Identification of Epigenetic Interactions between miRNA and Gene Expression as Potential Prognostic Markers in Bladder Cancer
Abstract
:1. Introduction
2. Patients and Methods
2.1. Patients and Sample Collection
2.2. Immunohistochemical Examination
2.3. Real-Time PCR for Studying Gene and Target miRNA
2.4. Outcome
2.5. Statistical Analysis
3. Results
3.1. Demographics
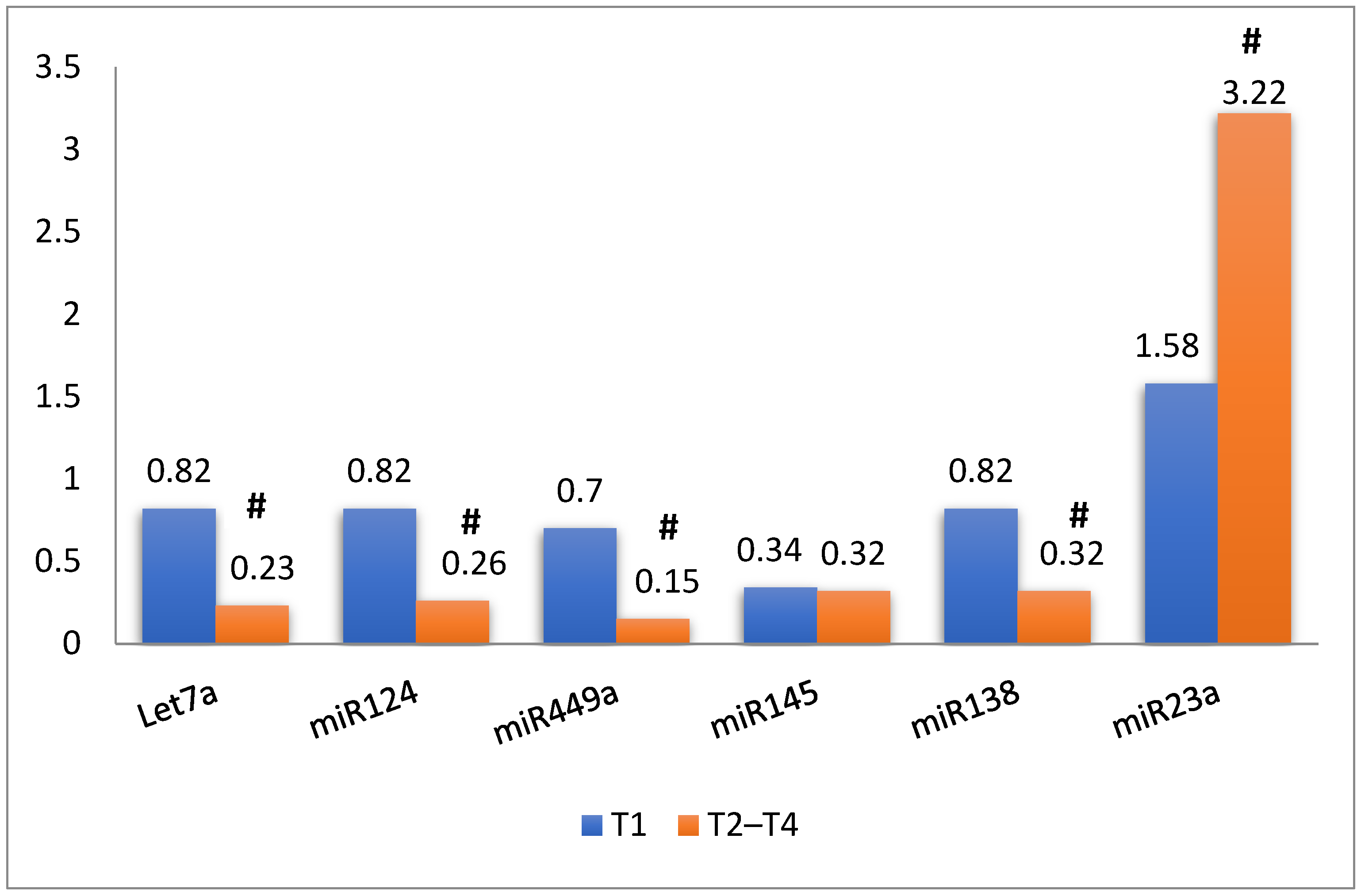
3.2. MiRNAs and Target mRNA Gene Expression
3.3. Correlation between miRNAs and Their Target Genes
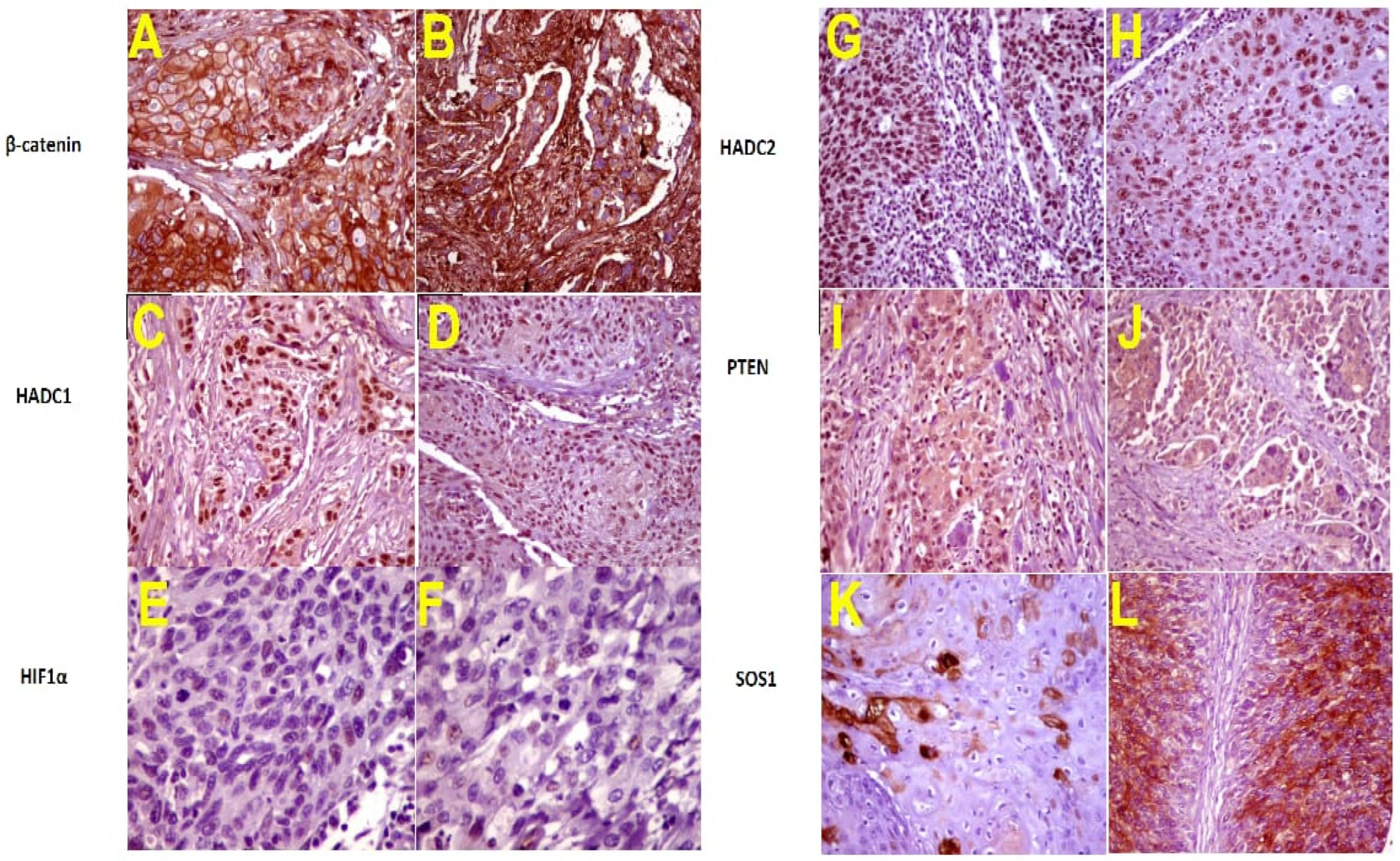
3.4. Immunohistochemistry
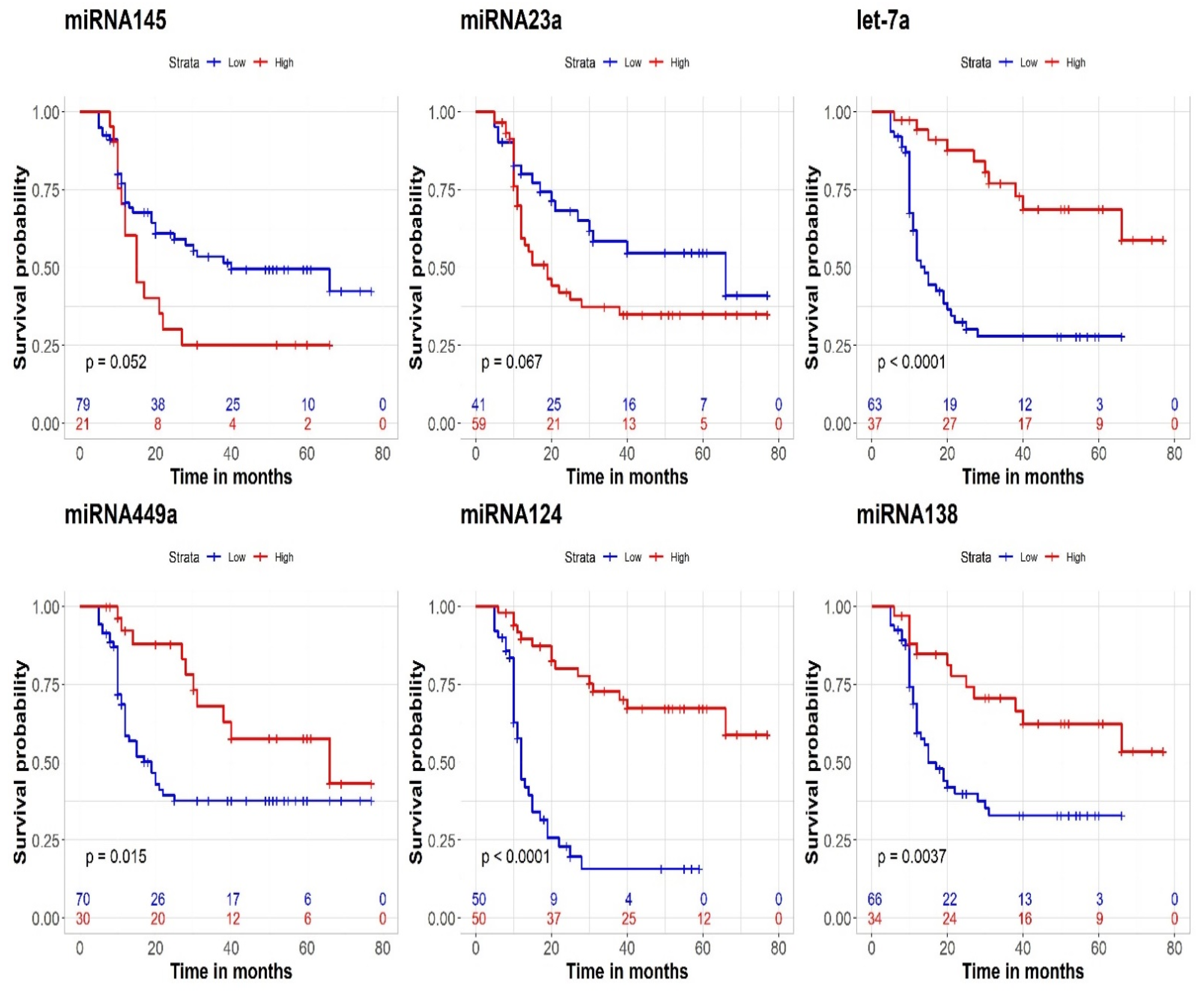
3.5. Prognostic Effect on CSS
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Antoni, S.; Ferlay, J.; Soerjomataram, I.; Znaor, A.; Jemal, A.; Bray, F. Bladder Cancer Incidence and Mortality: A Global Overview and Recent Trends. Eur. Urol. 2017, 71, 96–108. [Google Scholar] [CrossRef] [PubMed]
- Guo, C.C.; Czerniak, B. Bladder Cancer in the Genomic Era. Arch. Pathol. Lab. Med. 2019, 143, 695–704. [Google Scholar] [CrossRef] [PubMed]
- Robertson, A.G.; Kim, J.; Al-Ahmadie, H.; Bellmunt, J.; Guo, G.; Cherniack, A.D.; Hinoue, T.; Laird, P.W.; Hoadley, K.A.; Akbani, R.; et al. Comprehensive Molecular Characterization of Muscle-Invasive Bladder Cancer. Cell 2017, 171, 540–556.e25. [Google Scholar] [CrossRef] [PubMed]
- Braicu, C.; Cojocneanu-Petric, R.; Chira, S.; Truta, A.; Floares, A.; Achimas-Cadariu, P.; Berindan-Neagoe, I.; Petrut, B. Clinical and pathological implications of miRNA in bladder cancer. Int. J. Nanomed. 2015, 10, 791–800. [Google Scholar] [CrossRef] [PubMed]
- Veerla, S.; Lindgren, D.; Kvist, A.; Frigyesi, A.; Staaf, J.; Persson, H.; Liedberg, F.; Chebil, G.; Gudjonsson, S.; Borg, Å.; et al. MiRNA expression in urothelial carcinomas: Important roles of miR-10a, miR-222, miR-125b, miR-7 and miR-452 for tumor stage and metastasis, and frequent homozygous losses of miR-31. Int. J. Cancer 2009, 124, 2236–2242. [Google Scholar] [CrossRef]
- Zhu, H.; Ng, S.-C.; Segrè, A.V.; Shinoda, G.; Shah, S.P.; Einhorn, W.S.; Takeuchi, A.; Engreitz, J.M.; Hagan, J.P.; Kharas, M.G.; et al. The Lin28/let-7 Axis Regulates Glucose Metabolism. Cell 2011, 147, 81–94. [Google Scholar] [CrossRef]
