Persistence of Abscisic Acid Analogs in Plants: Chemical Control of Plant Growth and Physiology
Abstract
1. Introduction
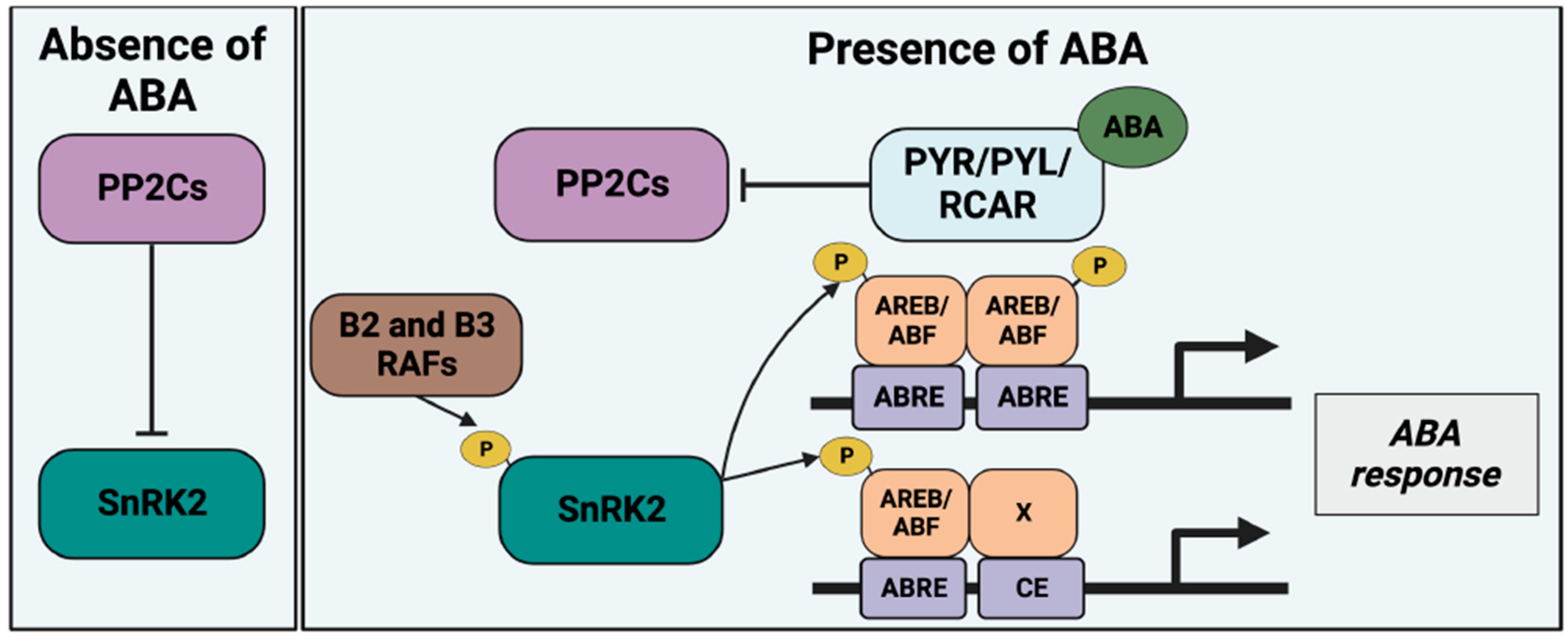
Introduction to ABA
2. ABA and Agriculture
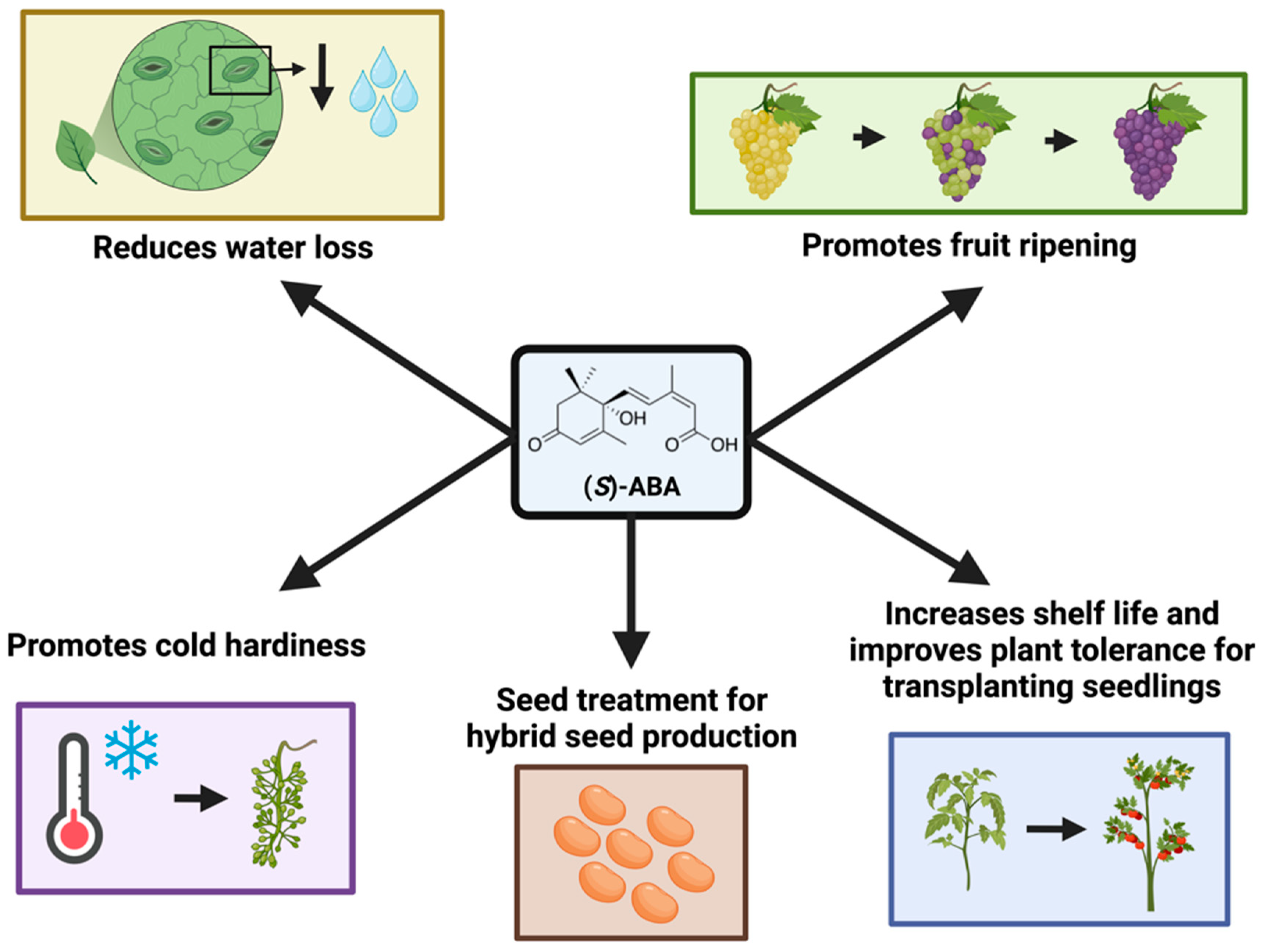
2.1. ABA as a Plant Growth Regulator
2.2. Registered Uses of (S)-ABA in Agriculture
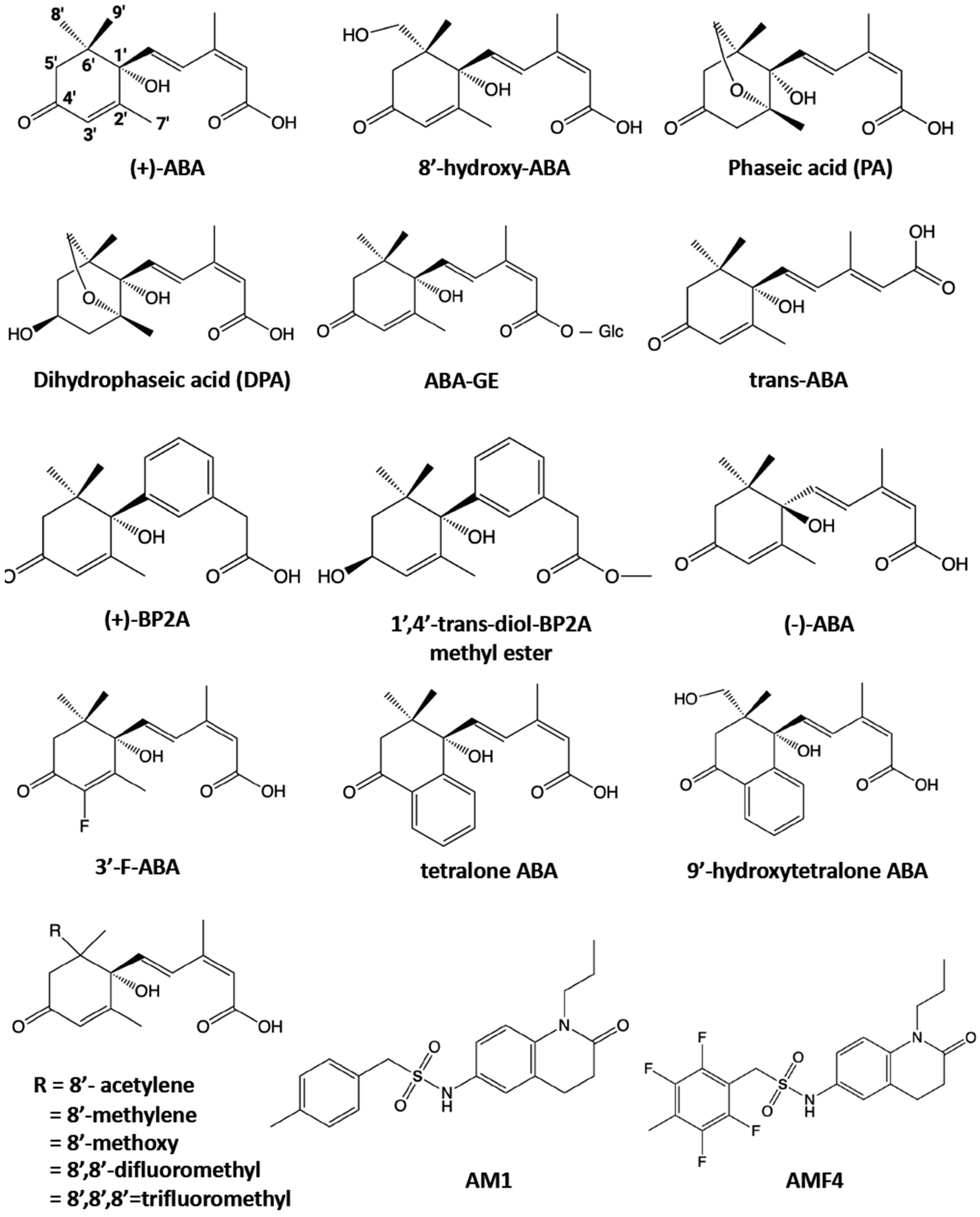
3. ABA Analogs
4. Persistence of ABA Analogs
4.1. Photostable ABA Analogs
4.2. Analogs Resistance to ABA 8′-Hydroxylase
4.3. ABA Analogs Resistant to Intramolecular Cyclization after C-8′ Hydroxylation
4.4. ABA Analogs and Functional Mimic Resistant to UGT and Xenobiotic Metabolism
5. Conclusions and Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kumar, A.; Memo, M.; Mastinu, A. Plant behaviour: An evolutionary response to the environment? Plant Biol. 2020, 22, 961–970. [Google Scholar] [CrossRef]
- Cutler, S.R.; Rodriguez, P.L.; Finkelstein, R.R.; Abrams, S.R. Abscisic acid: Emergence of a core signaling network. Annu. Rev. Plant Biol. 2010, 61, 651–679. [Google Scholar] [CrossRef] [PubMed]
- Seo, M.; Marion-Poll, A. Abscisic acid metabolism and transport. In Abscisic Acid in Plants; Advances in Botanical Research; Elsevier: Oxford, UK, 2019; Volume 92, pp. 1–49. ISBN 978-0-08-102620-5. [Google Scholar]
- Nambara, E.; Marion-Poll, A. Abscisic acid biosynthesis and catabolism. Annu. Rev. Plant Biol. 2005, 56, 165–185. [Google Scholar] [CrossRef] [PubMed]
- Saito, S.; Hirai, N.; Matsumoto, C.; Ohigashi, H.; Ohta, D.; Sakata, K.; Mizutani, M. Arabidopsis CYP707As encode (+)-abscisic acid 8′-hydroxylase, a key enzyme in the oxidative catabolism of abscisic acid. Plant Physiol. 2004, 134, 1439–1449. [Google Scholar] [CrossRef]
- Kushiro, T.; Okamoto, M.; Nakabayashi, K.; Yamagishi, K.; Kitamura, S.; Asami, T.; Hirai, N.; Koshiba, T.; Kamiya, Y.; Nambara, E. The Arabidopsis cytochrome P450 CYP707A Encodes ABA 8′-hydroxylases: Key enzymes in ABA catabolism. EMBO J. 2004, 23, 1647–1656. [Google Scholar] [CrossRef] [PubMed]
- Umezawa, T.; Okamoto, M.; Kushiro, T.; Nambara, E.; Oono, Y.; Seki, M.; Kobayashi, M.; Koshiba, T.; Kamiya, Y.; Shinozaki, K. CYP707A3, a major ABA 8′-hydroxylase involved in dehydration and rehydration response in Arabidopsis thaliana. Plant J. 2006, 46, 171–182. [Google Scholar] [CrossRef]
- Okamoto, M.; Kuwahara, A.; Seo, M.; Kushiro, T.; Asami, T.; Hirai, N.; Kamiya, Y.; Koshiba, T.; Nambara, E. CYP707A1 and CYP707A2, which encode abscisic acid 8′-hydroxylases, are indispensable for proper control of seed dormancy and germination in Arabidopsis. Plant Physiol. 2006, 141, 97–107. [Google Scholar] [CrossRef]
- Priest, D.M.; Ambrose, S.J.; Vaistij, F.E.; Elias, L.; Higgins, G.S.; Ross, A.R.S.; Abrams, S.R.; Bowles, D.J. Use of the glucosyltransferase UGT71B6 to disturb abscisic acid homeostasis in Arabidopsis thaliana. Plant J. 2006, 46, 492–502. [Google Scholar] [CrossRef]
