1. Introduction
Exposure to bioaerosols has become one of the major problems in indoor air quality (IAQ) for occupational and public health [
1]. The importance of assessment and control of contamination in critical environments, such as the physical environment of health care organizations [
2,
3,
4] is a requirement to protect patients and healthcare workers from hospital-acquired (nosocomial) infections and occupational diseases [
5].
Hospital-acquired infections contracted by patients after admission to health care facilities are a concern not only in terms of patient health, as they have a high impact on patient morbidity and mortality [
6,
7], lengthened hospital stays, as well as increased health care costs resulting from failed treatments [
8]. The scale of severity of the effects on human’s health, depends on their toxicity, exposure time, microbial load and also the immune system of the exposed individuals [
9,
10].
In the hospital context, the greatest concerns for airborne transmission are related to respiratory infections, including tuberculosis [
11], measles [
12], varicella [
13], influenza [
14], respiratory syncytial virus (RSV) [
15],
Bordetella pertussis [
16], as well as non-respiratory infections, such as norovirus [
17], methicillin-resistant
Staphylococcus aureus (MRSA) [
18] and
Clostridium difficile [
19,
20].
Presently, one of the most serious bacterial pathogens associated with indoor environments is
Legionella pneumophila that causes Legionnaire disease and Pontiac fever, which have become a leading public health concern [
21]. The health care facilities have some medical equipment (i.e., respiratory devices, such as CPAP machines, hydrotherapy equipment and bronchoscopes) [
22] that can originate the development of
Legionella and, potentially, emit contaminated aerosols. In view of the number of patients who visit health care facilities, as well as their immune status, the likelihood of exposure and infection is increased in these institutions [
23]. The rising incidences of
Legionella-associated diseases [
23], and the variety of sources of transmission, emphasize the need to understand the prevalence, survival and transmission of
Legionella in diverse public settings, namely in health care facilities.
Moreover, antimicrobial resistance in human pathogens, such as
Staphylococcus aureus, mostly associated with the inappropriate use of antibiotics, has led to emergence of antibiotic resistance strains (superbugs) at concerning rates worldwide [
24]. During its evolution,
S. aureus has acquired, via horizontal genetic transfer, the
Staphylococcal Cassette Chromosome mec complex (
SCCmec), in which is inserted the mecA gene which encodes the modified penicillin-binding protein 2 (PBP2), responsible for the resistance to
β-lactam antibiotics (oxacillin, streptomycin, tetracycline, erythromycin, among others.), by reduction of the affinity of the antibiotics binding to their receptor [
24,
25]. MRSA is currently acknowledged as one of the most relevant human pathogens, particularly in health associated clinical settings, due to the fact that it is a major cause of infections worldwide [
26] associated with extremely high mortality rates for invasive blood-stream and pneumonic infections [
27]. Relevantly, the ability demonstrated by this particular pathogen to develop new clones with the capacity to invade community settings and infect people without predisposing risk factors and also to buildup MRSA reservoirs are certainly major concerns.
Moreover, for the past few years,
Pseudomonas aeruginosa, an opportunistic pathogen, has also become a significant public health threat and one of the leading causes of nosocomial infections, particularly associated with immunocompromised patients [
28]. Due to the worldwide dissemination of high-risk clones which are characterized as multidrug-resistant or extensively drug-resistant (MDR/XDR) strains [
29] associated with severe hospital-acquired infections with very limited therapeutic options [
30] more attention has been given to this human pathogen. Considering the fact that antimicrobial resistance is currently considered one of the most relevant health care challenges of this century [
31], the identification of these particular strains, namely MRSA and
Pseudomonas aeruginosa, in health care facilities is imperative not only to secure exposed individuals health but also to avoid the spread of resistant strains to the community.
The presence of dynamic and highly diversified microbial communities in the air of urban and rural environments assumes a major impact on Public Health due to their dispersion and health consequences [
32,
33,
34,
35].
