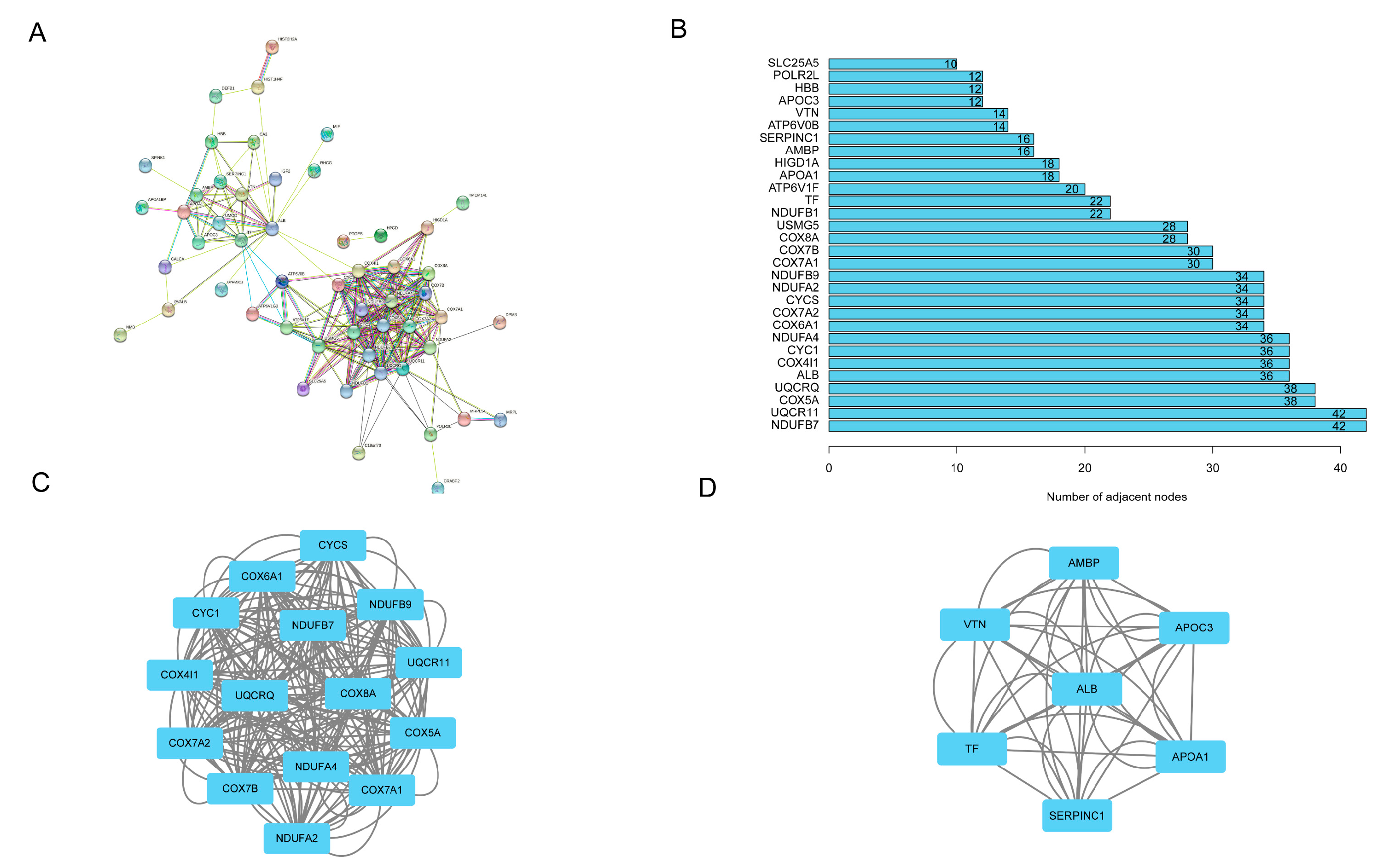
3.5. PPI Network Establishment, Hub Genes, and Submodules Screening
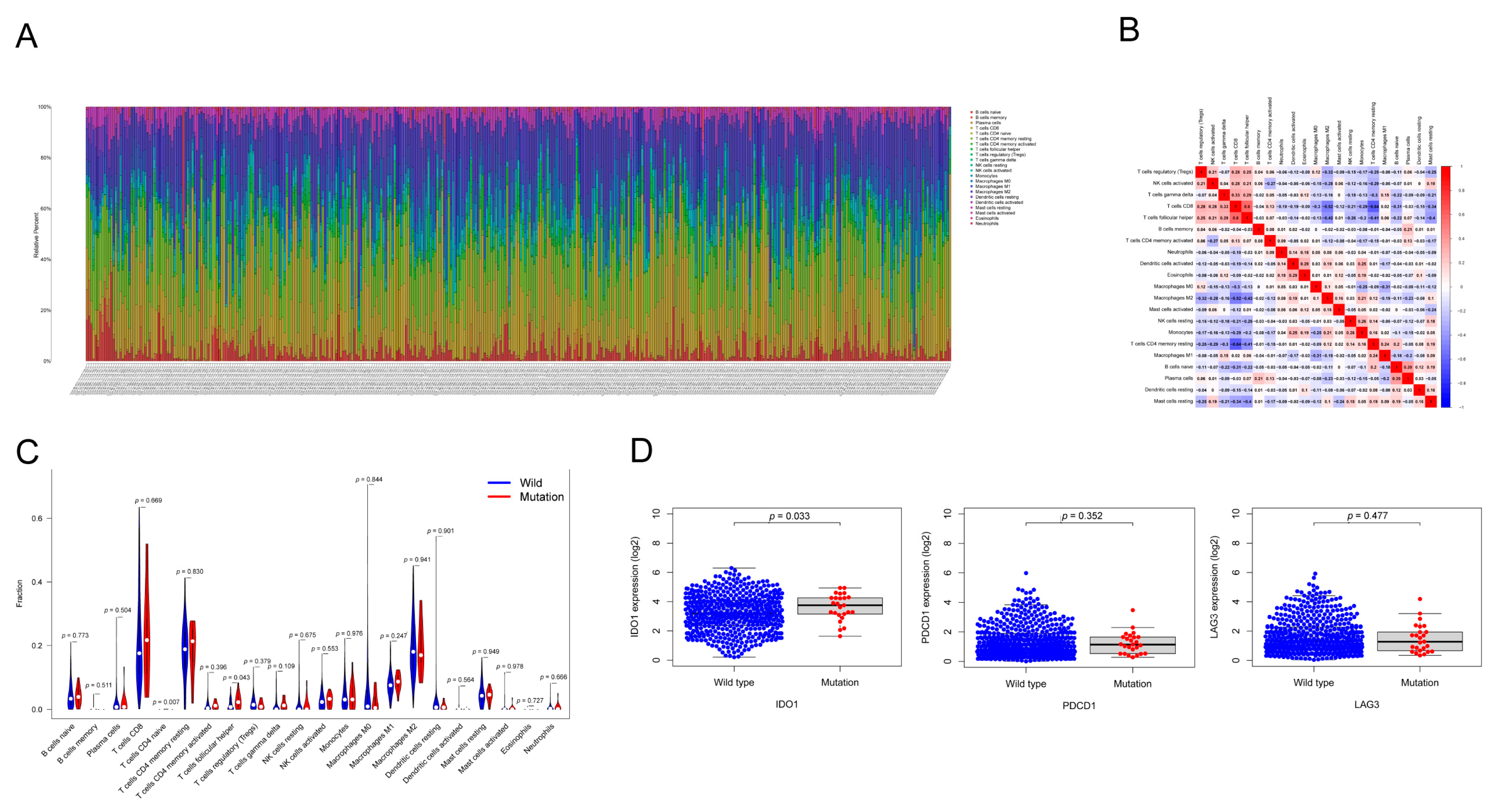
3.6. HMCN1 Mutation-Related Tumor Immune Microenvironment
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Linehan, W.M.; Ricketts, C.J. The Cancer Genome Atlas of renal cell carcinoma: Findings and clinical implications. Nat. Rev. Urol. 2019, 16, 539–552. [Google Scholar] [CrossRef] [PubMed]
- Deleuze, A.; Saout, J.; Dugay, F.; Peyronnet, B.; Mathieu, R.; Verhoest, G.; Bensalah, K.; Crouzet, L.; Laguerre, B.; Belaud-Rotureau, M.A.; et al. Immunotherapy in Renal Cell Carcinoma: The Future Is Now. Int. J. Mol. Sci. 2020, 21, 2532. [Google Scholar] [CrossRef]
- Hsieh, J.J.; Purdue, M.P.; Signoretti, S.; Swanton, C.; Albiges, L.; Schmidinger, M.; Heng, D.Y.; Larkin, J.; Ficarra, V. Renal cell carcinoma. Nat. Rev. Dis. Primers 2017, 3, 17009. [Google Scholar] [CrossRef] [PubMed]
- Jonasch, E.; Gao, J.; Rathmell, W.K. Renal cell carcinoma. BMJ 2014, 349, g4797. [Google Scholar] [CrossRef] [PubMed]
- Adibi, M.; Thomas, A.Z.; Borregales, L.D.; Matin, S.F.; Wood, C.G.; Karam, J.A. Surgical considerations for patients with metastatic renal cell carcinoma. Urol. Oncol. 2015, 33, 528–537. [Google Scholar] [CrossRef]
- Eskicorapci, S.Y.; Teber, D.; Schulze, M.; Ates, M.; Stock, C.; Rassweiler, J.J. Laparoscopic radical nephrectomy: The new gold standard surgical treatment for localized renal cell carcinoma. Sci. World J. 2007, 7, 825–836. [Google Scholar] [CrossRef]
- Attalla, K.; Weng, S.; Voss, M.H.; Hakimi, A.A. Epidemiology, Risk Assessment, and Biomarkers for Patients with Advanced Renal Cell Carcinoma. Urol. Clin. N. Am. 2020, 47, 293–303. [Google Scholar] [CrossRef]
- Motzer, R.J.; Russo, P.; Nanus, D.M.; Berg, W.J. Renal cell carcinoma. Curr. Probl. Cancer 1997, 21, 185–232. [Google Scholar] [CrossRef]
- Itsumi, M.; Tatsugami, K. Immunotherapy for renal cell carcinoma. Clin. Dev. Immunol. 2010, 2010, 284581. [Google Scholar] [CrossRef]
- Albiges, L.; Powles, T.; Staehler, M.; Bensalah, K.; Giles, R.H.; Hora, M.; Kuczyk, M.A.; Lam, T.B.; Ljungberg, B.; Marconi, L.; et al. Updated European Association of Urology Guidelines on Renal Cell Carcinoma: Immune Checkpoint Inhibition Is the New Backbone in First-line Treatment of Metastatic Clear-cell Renal Cell Carcinoma. Eur. Urol. 2019, 76, 151–156. [Google Scholar] [CrossRef]
