1. Introduction
Charcoal grilling is a popular method used worldwide for the dry cooking of meats such as beef and pork, especially in Korea, due to its pleasant smell and the charcoal taste that it imparts to cooked meat. Pan-broiling is another common method that is also used for cooking such meats.
However, it has been reported that a variety of hazardous compounds are formed during the cooking of meat and have been detected in food and air. Polycyclic aromatic hydrocarbons (PAHs) are compounds that are well known to form during the cooking of food. The generation of such compounds varies according to the cooking method used, with the highest concentration produced during grilling and smoking [
1]. The most extensively studied meats to date are beef, mutton, pork, and chicken. The concentrations of benzo[
a]pyrene (BaP), the most carcinogenic PAH, in cooked meat have been found to be in the range of 0.9 to 71 µg/kg [
2,
3,
4,
5,
6]. Pork belly, which contains the highest fat content among them, has been shown to produce the highest levels of PAHs [
5]. This is probably because when the pork is cooked, fat falls onto charcoal flames and is pyrolyzed at a high temperature (usually >500 °C), thus leading to the thermal synthesis of PAHs [
7,
8,
9,
10]. Therefore, the consumption of such cooked meat may add to a carcinogenic health risk.
Recently, concern about volatile
N-nitrosamines (NAs), in particular
N-nitrosodimethylamine (NDMA) in food and drinking water, has greatly increased due to its potent carcinogenicity in the liver and its frequent occurrence [
11,
12]. The United States Environmental Protection Agency (U.S. EPA) classified NDMA as a group B2 carcinogen and estimated the cancer slope factor of its oral ingestion exposure to be 51 (mg/kg/day)
−1 [
13]. This value is 51 times higher than that [1 (mg/kg/day)
−1] for BaP, which is classified as carcinogenic to humans (group A) [
14], indicating that NDMA may have very high carcinogenicity.
It has been reported that NAs, mainly NDMA, are formed in cured meat products through the reaction of secondary or tertiary amines with nitrite or nitrate [
15,
16]. NDMA and
N-nitrosodiethylamine (NDEA) were found in charcoal-grilled mutton, and their concentrations were observed to increase with cooking duration [
17]. NDMA and
N-nitrosopyrrolidine (NPYR) have also been found in fried bacon [
18]. These findings suggest that NDMA and other NAs may be released into the air during the dry-heat cooking of meat such as charcoal grilling and contribute to their elevated concentration in the air, in particular indoors. This is because indoor ventilation is usually operated too poorly in such charcoal grill restaurants to reduce their airborne levels.
To date, however, few studies on the release of airborne volatile NAs have been conducted, even though a potential health risk from human exposure to them via inhalation is highly expected. A report that NDMA, NPYR, and
N-nitrosopiperidine (NPIP) were found in the combustion smoke of some organic materials such as dried fish, hair, and cigarettes suggested that biological components such as secondary amines may be the major precursors for the formation of NAs [
19].
Airborne NAs were measured in the following ways. Particulate matter air samples were usually collected on quartz filters or PTFE membrane filters, and analyzed for NAs by chromatography (GC) coupled to tandem mass spectrometry following ultrasonication or pressurized solvent extraction [
20,
21,
22]. On the other hand, Thermosorb-N air sampling cartridges (Ellutia, Iselin, USA) have been used for collecting NAs in ambient air since they have the advantage of preventing the formation of NAs after sampling into the cartridges, and the concentrations of NAs were usually measured using GC with thermal energy analysis detection or MS [
23,
24,
25]. In the present study, high-performance liquid chromatography (HPLC) coupled to fluoresce detection (FLD) was employed instead for the quantitative determination of ambient NAs following their qualitative analysis by ultra-performance liquid chromatography-mass/mass (UPLC-MS/MS).
This study aimed to investigate the release of volatile NAs into indoor air as a result of the charcoal grilling and pan-broiling of meat. Three types of meat (beef sirloin, pork belly, and duck) were selected since they are the most frequently consumed meats.
