Application of Klebsiella oxytoca Biomass in the Biosorptive Treatment of PAH-Bearing Wastewater: Effect of PAH Hydrophobicity and Implications for Prediction
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Strain, Medium, and Biosorbent
2.3. Biodegradation and Sorption of Phenanthrene by a Live Bacterial Strain
2.4. Biosorption of PAHs by Inactive Bacterial Biomass
2.5. Analytical Methods
3. Results and Discussion
3.1. Biosorbent Characterization
3.2. Biosorption and Biodegradation of PHEN by Klebsiella oxytoca PYR-1
3.3. Biosorption of PAHs
3.3.1. Effects of Dosage
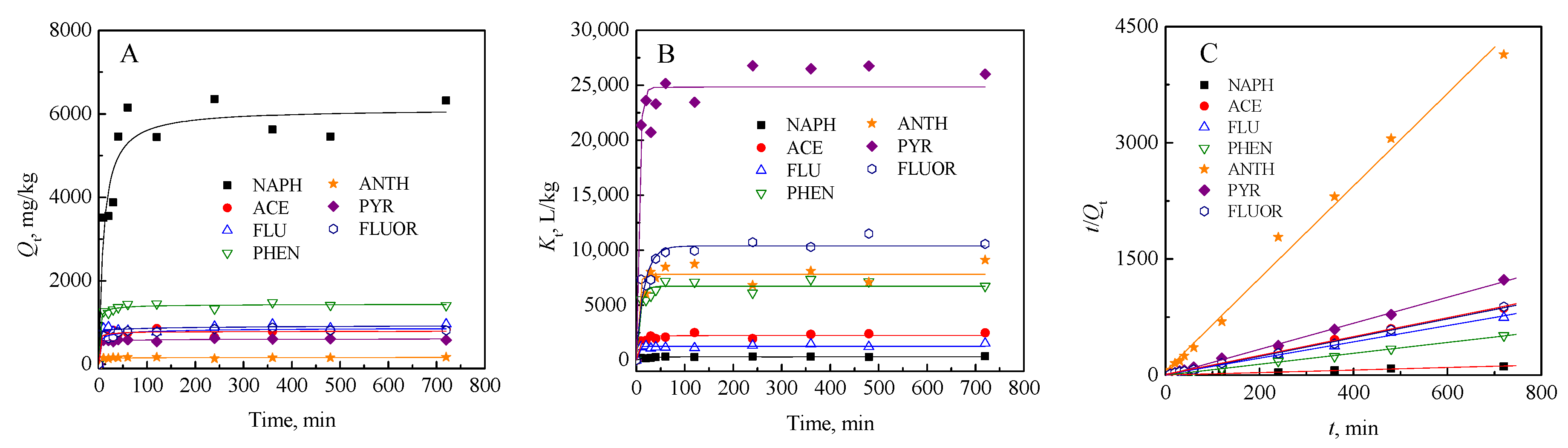
3.3.2. Biosorption Kinetics
3.3.3. Isotherm Biosorption
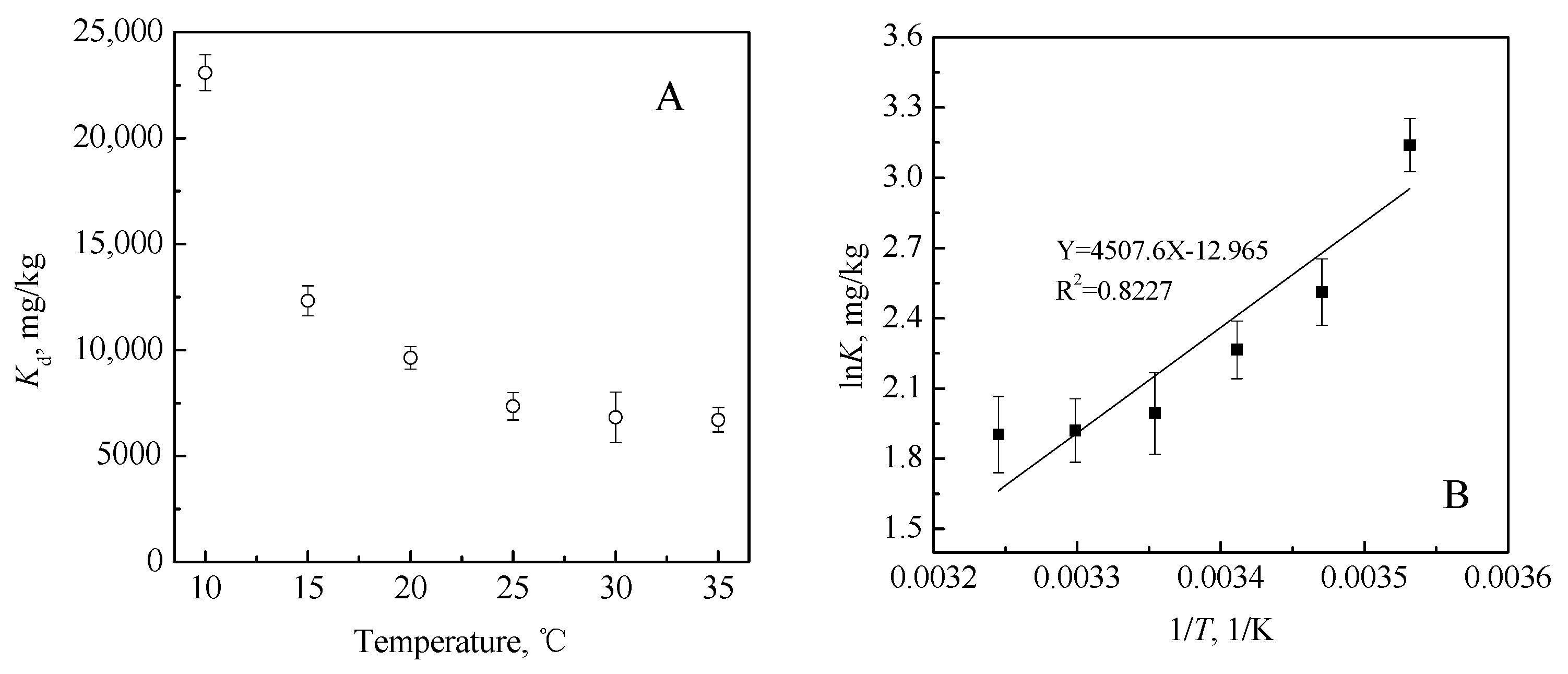
3.3.4. Thermodynamic Biosorption
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Stringfellow, W.T.; Alvarez-Cohen, L. Evaluating the relationship between the sorption of PAHs to bacterial biomass and biodegradation. Water Res. 1999, 33, 2535–2544. [Google Scholar] [CrossRef]
- Chen, B.L.; Wang, Y.S.; Hu, D.F. Biosorption and biodegradation of polycyclic aromatic hydrocarbons in aqueous solutions by a consortium of white-rot fungi. J. Hazard. Mater. 2010, 179, 845–851. [Google Scholar] [CrossRef] [PubMed]
- Tran, V.S.; Ngo, H.H.; Guo, W.S.; Zhang, J.; Liang, S.; Ton-That, C.; Zhang, X.B. Typical low cost biosorbents for adsorptive removal of specific organic pollutants from water. Bioresour. Technol. 2015, 182, 353–363. [Google Scholar] [CrossRef] [PubMed]
- US EPA (United States Environmental Protection Agency). Polycyclic Aromatic Hydrocarbons (PAHs); Office of Solid Waste: Washington, DC, USA, 2008.
- Zhang, D.; Zhu, L.Z. Controlling microbiological interfacial behaviors of hydrophobic organic compounds by surfactants in biodegradation process. Front. Environ. Sci. Eng. 2014, 8, 305–315. [Google Scholar] [CrossRef]
- Xu, N.-N.; Bao, M.-T.; Sun, P.-Y.; Li, Y.-M. Study on bioadsorption and biodegradation of petroleum hydrocarbons by a microbial consortium. Bioresour. Technol. 2013, 149, 22–30. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.-N.; Ran, C.-Y.; Yang, Y.; Ran, Y. Biosorption of phenanthrene by pure algae and field-collected planktons and their fractions. Chemosphere 2013, 93, 61–68. [Google Scholar] [CrossRef] [PubMed]