- Chan, T.A.; Yarchoan, M.; Jaffee, E.; Swanton, C.; Quezada, S.A.; Stenzinger, A.; Peters, S. Development of tumor mutation burden as an immunotherapy biomarker: Utility for the oncology clinic. Ann. Oncol. 2019, 30, 44–56. [Google Scholar] [CrossRef] [PubMed]
- Jardim, D.L.; Goodman, A.; de Melo Gagliato, D.; Kurzrock, R. The Challenges of Tumor Mutational Burden as an Immunotherapy Biomarker. Cancer cell 2021, 39, 154–173. [Google Scholar] [CrossRef] [PubMed]
- Qiao, M.; Jiang, T.; Liu, X.; Mao, S.; Zhou, F.; Li, X.; Zhao, C.; Chen, X.; Su, C.; Ren, S.; et al. Immune Checkpoint Inhibitors in EGFR-Mutated NSCLC: Dusk or Dawn? J. Thorac. Oncol. 2021, 16, 1267–1288. [Google Scholar] [CrossRef] [PubMed]
- Baba, Y.; Nomoto, D.; Okadome, K.; Ishimoto, T.; Iwatsuki, M.; Miyamoto, Y.; Yoshida, N.; Baba, H. Tumor immune microenvironment and immune checkpoint inhibitors in esophageal squamous cell carcinoma. Cancer Sci. 2020, 111, 3132–3141. [Google Scholar] [CrossRef] [PubMed]
- Lin, M.H.; Pope, B.D., 3rd; Sasaki, T.; Keeley, D.P.; Sherwood, D.R.; Miner, J.H. Mammalian hemicentin 1 is assembled into tracks in the extracellular matrix of multiple tissues. Dev. Dyn. 2020, 249, 775–788. [Google Scholar] [CrossRef]
- Schultz, D.W.; Klein, M.L.; Humpert, A.J.; Luzier, C.W.; Persun, V.; Schain, M.; Mahan, A.; Runckel, C.; Cassera, M.; Vittal, V.; et al. Analysis of the ARMD1 locus: Evidence that a mutation in HEMICENTIN-1 is associated with age-related macular degeneration in a large family. Hum. Mol. Genet. 2003, 12, 3315–3323. [Google Scholar] [CrossRef]
- Kikutake, C.; Yoshihara, M.; Sato, T.; Saito, D.; Suyama, M. Intratumor heterogeneity of HMCN1 mutant alleles associated with poor prognosis in patients with breast cancer. Oncotarget 2018, 9, 33337–33347. [Google Scholar] [CrossRef]
- The Cancer Genome Atlas. Available online: http://portal.gdc.cancer.gov/projects (accessed on 13 February 2022).
- Sato, Y.; Yoshizato, T.; Shiraishi, Y.; Maekawa, S.; Okuno, Y.; Kamura, T.; Shimamura, T.; Sato-Otsubo, A.; Nagae, G.; Suzuki, H.; et al. Integrated molecular analysis of clear-cell renal cell carcinoma. Nat. Genet. 2013, 45, 860–867. [Google Scholar] [CrossRef]
- cBio Cancer Genomics Portal. Available online: https://www.cbioportal.org/ (accessed on 13 February 2022).
- Skidmore, Z.L.; Campbell, K.M.; Cotto, K.C.; Griffith, M.; Griffith, O.L. Exploring the Genomic Landscape of Cancer Patient Cohorts with GenVisR. Curr. Protoc. 2021, 1, e252. [Google Scholar] [CrossRef]
- Chalmers, Z.R.; Connelly, C.F.; Fabrizio, D.; Gay, L.; Ali, S.M.; Ennis, R.; Schrock, A.; Campbell, B.; Shlien, A.; Chmielecki, J.; et al. Analysis of 100,000 human cancer genomes reveals the landscape of tumor mutational burden. Genome Med. 2017, 9, 34. [Google Scholar] [CrossRef]