- Catto, J.W.F.; Miah, S.; Owen, H.C.; Bryant, H.; Myers, K.; Dudziec, E.; Larré, S.; Milo, M.; Rehman, I.; Rosario, D.J.; et al. Distinct MicroRNA Alterations Characterize High- and Low-Grade Bladder Cancer. Cancer Res. 2009, 69, 8472–8481. [Google Scholar] [CrossRef]
- Giannopoulou, A.F.; Velentzas, A.D.; Konstantakou, E.G.; Avgeris, M.; Katarachia, S.A.; Papandreou, N.C.; Kalavros, N.I.; Mpakou, V.E.; Iconomidou, V.; Anastasiadou, E.; et al. Revisiting Histone Deacetylases in Human Tumorigenesis: The Paradigm of Urothelial Bladder Cancer. Int. J. Mol. Sci. 2019, 20, 1291. [Google Scholar] [CrossRef]
- Porkka, K.P.; Pfeiffer, M.J.; Waltering, K.K.; Vessella, R.L.; Tammela, T.L.; Visakorpi, T. MicroRNA Expression Profiling in Prostate Cancer. Cancer Res. 2007, 67, 6130–6135. [Google Scholar] [CrossRef]
- Chen, H.; Lin, Y.-W.; Mao, Y.-Q.; Wu, J.; Liu, Y.-F.; Zheng, X.-Y.; Xie, L.-P. MicroRNA-449a acts as a tumor suppressor in human bladder cancer through the regulation of pocket proteins. Cancer Lett. 2012, 320, 40–47. [Google Scholar] [CrossRef]
- Rosenberg, E.; Baniel, J.; Spector, Y.; Faerman, A.; Meiri, E.; Aharonov, R.; Margel, D.; Goren, Y.; Nativ, O. Predicting progression of bladder urothelial carcinoma using microRNA expression. Br. J. Urol. 2013, 112, 1027–1034. [Google Scholar] [CrossRef] [PubMed]
- Bastos, E.P.; Brentani, H.; Pereira, C.A.B.; Polpo, A.; Lima, L.; Puga, R.D.; Pasini, F.S.; Osorio, C.A.B.T.; Roela, R.A.; Achatz, M.I.; et al. A Set of miRNAs, Their Gene and Protein Targets and Stromal Genes Distinguish Early from Late Onset ER Positive Breast Cancer. PLoS ONE 2016, 11, e0154325. [Google Scholar] [CrossRef]
- Baek, D.; Villén, J.; Shin, C.; Camargo, F.D.; Gygi, S.P.; Bartel, D.P. The impact of microRNAs on protein output. Nature 2008, 455, 64–71. [Google Scholar] [CrossRef]
- Uhlmann, S.; Mannsperger, H.; Zhang, J.D.; Horvat, E.; Schmidt, C.; Küblbeck, M.; Henjes, F.; Ward, A.; Tschulena, U.; Zweig, K.; et al. Global microRNA level regulation of EGFR-driven cell-cycle protein network in breast cancer. Mol. Syst. Biol. 2012, 8, 570. [Google Scholar] [CrossRef] [PubMed]
- Epstein, J.I.; Amin, M.B.; Reuter, V.R.; Mostofi, F.K. The World Health Organization/International Society of Urological Pathology Consensus Classification of Urothelial (Transitional Cell) Neoplasms of the Urinary Bladder. Am. J. Surg. Pathol. 1998, 22, 1435–1448. [Google Scholar] [CrossRef]