The composition of bacteriota indoors is affected by ventilation, building design, environmental conditions (like temperature, humidity, presence of organic matter), ability to form biofilms, building occupancy, geographical location, outdoor environment [
1,
36,
37,
38,
39] and also by the activities of human occupants [
40,
41]. In clinical environments, the building design, ventilation systems, medical activity, occupancy rate, clinical staff and types of patients, frequency of cleaning and hospital cleaning procedures, as well as outdoor air penetrating indoors [
42,
43,
44,
45,
46], are decisive factors for the levels of aerial microbial concentration.
The airborne bioburden was previously identified in hospital environments [
1,
46,
47,
48,
49,
50], but until now, airborne bioburden assessment in Portuguese PHCCs has only been done in a few limited studies, although in this environment, patients may also present a vulnerable health. In a rural setting, located in the countryside, the population uses primary health centers due to the lack of other medical options and equipment available. On the other hand, in a city setting the population has a lot more options available to them. Likewise, the influence of urban versus rural locations on bioburden assessment has not been studied in these Primary Health Centers.
The World Health Organization publish air quality guidelines for specific pollutants, based on their health impacts [
51]. In Europe, improvements in ambient air quality are driven by Community directives (2008/50/EC and 2004/107/C). In Portugal there is only legislation regarding IAQ that establishes limit values for microbiological contamination, being applicable in all indoor environments, without any specification for health care facilities, or occupational context [
52]. Portuguese legal compliance differs in relation to the microorganism, for total bacteria, the indoor load should not exceed the outdoor load by 350 CFU.m
−3, there is a second criterion when this condition is not satisfied [
53]. Additionally, Portuguese legislation only considers air sampling to assess microbial contamination indoors that narrow the information regarding the exposure assessment [
3,
54]. As such, passive sampling methods such as surface swabs and electrostatics dust collectors should be included in the sampling approach to enrich the assessment and provide a more accurate risk characterization [
3,
54].
This study aims to assess the airborne bacteriota of rural and urban Portuguese Primary Health Care Centers, with a multi-approach protocol using active (air sampling by impaction) and passive (surface swabs, electrostatic dust cloth, settled dust and air-conditioning filters) sampling methods, while identifying the potential differences in airborne microbiota between both environments. The analysis of airborne microbiota in both PHCC consisted in the enumeration of mesophilic bacterial load, its phenotypic characterization and antibiotic resistance profile, and detection of Pseudomonas aeruginosa, Legionella sp. and Staphylococcus aureus.
2. Materials and Methods
This study was performed between May and September of 2018 in two selected Portuguese Primary Health Care Centers. One of the PHCC was in an urban setting and the other in a rural setting in Lisbon district. It is part of a larger exploratory study with financial support aiming to establish protocols to assess occupational exposure to bioburden in clinical environments.
2.1. PHCC Characterization and Measurement of Environmental Parameters
A walkthrough survey and checklist were applied to collect information regarding the characterization of the building, the procedures of the Health Center and sampling site characterization.
Both buildings were built as health units in 2016 and haven’t had modifications done in the last 12 months. However, during the walkthrough survey it was observed visible mold growth and cracks on some walls from the urban PHCC and cracks on the walls of the vaccination room in the rural PHCC. The urban PHCC has two floors above ground and the rural PHCC only has one.
The car parking, attached garage, direct access from basement car park, cooling towers and traffic in the urban PHCC and the outdoor car parking and waste management site (tip or garbage dumpsters) in the rural PHCC were identified as potential sources of air pollution that could influence the internal environment (up to 1 km).
The natural ventilation from the urban PHCC was only provided through the open door, while in the rural PHCC has natural ventilation from sliding windows, windows with tilting flag and full openness windows (usually open 1 h during the morning in the vaccination and treatments rooms, and after the cleaning procedures in all places). Both PHCC are provided with artificial ventilation provided by a Central HVAC system with air handing units and ductwork system, working from 8 am to 8 pm in the urban PHCC, whilst working all day in the rural PHCC. In Rural PHCC, these systems are located on the roof, while in Urban PHCC they are located next to the entrance, at 1.8 m from the ground level.
