Aberrantly Expressed tRNA-Val Fragments Can Distinguish Canine Hepatocellular Carcinoma from Canine Hepatocellular Adenoma
Abstract
:1. Introduction
2. Materials and Methods
2.1. Clinical Samples
2.2. Cell Lines and Cell Culture
2.3. Isolation of EVs
2.4. RNA Extraction
2.5. qRT-PCR Analysis for tRNA-Val
2.6. Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) Enrichment and Protein-Protein Interaction (PPI) Analysis
2.7. Statistical Analysis
3. Results
3.1. Expression of tRNA-Val Using qRT-PCR
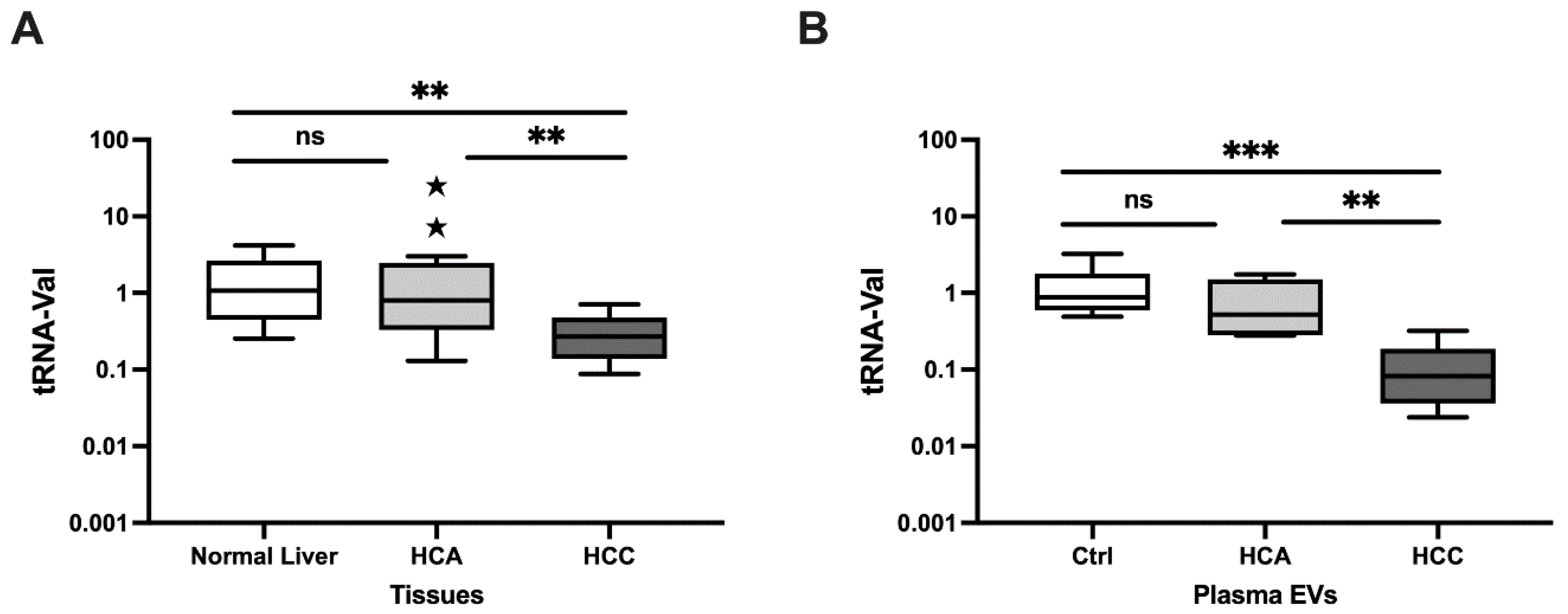
3.1.1. Relative Expression in Clinical Tissue Samples
3.1.2. Relative Expression in Plasma EVs
3.1.3. Relative Expression in Canine HCC Cell Lines
3.2. Diagnostic Value of tRNA-Val
3.3. Predicted Targets for tRNA-Val and Enrichment Analyses
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Zheng, J.; Sadot, E.; Vigidal, J.A.; Klimstra, D.S.; Balachandran, V.P.; Kingham, T.P.; Allen, P.J.; D’Angelica, M.I.; DeMatteo, R.P.; Jarnagin, W.R.; et al. Characterization of hepatocellular adenoma and carcinoma using microRNA profiling and targeted gene sequencing. PLoS ONE 2018, 13, e0200776. [Google Scholar] [CrossRef] [PubMed]
- Patnaik, A.K.; Hurvitz, A.I.; Lieberman, P.H.; Johnson, G.F. Canine hepatocellular carcinoma. Vet. Pathol. 1981, 18, 427–438. [Google Scholar] [CrossRef] [PubMed]
- Van Sprundel, R.G.; van den Ingh, T.S.; Guscetti, F.; Kershaw, O.; Kanemoto, H.; van Gils, H.M.; Rothuizen, J.; Roskams, T.; Spee, B. Classification of primary hepatic tumours in the dog. Vet. J. 2013, 197, 596–606. [Google Scholar] [CrossRef] [PubMed]
- Forner, A.; Reig, M.; Bruix, J. Hepatocellular carcinoma. Lancet 2018, 391, 1301–1314. [Google Scholar] [CrossRef] [PubMed]
- Patnaik, A.K.; Hurvitz, A.I.; Lieberman, P.H. Canine hepatic neoplasms: A clinicopathologic study. Vet. Pathol. 1980, 17, 553–564. [Google Scholar] [CrossRef]
- Zucman-Rossi, J.; Jeannot, E.; Nhieu, J.T.; Scoazec, J.Y.; Guettier, C.; Rebouissou, S.; Bacq, Y.; Leteurtre, E.; Paradis, V.; Michalak, S.; et al. Genotype-phenotype correlation in hepatocellular adenoma: New classification and relationship with HCC. Hepatology 2006, 43, 515–524. [Google Scholar] [CrossRef] [PubMed]
- Bioulac-Sage, P.; Laumonier, H.; Couchy, G.; Le Bail, B.; Sa Cunha, A.; Rullier, A.; Laurent, C.; Blanc, J.F.; Cubel, G.; Trillaud, H.; et al. Hepatocellular adenoma management and phenotypic classification: The Bordeaux experience. Hepatology 2009, 50, 481–489. [Google Scholar] [CrossRef]
- Zhu, L.; Ge, J.; Li, T.; Shen, Y.; Guo, J. tRNA-derived fragments and tRNA halves: The new players in cancers. Cancer Lett. 2019, 452, 31–37. [Google Scholar] [CrossRef] [PubMed]