- Barakat, L.A.; Barakat, N.; Zakaria, M.M.; Khirallah, S.M. Protective role of zinc oxide nanoparticles in kidney injury induced by cisplatin in rats. Life Sci. 2020, 262, 118503. [Google Scholar] [CrossRef]
- Delgado, J.; Pereira, A.; Villamor, N.; López-Guillermo, A.; Rozman, C. Survival analysis in hematologic malignancies: Recommendations for clinicians. Haematologica 2014, 99, 1410–1420. [Google Scholar] [CrossRef]
- Dyrskjøt, L.; Ostenfeld, M.S.; Bramsen, J.B.; Silahtaroglu, A.N.; Lamy, P.; Ramanathan, R.; Fristrup, N.; Jensen, J.L.; Andersen, C.L.; Zieger, K.; et al. Genomic Profiling of MicroRNAs in Bladder Cancer: miR-129 Is Associated with Poor Outcome and Promotes Cell Death In vitro. Cancer Res. 2009, 69, 4851–4860. [Google Scholar] [CrossRef]
- Wszolek, M.F.; Rieger-Christ, K.M.; Kenney, P.A.; Gould, J.J.; Neto, B.S.; LaVoie, A.K.; Logvinenko, T.; Libertino, J.A.; Summerhayes, I.C. A MicroRNA expression profile defining the invasive bladder tumor phenotype. Urol. Oncol. Semin. Orig. Investig. 2011, 29, 794–801.e1. [Google Scholar] [CrossRef]
- Vinall, R.L.; Tepper, C.G.; Ripoll, A.A.; Gandour-Edwards, R.F.; Durbin-Johnson, B.P.; Yap, S.A.; Ghosh, P.M.; White, R.W.D. Decreased expression of let-7c is associated with non-response of muscle-invasive bladder cancer patients to neoadjuvant chemotherapy. Genes Cancer 2016, 7, 86–97. [Google Scholar] [CrossRef] [Green Version]
- Xu, Z.; Yu, Y.-Q.; Ge, Y.-Z.; Zhu, J.-G.; Zhu, M.; Zhao, Y.-C.; Xu, L.-W.; Yang, X.-B.; Geng, L.-G.; Dou, Q.-L.; et al. MicroRNA expression profiles in muscle-invasive bladder cancer: Identification of a four-microRNA signature associated with patient survival. Tumor Biol. 2015, 36, 8159–8166. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Tang, K.; Xiao, H.; Zeng, J.; Guan, W.; Guo, X.; Xu, H.; Ye, Z. A panel of eight-miRNA signature as a potential biomarker for predicting survival in bladder cancer. J. Exp. Clin. Cancer Res. 2015, 34, 53. [Google Scholar] [CrossRef] [PubMed]
- Shi, X.-B.; Xue, L.; Ma, A.-H.; Tepper, C.G.; Gandour-Edwards, R.; Kung, H.-J.; White, R.W.D. Tumor suppressive miR-124 targets androgen receptor and inhibits proliferation of prostate cancer cells. Oncogene 2013, 32, 4130–4138. [Google Scholar] [CrossRef]