2. Materials and Methods
2.1. Reagents
Seven standard NAs, N-nitrosomorpholine (NMOR), NDMA, N-nitrosomethylethylamine (NMEA), NPYR, NDEA, NPIP, and N-nitrosodi-n-propylamine (NDPA), were purchased from Supelco (Bellefonte, USA) in concentrations of 5000 or 1000 mg/mL (for NMEA) and diluted to 1 mg/L with methanol for use as standard solutions. Dichloromethane (DCM), acetonitrile, methanol, and acetone were obtained from Burdick & Jackson (Morris Plains, USA). A denitrosating agent was prepared by mixing 1 mL of 48% hydrobromic acid (Wako, Osaka, Japan) and 10 mL of glacial acetic acid (Daejung, Siheung, Korea) and was stored in a 40 mL amber glass vial at 4 °C for 2 weeks prior to use. The agent for the derivatization of NAs (50 mL) was prepared by dissolving 25 mg of 5-(dimethylamino)naphthalene-1-sulfonyl (dansyl) chloride (Merck Millipore, Darmstadt, Germany) in acetone. A buffer solution (50 mL) of pH 10.5 was prepared by dissolving 0.32 g of NaOH (Kanto, Tokyo, Japan) and 2.0 g of NaHCO3 (Daejung) in ultrapure water.
2.2. Experimental Setting for the Dry-Heat Cooking of Meat
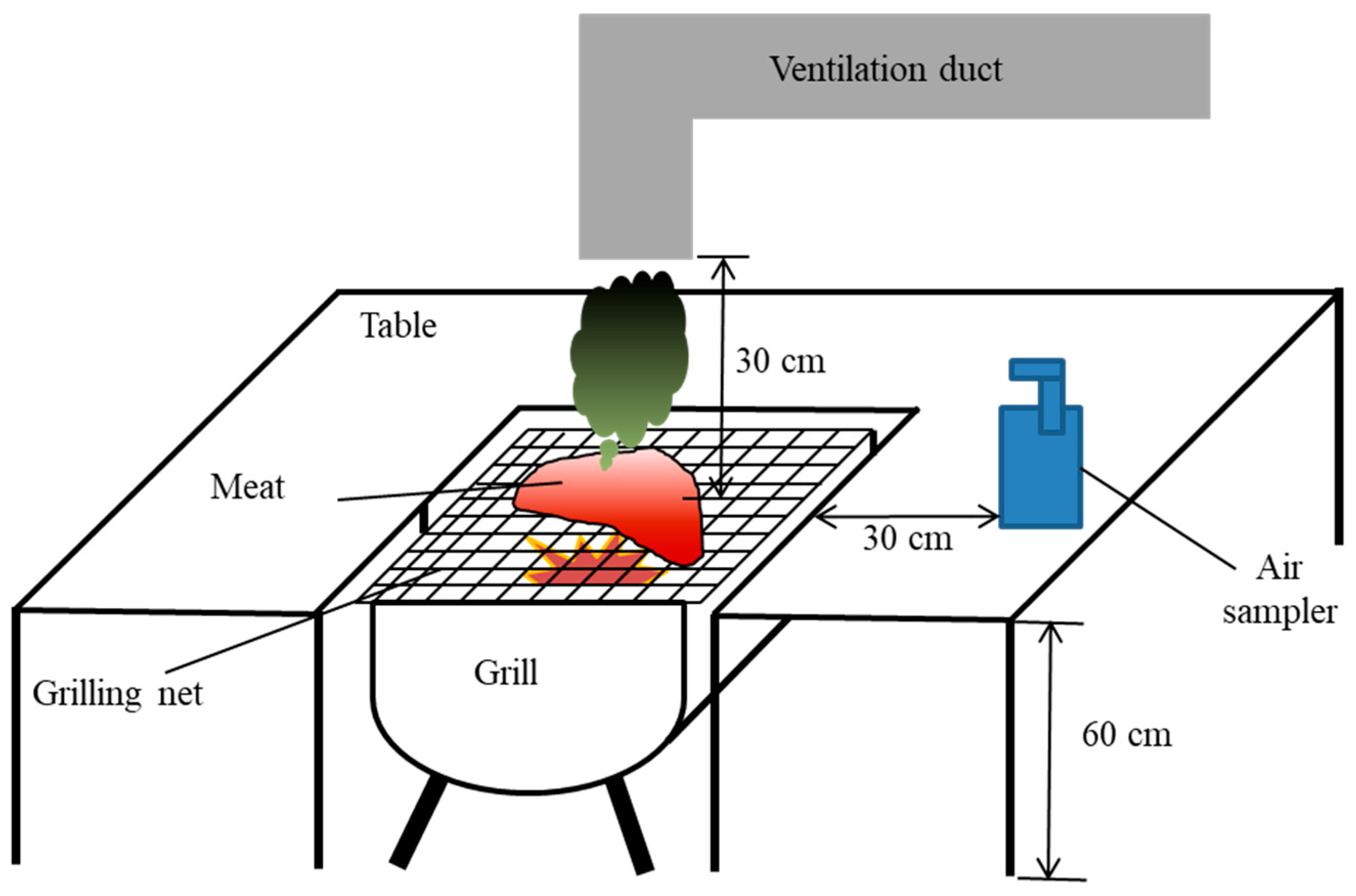
The dry-heat cooking experiments were conducted in a temporary building (
Figure 1) made of plywood. The building had dimensions of 2.4 m × 2.4 m × 2.4 m (width × length × height). An entrance door was installed 0.35 m from the left side with dimensions of 0.88 m × 1.75 m, and a window was installed 0.4 m from the top of the building with dimensions of 0.4 m × 0.4 m. A ventilation fan with a capacity of 11 m
3/min (0.3 m × 0.3 m, Shinil, Seoul, Korea) was placed on the opposite side of the building to the window.
Figure 2 shows the setup for the charcoal grilling of the meat and collection of air samples. A grill was erected at a height of 60 cm in the middle of the temporary building. To mimic a charcoal grill restaurant, one end of a ventilating pipe made from zinc-galvanized steel plate with an internal diameter of 10 cm was installed at a height of 30 cm over the top of the grill and the other end was placed close to the ventilation fan. During the experiment, the door was closed, the window was open, and the exhaust fan was continuously operated.