- Sanches, S.; Marins, M.; Silva, A.F.; Galinha, C.F.; Santos, M.A.; Pereira, I.A.C.; Crespo, M.T.B. Bioremoval of priority polycyclic aromatic hydrocarbons by a microbial community with high sorption ability. Environ. Sci. Pollut. Res. 2017, 24, 3550–3561. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Zhu, L.-Z. Effects of Tween 80 on the removal, sorption and biodegradation of pyrene by Klebsiealla oxytoca PYR-1. Environ. Pollut. 2012, 164, 169–174. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Zhu, L.-Z.; Li, F. Influence and mechanisms of surfactants on pyrene biodegradation based on interactions of surfactant with a Klebsiella oxytoca strain. Bioresour. Technol. 2013, 142, 454–461. [Google Scholar] [CrossRef] [PubMed]
- Volesky, B. Sorption and Biosorption; BV Sorbex: Quebec, QC, Canada, 2003. [Google Scholar]
- Aksu, Z. Application of biosorption for the removal of organic pollutants: A review. Process Biochem. 2005, 40, 997–1026. [Google Scholar] [CrossRef]
- Gu, H.-P.; Luo, X.-Y.; Wang, H.-Z.; Wu, L.-S.; Wu, J.-J.; Xu, J.-M. The characteristics of phenanthrene biosorption by chemically modified biomass of Phanerochaete chrysosporium. Environ. Sci. Pollut. Res. 2015, 22, 11850–11861. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.-L.; Ding, J. Biosorption and biodegradation of phenanthrene and pyrene in sterilized and unsterilized soil slurry systems stimulated by Phanerochaete chrysosporium. J. Hazard. Mater. 2012, 229, 159–169. [Google Scholar] [CrossRef] [PubMed]
- Gu, H.-P.; Luo, J.; Wang, H.-Z.; Yang, Y.; Wu, L.-S.; Wu, J.-J.; Xu, J.-M. Biodegradation, biosorption of phenanthrene and its trans-membrane transport by Massilia sp WF1 and Phanerochaete chrysosporium. Front. Microbiol. 2016, 7, 38. [Google Scholar] [CrossRef] [PubMed]
- Giri, A.K.; Patel, R.K.; Mahapatra, S.S.; Mishra, P.C. Biosorption of arsenic (III) from aqueous solution by living cells of Bacillus cereus. Environ. Sci. Pollut. Res. 2013, 20, 1281–1291. [Google Scholar] [CrossRef] [PubMed]
- Arias, A.H.; Souissi, A.; Glippa, O.; Roussin, M.; Dumoulin, D.; Net, S.; Ouddane, B.; Souissi, S. Removal and biodegradation of phenanthrene, fluoranthene and pyrene by the marine algae Rhodomonas baltica enriched from North Atlantic Coasts. Bull. Environ. Contam. Toxicol. 2017, 98, 392–399. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.-N.; Ran, Y.; Cao, X.-Y.; Mao, J.-D.; Cui, J.-F.; Schmidt-Rohr, K. Biosorption of nonylphenol by pure algae, field-collected planktons and their fractions. Environ. Pollut. 2015, 198, 61–69. [Google Scholar] [CrossRef] [PubMed]
- Ata, A.; Nalcaci, O.O.; Ovez, B. Macro algae Gravilaria verrucosa as a biosorbent: A study of sorption mechanisms. Algal Res. 2012, 1, 194–204. [Google Scholar] [CrossRef]
- Loukidou, M.X.; Zouboulis, A.I.; Karapantsios, T.D.; Matis, K.A. Equilibirum and kinetic modeling of chromium (VI) biosorption by Aeromonsa caviae. Colloid Surf. A 2004, 242, 93–104. [Google Scholar] [CrossRef]