- Zhang, T.; Wang, J.; Zhai, X.; Li, H.; Li, C.; Chang, J. MiR-124 retards bladder cancer growth by directly targeting CDK4. Acta Biochim. Biophys. Sin. 2014, 46, 1072–1079. [Google Scholar] [CrossRef] [PubMed]
- Bou Kheir, T.; Futoma-Kazmierczak, E.; Jacobsen, A.; Krogh, A.; Bardram, L.; Hother, C.; Grønbæk, K.; Federspiel, B.; Lund, A.H.; Friis-Hansen, L. miR-449 inhibits cell proliferation and is down-regulated in gastric cancer. Mol. Cancer 2011, 10, 29. [Google Scholar] [CrossRef] [PubMed]
- Lizé, M.; Pilarski, S.; Dobbelstein, M.; Liz, M. E2F1-inducible microRNA 449a/b suppresses cell proliferation and promotes apoptosis. Cell Death Differ. 2010, 17, 452–458. [Google Scholar] [CrossRef]
- Friedman, J.M.; Liang, G.; Liu, C.-C.; Wolff, E.M.; Tsai, Y.C.; Ye, W.; Zhou, X.; Jones, P.A. The Putative Tumor Suppressor microRNA-101 Modulates the Cancer Epigenome by Repressing the Polycomb Group Protein EZH2. Cancer Res. 2009, 69, 2623–2629. [Google Scholar] [CrossRef] [PubMed]
- Sachdeva, M.; Mo, Y.-Y. miR-145-mediated suppression of cell growth, invasion and metastasis. Am. J. Transl. Res. 2010, 2, 170–180. [Google Scholar]
- Chen, Y.; Cao, K.; Wang, S.; Chen, J.; He, B.; He, G.; Chen, Y.; Peng, B.; Zhou, J. MicroRNA-138 suppresses proliferation, invasion and glycolysis in malignant melanoma cells by targeting HIF-1α. Exp. Ther. Med. 2016, 11, 2513–2518. [Google Scholar] [CrossRef]
- Zaman, M.S.; Thamminana, S.; Shahryari, V.; Chiyomaru, T.; Deng, G.; Saini, S.; Majid, S.; Fukuhara, S.; Chang, I.; Arora, S.; et al. Inhibition of PTEN Gene Expression by Oncogenic miR-23b-3p in Renal Cancer. PLoS ONE 2012, 7, e50203. [Google Scholar] [CrossRef]
- Li, X.; Yu, X.; He, Y.; Meng, Y.; Liang, J.; Huang, L.; Du, H.; Wang, X.; Liu, W. Integrated Analysis of MicroRNA (miRNA) and mRNA Profiles Reveals Reduced Correlation between MicroRNA and Target Gene in Cancer. BioMed Res. Int. 2018, 2018, 1972606. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Niehrs, C. The complex world of WNT receptor signalling. Nat. Rev. Mol. Cell Biol. 2012, 13, 767–779. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Zhu, H.; Gao, Z.; Li, J.; Zhuang, J.; Dong, Y.; Shen, B.; Li, M.; Zhou, H.; Guo, H.; et al. Wnt7a activates canonical Wnt signaling, promotes bladder cancer cell invasion, and is suppressed by miR-370-3p. J. Biol. Chem. 2018, 293, 6693–6706. [Google Scholar] [CrossRef] [PubMed]
