When a Synonymous Variant Is Nonsynonymous
Abstract
:1. Introduction
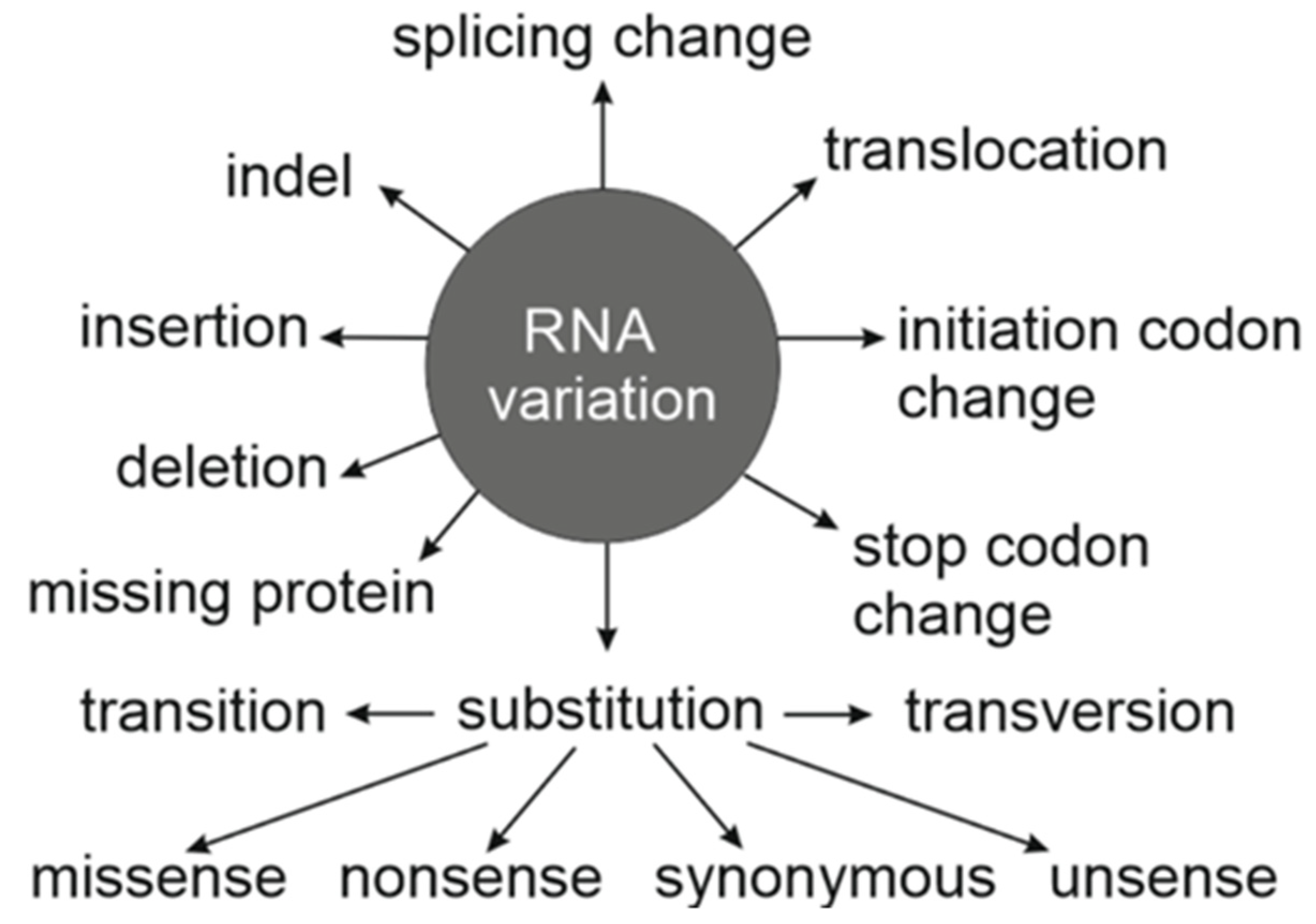
2. Consequences of Synonymous Variants
3. Problematic Databases and Annotations
4. Systematic Nomenclature
5. Discussion
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Vihinen, M. Muddled genetic terms miss and mess the message. Trends Genet. 2015, 31, 423–425. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vihinen, M. Variation Ontology for annotation of variation effects and mechanisms. Genome Res. 2014, 24, 356–364. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Den Dunnen, J.T.; Antonarakis, S.E. Mutation nomenclature extensions and suggestions to describe complex mutations: A discussion. Hum. Mutat. 2000, 15, 7–12. [Google Scholar] [CrossRef]
- Vihinen, M. Systematics for types and effects of RNA variations. RNA Biol. 2021, 18, 481–498. [Google Scholar] [CrossRef]
- Shabalina, S.A.; Spiridonov, N.A.; Kashina, A. Sounds of silence: Synonymous nucleotides as a key to biological regulation and complexity. Nucleic Acids Res. 2013, 41, 2073–2094. [Google Scholar] [CrossRef] [Green Version]
- Sauna, Z.E.; Kimchi-Sarfaty, C. Understanding the contribution of synonymous mutations to human disease. Nat. Rev. Genet. 2011, 12, 683–691. [Google Scholar] [CrossRef]
- Bali, V.; Bebok, Z. Decoding mechanisms by which silent codon changes influence protein biogenesis and function. Int. J. Biochem. Cell Biol. 2015, 64, 58–74. [Google Scholar] [CrossRef] [Green Version]
- Stergachis, A.B.; Haugen, E.; Shafer, A.; Fu, W.; Vernot, B.; Reynolds, A.; Raubitschek, A.; Ziegler, S.; LeProust, E.M.; Akey, J.M.; et al. Exonic transcription factor binding directs codon choice and affects protein evolution. Science 2013, 342, 1367–1372. [Google Scholar] [CrossRef] [Green Version]
- Kim, Y.J.; Kang, J.; Seymen, F.; Koruyucu, M.; Zhang, H.; Kasimoglu, Y.; Bayram, M.; Tuna-Ince, E.B.; Bayrak, S.; Tuloglu, N.; et al. Alteration of exon definition causes amelogenesis imperfecta. J. Dent. Res. 2020, 99, 410–418. [Google Scholar] [CrossRef]
- Nielsen, K.B.; Sørensen, S.; Cartegni, L.; Corydon, T.J.; Doktor, T.K.; Schroeder, L.D.; Reinert, L.S.; Elpeleg, O.; Krainer, A.R.; Gregersen, N.; et al. Seemingly neutral polymorphic variants may confer immunity to splicing-inactivating mutations: A synonymous SNP in exon 5 of MCAD protects from deleterious mutations in a flanking exonic splicing enhancer. Am. J. Hum. Genet. 2007, 80, 416–432. [Google Scholar] [CrossRef] [Green Version]
