3.1. Concentration Level Analysis of PM2.5’s Water-Soluble Ions
During the monitoring period, the total mass concentration value of the eight water-soluble ions of PM2.5 was 40.96 µg/m3, which accounted for 62% of the entire mass concentration. The sequence of the concentrations of water-soluble ions in order from high to low was SO42− > NO3− > NH4+ > Cl− > K+ > Ca2+ > Na+ > Mg2+, and the three secondary ions SO42−, NH4 + and NO3− were the main water-soluble ions, which were separately converted from gas precursors SO2, NOx and NH3 and accounted for 92% of the total measured water-soluble ions.
The concentration level of SO
42− was the highest of the eight water-soluble ions and was lower than the values for the northern cities Beijing and Tianjin and greater than the values for the southern cities Shanghai, Guangzhou and Hong Kong (
Table 1, [
15,
23,
24,
25,
26,
27]), mainly due to the emissions of industrial pollution sources and coal sources in Wuhan. The concentration levels of NO
3− and NH
4+ ions were basically identical to the concentration of SO
42−. The high concentration of NO
3− was based on the number of motor vehicles rising constantly in Wuhan in recent years. For example, take the NOx emissions (
Table 2), we can find that the industrial NOx emission (stationary source) was the main source of NOx. Among them, NOx emission from thermal power industry was the primary source of pollution and accounts for 35.06% in the total NOx emission, followed by vehicle exhaust emissions accounts for 34%, suggesting that NOx emissions have a tendency to increase gradually. In addition, as seen from the seasonal distribution of NO
3−, the concentration level in the winter and autumn was significantly higher than that in the spring and summer because the high temperatures in the spring and summer accelerated the volatilization loss of nitrate.
The annual average concentration of NH4+ in the study was second only to that of Beijing and was relatively high in the winter and low in the summer. NH4+, converted from NH3, is an important ion that reacts with SO42− and NO3− in the aerosol phase to form secondary particles. NH3 mainly comes from agricultural production, industrial emissions, vehicle exhaust emissions and other sources. Attributed to the sharp rise of motor vehicles in Wuhan, a large number of nitrogen compounds are emitted into atmosphere by vehicle exhaust and produce ammonium nitrate through a chemical reaction with NH3. Meanwhile, urban population growth (increasing the consumption of energy) and industrial economic expansion (such as thermal power industry, iron and steel industry and cement industry) are also important factors leading to an increase in ammonia emissions.
3.2. Seasonal Variation Characteristics of Water-Soluble Ions
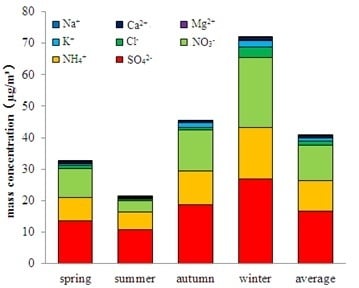
The mass concentration variation of water-soluble ions in PM
2.5 presented distinctly seasonal distribution features. The sequence of the mass concentration levels in the four seasons was winter > spring > autumn > summer. The seasonal distribution of the cumulative concentration of eight water-soluble ions is shown in
Figure 3. The concentration sum of the three main secondary ions (SO
42−, NO
3−, NH
4+) in the four seasons accounted for 79%, 46%, 67% and 85% of the total soluble-water ions, respectively, and was highest in the winter. The average mass concentration of the eight ions was 40.96 μg/m
3, which composed 63% of the total mass concentration of the water-soluble ions.
As shown in
Figure 4, the proportion of concentration contribution of the three main ions was SO
42− (31.64%) > NO
3− (26.27%) > NH
4+ (19.27%) in winter, and the same order in spring and autumn, but was SO
42− (23.11%) > NH
4+ (12.15%) > NO
3− (7.38%) in summer, implying concentration value of NH
4+ was ascending comparing with the value of NO
3−. High temperature in summer is advantageous for the decomposition of solid material NH
4NO
3 and forming into gaseous materials NH
3 and HNO
3. After two-step chemical reactions (step one: NH
3 + H
2O = NH
3·H
2O; step two: NH
3·H
2O = NH
4+ + OH
−) in the atmosphere, NH
3 transforms into NH
4+ compounds, causing the concentration level of NH
4+ to rise.
Similar to the seasonal variation tendency of all water-soluble ions, the concentration of SO42− was greatest in the winter, followed by the autumn, and was the lowest in the summer. The concentration value in the winter was 2.5 times that of the summer. One reason for the above situation is that citizens generally burn coal to keep warm in the winter. In addition, little rain and a dry climate in the winter cause SO42− to remain in the atmosphere for a long time, so its concentration is elevated. On the contrary, high temperatures and rainy weather in the summer are not conducive to the formation of SO42−.