- Cai, H.; Chen, Y.; Yang, X.; Ma, S.; Wang, Q.; Zhang, Y.; Niu, X.; Ding, G.; Yuan, Y. Let7b modulates the Wnt/β-catenin pathway in liver cancer cells via downregulated Frizzled4. Tumor Biol. 2017, 39, 1010428317716076. [Google Scholar] [CrossRef]
- Yang, L.; Wang, Y.; Luo, H.; Li, Y.; Chen, T.; Wu, S. hsa-miR-96 up-regulates MAP4K1 and IRS1 and may function as a promising diagnostic marker in human bladder urothelial carcinomas. Mol. Med. Rep. 2012, 5, 260–265. [Google Scholar] [CrossRef]
- Sharif, T.; Martell, E.; Dai, C.; Kennedy, B.E.; Murphy, P.; Clements, D.R.; Kim, Y.; Lee, P.W.K.; Gujar, S.A. Autophagic homeostasis is required for the pluripotency of cancer stem cells. Autophagy 2016, 13, 264–284. [Google Scholar] [CrossRef]
- Poyet, C.; Jentsch, B.; Hermanns, T.; Schweckendiek, D.; Seifert, H.-H.; Schmidtpeter, M.; Sulser, T.; Moch, H.; Wild, P.J.; Kristiansen, G. Expression of histone deacetylases 1, 2 and 3 in urothelial bladder cancer. BMC Clin. Pathol. 2014, 14, 10. [Google Scholar] [CrossRef]
- Noonan, E.J.; Place, R.F.; Pookot, D.; Basak, S.; Whitson, J.M.; Hirata, H.; Giardina, C.; Dahiya, R. miR-449a targets HDAC-1 and induces growth arrest in prostate cancer. Oncogene 2009, 28, 1714–1724. [Google Scholar] [CrossRef]
- Saponaro, C.; Malfettone, A.; Ranieri, G.; Danza, K.; Simone, G.; Paradiso, A.; Mangia, A. VEGF, HIF-1α Expression and MVD as an Angiogenic Network in Familial Breast Cancer. PLoS ONE 2013, 8, e53070. [Google Scholar] [CrossRef]
- Cheng, S.-H.; Liu, J.-M.; Liu, Q.-Y.; Luo, D.-Y.; Liao, B.-H.; Li, H.; Wang, K.-J. Prognostic role of microvessel density in patients with renal cell carcinoma: A meta-analysis. Int. J. Clin. Exp. Pathol. 2014, 7, 5855–5863. [Google Scholar]
- Ioachim, E.; Michael, M.; Salmas, M.; Stavropoulos, N.; Malamou-Mitsi, V. Hypoxia-Inducible Factors HIF-1α and HIF-2α Expression in Bladder Cancer and Their Associations with Other Angiogenesis-Related Proteins. Urol. Int. 2006, 77, 255–263. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.-T.; Hung, W.-C.; Kang, W.-Y.; Huang, Y.-C.; Su, Y.-C.; Yang, C.-H.; Chai, C.-Y. Overexpression of cyclooxygenase-2 in urothelial carcinoma in conjunction with tumor-associated-macrophage infiltration, hypoxia-inducible factor-1α expression, and tumor angiogenesis. APMIS 2009, 117, 176–184. [Google Scholar] [CrossRef] [PubMed]