- Xu, Z.-Y.; Lee, K.H.; Dong, T.; Jeong, J.C.; Jin, J.B.; Kanno, Y.; Kim, D.H.; Kim, S.Y.; Seo, M.; Bressan, R.A.; et al. A vacuolar β-glucosidase homolog that possesses glucose-conjugated abscisic acid hydrolyzing activity plays an important role in osmotic stress responses in Arabidopsis. Plant Cell 2012, 24, 2184–2199. [Google Scholar] [CrossRef]
- Lee, K.H.; Piao, H.L.; Kim, H.-Y.; Choi, S.M.; Jiang, F.; Hartung, W.; Hwang, I.; Kwak, J.M.; Lee, I.-J.; Hwang, I. Activation of glucosidase via stress-induced polymerization rapidly increases active pools of abscisic acid. Cell 2006, 126, 1109–1120. [Google Scholar] [CrossRef]
- Ma, Y.; Szostkiewicz, I.; Korte, A.; Moes, D.; Yang, Y.; Christmann, A.; Grill, E. Regulators of PP2C phosphatase activity function as abscisic acid sensors. Science 2009, 324, 1064–1068. [Google Scholar] [CrossRef] [PubMed]
- Park, S.-Y.; Fung, P.; Nishimura, N.; Jensen, D.R.; Fujii, H.; Zhao, Y.; Lumba, S.; Santiago, J.; Rodrigues, A.; Chow, T.F.; et al. Abscisic acid inhibits type 2C protein phosphatases via the PYR/PYL family of START proteins. Science 2009, 324, 1068–1071. [Google Scholar] [CrossRef] [PubMed]
- Vlad, F.; Rubio, S.; Rodrigues, A.; Sirichandra, C.; Belin, C.; Robert, N.; Leung, J.; Rodriguez, P.L.; Laurière, C.; Merlot, S. Protein phosphatases 2C regulate the activation of the Snf1-related kinase OST1 by abscisic acid in Arabidopsis. Plant Cell 2009, 21, 3170–3184. [Google Scholar] [CrossRef]
- Ng, L.-M.; Soon, F.-F.; Zhou, X.E.; West, G.M.; Kovach, A.; Suino-Powell, K.M.; Chalmers, M.J.; Li, J.; Yong, E.-L.; Zhu, J.-K.; et al. Structural basis for basal activity and autoactivation of abscisic acid (ABA) signaling SnRK2 kinases. Proc. Natl. Acad. Sci. USA 2011, 108, 21259–21264. [Google Scholar] [CrossRef] [PubMed]
- Soon, F.-F.; Ng, L.-M.; Zhou, X.E.; West, G.M.; Kovach, A.; Tan, M.H.E.; Suino-Powell, K.M.; He, Y.; Xu, Y.; Chalmers, M.J.; et al. Molecular mimicry regulates ABA signaling by SnRK2 kinases and PP2C phosphatases. Science 2012, 335, 85–88. [Google Scholar] [CrossRef] [PubMed]
- Umezawa, T.; Sugiyama, N.; Mizoguchi, M.; Hayashi, S.; Myouga, F.; Yamaguchi-Shinozaki, K.; Ishihama, Y.; Hirayama, T.; Shinozaki, K. Type 2C protein phosphatases directly regulate abscisic acid-activated protein kinases in Arabidopsis. Proc. Natl. Acad. Sci. USA 2009, 106, 17588–17593. [Google Scholar] [CrossRef]
- Melcher, K.; Ng, L.-M.; Zhou, X.E.; Soon, F.-F.; Xu, Y.; Suino-Powell, K.M.; Park, S.-Y.; Weiner, J.J.; Fujii, H.; Chinnusamy, V.; et al. A gate–latch–lock mechanism for hormone signalling by abscisic acid receptors. Nature 2009, 462, 602–608. [Google Scholar] [CrossRef]
- Miyazono, K.; Miyakawa, T.; Sawano, Y.; Kubota, K.; Kang, H.-J.; Asano, A.; Miyauchi, Y.; Takahashi, M.; Zhi, Y.; Fujita, Y.; et al. Structural basis of abscisic acid signalling. Nature 2009, 462, 609–614. [Google Scholar] [CrossRef] [PubMed]
- Nishimura, N.; Hitomi, K.; Arvai, A.S.; Rambo, R.P.; Hitomi, C.; Cutler, S.R.; Schroeder, J.I.; Getzoff, E.D. Structural mechanism of abscisic acid binding and signaling by dimeric PYR1. Science 2009, 326, 1373–1379. [Google Scholar] [CrossRef] [PubMed]