- Yin, W.; Jiang, X.; Tan, J.; Xin, Z.; Zhou, Q.; Zhan, C.; Fu, X.; Wu, Z.; Guo, Y.; Jiang, Z.; et al. Development and Validation of a Tumor Mutation Burden-Related Immune Prognostic Model for Lower-Grade Glioma. Front. Oncol. 2020, 10, 1409. [Google Scholar] [CrossRef] [PubMed]
- Parikh, K.; Huether, R.; White, K.; Hoskinson, D.; Beaubier, N.; Dong, H.; Adjei, A.A.; Mansfield, A.S. Tumor Mutational Burden From Tumor-Only Sequencing Compared With Germline Subtraction From Paired Tumor and Normal Specimens. JAMA Netw. Open 2020, 3, e200202. [Google Scholar] [CrossRef] [PubMed]
- Robinson, M.D.; McCarthy, D.J.; Smyth, G.K. edgeR: A Bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics 2010, 26, 139–140. [Google Scholar] [CrossRef] [PubMed]
- Yu, G.; Wang, L.G.; Han, Y.; He, Q.Y. clusterProfiler: An R package for comparing biological themes among gene clusters. OMICS J. Integr. Biol. 2012, 16, 284–287. [Google Scholar] [CrossRef] [PubMed]
- Subramanian, A.; Tamayo, P.; Mootha, V.K.; Mukherjee, S.; Ebert, B.L.; Gillette, M.A.; Paulovich, A.; Pomeroy, S.L.; Golub, T.R.; Lander, E.S.; et al. Gene set enrichment analysis: A knowledge-based approach for interpreting genome-wide expression profiles. Proc. Natl. Acad. Sci. USA 2005, 102, 15545–15550. [Google Scholar] [CrossRef]
- Liberzon, A.; Birger, C.; Thorvaldsdóttir, H.; Ghandi, M.; Mesirov, J.P.; Tamayo, P. The Molecular Signatures Database (MSigDB) hallmark gene set collection. Cell Syst. 2015, 1, 417–425. [Google Scholar] [CrossRef]
- von Mering, C.; Huynen, M.; Jaeggi, D.; Schmidt, S.; Bork, P.; Snel, B. STRING: A database of predicted functional associations between proteins. Nucleic Acids Res. 2003, 31, 258–261. [Google Scholar] [CrossRef]
- Smoot, M.E.; Ono, K.; Ruscheinski, J.; Wang, P.L.; Ideker, T. Cytoscape 2.8: New features for data integration and network visualization. Bioinformatics 2011, 27, 431–432. [Google Scholar] [CrossRef]
- Rhrissorrakrai, K.; Gunsalus, K.C. MINE: Module Identification in Networks. BMC Bioinform. 2011, 12, 192. [Google Scholar] [CrossRef]
- Newman, A.M.; Liu, C.L.; Green, M.R.; Gentles, A.J.; Feng, W.; Xu, Y.; Hoang, C.D.; Diehn, M.; Alizadeh, A.A. Robust enumeration of cell subsets from tissue expression profiles. Nat. Methods 2015, 12, 453–457. [Google Scholar] [CrossRef]
- Ritchie, M.E.; Phipson, B.; Wu, D.; Hu, Y.; Law, C.W.; Shi, W.; Smyth, G.K. limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res. 2015, 43, e47. [Google Scholar] [CrossRef] [PubMed]