The cleaning procedures and waste collection are performed by an external company. In the urban PHCC the floors and surface are cleaned during patient service and working hours, while in the rural PHCC the cleaning is performed daily after patient service and after working hours. Considering the type of activity realized in the treatment room, it is cleaned several times a day, four times in the urban PHCC and two in the rural PHCC. A mop is used to clean the floor while cloths are used for surfaces.
The same areas were evaluated once in both PHCC and the measurements were carried out during the normal working hours (
Table 1).
Measurements of temperature, relative humidity and carbon dioxide (CO2) were also performed in each sampling location in both PHCC. Temperature and relative humidity were measured by the Handheld Particle Counter from Lighthouse Worldwide Solutions —Model 3016/5016, and the CO2 was measured using the Graywolf equipment (Graywolf Sensing Solutions: Shelton, CT, USA). These measurements were performed in the same areas where bacteriota was evaluated and an average value for each place was obtained through a 5 min measurement.
2.2. Sampling and Characterization of Viable Bacteria
One indoor air sample in each evaluated area, and one outdoor sample (as reference), of 250 L were collected by impaction in both PHCC with a flow rate of 140 L/min (Millipore air Tester, Millipore, Billerica, MA, USA) onto each culture media plate according to the manufacturer’s instructions. Different culture media were used to ensure the selectivity for bacterial growth: Tryptic Soy Agar (TSA) supplemented with nystatin (0.2%) was used to assess the total aerobic mesophilic bacteria and Violet Red Bile Agar (VRBA) to total coliforms. For the air sampling was followed the guidelines of the national legislation [
53].
Surface samples were collected using a swab method with a 10 cm × 10 cm square stencil, disinfected with a 70% alcohol solution between samplings [
55]. Surface swab samples were extracted with 1 mL of 0.1% Tween™ 80 saline solution (NaCl 0.9%) for 30 min at 250 rpm on an orbital laboratory shaker (Edmund Bühler SM-30, Hechingen, Germany) and seeded on the selected culture media for bacterial growth (TSA and VRBA).
One piece of HVAC filter with approximately 2 cm
2 (1.4 cm × 1.4 cm) was collected in each PHCC, refrigerated at 4 °C during transport before analysis, and washed with 10 mL of 0.1% Tween™ 80 saline solution (NaCl 0.9%) for 30 min at 250 rpm on an orbital laboratory shaker (Edmund Bühler SM-30, Hechingen, Germany) [
56] and plated onto the selected media (TSA and VRBA) for bacterial growth.
All the inoculated plates from the different environmental parameters were incubated at 30 °C (TSA) and at 35 °C (VRBA) for 7 days, for bacterial growth. After the incubation period, quantitative (colony-forming units—CFU.m
−3 and CFU.m
−2) results for bacteria were obtained. Bacterial phenotyping was based primarily on morphology, Gram-staining, endospore formation, catalase activity (catalase test) and cytochrome oxidase presence (oxidase test). Bacteria was grouped into morphologic groups as Gram-positive cocci, Gram-negative cocci, Gram-positive rods and Gram-negative rods according to its phenotype based on Bergey manual of determinative bacteriology [
57]. The most frequent bacterial isolates were identified using a miniaturized biochemical test (RapID, Remel; Santa Fe, Lenexa, KS, USA).
2.3. Electrostatic Dust Collector
One EDC with a surface exposure area of 0.00942 m
2 was added in each sampled area at a minimum 0.93 m above floor level, and dust was left to settle for 15 days before analysis. EDC samples were submitted to extraction and bioburden characterized by culture-based methods described previously [
58,
59]. The EDC were weighed, before and after being exposed, and the mass of the collected dust was determined by subtracting the values. Each EDC cloth was then washed with 20 mL 0.9% NaCl with 0.05% Tween80™ (Merck S.A, Lisbon, Portugal) by orbital shaking (250 rpm, 60 min, at room temperature), and 150 μL of the wash suspension was inoculated onto two different culture media: TSA with 0.2% nystatin, and VRBA. After incubation of TSA and VRBA plates at 30 and 35 °C for 7 days, respectively, quantitative colony-forming units were obtained.