- Pozzoli, U.; Sironi, M. Silencers regulate both constitutive and alternative splicing events in mammals. Cell Mol. Life Sci. 2005, 62, 1579–1604. [Google Scholar] [CrossRef] [PubMed]
- Kashima, T.; Manley, J.L. A negative element in SMN2 exon 7 inhibits splicing in spinal muscular atrophy. Nat. Genet. 2003, 34, 460–463. [Google Scholar] [CrossRef] [PubMed]
- Singh, N.N.; Androphy, E.J.; Singh, R.N. An extended inhibitory context causes skipping of exon 7 of SMN2 in spinal muscular atrophy. Biochem. Biophys. Res. Commun. 2004, 315, 381–388. [Google Scholar] [CrossRef] [PubMed]
- Kurosaki, T.; Popp, M.W.; Maquat, L.E. Quality and quantity control of gene expression by nonsense-mediated mRNA decay. Nat. Rev. Mol. Cell Biol. 2019, 20, 406–420. [Google Scholar] [CrossRef]
- Supek, F.; Miñana, B.; Valcárcel, J.; Gabaldón, T.; Lehner, B. Synonymous mutations frequently act as driver mutations in human cancers. Cell 2014, 156, 1324–1335. [Google Scholar] [CrossRef] [Green Version]
- Lindeboom, R.G.; Supek, F.; Lehner, B. The rules and impact of nonsense-mediated mRNA decay in human cancers. Nat. Genet. 2016, 48, 1112–1118. [Google Scholar] [CrossRef] [Green Version]
- Nackley, A.G.; Shabalina, S.A.; Tchivileva, I.E.; Satterfield, K.; Korchynskyi, O.; Makarov, S.S.; Maixner, W.; Diatchenko, L. Human catechol-O-methyltransferase haplotypes modulate protein expression by altering mRNA secondary structure. Science 2006, 314, 1930–1933. [Google Scholar] [CrossRef] [Green Version]
- Forman, J.J.; Coller, H.A. The code within the code: MicroRNAs target coding regions. Cell Cycle 2010, 9, 1533–1541. [Google Scholar] [CrossRef] [Green Version]
- Brest, P.; Lapaquette, P.; Souidi, M.; Lebrigand, K.; Cesaro, A.; Vouret-Craviari, V.; Mari, B.; Barbry, P.; Mosnier, J.F.; Hébuterne, X.; et al. A synonymous variant in IRGM alters a binding site for miR-196 and causes deregulation of IRGM-dependent xenophagy in Crohn’s disease. Nat. Genet. 2011, 43, 242–245. [Google Scholar] [CrossRef]
- Plotkin, J.B.; Kudla, G. Synonymous but not the same: The causes and consequences of codon bias. Nat. Rev. Genet. 2011, 12, 32–42. [Google Scholar] [CrossRef] [Green Version]