- Hu, K. Become Competent within One Day in Generating Boxplots and Violin Plots for a Novice without Prior R Experience. Methods Protoc. 2020, 3, 64. [Google Scholar] [CrossRef] [PubMed]
- Peña-Llopis, S.; Vega-Rubín-de-Celis, S.; Liao, A.; Leng, N.; Pavía-Jiménez, A.; Wang, S.; Yamasaki, T.; Zhrebker, L.; Sivanand, S.; Spence, P.; et al. BAP1 loss defines a new class of renal cell carcinoma. Nat. Genet. 2012, 44, 751–759. [Google Scholar] [CrossRef] [PubMed]
- Jin, S.; Wu, J.; Zhu, Y.; Gu, W.; Wan, F.; Xiao, W.; Dai, B.; Zhang, H.; Shi, G.; Shen, Y.; et al. Comprehensive Analysis of BAP1 Somatic Mutation in Clear Cell Renal Cell Carcinoma to Explore Potential Mechanisms in Silico. J. Cancer 2018, 9, 4108–4116. [Google Scholar] [CrossRef]
- Li, P.; Xiao, J.; Zhou, B.; Wei, J.; Luo, J.; Chen, W. SYNE1 mutation may enhance the response to immune checkpoint blockade therapy in clear cell renal cell carcinoma patients. Aging 2020, 12, 19316–19324. [Google Scholar] [CrossRef]
- Welcker, D.; Stein, C.; Feitosa, N.M.; Armistead, J.; Zhang, J.L.; Lütke, S.; Kleinridders, A.; Brüning, J.C.; Eming, S.A.; Sengle, G.; et al. Hemicentin-1 is an essential extracellular matrix component of the dermal-epidermal and myotendinous junctions. Sci. Rep. 2021, 11, 17926. [Google Scholar] [CrossRef]
- Fisher, S.A.; Rivera, A.; Fritsche, L.G.; Keilhauer, C.N.; Lichtner, P.; Meitinger, T.; Rudolph, G.; Weber, B.H. Case-control genetic association study of fibulin-6 (FBLN6 or HMCN1) variants in age-related macular degeneration (AMD). Hum. Mutat. 2007, 28, 406–413. [Google Scholar] [CrossRef]
- Liu, C.L.; Pan, H.W.; Torng, P.L.; Fan, M.H.; Mao, T.L. SRPX and HMCN1 regulate cancer-associated fibroblasts to promote the invasiveness of ovarian carcinoma. Oncol. Rep. 2019, 42, 2706–2715. [Google Scholar] [CrossRef]
- Elmas, A.; Lujambio, A.; Huang, K.L. Proteomic Analyses Identify Therapeutic Targets in Hepatocellular Carcinoma. Front. Oncol. 2022, 12, 814120. [Google Scholar] [CrossRef]
- Lee, S.H.; Je, E.M.; Yoo, N.J.; Lee, S.H. HMCN1, a cell polarity-related gene, is somatically mutated in gastric and colorectal cancers. Pathol. Oncol. Res. 2015, 21, 847–848. [Google Scholar] [CrossRef]
- Zhao, X.; Lei, Y.; Li, G.; Cheng, Y.; Yang, H.; Xie, L.; Long, H.; Jiang, R. Integrative analysis of cancer driver genes in prostate adenocarcinoma. Mol. Med. Rep. 2019, 19, 2707–2715. [Google Scholar] [CrossRef] [PubMed]