- Xue, S.; Hu, H.; Ries, A.; Merilo, E.; Kollist, H.; Schroeder, J.I. Central functions of bicarbonate in S-type anion channel activation and OST1 protein kinase in CO 2 signal transduction in guard cell: CO 2 signalling in guard cells. EMBO J. 2011, 30, 1645–1658. [Google Scholar] [CrossRef]
- Fujii, H.; Chinnusamy, V.; Rodrigues, A.; Rubio, S.; Antoni, R.; Park, S.-Y.; Cutler, S.R.; Sheen, J.; Rodriguez, P.L.; Zhu, J.-K. In vitro reconstitution of an abscisic acid signalling pathway. Nature 2009, 462, 660–664. [Google Scholar] [CrossRef]
- Nakashima, K.; Fujita, Y.; Kanamori, N.; Katagiri, T.; Umezawa, T.; Kidokoro, S.; Maruyama, K.; Yoshida, T.; Ishiyama, K.; Kobayashi, M.; et al. Three Arabidopsis SnRK2 protein kinases, SRK2D/SnRK2.2, SRK2E/SnRK2.6/OST1 and SRK2I/SnRK2.3, involved in ABA signaling are essential for the control of seed development and dormancy. Plant Cell Physiol. 2009, 50, 1345–1363. [Google Scholar] [CrossRef] [PubMed]
- Geiger, D.; Scherzer, S.; Mumm, P.; Stange, A.; Marten, I.; Bauer, H.; Ache, P.; Matschi, S.; Liese, A.; Al-Rasheid, K.A.S.; et al. Activity of guard cell anion channel SLAC1 is controlled by drought-stress signaling kinase-phosphatase pair. Proc. Natl. Acad. Sci. USA 2009, 106, 21425–21430. [Google Scholar] [CrossRef] [PubMed]
- Soma, F.; Takahashi, F.; Suzuki, T.; Shinozaki, K.; Yamaguchi-Shinozaki, K. Plant Raf-like kinases regulate the mRNA population upstream of ABA-unresponsive SnRK2 kinases under drought stress. Nat. Commun. 2020, 11, 1373. [Google Scholar] [CrossRef]
- Hao, Q.; Yin, P.; Li, W.; Wang, L.; Yan, C.; Lin, Z.; Wu, J.Z.; Wang, J.; Yan, S.F.; Yan, N. The molecular basis of ABA-independent inhibition of PP2Cs by a subclass of PYL proteins. Mol. Cell 2011, 42, 662–672. [Google Scholar] [CrossRef]
- Dupeux, F.; Santiago, J.; Betz, K.; Twycross, J.; Park, S.-Y.; Rodriguez, L.; Gonzalez-Guzman, M.; Jensen, M.R.; Krasnogor, N.; Blackledge, M.; et al. A thermodynamic switch modulates abscisic acid receptor sensitivity: A thermodynamic switch in the ABA pathway. EMBO J. 2011, 30, 4171–4184. [Google Scholar] [CrossRef]
- Melcher, K.; Zhou, X.E.; Xu, H.E. Thirsty plants and beyond: Structural mechanisms of abscisic acid perception and signaling. Curr. Opin. Struct. Biol. 2010, 20, 722–729. [Google Scholar] [CrossRef]
- Soma, F.; Takahashi, F.; Yamaguchi-Shinozaki, K.; Shinozaki, K. Cellular phosphorylation signaling and gene expression in drought stress responses: ABA-dependent and ABA-independent regulatory systems. Plants 2021, 10, 756. [Google Scholar] [CrossRef]
- Shen, Q.; Zhang, P.; Ho, T.H. Modular nature of abscisic acid (ABA) response complexes: Composite promoter units that are necessary and sufficient for ABA induction of gene expression in barley. Plant Cell 1996, 8, 1107–1119. [Google Scholar] [CrossRef] [PubMed]
- Ndathe, R.; Dale, R.; Kato, N. Dynamic modeling of ABA-dependent expression of the Arabidopsis RD29A gene. Front. Plant Sci. 2022, 13, 928718. [Google Scholar] [CrossRef]