2.4. Settled Dust Sampling
Sampling of the settled dust was accomplished by a vacuum cleaner (HOOVER Brave BV71_BV10 A2, Brugherio (MB), Itália, USA) equipped with a 40-micron nylon mesh inserted in a DUSTREAM™ collector (Indoor Biotechnologies, Charlottesville, VA, USA). Vacuuming was performed for a period of 10 min in different selected areas [
60] and the dust collected from several surfaces and according to previously described procedure [
60]. Sampling duration was the same in all PHCCs in order to standardize sampling procedures and also to ensure a sufficient amount of dust for analysis [
60]. A DUSTREAM™ filter tube was collected in each PHCC. The samples were measured and analyzed for bacterial load.
The samples were weighted and washed in a ratio of 1 g (dust) per 9.1 mL of NaCl 0.9% with 0.05% Tween 80 (10 μL) for 60 min at 250 rpm, and 0.15 mL of this suspension were inoculated onto two different media: TSA with 0.2% nystatin and VRBA. After incubation of TSA and VRBA at 30 °C and 35 °C for 7 days for mesophilic bacteria and coliforms, respectively, bioburden densities (CFU·g−1 of dust) were determined.
2.5. Antibiotic Susceptibility Testing
Antimicrobial susceptibility testing was performed on isolates based on the disk diffusion method on Mueller-Hinton agar [
61,
62]. The grades of susceptibility pattern were recognized as sensitive and resistant by comparison of the zone of inhibition as indicated in European Committee on Antimicrobial Susceptibility Testing standard 2020 [
63]. The antibiotics tested for both gram-negative and gram-positive bacteria (Oxoid, Basingtone, UK) were aztreonam (30 μg), cefotaxime (30 μg), gentamicin (30 μg), imipenem (10 μg) and ticarcillin (75 μg). Antibiotics were selected based on literature, effectiveness and EUCAST Guidelines. In Rural PHCC, 130 isolates out of a total of 540 isolates were tested for antibiotic resistance and in Urban PHCC, 84 isolates out of a total of 440 isolates were tested.
2.6. Detection of the Presence of Pseudomonas aeruginosa, Staphylococcus aureus and Legionella sp.
The presence of P. aeruginosa was investigated in different environmental parameters such as surfaces, EDC, settled dust an HVAC filter, while S. aureus assessment was performed exclusively on surfaces. For P. aeruginosa and S. aureus detection, samples were inoculated in Tryptic Soy Broth for 24 h at 37 °C and seeded in non-selective Columbia blood agar and selective culture media plates Cetrimide Selective Agar and Chromogen MRSA, respectively for 24 h at 37 °C. After incubation presumptive colonies were isolated and MRSA isolates confirmed by immunologic assays.
Legionella samples were collected in December 2018 according to the guidelines from Portugal Health General Direction and the culture method described in ISO 11731:2017 [
64] was applied for the isolation and quantification of
Legionella in water samples. Water samples (1 L/PHCC) were collected from worker’s toilets showers and condensation water from the HVAC system tray (10 mL for PHCC), filtered and analyzed by the culture method for the detection of
Legionella on the same day of sampling. The water sample (1 L) and condensation water (10 mL) were filtered through a 0.2 μm polycarbonate membrane filter followed by a washing procedure after added 10 mL of PBS and was divided into 3 portions. One portion was used for treatment with heat (5 mL of the concentrated sample in a sterile container and placed it in a water bath at 50 °C for 30 min), another portion used for treatment with acid solution (added 1 mL 0.2 M HCl-KCl (pH 2.2) at 1 mL of the concentrated sample in a sterile container) and the last portion untreated. Three aliquots of 0.5 mL water from each portion were inoculated to GVPC agar (Oxoid) and BCYE agar (Oxoid). The cultures were incubated at 37 ± 1 °C for 7 to 10 days in a humidified atmosphere and examined after two, three, four or five days, followed by a final inspection at the end of the incubation period. Identification was conducted by performing subcultures onto a plate of BCYE agar and a plate of BCYE–cys agar and incubate at 37 ± 1 °C for 2 to 5 days. If bacterial isolates grew on BCYE agar but fail to grow on the plate of BCYE–cys agar, biochemical confirmation/identification tests were performed.