- Kuscu, C.; Kumar, P.; Kiran, M.; Su, Z.; Malik, A.; Dutta, A. tRNA fragments (tRFs) guide Ago to regulate gene expression post-transcriptionally in a Dicer-independent manner. RNA 2018, 24, 1093–1105. [Google Scholar] [CrossRef]
- Kumar, P.; Anaya, J.; Mudunuri, S.B.; Dutta, A. Meta-analysis of tRNA derived RNA fragments reveals that they are evolutionarily conserved and associate with AGO proteins to recognize specific RNA targets. BMC Biol. 2014, 12, 78. [Google Scholar] [CrossRef]
- Gong, M.; Deng, Y.; Xiang, Y.; Ye, D. The role and mechanism of action of tRNA-derived fragments in the diagnosis and treatment of malignant tumors. Cell Commun. Signal 2023, 21, 62. [Google Scholar] [CrossRef] [PubMed]
- Fu, M.; Gu, J.; Wang, M.; Zhang, J.; Chen, Y.; Jiang, P.; Zhu, T.; Zhang, X. Emerging roles of tRNA-derived fragments in cancer. Mol. Cancer 2023, 22, 30. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Liu, D.; Zhang, L.; Wang, J.; Ding, Y.; Sun, Z.; Wang, W. 5′-tiRNA-Gln inhibits hepatocellular carcinoma progression by repressing translation through the interaction with eukaryotic initiation factor 4A-I. Front. Med. 2023, 17, 476–492. [Google Scholar] [CrossRef] [PubMed]
- Panoutsopoulou, K.; Magkou, P.; Dreyer, T.; Dorn, J.; Obermayr, E.; Mahner, S.; van Gorp, T.; Braicu, I.; Magdolen, V.; Zeillinger, R.; et al. tRNA-derived small RNA 3′U-tRF(ValCAC) promotes tumour migration and early progression in ovarian cancer. Eur. J. Cancer 2023, 180, 134–145. [Google Scholar] [CrossRef] [PubMed]
- Cui, H.; Li, H.; Wu, H.; Du, F.; Xie, X.; Zeng, S.; Zhang, Z.; Dong, K.; Shang, L.; Jing, C.; et al. A novel 3′tRNA-derived fragment tRF-Val promotes proliferation and inhibits apoptosis by targeting EEF1A1 in gastric cancer. Cell Death Dis. 2022, 13, 471. [Google Scholar] [CrossRef] [PubMed]
- Gu, X.; Ma, S.; Liang, B.; Ju, S. Serum hsa_tsr016141 as a Kind of tRNA-Derived Fragments Is a Novel Biomarker in Gastric Cancer. Front. Oncol. 2021, 11, 679366. [Google Scholar] [CrossRef] [PubMed]
- Zheng, J.; Li, C.; Zhu, Z.; Yang, F.; Wang, X.; Jiang, P.; Yan, F. A 5′-tRNA Derived Fragment NamedtiRNA-Val-CAC-001 Works as a Suppressor in Gastric Cancer. Cancer Manag. Res. 2022, 14, 2323–2337. [Google Scholar] [CrossRef] [PubMed]