- Mitra, S.; Ray, S.K.; Banerjee, R. Synonymous codons influencing gene expression in organisms. Res. Rep. Biochem. 2016, 6, 57–65. [Google Scholar] [CrossRef] [Green Version]
- Pechmann, S.; Frydman, J. Evolutionary conservation of codon optimality reveals hidden signatures of cotranslational folding. Nat. Struct. Mol. Biol. 2013, 20, 237–243. [Google Scholar] [CrossRef] [Green Version]
- Buhr, F.; Jha, S.; Thommen, M.; Mittelstaet, J.; Kutz, F.; Schwalbe, H.; Rodnina, M.V.; Komar, A.A. Synonymous codons direct cotranslational folding toward different protein conformations. Mol. Cell 2016, 61, 341–351. [Google Scholar] [CrossRef] [Green Version]
- Kimchi-Sarfaty, C.; Oh, J.M.; Kim, I.W.; Sauna, Z.E.; Calcagno, A.M.; Ambudkar, S.V.; Gottesman, M.M. A “silent” polymorphism in the MDR1 gene changes substrate specificity. Science 2007, 315, 525–528. [Google Scholar] [CrossRef] [Green Version]
- Hunt, R.; Hettiarachchi, G.; Katneni, U.; Hernandez, N.; Holcomb, D.; Kames, J.; Alnifaidy, R.; Lin, B.; Hamasaki-Katagiri, N.; Wesley, A.; et al. A single synonymous variant (c.354G>A [p.P118P]) in ADAMTS13 confers enhanced specific activity. Int. J. Mol. Sci. 2019, 20, 5734. [Google Scholar] [CrossRef] [Green Version]
- Simhadri, V.L.; Hamasaki-Katagiri, N.; Lin, B.C.; Hunt, R.; Jha, S.; Tseng, S.C.; Wu, A.; Bentley, A.A.; Zichel, R.; Lu, Q.; et al. Single synonymous mutation in factor IX alters protein properties and underlies haemophilia B. J. Med. Genet. 2017, 54, 338–345. [Google Scholar] [CrossRef]
- Karakostis, K.; Vadivel Gnanasundram, S.; López, I.; Thermou, A.; Wang, L.; Nylander, K.; Olivares-Illana, V.; Fåhraeus, R. A single synonymous mutation determines the phosphorylation and stability of the nascent protein. J. Mol. Cell Biol. 2019, 11, 187–199. [Google Scholar] [CrossRef]
- Vihinen, M. Functional effects of protein variants. Biochimie 2021, 180, 104–120. [Google Scholar] [CrossRef]
- Wang, K.; Li, M.; Hakonarson, H. ANNOVAR: Functional annotation of genetic variants from high-throughput sequencing data. Nucleic Acids Res. 2010, 38, e164. [Google Scholar] [CrossRef]
- Cingolani, P.; Platts, A.; Wang, L.L.; Coon, M.; Nguyen, T.; Wang, L.; Land, S.J.; Lu, X.; Ruden, D.M. A program for annotating and predicting the effects of single nucleotide polymorphisms, SnpEff: SNPs in the genome of Drosophila melanogaster strain w1118; iso-2; iso-3. Fly 2012, 6, 80–92. [Google Scholar] [CrossRef] [Green Version]