- Nakabayashi, K.; Okamoto, M.; Koshiba, T.; Kamiya, Y.; Nambara, E. Genome-wide profiling of stored mRNA in Arabidopsis thaliana seed germination: Epigenetic and genetic regulation of transcription in seed. Plant J. 2005, 41, 697–709. [Google Scholar] [CrossRef] [PubMed]
- Hobo, T.; Asada, M.; Kowyama, Y.; Hattori, T. ACGT-containing abscisic acid response element (ABRE) and coupling element 3 (CE3) are functionally equivalent. Plant J. 1999, 19, 679–689. [Google Scholar] [CrossRef]
- Uno, Y.; Furihata, T.; Abe, H.; Yoshida, R.; Shinozaki, K.; Yamaguchi-Shinozaki, K. Arabidopsis basic leucine zipper transcription factors involved in an abscisic acid-dependent signal transduction pathway under drought and high-salinity conditions. Proc. Natl. Acad. Sci. USA 2000, 97, 11632–11637. [Google Scholar] [CrossRef]
- Sun, Y.; Oh, D.-H.; Duan, L.; Ramachandran, P.; Ramirez, A.; Bartlett, A.; Tran, K.-N.; Wang, G.; Dassanayake, M.; Dinneny, J.R. Divergence in the ABA gene regulatory network underlies differential growth control. Nat. Plants 2022, 8, 549–560. [Google Scholar] [CrossRef]
- Urano, K.; Maruyama, K.; Ogata, Y.; Morishita, Y.; Takeda, M.; Sakurai, N.; Suzuki, H.; Saito, K.; Shibata, D.; Kobayashi, M.; et al. Characterization of the ABA-regulated global responses to dehydration in Arabidopsis by metabolomics. Plant J. 2009, 57, 1065–1078. [Google Scholar] [CrossRef] [PubMed]
- Nemhauser, J.L.; Hong, F.; Chory, J. Different plant hormones regulate similar processes through largely nonoverlapping transcriptional responses. Cell 2006, 126, 467–475. [Google Scholar] [CrossRef]
- Hussain, Q.; Asim, M.; Zhang, R.; Khan, R.; Farooq, S.; Wu, J. Transcription factors interact with ABA through gene expression and signaling pathways to mitigate drought and salinity stress. Biomolecules 2021, 11, 1159. [Google Scholar] [CrossRef]
- Sirichandra, C.; Gu, D.; Hu, H.-C.; Davanture, M.; Lee, S.; Djaoui, M.; Valot, B.; Zivy, M.; Leung, J.; Merlot, S.; et al. Phosphorylation of the Arabidopsis AtrbohF NADPH oxidase by OST1 protein kinase. FEBS Lett. 2009, 583, 2982–2986. [Google Scholar] [CrossRef]
- Leymarie, J.; Vitkauskaité, G.; Hoang, H.H.; Gendreau, E.; Chazoule, V.; Meimoun, P.; Corbineau, F.; El-Maarouf-Bouteau, H.; Bailly, C. Role of reactive oxygen species in the regulation of Arabidopsis seed dormancy. Plant Cell Physiol. 2012, 53, 96–106. [Google Scholar] [CrossRef]
- Hasanuzzaman, M.; Bhuyan, M.H.M.B.; Parvin, K.; Bhuiyan, T.F.; Anee, T.I.; Nahar, K.; Hossen, M.S.; Zulfiqar, F.; Alam, M.M.; Fujita, M. Regulation of ROS metabolism in plants under environmental stress: A review of recent experimental evidence. Int. J. Mol. Sci. 2020, 21, 8695. [Google Scholar] [CrossRef] [PubMed]
- Cutler, S.R.; Wendeborn, S.V.; Jung, P.J.; Lachia, M.D.; Dumeunier, R. Compounds That Induce ABA Responses. U.S. Patent 2016/0280651 A1, 2016. [Google Scholar]
- Racsko, J.; Marmor, F.; Hopkins, C.R.; Petracek, P.; Silverman, F.P.; Jr, R.F.; Liu, X.; Woolard, D.; Lopez, J.; Leep, D.; et al. Use of S-abscisic acid (ConTegoTM SL) in vegetable production. Acta Hort. 2014, 1042, 243–245. [Google Scholar] [CrossRef]