The concentration levels of Ca
2+ and Mg
2+ experienced similar seasonal varying trends, such that the values decreased as follows: winter > autumn > spring > summer. The concentrations of Ca
2+ and Mg
2+ in the winter were 1.9 times and 4.3 times those of the summer, respectively. As typical ions of flowing dust [
29], the concentrations of Ca
2+ and Mg
2+ are immensely influenced by seasons and anthropic actions. On one hand, the winter climate with dry weather and little rain reduces wet subsidence of Ca
2+ and Mg
2+; on the other hand, with accelerating urbanization processes in recent years in Wuhan, a large number of surfaces from construction operation are emerging every year, thus increasing dust sources and resulting in the rise in the concentrations of Ca
2+ and Mg
2+ ions. Conversely, high temperatures and rainy weather in the summer provide beneficial conditions for the settlement of Ca
2+ and Mg
2+ compounds, which causes the concentrations of Ca
2+ and Mg
2+ ions to drop.
3.3. Concentration Equivalent Ratio Analysis of NO3−/SO42−
Concentration equivalent normality is defined as the number of equivalents per liter of solution, where the definition of an equivalent depends on the reaction taking place in the solution. For an acid-base reaction, the equivalent is the mass of the acid or base that can furnish or accept exactly 1 mole of protons (H+ ions). The mass concentration equivalent ratio of NO
3− and SO
42− could be used as relative significant index to measure the relative contribution of mobile source (vehicles) and fixed sources (coal) for nitrogen pollution and sulfur pollution in the atmosphere [
24]. Arimoto
et al. (1996) attributed the high ratio of NO
3−/SO
42− to mobile sources, which had a greater contribution to the concentrations of regional atmospheric pollutants [
30]. The sulfur contents in gasoline and diesel in China were 0.12% and 0.2%, respectively. The NO
x/SO
x ratios from comburent of gasoline and diesel fuel were approximately 13:1 and 8:1, respectively. Coal’s sulfur content is 1%; the ratio of NO
X/SO
X from coal’s combustion is approximately 1:2. Therefore, NO
X and SO
X can act as tracers of mobile sources and fixed sources separately. When the concentration equivalent ratio of NO
3−/SO
42− exceeds 1, it means that pollution sources of the observation point are dominated by mobile sources, while fixed sources play major roles when the ratio is below 1 [
30]. The equivalent ratios of NO
3−/SO
42− in Wuhan were 0.73, 0.32, 0.70 and 0.83 in the spring, summer, autumn and winter, respectively. The annual average equivalent ratio of NO
3−/SO
42− in Wuhan was 0.64, which is higher than the value of 0.73 in Changbai Mountain and the value of 0.46 in Nanjing, lower than the value of 0.83 in Shanghai, and essentially consistent with the value of 0.64 in Beijing [
31]. The results revealed that the main pollution source in Wuhan was a fixed pollution source, which was consistent with the research of Zhang
et al. [
22].
3.4. Charge Balance Analysis of Water-Soluble Ions
Previous studies showed that the charge balance of water-soluble ions in PM
2.5 could be used to analyze the importance of the contribution of water-soluble ions to the mass concentration of PM
2.5 [
14,
32,
33]. According to the analysis of data from the experiments, the charge balance figures of PM
2.5’s anions and cations in the four seasons in 2013 are drawn in
Figure 5.
The slope value of the linear fitting lines reached 0.9319 (R
2 = 0.9887), 0.9279 (R
2 = 0.9459) and 1.0158 (R
2 = 0.9844) in spring, summer and autumn, respectively. All values were nearly 1, while the slope value in winter only reached 0.8888 (R
2 = 0.9688), and had a relatively large gap with 1. These results revealed that the main ionic compositions in PM
2.5 in spring, summer and autumn were SO
42−, NO
3−, Cl
−, Na
+, K
+, NH
4+, Mg
2+ and Ca
2+, the eight ions that the experiments analyzed. By contrast, cationic charge numbers were slightly low in winter, revealing that there were some other cationic ions not detected except those had been measured in this study (Na
+, K
+, NH
4+, Mg
2+ and Ca
2+), such as H
+ [
34], organic cations or heavy metal ions (Zn
2+, Cu
2+,
etc.), which reflected that the ion components of PM
2.5 in winter were much more complicated than that in spring, summer and autumn, and resulted from the more serious air pollution problems in winter compared with other seasons. Morever, existing research have shown that the mass concentrations of PM were higher in winter than other seasons, hence it carried a certain probability that PM
2.5 contained organic cations [
7] or heavy metal ions (Zn
2+, Cu
2+,
etc.) in winter [
35]. This is not only a significant feature of the PM
2.5 in winter, but also one of the reasons that the days of heavy pollution weather in winter were more than the days in the other three seasons.