2.7. Statistical Analysis
The data was analyzed in statistical software, SPSS 26.0 for Windows (Redmond, WA, USA). The results were considered significant at the 5% significance level. The Shapiro-Wilk test was used to test the normality of the data. Frequency analysis (n; %), and the calculation of the minimum, maximum and mean, were performed for the qualitative and quantitative data, respectively. Since the assumption of normality was not verified, the Mann-Whitney test was used to compare the rural and urban PHCC environment, regarding the total aerobic mesophilic bacteria and total coliforms. The Spearman correlation coefficient was used to study the relationship between total aerobic mesophilic bacteria, total coliforms and environmental parameters measured (temperature, relative humidity and CO2). To compare the settings, graphical representations were used, since the data did not allow any other type of analysis to be performed.
4. Discussion
The most frequently available data on airborne microbial concentration refers to studies carried out in a hospital context, namely in operating rooms, lobbies [
48], wards [
49,
67], operating theaters [
42,
68], surrounding areas, patient room and nursing care room [
67,
69,
70]. However, there is a gap in evidence about microbiological assessment in primary health service facilities, albeit the potential impact on human health being comparable to that of hospital environment [
71]. Both the characterization of microbial contamination on this type of health units, and the analysis of data in different geographical locations, were not yet subject of extensive investigation. Therefore, this study aims to highlight the bacterial profile in two types of health care centers, urban and rural. Health units serve the purpose of public health preservation, but they are also potential environments with negative effects and risks to human health and to the well-being of their users, workers and patients [
3]. This effect is of particular concern for immunocompromised patients and other vulnerable populations, since those with a debilitated immune function are more susceptible to contract nosocomial infections [
5,
72].
The IAQ in healthcare facilities requires a higher degree of standards compared to other building types. Although the concern with indoor air was assumed in Portugal, with the creation of specific legislation for IAQ [
52,
53], it does not take in consideration the type of environment and the correlation between the clinical relevance/pathogenicity of the biological agent (bacteria) with the concentration observed in indoor air. Currently, the Portuguese legislation only defines a quantitative reference value for bacterial contamination in outdoor air quality, correlating it to all types of building and without reference to the kind of indoor environment. Furthermore, it appears that there are no adequate methodologies and reference standards for the exposure assessment of health professionals to biological risks [
73].
The importance of applying different sampling methods allowing a more complete characterization of the exposure has already been mentioned in other studies [
74,
75,
76], emphasizing the temporal variation as a factor to obtain a more accurate risk assessment. Thus, although there is a legal reference in Portugal on the assessment of IAQ through the use of air sampling as stand-alone method, passive methods (surface swabs, electrostatic dust cloth, vacuum settled dust and filters for HVAC systems) were also applied in this study, in order to assure a higher accuracy in the assessment of IAQ. Although passive sampling by vacuuming may be a limitation to assess the special and temporal variations, dust samples are easy to collect and may represent a long-term integrated sample of exposure to bacteria [
77,
78].