- Quaghebeur, K. Use of S-Abscisic Acid for Improving Fruit Set and Producing Parthenovarpic Fruits and as a Growth Inhibitor. U.S. Patent 2008/0318787 A1, 25 December 2008. [Google Scholar]
- Bowen, P.; Shellie, K.C.; Mills, L.; Willwerth, J.; Bogdanoff, C.; Keller, M. Abscisic acid form, concentration, and application timing influence phenology and bud cold hardiness in merlot grapevines. Can. J. Plant Sci. 2016, 96, 347–359. [Google Scholar] [CrossRef]
- Wilson, D.O.; Higgs, N.; Silverman, F.P.; Petracek, P.D.; Warrior, P. Use of Abscisic Acid Seed Treatment to Enhance Corn Emergence after Early Planting. U.S. Patent WO 2008/156859 A1, 2008. [Google Scholar]
- Kou, X.; Yang, S.; Chai, L.; Wu, C.; Zhou, J.; Liu, Y.; Xue, Z. Abscisic acid and fruit ripening: Multifaceted analysis of the effect of abscisic acid on fleshy fruit ripening. Sci. Hortic. 2021, 281, 109999. [Google Scholar] [CrossRef]
- Bai, Q.; Huang, Y.; Shen, Y. The physiological and molecular mechanism of abscisic acid in regulation of fleshy fruit ripening. Front. Plant Sci. 2021, 11, 619953. [Google Scholar] [CrossRef] [PubMed]
- McArtney, S.J.; Abrams, S.R.; Woolard, D.D.; Petracek, P.D. Effects of S-abscisic acid and (+)-8′-acetylene abscisic acid on fruit set and stomatal conductance in apple. HortScience 2014, 49, 763–768. [Google Scholar] [CrossRef]
- Abrams, S.R.; Loewen, M.C. Chemistry and chemical biology of ABA. In Advances in Botanical Research; Abscisic acid in, plants, Seo, M., Marion-Poll, A., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; Volume 92, pp. 315–340. [Google Scholar]
- Walton, D.C. Structure-activity relationships of abscisic acid analogs and metabolites. In Abscisic Acid; Praeger: New York, NY, USA, 1983; pp. 113–146. ISBN 0-03-055831-X. [Google Scholar]
- Todoroki, Y.; Hirai, N. Abscisic acid analogs for probing the mechanism of abscisic acid reception and inactivation. In Studies in Natural Products Chemistry; Elsevier: Amsterdam, The Netherlands, 2002; Volume 27, pp. 321–360. ISBN 978-0-444-51230-7. [Google Scholar]
- Dejonghe, W.; Okamoto, M.; Cutler, S.R. Small molecule probes of ABA biosynthesis and signaling. Plant Cell Physiol. 2018, 59, 1490–1499. [Google Scholar] [CrossRef]
- Gupta, M.K.; Lenka, S.K.; Gupta, S.; Rawal, R.K. Agonist, antagonist and signaling modulators of ABA receptor for agronomic and post-harvest management. Plant Physiol. Biochem. 2020, 148, 10–25. [Google Scholar] [CrossRef] [PubMed]
- Okamoto, M.; Cutler, S.R. Chemical control of ABA receptors to enable plant protection against water stress. In Plant Chemical Genomics; Fauser, F., Jonikas, M., Eds.; Methods in Molecular Biology; Springer: New York, NY, USA, 2018; Volume 1795, pp. 127–141. ISBN 978-1-4939-7873-1. [Google Scholar]
- Ueno, K.; Araki, Y.; Hirai, N.; Saito, S.; Mizutani, M.; Sakata, K.; Todoroki, Y. Differences between the structural requirements for ABA 8′-hydroxylase inhibition and for ABA activity. Bioorg. Med. Chem. 2005, 13, 3359–3370. [Google Scholar] [CrossRef]