- Xu, W.; Zhou, B.; Wang, J.; Tang, L.; Hu, Q.; Wang, J.; Chen, H.; Zheng, J.; Yan, F.; Chen, H. tRNA-Derived Fragment tRF-Glu-TTC-027 Regulates the Progression of Gastric Carcinoma via MAPK Signaling Pathway. Front. Oncol. 2021, 11, 733763. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Li, Z.; Yu, X.; Ruan, Y.; Shen, Y.; Shao, Y.; Zhang, X.; Ye, G.; Guo, J. The tRNA-derived fragment 5026a inhibits the proliferation of gastric cancer cells by regulating the PTEN/PI3K/AKT signaling pathway. Stem Cell Res. Ther. 2021, 12, 418. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Bi, Z.; Dong, X.; Yu, M.; Wang, K.; Song, X.; Xie, L.; Song, X. tRNA-derived fragments: tRF-Gly-CCC-046, tRF-Tyr-GTA-010 and tRF-Pro-TGG-001 as novel diagnostic biomarkers for breast cancer. Thorac. Cancer 2021, 12, 2314–2323. [Google Scholar] [CrossRef]
- Tong, L.; Zhang, W.; Qu, B.; Zhang, F.; Wu, Z.; Shi, J.; Chen, X.; Song, Y.; Wang, Z. The tRNA-Derived Fragment-3017A Promotes Metastasis by Inhibiting NELL2 in Human Gastric Cancer. Front. Oncol. 2020, 10, 570916. [Google Scholar] [CrossRef]
- Mo, D.; Jiang, P.; Yang, Y.; Mao, X.; Tan, X.; Tang, X.; Wei, D.; Li, B.; Wang, X.; Tang, L.; et al. A tRNA fragment, 5′-tiRNA(Val), suppresses the Wnt/β-catenin signaling pathway by targeting FZD3 in breast cancer. Cancer Lett. 2019, 457, 60–73. [Google Scholar] [CrossRef] [PubMed]
- Sahlolbei, M.; Fattahi, F.; Vafaei, S.; Rajabzadeh, R.; Shiralipour, A.; Madjd, Z.; Kiani, J. Relationship Between Low Expressions of tRNA-Derived Fragments with Metastatic Behavior of Colorectal Cancer. J. Gastrointest. Cancer 2022, 53, 862–869. [Google Scholar] [CrossRef] [PubMed]
- Rahman, M.M.; Lai, Y.C.; Husna, A.A.; Chen, H.W.; Tanaka, Y.; Kawaguchi, H.; Hatai, H.; Miyoshi, N.; Nakagawa, T.; Fukushima, R.; et al. Aberrantly expressed snoRNA, snRNA, piRNA and tRFs in canine melanoma. Vet. Comp. Oncol. 2020, 18, 353–361. [Google Scholar] [CrossRef] [PubMed]
- Hasan, M.N.; Rahman, M.M.; Husna, A.A.; Nozaki, N.; Yamato, O.; Miura, N. YRNA and tRNA fragments can differentiate benign from malignant canine mammary gland tumors. Biochem. Biophys. Res. Commun. 2024, 691, 149336. [Google Scholar] [CrossRef]
- Zaborowski, M.P.; Balaj, L.; Breakefield, X.O.; Lai, C.P. Extracellular Vesicles: Composition, Biological Relevance, and Methods of Study. Bioscience 2015, 65, 783–797. [Google Scholar] [CrossRef]
- Zheng, B.; Song, X.; Wang, L.; Zhang, Y.; Tang, Y.; Wang, S.; Li, L.; Wu, Y.; Song, X.; Xie, L. Plasma exosomal tRNA-derived fragments as diagnostic biomarkers in non-small cell lung cancer. Front. Oncol. 2022, 12, 1037523. [Google Scholar] [CrossRef]
- Xi, J.; Zeng, Z.; Li, X.; Zhang, X.; Xu, J. Expression and Diagnostic Value of tRNA-Derived Fragments Secreted by Extracellular Vesicles in Hypopharyngeal Carcinoma. Onco Targets Ther. 2021, 14, 4189–4199. [Google Scholar] [CrossRef]