- Lefter, M.; Vis, J.K.; Vermaat, M.; den Dunnen, J.T.; Taschner, P.E.M.; Laros, J.F.J. Next generation HGVS nomenclature checker. Bioinformatics 2021, 37, 2811–2817. [Google Scholar] [CrossRef]
- Freeman, P.J.; Hart, R.K.; Gretton, L.J.; Brookes, A.J.; Dalgleish, R. VariantValidator: Accurate validation, mapping, and formatting of sequence variation descriptions. Hum. Mutat. 2018, 39, 61–68. [Google Scholar] [CrossRef] [Green Version]
- Hart, R.K.; Rico, R.; Hare, E.; Garcia, J.; Westbrook, J.; Fusaro, V.A. A Python package for parsing, validating, mapping and formatting sequence variants using HGVS nomenclature. Bioinformatics 2015, 31, 268–270. [Google Scholar] [CrossRef] [Green Version]
- McLaren, W.; Gil, L.; Hunt, S.E.; Riat, H.S.; Ritchie, G.R.; Thormann, A.; Flicek, P.; Cunningham, F. The Ensembl Variant Effect Predictor. Genome Biol. 2016, 17, 122. [Google Scholar] [CrossRef] [Green Version]
- Miao, X.; Li, X.; Wang, L.; Zheng, C.; Cai, J. DSMNC: A database of somatic mutations in normal cells. Nucleic Acids Res. 2019, 47, D971–D975. [Google Scholar] [CrossRef]
- Sharma, Y.; Miladi, M.; Dukare, S.; Boulay, K.; Caudron-Herger, M.; Groß, M.; Backofen, R.; Diederichs, S. A pan-cancer analysis of synonymous mutations. Nat. Commun. 2019, 10, 2569. [Google Scholar] [CrossRef] [Green Version]
- Wen, P.; Xiao, P.; Xia, J. dbDSM: A manually curated database for deleterious synonymous mutations. Bioinformatics 2016, 32, 1914–1916. [Google Scholar] [CrossRef]
- Fokkema, I.; Kroon, M.; López Hernández, J.A.; Asscheman, D.; Lugtenburg, I.; Hoogenboom, J.; den Dunnen, J.T. The LOVD3 platform: Efficient genome-wide sharing of genetic variants. Eur. J. Hum. Genet. 2021, 29, 1796–1803. [Google Scholar] [CrossRef]
- Bin, Y.; Wang, X.; Zhao, L.; Wen, P.; Xia, J. An analysis of mutational signatures of synonymous mutations across 15 cancer types. BMC Med. Genet. 2019, 20, 190. [Google Scholar] [CrossRef]
- Gelfman, S.; Wang, Q.; McSweeney, K.M.; Ren, Z.; La Carpia, F.; Halvorsen, M.; Schoch, K.; Ratzon, F.; Heinzen, E.L.; Boland, M.J.; et al. Annotating pathogenic non-coding variants in genic regions. Nat. Commun. 2017, 8, 236. [Google Scholar] [CrossRef]
- Tang, X.; Zhang, T.; Cheng, N.; Wang, H.; Zheng, C.H.; Xia, J.; Zhang, T. usDSM: A novel method for deleterious synonymous mutation prediction using undersampling scheme. Brief. Bioinform. 2021, 22, bbab123. [Google Scholar] [CrossRef]