Three types of meat were chosen for the experiments: beef sirloin, pork belly, and duck since they are the most commonly consumed in Korea. Approximately 500 g of each type of meat, which is an average serving for two to three people, was used for each cooking experiment. Approximately 500 g of white charcoal was used as a fuel for grilling and was burned for ca. 30 min prior to grilling. Each type of meat was cut into small pieces suitable for eating and heat cooked for approximately 5, 16, and 25 min for duck, beef, and pork, respectively. In addition, pan-broiling was also conducted in the same manner as charcoal grilling using a portable butane range.
Charcoal grilling experiments were conducted with beef sirloin and pork belly 12 times each, while duck meat was grilled seven times. The pan-broiling experiments were repeated four times for each type of meat (
Table 1). Surface temperatures on the burning charcoal, grill, and pan were measured at the beginning of the experiments and 25 min after the start of cooking.
2.3. Ambient Air Sampling
Ambient air samples were collected in Thermosorb-N air sampling cartridges at 2 L/min at a distance of 30 cm from the edge of the grill or pan for 50 min to mimic human exposure to air pollutants through inhalation while staying in the restaurant, starting at the beginning of heat cooking, using a PCXR4 Universal Sample Pump (SKC, Eighty Four, USA) (
Figure 2). One to three replicate samples were collected during each experiment. The building was ventilated for at least 2 h between two consecutive experiments by opening the entrance door and window and continuously operating the exhaust fan. Background air samples were collected prior to each cooking experiment. Additional air samples were collected without meat cooking for both grilling and broiling to check the emission of NAs from the burning of fuel alone (white charcoal and butane gas).