- Lin, C.; Zheng, L.; Huang, R.; Yang, G.; Chen, J.; Li, H. tRFs as Potential Exosome tRNA-Derived Fragment Biomarkers for Gastric Carcinoma. Clin. Lab. 2020, 66, 961. [Google Scholar] [CrossRef]
- Husna, A.A.; Rahman, M.M.; Lai, Y.C.; Chen, H.W.; Hasan, M.N.; Nakagawa, T.; Miura, N. Identification of melanoma-specific exosomal miRNAs as the potential biomarker for canine oral melanoma. Pigment. Cell Melanoma Res. 2021, 34, 1062–1073. [Google Scholar] [CrossRef]
- Gustafson, D.L.; Duval, D.L.; Regan, D.P.; Thamm, D.H. Canine sarcomas as a surrogate for the human disease. Pharmacol. Ther. 2018, 188, 80–96. [Google Scholar] [CrossRef] [PubMed]
- Gardner, H.L.; Fenger, J.M.; London, C.A. Dogs as a Model for Cancer. Annu. Rev. Anim. Biosci. 2016, 4, 199–222. [Google Scholar] [CrossRef] [PubMed]
- Lai, Y.C.; Ushio, N.; Rahman, M.M.; Katanoda, Y.; Ogihara, K.; Naya, Y.; Moriyama, A.; Iwanaga, T.; Saitoh, Y.; Sogawa, T.; et al. Aberrant expression of microRNAs and the miR-1/MET pathway in canine hepatocellular carcinoma. Vet. Comp. Oncol. 2018, 16, 288–296. [Google Scholar] [CrossRef] [PubMed]
- Fujimoto, A.; Neo, S.; Ishizuka, C.; Kato, T.; Segawa, K.; Kawarai, S.; Ogihara, K.; Hisasue, M.; Tsuchiya, R. Identification of cell surface antigen expression in canine hepatocellular carcinoma cell lines. J. Vet. Med. Sci. 2013, 75, 831–835. [Google Scholar] [CrossRef] [PubMed]
- Hino, Y.; Rahman, M.M.; Lai, Y.C.; Husna, A.A.; Chen, H.W.; Hasan, M.N.; Nakagawa, T.; Miura, N. Hypoxic miRNAs expression are different between primary and metastatic melanoma cells. Gene 2021, 782, 145552. [Google Scholar] [CrossRef] [PubMed]
- Husna, A.A.; Rahman, M.M.; Chen, H.W.; Hasan, M.N.; Nakagawa, T.; Miura, N. Long non-coding RNA and transfer RNA-derived small fragments in exosomes are potential biomarkers for canine oral melanoma. Vet. Comp. Oncol. 2022, 20, 653–663. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Li, J.; Gong, Y.; Wu, Q.; Tan, S.; Sun, D.; Xu, X.; Zuo, Y.; Zhao, Y.; Wei, Y.Q.; et al. Exosomal tRNA-derived small RNA as a promising biomarker for cancer diagnosis. Mol. Cancer 2019, 18, 74. [Google Scholar] [CrossRef] [PubMed]
- Weng, Q.; Wang, Y.; Xie, Y.; Yu, X.; Zhang, S.; Ge, J.; Li, Z.; Ye, G.; Guo, J. Extracellular vesicles-associated tRNA-derived fragments (tRFs): Biogenesis, biological functions, and their role as potential biomarkers in human diseases. J. Mol. Med. 2022, 100, 679–695. [Google Scholar] [CrossRef]
- Suresh, P.S.; Thankachan, S.; Venkatesh, T. Landscape of Clinically Relevant Exosomal tRNA-Derived Non-coding RNAs. Mol. Biotechnol. 2023, 65, 300–310. [Google Scholar] [CrossRef]