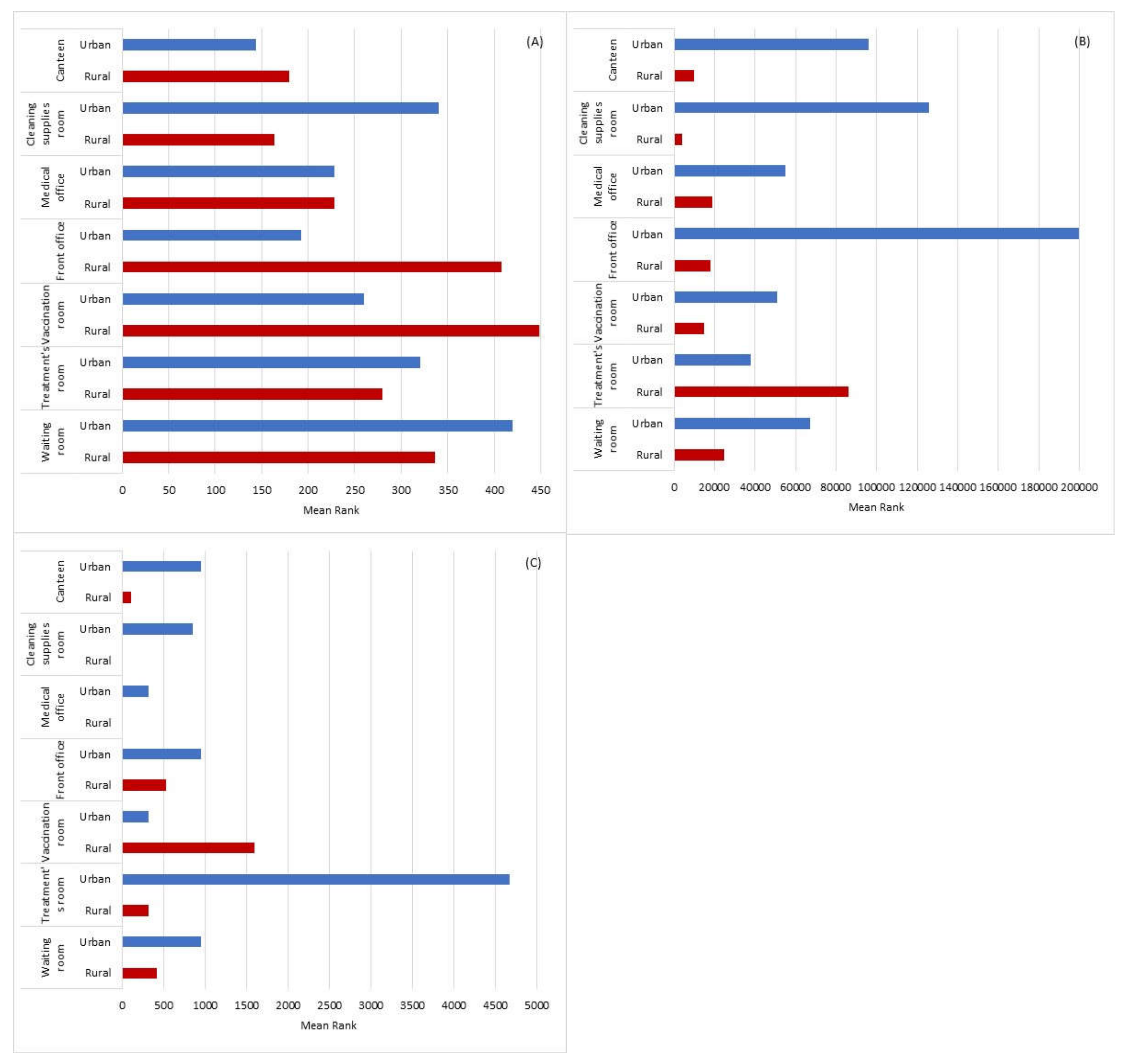
Our results show the same quantitative trend on results between active and passive methods. In fact, the vaccination room was the sampled area with the greatest bacterial contamination in Rural PHCC in both sampling methods (active-air and passive-EDC).
Although the treatment room presented the highest concentration in both passive methods, such as in the surfaces of Rural PHCC and in EDC of Urban PHCC, that was not the tendency in the active method. These results highlight the complementation of both types of sampling methods in IAQ assessment.
The indoor-to-outdoor bioaerosol concentration ratio (I/O) is used to estimate emissions sources of bioaerosol [
79]. For situations in which I/O > 1.0, indoor air is considered the main source of indoor bioaerosols, and when I/O < 1.0, outdoor air is considered the main source of indoor bioaerosols. The I/O ratio in both Rural and Urban PHCC was superior to 1.0, only in the canteen from the Urban PHCC (0.8) was under, which suggests that air bacterial load may be derived from the indoor environment, following the trend of previous studies [
1,
3]. Indeed, the airborne microbial load from the Urban PHCC was under the limits recommended by IAQ Portuguese legislation, while in the Rural PHCC two sampled areas did not comply with the first and the second Portuguese legislation criteria. Moreover, the level of air bacteriota observed in both PHCC was like those described in the literature concerning similar sampled area from hospitals [
1,
9,
48,
80,
81], highlighting the importance of assess bacteriota also in this type of health care facilities.