- Saravia, C.H.; Flores, C.; Schwarz, L.J.; Bravo, L.; Zavaleta, J.; Araujo, J.; Neciosup, S.; Pinto, J.A. Patterns of Mutation Enrichment in Metastatic Triple-Negative Breast Cancer. Clin. Med. Insights Oncol. 2019, 13, 1179554919868482. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Liu, F.; Zhang, Y.; Wu, X.; Wu, W.; Wang, X.A.; Zhao, S.; Liu, S.; Liang, H.; Zhang, F.; et al. Whole-genome sequencing reveals the mutational landscape of metastatic small-cell gallbladder neuroendocrine carcinoma (GB-SCNEC). Cancer Lett. 2017, 391, 20–27. [Google Scholar] [CrossRef]
- Chen, C.; Shi, C.; Huang, X.; Zheng, J.; Zhu, Z.; Li, Q.; Qiu, S.; Huang, Z.; Zhuang, Z.; Wu, R.; et al. Molecular Profiles and Metastasis Markers in Chinese Patients with Gastric Carcinoma. Sci. Rep. 2019, 9, 13995. [Google Scholar] [CrossRef]
- Januchowski, R.; Zawierucha, P.; Ruciński, M.; Zabel, M. Microarray-based detection and expression analysis of extracellular matrix proteins in drug-resistant ovarian cancer cell lines. Oncol. Rep. 2014, 32, 1981–1990. [Google Scholar] [CrossRef]
- Thompson, C.L.; Klein, B.E.; Klein, R.; Xu, Z.; Capriotti, J.; Joshi, T.; Leontiev, D.; Lee, K.E.; Elston, R.C.; Iyengar, S.K. Complement factor H and hemicentin-1 in age-related macular degeneration and renal phenotypes. Hum. Mol. Genet. 2007, 16, 2135–2148. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Abboud, H.E.; Pahl, M.V.; Tayek, J.; Snyder, S.; Tamkin, J.; Alcorn, H., Jr.; Ipp, E.; Nast, C.C.; Elston, R.C.; et al. Examination of association with candidate genes for diabetic nephropathy in a Mexican American population. Clin. J. Am. Soc. Nephrol. CJASN 2010, 5, 1072–1078. [Google Scholar] [CrossRef]
- Toffoli, B.; Zennaro, C.; Winkler, C.; Giordano Attianese, G.M.P.; Bernardi, S.; Carraro, M.; Gilardi, F.; Desvergne, B. Hemicentin 1 influences podocyte dynamic changes in glomerular diseases. Am. J. Physiol. Renal. Physiol. 2018, 314, F1154–F1165. [Google Scholar] [CrossRef]
- Labochka, D.; Moszczuk, B.; Kukwa, W.; Szczylik, C.; Czarnecka, A.M. Mechanisms through which diabetes mellitus influences renal cell carcinoma development and treatment: A review of the literature. Int. J. Mol. Med. 2016, 38, 1887–1894. [Google Scholar] [CrossRef][Green Version]
- Lee, H.; Kwak, C.; Kim, H.H.; Byun, S.S.; Lee, S.E.; Hong, S.K. Diabetes Mellitus as an Independent Predictor of Survival of Patients Surgically Treated for Renal Cell Carcinoma: A Propensity Score Matching Study. J. Urol. 2015, 194, 1554–1560. [Google Scholar] [CrossRef]