3.5. Correlation and Seasonal Difference Analysis of Water-Soluble Ions
The existing forms of water-soluble ions in PM
2.5 are diverse in different air pollution extents or different seasons, which have certain effects on atmospheric visibility, the PH of particulate matter, the viability of chemical reactions,
etc. The correlation analysis method is usually used to study the existing forms of water-soluble ions [
36]. As the correlation coefficient between water-soluble ions increases, the correlation between water-soluble ions increases.
The Pearson correlation coefficients of the water-soluble ions of PM
2.5 in all four seasons are shown in
Table 3,
Table 4,
Table 5 and
Table 6 below. High correlations existed between NH
4+ and SO
42−, NH
4+ and NO
3−, Mg
2+ and SO
42−, Ca
2+ and SO
42−, K
+ and Cl
−, Na
+ and Cl
−, which were consistent overall in one season. Nevertheless, seasonal differences lie in water-soluble ions. The correlation levels between NH
4+ and SO
42−, NH
4+ and NO
3− were significantly higher than the level in the summer, slightly exceeding the value in the autumn, while distinctly lower than the degree in the winter. The correlations between Mg
2+ and SO
42− were higher in the spring, summer and autumn, but not in the winter, according to the sequence that the correlation coefficient spring > summer > autumn > winter. The correlation between Mg
2+ and Cl
− was higher than the level between Mg
2+ and SO
42−. The correlation of Ca
2+ and SO
42− followed the order of spring > autumn > summer > winter, and the correlation between Ca
2+ and NO
3− was higher than that between Ca
2+ and SO
42−. The correlation between K
+ and Cl
− followed the order autumn > winter > spring, and the correlation level of K
+ and SO
42− was obvious than the level of K
+ and Cl
−. NH
4+, SO
42− and NO
3− in the weak acid environment is reversible reaction, and reaction process is as follows:
NH
4+ as a kind of weak acid ion, is an incomplete reaction in aqueous solution, which existing in free form has not been involved in the charge balance in the solution. In the acidic environment, we can ignore the effects of free NH
4+ on the balance, the results as shown in
Figure 6.
Figure 6 presents the positive and negative charge balances of NH
4+, SO
42− and NO
3− in all four seasons. As is shown in these figures, the slope values (k) of the fitting line between the charge equivalent of NH
4+ and the charge equivalent of SO
42−+NO
3− were all less, but very close to, 1; meanwhile, the goodness of fit values (R
2) approximated 1. As a consequence, NH
4+ in PM
2.5 in Wuhan was neutralized by SO
42− and NO
3− in all four seasons in 2013, which then existed with the forms of (NH
4)
2SO
4, (NH
4)
3H(SO
4)
2and NH
4NO
3 in PM
2.5.
Synthetically, diverse forms of inorganic water-soluble ions in PM
2.5 not only have some similar states or common characteristics but also exists some variation in four different seasons in Wuhan. The similarity or consistency was revealed at the aspect that the main compositions of PM
2.5 were basically identical in four seasons, with their cations consisted of NH
4+, Mg
2+, Ca
2+, K
+ and Na
+. In addition, there were several kinds of same particles in the four seasons, including (NH
4)
2SO
4, NH
4NO
3 and CaSO
4. The variation or diversity was reflected by the types of main particles compositions of PM
2.5 in four seasons. Among them, Na
+ ion mainly composited to form NaCl in spring (correlation coefficient between Na
+ and Cl
− reached 0.458 in
Table 3), while forming NaNO
3 in summer, autumn and winter (correlation coefficients between Na
+ and NO
3− reached 0.423, 0.331 and 0.706 in
Table 4,
Table 5 and
Table 6, respectively); K
+ composed to be K
2SO
4 in summer (correlation coefficient between K
+ and SO
42− reached 0.631 in
Table 4), and then KCl in spring, autumn and winter (correlation coefficients between K
+ and Cl
− reached 0.537, 0.632 and 0.612 in
Table 3, and
Table 5 and
Table 6, respectively); K
+ also formed KNO
3 only in autumn (correlation coefficient between K
+ and NO
3− reached 0.586 in
Table 5); Mg
2+ composited MgCl
2 in winter (correlation coefficient between Mg
2+ and Cl
− reached 0.331 in
Table 6) while MgSO
4 in spring, summer and autumn (correlation coefficients between Mg
2+ and SO
42− reached 0.590, 0.469 and 0.441 in
Table 3,
Table 4 and
Table 5, respectively); furthermore, Ca(NO
3)
2 also came into being in winter as a compound of Ca
2+, with correlation coefficient between Ca
2+ and NO
3− reached 0.418 in
Table 6, unlike other seasons that CaSO
4 was the main existing form.