The identification of bacterial isolates was performed based on conventional methods through culture techniques and biochemical tests, following the same culturability assessment approach applied for the methodology of this study. Although having limitations (e.g., time consuming and lack to identify non-culturable microorganisms), culture and microscopy are still two of the most utilized techniques [
82]. Molecular approaches (e.g., NGS, qPCR) are particularly important for non-culturable microorganisms, but need demanding equipment and resources. Considering the applied methodology, the bacterial phenotypic identification indicated that Gram-positive cocci were the most dominant group of genera (53% Rural PHCC and 63% Urban PHCC) identified in the air, followed by gram-negative cocci (37% Rural PHCC ad 20%/ Urban PHCC), gram-negative rods (4% Rural PHCC and 3% Urban PHCC) and gram-positive rods (1% Urban PHCC). Our results are consistent with previous studies [
1,
9,
49,
67,
68,
83], both in the rate of isolated bacterial species, and indicating Gram-positive cocci as the predominant microbial phenotype in hospital indoor air. The microorganisms found in hospital environments are diverse and can be defined based on their origin, some of these microorganisms can be traced to a human origin (skin and mucous membranes), including
Staphylococcus aureus,
Enterobacteriaceae or
Enterococcus, whilst others are from environmental origin. From the latter, some have natural resistance to antibiotics, namely Gram-negative rods such as
Pseudomonas aeruginosa,
Acinetobacteriaceae,
Burkholderia cepacia,
Legionella pneumophilia,
Stenotrophomonas maltophilia or atypical mycobacteria [
80].
In fact, the airborne Gram-positive cocci (
Staphylococcus and Micrococcus) were also the most detected bacterial genera in a sensitive ward of an academic pediatric hospital in Iran [
84], in hospitals in India [
85], in a hospital ward in India [
67], in five educational Hospital wards air in Iran [
86], in General Hospitals of Korea [
87], in a regional Danish hospital [
88] and in tertiary university hospital in NE Greece [
89]. In our study, higher gram-positive cocci isolates were found in the treatment rooms and vaccination rooms of both PHCC, smaller spaces where they perform nursing treatments, shared between workers and patients, exposing them to both. The higher concentrations of Gram-positive cocci may be due to bioaerosol dispersal during skin shedding, their higher adaptability to environmental conditions, inadequate ventilation and occupancy rate [
90,
91].
In this study, even though the Urban PHCC has higher concentrations of bacteria in most types of samples, it was the Rural PHCC that presented the highest concentration of bacterial pathogens in the air. Different locations have their own unique bacterial communities, local sources of bacteria and the influence of other environmental factors that affect bacterial composition, diversity, abundance, and predominant genera [
34]. The obtained results reflects that the compliance of quantitative results could not evidence the pathogenic potential of airborne bacterial population. Taking this in consideration, the legislation regarding Portuguese indoor air quality should be revised, not only to be focused on quantitative results, such as concentrations of bacteria, but also on the presence of specific bacterial species (e.g.,
Staphylococcus sp.,
Neisseria sp.,
Pseudomonas aeruginosa,
Acinetobacter spp.
Enterobacteriacea family members).
The most common indoor species documented are:
Staphylococcus auricularius,
S. capitis,
S. epidermidis,
S. haemolyticus,
S. hominis,
S. saprophyticus and
S. warneri [
92,
93,
94], all commonly found on human skin flora [
95] and some with antibiotic resistant phenotypes and an opportunistic behavior as bacterial pathogens [