- Smith, D.A.; Beaumont, K.; Maurer, T.S.; Di, L. Relevance of half-life in drug design: Miniperspective. J. Med. Chem. 2018, 61, 4273–4282. [Google Scholar] [CrossRef]
- Takeuchi, J.; Mimura, S.; Ohnishi, T.; Todoroki, Y. Photostable abscisic acid agonists with a geometrically rigid cyclized side chain. J. Agric. Food Chem. 2022, 70, 869–876. [Google Scholar] [CrossRef]
- Milborrow, B.V. Pathways to and from abscisic acid. In Abscisic Acid; Praeger: New York, NY, USA, 1983; pp. 79–112. [Google Scholar]
- Todoroki, Y.; Tanaka, T.; Kisamori, M.; Hirai, N. 3′-azidoabscisic acid as a photoaffinity reagent for abscisic acid binding proteins. Bioorg. Med. Chem. Lett. 2001, 11, 2381–2384. [Google Scholar] [CrossRef] [PubMed]
- Szostkiewicz, I.; Richter, K.; Kepka, M.; Demmel, S.; Ma, Y.; Korte, A.; Assaad, F.F.; Christmann, A.; Grill, E. Closely related receptor complexes differ in their ABA selectivity and sensitivity. Plant J. 2010, 61, 25–35. [Google Scholar] [CrossRef]
- Benson, C.L.; Kepka, M.; Wunschel, C.; Rajagopalan, N.; Nelson, K.M.; Christmann, A.; Abrams, S.R.; Grill, E.; Loewen, M.C. Abscisic acid analogs as chemical probes for dissection of abscisic acid responses in Arabidopsis thaliana. Phytochemistry 2015, 113, 96–107. [Google Scholar] [CrossRef] [PubMed]
- Lin, B.-L.; Wang, H.-J.; Wang, J.-S.; Zaharia, L.I.; Abrams, S.R. Abscisic acid regulation of heterophylly in Marsilea quadrifolia L.: Effects of R-(−) and S-(+) isomers. J. Exp. Bot. 2005, 56, 2935–2948. [Google Scholar] [CrossRef]
- Huang, D.; Jaradat, M.R.; Wu, W.; Ambrose, S.J.; Ross, A.R.; Abrams, S.R.; Cutler, A.J. Structural analogs of ABA reveal novel features of ABA perception and signaling in Arabidopsis: ABA signaling revealed by ABA analogs. Plant J. 2007, 50, 414–428. [Google Scholar] [CrossRef]
- Todoroki, Y.; Nakano, S.; Hirai, N.; Mitsui, T.; Ohigashi, H. Synthesis, biological activity, and metabolism of 8′,8′,8′-trideuteroabscisic acid. Biosci. Biotechnol. Biochem. 1997, 61, 1872–1876. [Google Scholar] [CrossRef] [PubMed]
- Rose, P.A.; Cutler, A.J.; Irvine, N.M.; Shaw, A.C.; Squires, T.M.; Loewen, M.K.; Abrams, S.R. 8’-acetylene ABA: An irreversible inhibitor of ABA 8’-hydroxylase. Bioorg. Med. Chem. Lett. 1997, 7, 2543–2546. [Google Scholar] [CrossRef]
- Cutler, A.J.; Rose, P.A.; Squires, T.M.; Loewen, M.K.; Shaw, A.C.; Quail, J.W.; Krochko, J.E.; Abrams, S.R. Inhibitors of abscisic acid 8‘-hydroxylase. Biochemistry 2000, 39, 13614–13624. [Google Scholar] [CrossRef]
- Abrams, S.R.; Rose, P.A.; Cutler, A.J.; Walker-Simmons, M.K. 8’-Methylene abscisic acid. Plant Physiol. 1997, 114, 89–97. [Google Scholar] [CrossRef]
- Todoroki, Y.; Sawada, M.; Matsumoto, M.; Tsukada, S.; Ueno, K.; Isaka, M.; Owaki, M.; Hirai, N. Metabolism of 5′α,8′-cycloabscisic acid, a highly potent and long-lasting abscisic acid analogue, in radish seedlings. Bioorg. Med. Chem. 2004, 12, 363–370. [Google Scholar] [CrossRef]
- Todoroki, Y.; Hirai, N.; Koshimizu, K. 8′- and 9′-Methoxyabscisic acids as antimetabolic analogs of abscisic acid. Biosci. Biotechnol. Biochem. 1994, 58, 707–715. [Google Scholar] [CrossRef]