2.4. Qualitative and Quantitative Analyses for N-Nitrosamines
Twenty microliters of a nitrosamine standard solution (20 ng/L) were injected into the HPLC-FLD after dansyl derivatization to identify the chromatographic retention times of the seven analytes. An ultra-performance liquid chromatograph-tandem mass spectrometer (UPLC-MS/MS) was used to identify NAs in the collected air samples, as described below. Two milliliters of a mixed eluent of dichloromethane (DCM) and methanol (95:5, v/v) were passed through the Thermosorb-N cartridge at a flow rate of 0.5 mL/min in the opposite direction to the air sampling, and the eluate was concentrated to 1 mL using a mild stream of N2 gas. Two microliters of the concentrate were injected into the UPLC system (Nanospace SI-2, Shiseido, Tokyo, Japan) coupled to a mass spectrometer (TSQ Quantum UltraTM Triple Quadrupole, Thermo Fisher Scientific, Waltham, USA). The stationary phase used was an Acquity UPLC HSS C18 column (100 mm × 2.1 mm × 1.8 µm, Waters, Milford, USA). The mobile phase was composed of methanol (A) and 5 mM ammonium formate (B), with a flow rate of 0.2 mL/min using the following gradients: 45% A from 0 to 3 min, 45% to 95% A from 3 to 5 min, 95% A from 5 to 7.5 min, and 95% to 5% A from 7.5 to 13 min. The column temperature was maintained at 40 °C during the analysis. The mass spectrometer was operated in selected reaction monitoring (SRM) mode.
The Thermosorb-N cartridge eluate (2 mL), which was obtained using the same procedure as that for the qualitative analysis, was washed with 0.5 mL of 0.1 N aqueous HCl solution to remove secondary amines possibly included in the eluate, and the organic layer was concentrated to 1 mL using a mild stream of N2 gas. The NAs were then analyzed following dansyl derivatization. Ten microliters of the denitrosating agent were added to the concentrate, and the mixture was heated at 40 °C for 30 min, after which it was evaporated to dryness at 60 °C using a mild stream of N2 gas. The residue was then heated to 40 °C for 30 min with 150 µL of a pH 10.5 buffer solution and 150 µL of the dansyl chloride solution. After adding 50 µL of ultrapure water to the cooled mixture, it was filtered through a Pasteur pipette containing fat-removed cotton wool. The filtrate was transferred to an autosampler vial. For quantitative analysis, a Varian ProStar pump (Palo Alto, USA) and a Waters 470 FLD (Waters, Milford, USA) were used. The stationary phase used was a SKYPAK C18 column (250 mm × 4.6 mm × 3 μm; SK Chemicals, Seongnam, Korea), and the mobile phase, consisting of acetonitrile and water (55:45, v/v), was set at a flow rate of 1 mL/min. Twenty microliters of the analytical sample were injected into the HPLC system, and the wavelengths used for the emission and excitation of the FLD were 340 and 530 nm, respectively.
2.5. Method Validation and Data Analysis
A five-point calibration curve for NDMA was prepared by spiking 1, 10, 20, 30, and 40 µL of a 1 mg/L standard solution, which corresponded to air NDMA concentrations of 10, 100, 200, 300, and 400 ng/m
3, respectively, for an air volume of 0.1 m
3, into separate Thermosorb-N cartridges and analyzing them following the same procedure as that used for the air samples. The method detection limit (MDL) and limit of quantitation (LOQ) for NDMA were estimated according to the EPA method [
26]. Repeatability, defined as analytical precision, was expressed as the relative standard deviation (RSD) of the measurements of ten 200 ng/m
3 standard cartridges. The accuracy was evaluated as both an average recovery (%) of the measurements of three 200 ng/m
3 standard cartridges and the percentage error (
) of three cartridge measurements for the same concentration. An air sampling breakthrough test was conducted in duplicate by withdrawing air through two cartridges connected in series using Tygon
® tubing and comparing the chromatographic peak areas in the front and back cartridges.