- Wang, J.; Ma, G.; Li, M.; Han, X.; Xu, J.; Liang, M.; Mao, X.; Chen, X.; Xia, T.; Liu, X.; et al. Plasma tRNA Fragments Derived from 5′ Ends as Novel Diagnostic Biomarkers for Early-Stage Breast Cancer. Mol. Ther. Nucleic Acids 2020, 21, 954–964. [Google Scholar] [CrossRef]
- Majumder, A.; Syed, K.M.; Mukherjee, A.; Lankadasari, M.B.; Azeez, J.M.; Sreeja, S.; Harikumar, K.B.; Pillai, M.R.; Dutta, D. Enhanced expression of histone chaperone APLF associate with breast cancer. Mol. Cancer 2018, 17, 76. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.; Wang, G.; Liu, Y.; Mei, C.; Yao, Y.; Wu, X.; Chen, X.; Ma, W.; Li, K.; Zhang, Z.; et al. A novel long non-coding RNA RP11-286H15.1 represses hepatocellular carcinoma progression by promoting ubiquitination of PABPC4. Cancer Lett. 2021, 499, 109–121. [Google Scholar] [CrossRef] [PubMed]
- Zhu, W.; Zhang, Q.; Liu, M.; Yan, M.; Chu, X.; Li, Y. Identification of DNA repair-related genes predicting pathogenesis and prognosis for liver cancer. Cancer Cell Int. 2021, 21, 81. [Google Scholar] [CrossRef] [PubMed]
- Hernanda, P.Y.; Chen, K.; Das, A.M.; Sideras, K.; Wang, W.; Li, J.; Cao, W.; Bots, S.J.; Kodach, L.L.; de Man, R.A.; et al. SMAD4 exerts a tumor-promoting role in hepatocellular carcinoma. Oncogene 2015, 34, 5055–5068. [Google Scholar] [CrossRef] [PubMed]
- Kang, A.R.; An, H.T.; Ko, J.; Choi, E.J.; Kang, S. Ataxin-1 is involved in tumorigenesis of cervical cancer cells via the EGFR-RAS-MAPK signaling pathway. Oncotarget 2017, 8, 94606–94618. [Google Scholar] [CrossRef] [PubMed]
- Ray, D.; Yun, Y.C.; Idris, M.; Cheng, S.; Boot, A.; Iain, T.B.H.; Rozen, S.G.; Tan, P.; Epstein, D.M. A tumor-associated splice-isoform of MAP2K7 drives dedifferentiation in MBNL1-low cancers via JNK activation. Proc. Natl. Acad. Sci. USA 2020, 117, 16391–16400. [Google Scholar] [CrossRef] [PubMed]
- Luo, C.; Cheng, Y.; Liu, Y.; Chen, L.; Liu, L.; Wei, N.; Xie, Z.; Wu, W.; Feng, Y. SRSF2 Regulates Alternative Splicing to Drive Hepatocellular Carcinoma Development. Cancer Res. 2017, 77, 1168–1178. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Huang, W.L.; Xu, Q.G.; Zhang, L.; Sun, S.H.; Zhou, W.P.; Yang, F. Overactivated neddylation pathway in human hepatocellular carcinoma. Cancer Med. 2018, 7, 3363–3372. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Yang, J.; Song, Q.; Ding, X.; Sun, F.; Yang, L. UBA3 promotes the occurrence and metastasis of intrahepatic cholangiocarcinoma through MAPK signaling pathway. Acta Biochim. Biophys. Sin. 2024, 56, 199–209. [Google Scholar] [CrossRef] [PubMed]