- DeBerardinis, R.J.; Chandel, N.S. Fundamentals of cancer metabolism. Sci. Adv. 2016, 2, e1600200. [Google Scholar] [CrossRef] [PubMed]
- Park, J.H.; Pyun, W.Y.; Park, H.W. Cancer Metabolism: Phenotype, Signaling and Therapeutic Targets. Cells 2020, 9, 2308. [Google Scholar] [CrossRef] [PubMed]
- Wettersten, H.I.; Aboud, O.A.; Lara, P.N., Jr.; Weiss, R.H. Metabolic reprogramming in clear cell renal cell carcinoma. Nat. Rev. Nephrol. 2017, 13, 410–419. [Google Scholar] [CrossRef] [PubMed]
- Ashton, T.M.; McKenna, W.G.; Kunz-Schughart, L.A.; Higgins, G.S. Oxidative Phosphorylation as an Emerging Target in Cancer Therapy. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2018, 24, 2482–2490. [Google Scholar] [CrossRef] [PubMed]
- Bonnay, F.; Veloso, A.; Steinmann, V.; Köcher, T.; Abdusselamoglu, M.D.; Bajaj, S.; Rivelles, E.; Landskron, L.; Esterbauer, H.; Zinzen, R.P.; et al. Oxidative Metabolism Drives Immortalization of Neural Stem Cells during Tumorigenesis. Cell 2020, 182, 1490–1507.e1419. [Google Scholar] [CrossRef]
- Martínez-Reyes, I.; Cardona, L.R.; Kong, H.; Vasan, K.; McElroy, G.S.; Werner, M.; Kihshen, H.; Reczek, C.R.; Weinberg, S.E.; Gao, P.; et al. Mitochondrial ubiquinol oxidation is necessary for tumour growth. Nature 2020, 585, 288–292. [Google Scholar] [CrossRef]
- Romani, P.; Valcarcel-Jimenez, L.; Frezza, C.; Dupont, S. Crosstalk between mechanotransduction and metabolism. Nat. Rev. Mol. Cell Biol. 2021, 22, 22–38. [Google Scholar] [CrossRef]
- Johnston, R.J.; Poholek, A.C.; DiToro, D.; Yusuf, I.; Eto, D.; Barnett, B.; Dent, A.L.; Craft, J.; Crotty, S. Bcl6 and Blimp-1 are reciprocal and antagonistic regulators of T follicular helper cell differentiation. Science 2009, 325, 1006–1010. [Google Scholar] [CrossRef]
- Nurieva, R.I.; Chung, Y.; Martinez, G.J.; Yang, X.O.; Tanaka, S.; Matskevitch, T.D.; Wang, Y.H.; Dong, C. Bcl6 mediates the development of T follicular helper cells. Science 2009, 325, 1001–1005. [Google Scholar] [CrossRef]
- Song, W.; Craft, J. T follicular helper cell heterogeneity: Time, space, and function. Immunol. Rev. 2019, 288, 85–96. [Google Scholar] [CrossRef]
- Crotty, S. T follicular helper cell differentiation, function, and roles in disease. Immunity 2014, 41, 529–542. [Google Scholar] [CrossRef] [PubMed]
- Hodi, F.S.; O’Day, S.J.; McDermott, D.F.; Weber, R.W.; Sosman, J.A.; Haanen, J.B.; Gonzalez, R.; Robert, C.; Schadendorf, D.; Hassel, J.C.; et al. Improved survival with ipilimumab in patients with metastatic melanoma. N. Engl. J. Med. 2010, 363, 711–723. [Google Scholar] [CrossRef] [PubMed]