Values measuring less than the MDL were regarded as the MDL/2 in the data analysis [
27], and IBM SPSS Statistics (version 24.0, IBM Corp., Armonk, USA) was used for the following statistical analyses. Student’s
t-tests were conducted to determine the statistical difference of the PAH concentrations arising from the charcoal grilling and pan-broiling of each type of meat. The differences between the three different types of meat for each cooking method were tested using one-way analysis of variance (ANOVA) tests.
2.6. Inhalation Cancer Risk Assessment
Inhalation cancer risk assessment for NDMA exposure while eating meat in a charcoal grill restaurant was conducted according to the U.S. EPA procedure [
28]. Excess lifetime cancer risks (ELCR) were estimated for NDMA exposure levels corresponding to the average and 95th percentile values, assuming that people eat dry-heat cooked meat in charcoal grill restaurants once a week. Exposure factors in the Exposure Factors Handbook [
29] and the Korean Statistical Information Service [
30] were used. The values of meat consumption frequency were obtained from the Korean Rural Economic Institute [
31]. Using the above data, annual meat consumption amounts per capita were estimated to be 10.8, 21.8 and 2.2 kg for beef, pork, and duck, respectively. From these results, daily meat consumption rates were estimated to be 29.6, 59.7, and 6.00 g for beef, pork, and duck, respectively. Assuming the consumption of 150 g of meat during each meal, beef, pork, and duck meats are eaten every 5.1, 2.5, and 24.9 days. Therefore, beef, pork, and duck meats were estimated to be consumed with exposure frequencies (EF) of 72, 145, and 15 days per year, respectively. It was assumed that beef sirloin and pork belly were consumed as sole sources of beef and pork meats, respectively. The exposure time (ET) for which restaurant customers stay indoors was assumed to be 1.5 h/day. The exposure duration (ED) was assumed to be 79 yr (from 3 to 82 yr of age). The inhalation cancer slope factor (CSF) for NDMA was calculated to be 49 (mg/kg/day)
−1 by multiplying its inhalation unit risk, 1.4 × 10
−2 per µg/m
3 [
13], by a conversion factor (3.5 × 10
3) [
32]. Lifetime average daily doses and ELCRs were estimated using the following equations:
where,
LADDinh: Lifetime average daily dose via inhalation (mg/kg-day)
Cair: NDMA concentration in the air (mg/m3)
IR: Inhalation rate (17.65 m3/day)
ET: Exposure time (1.5 h/day)
EF: Exposure frequency (72, 145, and 55 day/yr for beef, pork, and duck meats, respectively)
ED: Exposure duration (79 yr)
BW: Body weight (62.8 kg)
LT: Lifetime (82 yr)
ELCRinh: Excess lifetime cancer risk via inhalation
CSF: cancer slope factor for NDMA [49 (mg/kg/day)−1].
4. Discussion
This study has shown that the charcoal grilling of meat, in particular beef and pork, is an important source of airborne NDMA in charcoal grill restaurants. Therefore, it suggested that people can be exposed to the carcinogenic compound while eating such cooked meat by inhaling the indoor air. Pan-broiling was also found to be a contributor to indoor NDMA concentration elevation, but to a small degree compared to charcoal grilling.
NAs other than NDMA might also be formed during the heat cooking of meat, but they were not detected in the air samples, probably because of their lower formation and/or lower volatility than NDMA.
Although the surface temperature values of the charcoal grill and frying pan were not very different from each other, there was a significant difference in the airborne NDMA concentrations between the two cooking methods for all types of meat. The major difference between the two cooking methods is that most of the meat is heated directly by open flame with the charcoal grilling method, although the other part is heated indirectly on the grill surface. Contrarily, with the pan-broiling method, the meat is heated indirectly through the pan surface.