- Bieberich, E. Synthesis, Processing, and Function of N-glycans in N-glycoproteins. Adv. Neurobiol. 2014, 9, 47–70. [Google Scholar]
- Tu, Y.; Chen, C.; Pan, J.; Xu, J.; Zhou, Z.G.; Wang, C.Y. The Ubiquitin Proteasome Pathway (UPP) in the regulation of cell cycle control and DNA damage repair and its implication in tumorigenesis. Int. J. Clin. Exp. Pathol. 2012, 5, 726–738. [Google Scholar] [PubMed]
- Shang, F.; Taylor, A. Roles for the ubiquitin-proteasome pathway in protein quality control and signaling in the retina: Implications in the pathogenesis of age-related macular degeneration. Mol. Asp. Med. 2012, 33, 446–466. [Google Scholar] [CrossRef] [PubMed]
- Hu, M.; Zhang, R.; Yang, J.; Zhao, C.; Liu, W.; Huang, Y.; Lyu, H.; Xiao, S.; Guo, D.; Zhou, C.; et al. The role of N-glycosylation modification in the pathogenesis of liver cancer. Cell Death Dis. 2023, 14, 222. [Google Scholar] [CrossRef] [PubMed]
- DelaCourt, A.; Black, A.; Angel, P.; Drake, R.; Hoshida, Y.; Singal, A.; Lewin, D.; Taouli, B.; Lewis, S.; Schwarz, M.; et al. N-Glycosylation Patterns Correlate with Hepatocellular Carcinoma Genetic Subtypes. Mol. Cancer Res. 2021, 19, 1868–1877. [Google Scholar] [CrossRef] [PubMed]
- Park, J.S.; Ma, H.; Roh, Y.S. Ubiquitin pathways regulate the pathogenesis of chronic liver disease. Biochem. Pharmacol. 2021, 193, 114764. [Google Scholar] [CrossRef]
- Oh, J.H.; Cho, J.Y. Comparative oncology: Overcoming human cancer through companion animal studies. Exp. Mol. Med. 2023, 55, 725–734. [Google Scholar] [CrossRef]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hashimoto, S.; Hasan, M.N.; Arif, M.; Nozaki, N.; Husna, A.A.; Furusawa, Y.; Sogawa, T.; Takahashi, K.; Kuramoto, T.; Noguchi, A.; et al. Aberrantly Expressed tRNA-Val Fragments Can Distinguish Canine Hepatocellular Carcinoma from Canine Hepatocellular Adenoma. Genes 2024, 15, 1024. https://doi.org/10.3390/genes15081024
Hashimoto S, Hasan MN, Arif M, Nozaki N, Husna AA, Furusawa Y, Sogawa T, Takahashi K, Kuramoto T, Noguchi A, et al. Aberrantly Expressed tRNA-Val Fragments Can Distinguish Canine Hepatocellular Carcinoma from Canine Hepatocellular Adenoma. Genes. 2024; 15(8):1024. https://doi.org/10.3390/genes15081024
Chicago/Turabian StyleHashimoto, Saki, MD Nazmul Hasan, Mohammad Arif, Nobuhiro Nozaki, Al Asmaul Husna, Yu Furusawa, Takeshi Sogawa, Kaori Takahashi, Tomohide Kuramoto, Aki Noguchi, and et al. 2024. "Aberrantly Expressed tRNA-Val Fragments Can Distinguish Canine Hepatocellular Carcinoma from Canine Hepatocellular Adenoma" Genes 15, no. 8: 1024. https://doi.org/10.3390/genes15081024