- Livingstone, M.; Folkman, L.; Yang, Y.; Zhang, P.; Mort, M.; Cooper, D.N.; Liu, Y.; Stantic, B.; Zhou, Y. Investigating DNA-, RNA-, and protein-based features as a means to discriminate pathogenic synonymous variants. Hum. Mutat. 2017, 38, 1336–1347. [Google Scholar] [CrossRef]
- Mueller, W.F.; Larsen, L.S.; Garibaldi, A.; Hatfield, G.W.; Hertel, K.J. The silent sway of splicing by synonymous substitutions. J. Biol. Chem. 2015, 290, 27700–27711. [Google Scholar] [CrossRef] [Green Version]
- Rhine, C.L.; Neil, C.; Wang, J.; Maguire, S.; Buerer, L.; Salomon, M.; Meremikwu, I.C.; Kim, J.; Strande, N.T.; Fairbrother, W.G. Massively parallel reporter assays discover de novo exonic splicing mutants in paralogs of Autism genes. PLoS Genet. 2022, 18, e1009884. [Google Scholar] [CrossRef]
- Wehr, C.; Grotius, K.; Casadei, S.; Bleckmann, D.; Bode, S.F.N.; Frye, B.C.; Seidl, M.; Gulsuner, S.; King, M.C.; Percival, M.B.; et al. A novel disease-causing synonymous exonic mutation in GATA2 affecting RNA splicing. Blood 2018, 132, 1211–1215. [Google Scholar] [CrossRef]
- Tonin, R.; Catarzi, S.; Caciotti, A.; Procopio, E.; Marini, C.; Guerrini, R.; Morrone, A. Progressive myoclonus epilepsy in Gaucher Disease due to a new Gly-Gly mutation causing loss of an Exonic Splicing Enhancer. J. Neurol. 2019, 266, 92–101. [Google Scholar] [CrossRef] [Green Version]
- Tay, Y.; Zhang, J.; Thomson, A.M.; Lim, B.; Rigoutsos, I. MicroRNAs to Nanog, Oct4 and Sox2 coding regions modulate embryonic stem cell differentiation. Nature 2008, 455, 1124–1128. [Google Scholar] [CrossRef]
- Sant, D.W.; Sinclair, M.; Mungall, C.J.; Schulz, S.; Zerbino, D.; Lovering, R.C.; Logie, C.; Eilbeck, K. Sequence Ontology terminology for gene regulation. Biochim. Biophys. Acta Gene Regul. Mech. 2021, 1864, 194745. [Google Scholar] [CrossRef]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vihinen, M. When a Synonymous Variant Is Nonsynonymous. Genes 2022, 13, 1485. https://doi.org/10.3390/genes13081485
Vihinen M. When a Synonymous Variant Is Nonsynonymous. Genes. 2022; 13(8):1485. https://doi.org/10.3390/genes13081485
Chicago/Turabian StyleVihinen, Mauno. 2022. "When a Synonymous Variant Is Nonsynonymous" Genes 13, no. 8: 1485. https://doi.org/10.3390/genes13081485