In some previous studies, the NAs, mainly NDMA, were found in food cooked by the direct flame heating method. The NAs, especially NDMA, were present in barley used for making beer and coffee roasted in direct flame heated air as well as in meat that was roasted, broiled, grilled, baked or deep fried in open furnaces [
33,
34,
35]. The following mechanisms for the formation of NDMA were proposed. Nitrogen oxides (NO
x), which are formed during the combustion of fuels, can react with secondary amines to form nitrosamines, mainly NDMA [
34]. The secondary amines were reported to be formed by the thermal decomposition of amino acids such as lysine in proteins [
36,
37], which suggested that the protein content of the meat is an important factor in the formation of NDMA. Beef sirloin generally has the highest protein content among the types of meat examined (9.3%, 17%, and 21% for pork belly, duck, and beef sirloin, respectively) [
38]. Duck contains higher protein content than pork belly, but released less NDMA than other meats, probably due to the shorter cooking time for duck (5 min for duck vs. 25 min for pork belly).
NDMA concentrations within each set of experiments varied greatly (e.g., 179–906 ng/m3 for charcoal-grilled beef sirloin). This might be caused by the following factors: (1) the meat slices were not homogenous in composition and thus the protein content varied within each experimental set; and (2) the meat was not consistently turned over by hand during cooking, and thus the degree of cooking might be different.
Thermosorb-N cartridges were employed for the sampling of airborne NDMA in this study. These samplers do not collect gaseous and particulate phases separately, but both phases together. NDMA has a relatively low boiling point (153 °C) and high vapor pressure (700 Pa at 20 °C). In addition, sampling was conducted near the hot cooking appliance during cooking. Therefore, most of the NDMA measured was expected to be present in the gaseous phase in the air [
39].
NDMA was also found outdoors at a few ng/m
3 levels in PM
2.5 (particulate matter less than 2.5 μm in average aerodynamic diameter) samples collected from urban areas in the United Kingdom and Korea [
21,
22], and the concentrations were found to be higher in winter than in summer. Other NAs such as NDEA, NDBA, NPIP, NPYR, and NMOR were also found in these particulate samples, implying that the NAs are ubiquitous environmental pollutants present in air as well as food and water. Further studies on the distribution of NAs between gaseous and particulate phases need to be conducted in order to investigate their transport and fate in the air.
Cancer risks from the inhalation exposure to NDMA in charcoal grill restaurants were greater than the acceptable risk level for all types of meat, suggesting that meat charcoal grilling indoors can pose a significant cancer risk. Pan-broiling is also a contributor to NDMA formation, although it leads to much less formation than charcoal grilling.
It should be noted that the cancer risks associated with exposure to NDMA in charcoal grilling restaurants would vary greatly depending on its concentration near the human breathing zone. The flow rate of the direct intake of air into the ventilation pipe would be the most important factor since it would mainly determine a person’s level of exposure to NDMA emitted from the grill. The actual intake flow rate of air was not measured in this study, however. Other factors in restaurants such as natural ventilation, the size of the restaurant, the number of grills, etc. would affect the levels of exposure to NDMA. Therefore, the cancer risk values estimated in this study may not accurately reflect those for such restaurants. Nonetheless, the results of this study may be helpful to recognize the risk of the formation of the carcinogen from charcoal grilling of meat.
Proper ventilation is therefore necessary to reduce the exposure to indoor air pollutants, including NDMA. However, the ventilation systems installed in most Korean charcoal grill restaurants consist of a long duct without a hood covering the entire charcoal grill. Hence, the gaseous and particulate pollutants emitted from the surroundings of the grill cannot be sufficiently drawn away by the system, leading to the elevation of their indoor concentrations.
What should be noted is that there are usually more than 10 tables and grills in such restaurants, and customers may actually be exposed to NDMA emitted from other grills as well as their owns. Consequently, much higher NDMA exposure is expected than that measured in this study, which may cause serious health problems, including carcinogenicity. Field measurements in such restaurants need to be conducted for more accurate and realistic risk assessment.