- Todoroki, Y.; Hirai, N.; Koshimizu, K. 8′,8′-Difluoro- and 8′,8′,8′-trifluoroabscisic acids as highly potent, long-lasting analogues of abscisic acid. Phytochemistry 1995, 38, 561–568. [Google Scholar] [CrossRef]
- Arai, S.; Todoroki, Y.; Ibaraki, S.; Naoe, Y.; Hirai, N.; Ohigashi, H. Synthesis and biological activity of 3’-chloro, -bromo, and -iodoabscisic acids, and biological activity of 3’-fluoro- 8’-hydroxyabscisic acid. Phytochemistry 1999, 52, 1185–1193. [Google Scholar] [CrossRef]
- Zou, J.; Abrams, G.D.; Barton, D.L.; Taylor, D.C.; Pomeroy, M.K.; Abrams, S.R. Induction of lipid and oleosin biosynthesis by (+)-abscisic acid and its metabolites in microspore-derived embryos of Brassica napus L. Cv Reston. Plant Physiol. 1995, 108, 563–571. [Google Scholar] [CrossRef]
- Jadhav, A.S.; Taylor, D.C.; Giblin, M.; Ferrie, A.M.R.; Ambrose, S.J.; Ross, A.R.S.; Nelson, K.M.; Irina Zaharia, L.; Sharma, N.; Anderson, M.; et al. Hormonal regulation of oil accumulation in Brassica seeds: Metabolism and biological activity of ABA, 7′-, 8′- and 9′-hydroxy ABA in microspore derived embryos of B. napus. Phytochemistry 2008, 69, 2678–2688. [Google Scholar] [CrossRef]
- Todoroki, Y.; Hirai, N.; Ohigashi, H. Analysis of isomerization process of 8′-hydroxyabscisic acid and its 3′-fluorinated analog in aqueous solutions. Tetrahedron 2000, 56, 1649–1653. [Google Scholar] [CrossRef]
- Nyangulu, J.M.; Nelson, K.M.; Rose, P.A.; Gai, Y.; Loewen, M.; Lougheed, B.; Quail, J.W.; Cutler, A.J.; Abrams, S.R. Synthesis and biological activity of tetralone abscisic acid analogues. Org. Biomol. Chem. 2006, 4, 1400. [Google Scholar] [CrossRef]
- Vaidya, A.S.; Helander, J.D.M.; Peterson, F.C.; Elzinga, D.; Dejonghe, W.; Kaundal, A.; Park, S.-Y.; Xing, Z.; Mega, R.; Takeuchi, J.; et al. Dynamic control of plant water use using designed ABA receptor agonists. Science 2019, 366, eaaw8848. [Google Scholar] [CrossRef]
- Kepka, M.; Benson, C.L.; Gonugunta, V.K.; Nelson, K.M.; Christmann, A.; Grill, E.; Abrams, S.R. Action of natural abscisic acid precursors and catabolites on abscisic acid receptor complexes. Plant Physiol. 2011, 157, 2108–2119. [Google Scholar] [CrossRef]
- Takeuchi, J.; Ohnishi, T.; Okamoto, M.; Todoroki, Y. Conformationally restricted 3′-modified ABA analogs for controlling ABA receptors. Org. Biomol. Chem. 2015, 13, 4278–4288. [Google Scholar] [CrossRef]
- Jugulam, M.; Shyam, C. Non-target-site resistance to herbicides: Recent developments. Plants 2019, 8, 417. [Google Scholar] [CrossRef]
- Milborrow, B.V. The chemistry and physiology of abscisic acid. Annu. Rev. Plant Physiol. 1974, 25, 259–307. [Google Scholar] [CrossRef]
- Priest, D.M.; Jackson, R.G.; Ashford, D.A.; Abrams, S.R.; Bowles, D.J. The use of abscisic acid analogues to analyse the substrate selectivity of UGT71B6, a UDP-glycosyltransferase of Arabidopsis Thaliana. FEBS Lett. 2005, 579, 4454–4458. [Google Scholar] [CrossRef]