96]. In our study,
Staphylococcus (including
S. capitis subsp.
capitis,
S. cohnii ssp
cohnii,
S. haemolyticus,
S. hominis ssp
hominis,
S. epidermidis,
S. simulans,
S. warner,
S. uralyticus) isolates showed resistance for Ticarcillin antibiotic in both PHCC. The ticarcillin antibiotic is used to treat infections of the skin and skin structure, infections of the lower respiratory tract, infections of the urinary tract and intra-abdominal. Ticarcillin is used combined with clavulanic acid, a beta-lactamase inhibitor, to eradicate some penicillin-resistant organisms [
97]. Despite the fact that no MRSA were isolated in the analyzed samples, it is importance to notice that coagulase-negative staphylococci (CoNS) are opportunistic pathogens with the ability to produce several toxins, enzymes and enclose resistance genes, which can be further transferred to more virulent strains such as
S. aureus [
98]. It is relevant to acknowledge that, MRSA has acquired, via horizontal genetic transfer, the SCCmec, which is responsible for the resistance to
β-lactam antibiotics [
24,
25]. In fact, molecular typing techniques have demonstrated that CoNS can enclose high levels of multidrug resistance that may be transferable to
S. aureus [
99] which is particularly concerning considering
S. aureus ability to develop new clones with capacity to invade community settings [
24].
Long-term care institutions were highlighted as important risk factors to emerging pathogens [
100] in our study
Pseudomonas aeruginosa it was not detected, probably due to the character more ambulatory use of this type of health facilities.
The absence of
Legionella in the both PHCC, perhaps is due to the fact that in Portugal to minimize the spread of
Legionella Pneumophila and the associated risk of Legionnaires’ Disease, it was established from 2017 the compliance with a strict program for the prevention and environmental control of the
Legionella bacterium, in which measures for monitoring, surveillance, risk assessment and prevention and control (physical, chemical and microbiological) must be adopted to promote the adequate maintenance of the water and air systems in the health units [
101].
It is recognized that contamination of surfaces with bacteria can increase air contamination by 15% [
102]. Considering the obtained results, statistically significant differences were detected in relation to bacterial counts on the surface samples. Urban PHCC presented higher aerobic mesophilic bacteria counts than the Rural PHCC, which may be related to the cleaning procedures used for the correct disinfection of surfaces in Rural PHCC. Compared with previous studies, both PHCC results were higher than those assessed in other environments (86 CFU/25 cm
2 in offices with synthetic floor, 120 CFU/25 cm
2 in classrooms and 250 CFU/25 cm
2 in training rooms) [
103].
The higher values of contamination assessed by EDC were found in the vaccination room in Rural PHCC and in the treatment rooms of Urban PHCC. These two locations were also identified with higher indoor air contamination in both PHCC, showing a relationship in the sampling air at that time and in the long-term sampling since a single EDC analysis is equivalent to the sum of several air-impaction measurements [
104].
Concerning settled dust samples, our results demonstrate a higher value for bacterial contamination in Urban PHCC, when compared with the Rural PHCC. However, these values were lower when comparing with other environments, namely in homes with a value averaging of 16 × 10
5 CFU.g
−1 [
105] and 2.9 × 10
5 to 4.9 × 10
7 CFU.g
−1 [
106], and in agricultural environments, with bacterial load ranging from 19 CFU.g
−1 to 109 CFU.g
−1 [
107].
The positive correlation observed between temperature and aerobic mesophilic bacteria concentration in Urban PHCC, and the fact that higher temperatures may facilitate increased growth of bacteria in the environment, may be related to the higher aerobic mesophilic bacteria load in Urban PHCC [
108]. Furthermore, the positive correlation detected between temperature, relative humidity and bacterial densities in air and surfaces are similar to the results observed in previous studies [
3,
109]. The use of the HVAC systems may increase the dispersion of all bacterial contamination that can be transmitted in the air. However, if efficient filters are used, 99.9% of all bacteria present in the air can be removed [
108]. In our study, total mesophilic aerobic bacteria detected in the HVAC filters were higher in the urban PHCC compared to the rural PHCC, which may be related to the location of the outdoor air intakes. In the latter case, adjacent buildings and proximity to a roadway could be an increment of contamination. Therefore, it is crucial that the HVAC system in healthcare facilities maintains ideal temperature and humidity conditions for all its occupants, improving indoor air quality and decreasing the probability of infections [
108].