- Fus, Ł.P.; Pihowicz, P.; Koperski, Ł.; Marczewska, J.M.; Górnicka, B. HIF-1α expression is inversely associated with tumor stage, grade and microvessel density in urothelial bladder carcinoma. Pol. J. Pathol. 2018, 69, 395–404. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Miao, Y.; Shan, Y.; Liu, B.; Li, Y.; Zhao, L.; Jia, L. MiR-106b and miR-93 regulate cell progression by suppression of PTEN via PI3K/Akt pathway in breast cancer. Cell Death Dis. 2017, 8, e2796. [Google Scholar] [CrossRef] [PubMed]
- Selbach, M.; Schwanhäusser, B.; Thierfelder, N.; Fang, Z.; Khanin, R.; Rajewsky, N. Widespread changes in protein synthesis induced by microRNAs. Nature 2008, 455, 58–63. [Google Scholar] [CrossRef]
- Östling, P.; Leivonen, S.-K.; Aakula, A.; Kohonen, P.; Mäkelä, R.; Hagman, Z.; Edsjö, A.; Kangaspeska, S.; Edgren, H.; Nicorici, D.; et al. Systematic Analysis of MicroRNAs Targeting the Androgen Receptor in Prostate Cancer Cells. Cancer Res. 2011, 71, 1956–1967. [Google Scholar] [CrossRef]
- Kazem, A.; El Sayed, K.; El Kerm, Y. Prognostic significance of COX-2 and β-catenin in colorectal carcinoma. Alex. J. Med. 2014, 50, 211–220. [Google Scholar] [CrossRef]
- Timofeeva, O.A.; Zhang, X.; Ressom, H.W.; Varghese, R.S.; Kallakury, B.V.S.; Wang, K.; Ji, Y.; Cheema, A.; Jung, M.; Brown, M.L.; et al. En-hanced expression of SOS1 is detected in prostate cancer epithelial cells from African-American men. Int. J. Oncol. 2009, 35, 751–760. [Google Scholar]


| Common Name | Sequence | Accession No. |
|---|---|---|
| WNT7a | F-5′-ACTTAGGGGTAAGGAGGGGC-3′ R-5′-GCTGGACCCAAAGCAAAGTG-3′ | NM_004625.4 |
| β-catenin | F-5′-CTGAGGAGCAGCTTCAGTCC-3′ R-5′-ATTGCACGTGTGGCAAGTTC-3′ | NM_001098209.2 |
| IRS2 | F-5′-CCACTGACAACGAGAGCCAT-3′ R-5′-CCCATGGCCCCAGTGTTTAT-3′ | NM_005544.2 |
| FZD4 | F-5′-CCAACTGGGCACTTTTTCGG-3′ R-5′-TCTAAACAGCAGACAGCGCA-3′ | NM_012193.4 |
| SOS1 | F-5′-TGTGCAAGGCCATGGATACC-3′ R-5′-CCTTGTCAGCACACATTGCC-3′ | NM_001382394.1 |
| HDAC1 | F-5′-ACTGCTAAAGTATCACCAGAGGG-3′ R-5′-CACACTTGGCGTGTCCTTTG-3′ | NM_004964.3 |
| HDAC2 | F-5′-GAGGTGGCTACACAATCCGT-3′ R-5′-TAGCCACTGAAACAAGACTTCA-3′ | NM_001527.4 |