- Borghaei, H.; Paz-Ares, L.; Horn, L.; Spigel, D.R.; Steins, M.; Ready, N.E.; Chow, L.Q.; Vokes, E.E.; Felip, E.; Holgado, E.; et al. Nivolumab versus Docetaxel in Advanced Nonsquamous Non-Small-Cell Lung Cancer. N. Engl. J. Med. 2015, 373, 1627–1639. [Google Scholar] [CrossRef] [PubMed]
- Garon, E.B.; Rizvi, N.A.; Hui, R.; Leighl, N.; Balmanoukian, A.S.; Eder, J.P.; Patnaik, A.; Aggarwal, C.; Gubens, M.; Horn, L.; et al. Pembrolizumab for the treatment of non-small-cell lung cancer. N. Engl. J. Med. 2015, 372, 2018–2028. [Google Scholar] [CrossRef]
- Darvin, P.; Toor, S.M.; Sasidharan Nair, V.; Elkord, E. Immune checkpoint inhibitors: Recent progress and potential biomarkers. Exp. Mol. Med. 2018, 50, 1–11. [Google Scholar] [CrossRef]
- Zhai, L.; Ladomersky, E.; Lenzen, A.; Nguyen, B.; Patel, R.; Lauing, K.L.; Wu, M.; Wainwright, D.A. IDO1 in cancer: A Gemini of immune checkpoints. Cell. Mol. Immunol. 2018, 15, 447–457. [Google Scholar] [CrossRef]
- Werner-Felmayer, G.; Werner, E.R.; Fuchs, D.; Hausen, A.; Reibnegger, G.; Wachter, H. Characteristics of interferon induced tryptophan metabolism in human cells in vitro. Biochim. Biophys. Acta 1989, 1012, 140–147. [Google Scholar] [CrossRef]
- Jitschin, R.; Braun, M.; Büttner, M.; Dettmer-Wilde, K.; Bricks, J.; Berger, J.; Eckart, M.J.; Krause, S.W.; Oefner, P.J.; Le Blanc, K.; et al. CLL-cells induce IDOhi CD14+HLA-DRlo myeloid-derived suppressor cells that inhibit T-cell responses and promote TRegs. Blood 2014, 124, 750–760. [Google Scholar] [CrossRef]
- Yu, J.; Du, W.; Yan, F.; Wang, Y.; Li, H.; Cao, S.; Yu, W.; Shen, C.; Liu, J.; Ren, X. Myeloid-derived suppressor cells suppress antitumor immune responses through IDO expression and correlate with lymph node metastasis in patients with breast cancer. J. Immunol. 2013, 190, 3783–3797. [Google Scholar] [CrossRef]
- Tang, K.; Wu, Y.H.; Song, Y.; Yu, B. Indoleamine 2,3-dioxygenase 1 (IDO1) inhibitors in clinical trials for cancer immunotherapy. J. Hematol. Oncol. 2021, 14, 68. [Google Scholar] [CrossRef]
- Munn, D.H.; Mellor, A.L. Indoleamine 2,3 dioxygenase and metabolic control of immune responses. Trends Immunol. 2013, 34, 137–143. [Google Scholar] [CrossRef] [PubMed]
- Ball, H.J.; Sanchez-Perez, A.; Weiser, S.; Austin, C.J.; Astelbauer, F.; Miu, J.; McQuillan, J.A.; Stocker, R.; Jermiin, L.S.; Hunt, N.H. Characterization of an indoleamine 2,3-dioxygenase-like protein found in humans and mice. Gene 2007, 396, 203–213. [Google Scholar] [CrossRef] [PubMed]