- Bano, A.; Hussain, J.; Akbar, A.; Mehmood, K.; Anwar, M.; Hasni, M.S.; Ullah, S.; Sajid, S.; Ali, I. Biosorption of heavy metals by obligate halophilic fungi. Chemosphere 2018, 199, 218–222. [Google Scholar] [CrossRef] [PubMed]
- Park, J.H.; Chon, H.T. Characterization of cadmium biosorption by Exiguobacterium sp. Isolated from farmland soil near Cu-Pb-Zn mine. Environ. Sci. Pollut. Res. 2016, 23, 11814–11822. [Google Scholar] [CrossRef] [PubMed]
- Albert, Q.; Leleyter, L.; Lemoine, M.; Heutte, N.; Rioult, J.P.; Sage, L.; Baraud, F.; Garon, D. Comparison of tolerance and biosorption of three trace metals (Cd, Cu, Pb) by soil fungus Absidia cylindrospora. Chemosphere 2018, 196, 386–392. [Google Scholar] [CrossRef] [PubMed]
- Yu, Q.; Fein, J.B. Enhanced removal of dissolved Hg(II), Cd(II), and Au(III) from water by Bacillus subtilis bacterial biomass containing an elevated concentration of sulfhydryl sites. Environ. Sci. Technol. 2017, 51, 14360–14367. [Google Scholar] [CrossRef] [PubMed]
- Sulaymon, A.H.; Mohammed, A.A.; Al-Musawi, T.J. Competitive biosorption of lead, cadmium, copper, and arsenic ions using algae. Environ. Sci. Pollut. Res. 2013, 20, 3011–3023. [Google Scholar] [CrossRef] [PubMed]
- Cheng, N.; Li, Q.-Y.; Tang, A.-X.; Su, W.; Liu, Y.-Y. Decolorization of a variety of dyes by Aspergillus flavus A5p1. Bioprocess Biosyst. Eng. 2018, 41, 511–518. [Google Scholar]
- Zhao, S.-X.; Zhou, T.-S. Biosorption of methylene blue from wastewater by an extraction residue of Salvia miltiorrhiza Bge. Bioresour. Technol. 2016, 219, 330–337. [Google Scholar] [CrossRef] [PubMed]
- Chandra, T.S.; Mudliar, S.N.; Vidyashankar, S.; Mukherji, S.; Sarada, R.; Krishnamurthi, K.; Chauhan, V.S. Defatted algal biomass as a non-conventional low-cost adsorbent: Surface characterization and methylene blue adsorption characteristics. Bioresour. Technol. 2015, 184, 395–404. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.; Ye, J.-S.; Ma, J.-W.; Tang, L.-T.; Huang, J. Biosorption and biodegradation of triphenyltin by Stenotrophomonas maltophilia and their influence on cellular metabolism. J. Hazard. Mater. 2014, 276, 112–119. [Google Scholar] [CrossRef] [PubMed]
- Tsezos, M.; Bell, J.P. Comparison of the biosorption and desorption of hazardous organic pollutants by live and dead biomass. Water Res. 1989, 23, 561–568. [Google Scholar] [CrossRef]
- Li, Y.-R.; Zhang, J.; Liu, H. Removal of chloramphenicol from aqueous solution using low-cost activated carbon prepared from Typha orientalis. Water 2018, 10, 351. [Google Scholar]