| HIF1α | F-5′-ACTTGGCAACCTTGGATTGGA-3′ R-5′-GCACCAAGCAGGTCATAGGT-3′ | NM_001243084.1 |
| PTEN | F-5′-AGCTGGAAAGGGACGAACTG-3′ R-5′-ACACACAGGTAACGGCTGAG-3′ | NM_001304717.5 |
| GAPDH | F-5′-GAAGGTGAAGGTCGTAGTC-3′ R-5′-GAAGATGGTGATGGGATTTC-3′ | NM_001357943.2 |
| miRNA | miRNA Regulated Status | Target Gene | Gene Regulation Status | Pearson’s r (p-Value) | |
|---|---|---|---|---|---|
| T1 | T2–T4 | ||||
| Let-7a-5p | down | WNT7A | up | −0.530 (p < 0.001) | −0.687 (p < 0.001) |
| Let-7a-5p | down | β-catenin | up | 0.418 (p = 0.007) | −0.687 (p < 0.001) |
| Let-7a-5p | down | IRS2 | up | 0.374 (p = 0.018) | −0.664 (p < 0.001) |
| Let-7a-5p | down | FZD4 | up | −0.323 (p = 0.042) | −0.733 (p < 0.001) |
| miR124-3p | down | SOS1 | up | −0.630 (p < 0.001) | −0.579 (p < 0.001) |
| miR449a-5p | down | HDAC1 | up | −0.326 (p = 0.04) | 0.369 (p < 0.001) |
| miR145-3p | down | HDAC2 | up | 0.271 (p = 0.091) | −0.123 (p = 0.186) |
| miR138-5p | down | HIF1α | up | −0.601 (p < 0.001) | 0.510 (p < 0.001) |
| miR23a-5p | up | PTEN | down | 0.660 (p < 0.001) | −0.513 (p < 0.001) |
| Gender | Age | Tumor Stage | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Male | Female | p-Value | <70 Years | ≥70 Years | p-Value | T1 | T2–T4 | p-Value | ||
| β-Catenin | High | 96 (72.72%) | 17 (68%) | 0.464 | 86 (61.87%) | 11 (61.11%) | 0.239 | 9 (22.5%) | 88 (75.21%) | 0.003 |
| Low | 36 (25.89%) | 8 (32%) | 53 (38.12%) | 7 (38.88%) | 31 (77.5%) | 29 (24.78) | ||||
| HDAC1 | High | 72 (58.5%) | 11 (44%) | 0.182 | 77 (57.5%) | 6 (42.9%) | 0.295 | 34 (85%) | 45 (38.46%) | 0.001 |
| Low | 51 (41.5%) | 14 (56%) | 57 (42.5%) | 8 (57.1%) | 6 (15%) | 72 (61.54%) | ||||
| HIF1α | High | 50 (37.9%) | 7 (28%) | 0.346 | 54 (40.3%) | 3 (21.4%) | 0.167 | 29 (72.5%) | 28 (23.9%) | 0.000 |
| Low | 82 (62.1%) | 18 (72%) | 80 (59.7%) | 11 (78.6%) | 11 (27.5%) | 89 (76.1%) | ||||
| SOS1 | High | 81 (61.4%) | 20 (80%) | 0.074 | 94 (70.1%) | 7 (50%) | 0.123 | 16 (40%) | 85 (72.6%) | 0.000 |
| Low | 51 (38.6%) | 5 (20%) | 40 (29.9%) | 7 (50%) | 24 (60%) | 32 (27.4%) | ||||
| PTEN | High | 46 (34.8%) | 7 (28%) | 0.507 | 50 (37.3%) | 3 (21.4%) | 0.238 | 28 (70%) | 25 (21.4%) | 0.000 |
| Low | 86 (65.2%) | 18 (72%) | 84 (62.7%) | 11 (78.6%) | 12 (30%) | 92 (78.6%) | ||||
| HDAC2 | High | 64 (48.5%) | 9 (36%) | 0.251 | 67 (50%) | 67 (50%) | 0.309 | 32 (80%) | 41 (35.04%) | 0.000 |