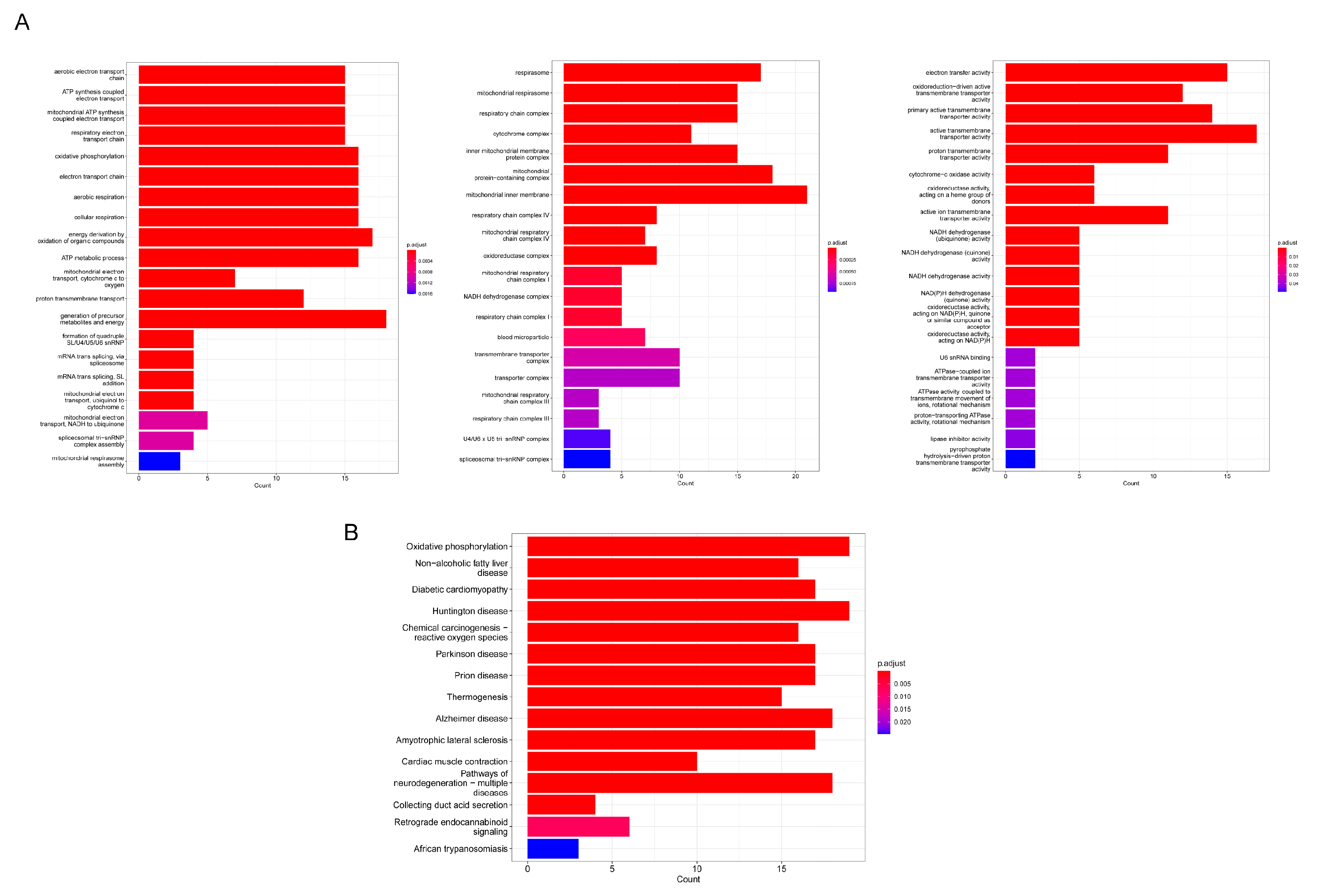
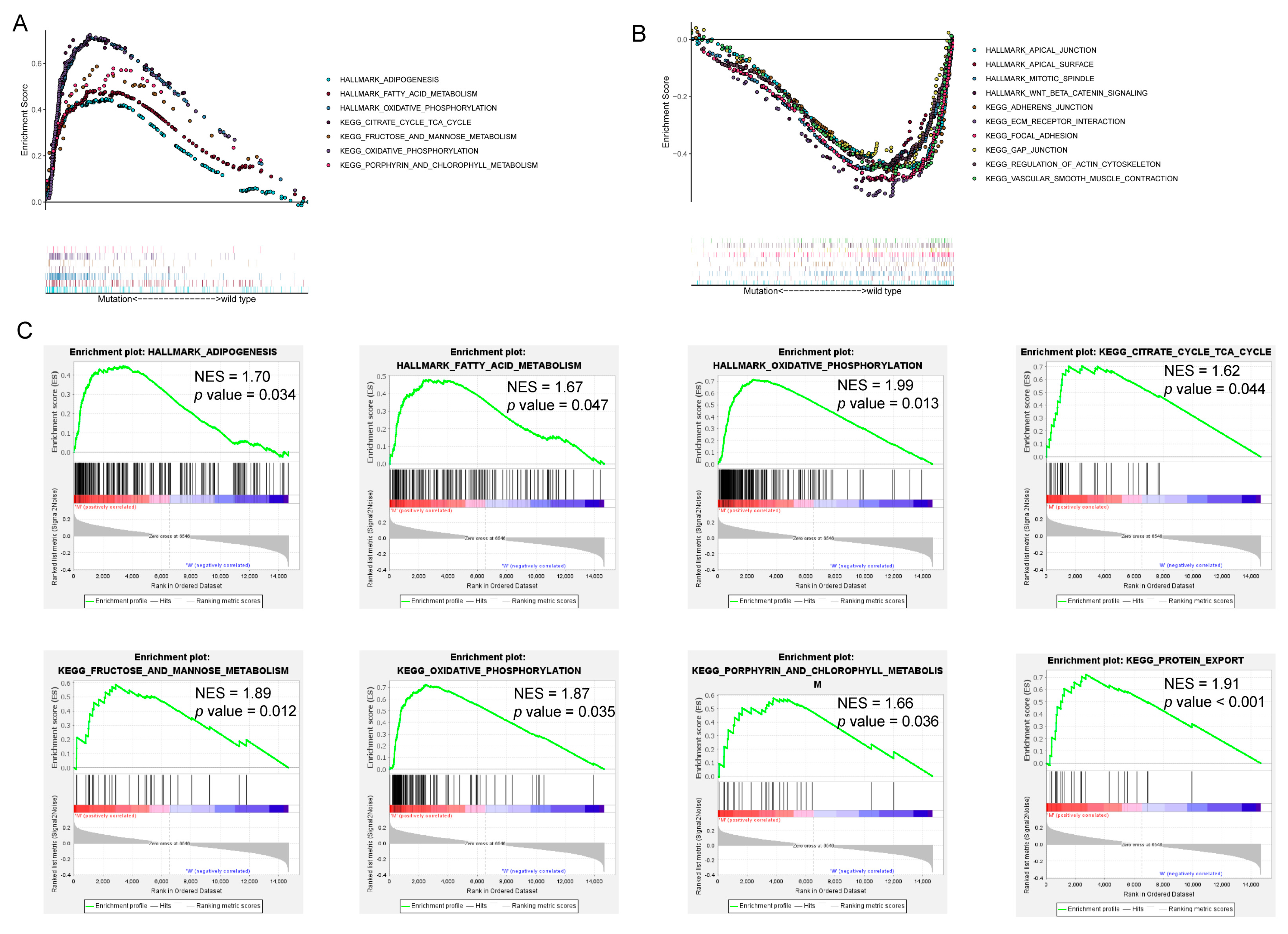
| ID | Description | p-Value | Genes |
|---|---|---|---|
| GO | |||
| GO:0006119 | Oxidative phosphorylation | 6.61 × 10−33 | COX7B/COX8A/COX5A/CYC1/COX4I1/CYCS/NDUFB9/COX6A1/COX7A2/COX7A1/NDUFB7/UQCR11/NDUFA4/NDUFA2/UQCRQ |
| GO:0019646 | Aerobic electron transport chain | 1.09 × 10−32 | COX8A/COX5A/CYC1/COX4I1/CYCS/NDUFB9/COX6A1/COX7A2/COX7A1/NDUFB7/UQCR11/NDUFA4/NDUFA2/UQCRQ |
| GO:0042773 | ATP synthesis coupled electron transport | 4.11 × 10−32 | COX8A/COX5A/CYC1/COX4I1/CYCS/NDUFB9/COX6A1/COX7A2/COX7A1/NDUFB7/UQCR11/NDUFA4/NDUFA2/UQCRQ |
| KEGG | |||
| hsa00190 | Oxidative phosphorylation | 7.91 × 10−28 | COX7B/COX8A/COX5A/CYC1/COX4I1/CYCS/NDUFB9/COX6A1/COX7A2/COX7A1/NDUFB7/UQCR11/NDUFA4/NDUFA2/UQCRQ |
| hsa04932 | Non-alcoholic fatty liver disease | 7.87 × 10−27 | COX7B/COX8A/COX5A/CYC1/COX4I1/CYCS/NDUFB9/COX6A1/COX7A2/COX7A1/NDUFB7/UQCR11/NDUFA4/NDUFA2/UQCRQ |
| hsa05012 | Parkinson disease | 3.49 × 10−23 | COX7B/COX8A/COX5A/CYC1/COX4I1/CYCS/NDUFB9/COX6A1/COX7A2/COX7A1/NDUFB7/UQCR11/NDUFA4/NDUFA2/UQCRQ |
| ID | Description | p-Value | Genes |
|---|---|---|---|
| GO | |||
| GO:0051346 | Negative regulation of hydrolase activity | 6.72 × 10−8 | VTN/SERPINC1/APOA1/APOC3/AMBP |
| GO:0035376 | Sterol import | 5.38 × 10−6 | APOA1/APOC3 |
| GO:0070508 | Cholesterol import | 5.38 × 10−6 | APOA1/APOC3 |
| KEGG | |||
| hsa04979 | Cholesterol metabolism | 0.00054577 | APOA1/APOC3 |
| hsa03320 | PPAR signaling pathway | 0.00122622 | APOA1/APOC3 |
| hsa04610 | Complement and coagulation cascades | 0.001572335 | VTN/SERPINC1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gong, Z.; Wu, X.; Guo, Q.; Du, H.; Zhang, F.; Kong, Y. Comprehensive Analysis of HMCN1 Somatic Mutation in Clear Cell Renal Cell Carcinoma. Genes 2022, 13, 1282. https://doi.org/10.3390/genes13071282
Gong Z, Wu X, Guo Q, Du H, Zhang F, Kong Y. Comprehensive Analysis of HMCN1 Somatic Mutation in Clear Cell Renal Cell Carcinoma. Genes. 2022; 13(7):1282. https://doi.org/10.3390/genes13071282
Chicago/Turabian StyleGong, Ziqi, Xiaowen Wu, Qian Guo, Haizhen Du, Fenghao Zhang, and Yan Kong. 2022. "Comprehensive Analysis of HMCN1 Somatic Mutation in Clear Cell Renal Cell Carcinoma" Genes 13, no. 7: 1282. https://doi.org/10.3390/genes13071282
APA StyleGong, Z., Wu, X., Guo, Q., Du, H., Zhang, F., & Kong, Y. (2022). Comprehensive Analysis of HMCN1 Somatic Mutation in Clear Cell Renal Cell Carcinoma. Genes, 13(7), 1282. https://doi.org/10.3390/genes13071282