- Lim, E.-K.; Doucet, C.J.; Hou, B.; Jackson, R.G.; Abrams, S.R.; Bowles, D.J. Resolution of (+)-abscisic acid using an Arabidopsis glycosyltransferase. Tetrahedron Asymmetry 2005, 16, 143–147. [Google Scholar] [CrossRef]
- Liu, Z.; Yan, J.P.; Li, D.K.; Luo, Q.; Yan, Q.; Liu, Z.B.; Ye, L.M.; Wang, J.M.; Li, X.F.; Yang, Y. UDP-glucosyltransferase71C5, a major glucosyltransferase, mediates abscisic acid homeostasis in Arabidopsis. Plant Physiol. 2015, 167, 1659–1670. [Google Scholar] [CrossRef]
- Cao, M.-J.; Zhang, Y.-L.; Liu, X.; Huang, H.; Zhou, X.E.; Wang, W.-L.; Zeng, A.; Zhao, C.-Z.; Si, T.; Du, J.; et al. Combining chemical and genetic approaches to increase drought resistance in plants. Nat. Commun. 2017, 8, 1183. [Google Scholar] [CrossRef] [PubMed]
- Okamoto, M.; Peterson, F.C.; Defries, A.; Park, S.-Y.; Endo, A.; Nambara, E.; Volkman, B.F.; Cutler, S.R. Activation of dimeric ABA receptors elicits guard cell closure, ABA-regulated gene expression, and drought tolerance. Proc. Natl. Acad. Sci. USA 2013, 110, 12132–12137. [Google Scholar] [CrossRef]
- Dejonghe, W.; Cutler, S.R. Abscisic acid as a gateway for the crops of tomorrow. In Advances in Botanical Research; Elsevier: Amsterdam, The Netherlands, 2019; Volume 92, pp. 341–370. ISBN 978-0-08-102620-5. [Google Scholar]
- Takeuchi, J.; Nagamiya, H.; Moroi, S.; Ohnishi, T.; Todoroki, Y. Design of potent ABA receptor antagonists based on a conformational restriction approach. Org. Biomol. Chem. 2020, 18, 4988. [Google Scholar] [CrossRef]
- Diddi, N.; Lai, L.; Brookbank, B.P.; Hussain, S.; Nambara, E.; Todd, C.; Nourimand, M.; Tar’an, B.; Song, D.; Holbrook, L.; et al. 3′-(Phenyl alkynyl) analogs of abscisic acid: Synthesis and biological activity of potent ABA antagonists. Org. Biomol. Chem. 2021, 19, 2978–2985. [Google Scholar] [CrossRef] [PubMed]
- Abrams, S.R.; Lai, L.; Diddi, N. 3′-Unsaturated abscisic acid derivatives as ABA antagonists. U.S. Patent WO 2020/102892 A1, 2020. [Google Scholar]
- Diddi, N.; Lai, L.; Nguyen, C.H.; Yan, D.; Nambara, E.; Abrams, S.R. An efficient and scalable synthesis of the potent analog (+)-tetralone ABA from natural ABA. Org. Biomol. Chem. 2023, 21, 3014–3019. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nguyen, C.H.; Yan, D.; Nambara, E. Persistence of Abscisic Acid Analogs in Plants: Chemical Control of Plant Growth and Physiology. Genes 2023, 14, 1078. https://doi.org/10.3390/genes14051078
Nguyen CH, Yan D, Nambara E. Persistence of Abscisic Acid Analogs in Plants: Chemical Control of Plant Growth and Physiology. Genes. 2023; 14(5):1078. https://doi.org/10.3390/genes14051078
Chicago/Turabian StyleNguyen, Christine H., Dawei Yan, and Eiji Nambara. 2023. "Persistence of Abscisic Acid Analogs in Plants: Chemical Control of Plant Growth and Physiology" Genes 14, no. 5: 1078. https://doi.org/10.3390/genes14051078
APA StyleNguyen, C. H., Yan, D., & Nambara, E. (2023). Persistence of Abscisic Acid Analogs in Plants: Chemical Control of Plant Growth and Physiology. Genes, 14(5), 1078. https://doi.org/10.3390/genes14051078