| Low | 68 (51.5%) | 16 (64%) | 5 (35.7%) | 9 (64.3%) | 8 (20%) | 76 (64.95%) | ||||
| Expression | 1-Year CSS (%) | 5-Year CSS (%) | p-Value | |
|---|---|---|---|---|
| miRNA | ||||
| Let-7a-5p | Low | 61.9 | 27.9 | <0.001 |
| High | 94.3 | 68.9 | ||
| miRNA-449a-5p | Low | 57.7 | 15.7 | <0.001 |
| High | 89.5 | 67.2 | ||
| miRNA-145-3P | Low | 77 | 49.5 | 0.05 |
| High | 70.4 | 25 | ||
| miRNA-124-3P | Low | 68.5 | 37.6 | 0.01 |
| High | 92.3 | 57.7 | ||
| miRNA-138-5p | Low | 68.7 | 32.8 | 0.004 |
| High | 84.7 | 62.3 | ||
| miRNA-23a-5p | Low | 80 | 54.7 | 0.06 |
| High | 69.9 | 34.8 | ||
| Gene products | ||||
| β-catenin | Low | 86.5 | 54 | <0.001 |
| High | 32 | - | ||
| FZD | Low | 87 | 55 | <0.001 |
| High | 41.9 | - | ||
| HDAC1 | Low | 62.4 | 31 | <0.001 |
| High | 94 | 64 | ||
| HDAC2 | Low | 67.6 | 32 | 0.004 |
| High | 87 | 66.6 | ||
| HIF | Low | 71.9 | 37.4 | 0.02 |
| High | 89.5 | 68 | ||
| IRS | Low | 91.5 | 68.8 | <0.001 |
| High | 64.8 | 27.9 | ||
| PTEN | Low | 58.3 | 36 | 0.01 |
| High | 85 | 48 | ||
| SOS-1 | Low | 93.5 | 57 | 0.01 |
| High | 59.8 | 29.7 | ||
| WNT7a | Low | 92.9 | 59.4 | <0.001 |
| High | 50.7 | 19.7 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Awadalla, A.; Abol-Enein, H.; Hamam, E.T.; Ahmed, A.E.; Khirallah, S.M.; El-Assmy, A.; Mostafa, S.A.; Babalghith, A.O.; Ali, M.; Abdel-Rahim, M.; et al. Identification of Epigenetic Interactions between miRNA and Gene Expression as Potential Prognostic Markers in Bladder Cancer. Genes 2022, 13, 1629. https://doi.org/10.3390/genes13091629
Awadalla A, Abol-Enein H, Hamam ET, Ahmed AE, Khirallah SM, El-Assmy A, Mostafa SA, Babalghith AO, Ali M, Abdel-Rahim M, et al. Identification of Epigenetic Interactions between miRNA and Gene Expression as Potential Prognostic Markers in Bladder Cancer. Genes. 2022; 13(9):1629. https://doi.org/10.3390/genes13091629
Chicago/Turabian StyleAwadalla, Amira, Hassan Abol-Enein, Eman T. Hamam, Asmaa E. Ahmed, Salma M. Khirallah, Ahmed El-Assmy, Sally Abdallah Mostafa, Ahmed O. Babalghith, Mohamed Ali, Mona Abdel-Rahim, and et al. 2022. "Identification of Epigenetic Interactions between miRNA and Gene Expression as Potential Prognostic Markers in Bladder Cancer" Genes 13, no. 9: 1629. https://doi.org/10.3390/genes13091629