- Vijayaraghavan, K.; Yun, Y.-S. Utilization of fermentation waste (Corynebacterium glutamicum) for biosorption of Reactive Black 5 from aqueous solution. J. Hazard. Mater. 2007, 141, 45–52. [Google Scholar] [CrossRef] [PubMed]
- Chan, S.M.N.; Luan, T.; Wong, M.H.; Tam, N.F.Y. Removal and biodegradation of polycyclic aromatic hydrocarbons by Selenastrum capricornutum. Environ. Toxicol. Chem. 2006, 25, 1772–1779. [Google Scholar] [CrossRef] [PubMed]
- Xiao, L.; Qu, X.; Zhu, D.-Q. Biosorption of nonpolar hydrophobic organic compounds to Escherichia coli facilitated by metal and proton surface binding. Environ. Sci. Technol. 2007, 41, 2750–2755. [Google Scholar] [CrossRef] [PubMed]
- Xi, Z.-M.; Chen, B.-L. The effect of structural compositions on the biosorption of phenanthrene and pyrene by tea leaf residues fractions as model biosorbents. Environ. Sci. Pollut. Res. 2014, 21, 3318–3330. [Google Scholar] [CrossRef] [PubMed]
- Bai, L.-L.; Xu, H.-C.; Wang, C.-H.; Deng, J.-C.; Jiang, H.-L. Extracellular polymeric substances facilitate the biosorption of phenanthrene on cyanobacteria Microcystis aeruginosa. Chemosphere 2016, 162, 172–180. [Google Scholar] [CrossRef] [PubMed]
- Mahmoud, M.E.; El Zokm, G.E.; Farag, A.E.M.; Abdelwahab, M.S. Assessment of heat-inactivated marine Aspergillus flavus as a novel biosorbent for removal of Cd(II), Hg(II), and Pb(II) from water. Environ. Sci. Pollut. Res. 2017, 24, 18218–18228. [Google Scholar] [CrossRef] [PubMed]
- Puchana-Rosero, M.J.; Lima, E.C.; Ortiz-Monsalve, S.; Mella, B.; Costa, D.; Poll, E.; Gutterres, M. Fungal biomass as biosorbent for the removal of Acid Blue 161 dye in aqueous solution. Environ. Sci. Pollut. Res. 2017, 24, 4200–4209. [Google Scholar] [CrossRef] [PubMed]
- Noinaj, N.; Kuszak, A.J.; Gumbart, J.C.; Lukacik, P.; Chang, H.; Easley, N.C.; Lithgow, T.; Buchanan, S.K. Structural insight into the biogenesis of β-barrel membrane proteins. Nature 2013, 501, 385–390. [Google Scholar] [CrossRef] [PubMed]
- Xi, Z.-M.; Chen, B.-L. Removal of polycyclic aromatic hydrocarbons from aqueous solution by raw and modified plant residue materials as biosorbents. J. Environ. Sci. (China) 2014, 26, 737–748. [Google Scholar] [CrossRef]
- Olivella, M.A.; Jove, P.; Oliveras, A. The use of cork waste as a biosorbent for persistent organic pollutants-study of adsorption/desorption of polycyclic aromatic hydrocarbons. J. Environ. Sci. Health A 2011, 46, 824–832. [Google Scholar] [CrossRef] [PubMed]
- Yang, K.; Zhu, L.-Z.; Xing, B.-S. Enhanced soil washing of phenanthrene by mixed solutions of TX100 and SDBS. Environ. Sci. Technol. 2006, 40, 4274–7280. [Google Scholar] [CrossRef] [PubMed]
- Zhou, W.-J.; Zhu, L.-Z. Enhanced desorption of phenanthrene from contaminated soil using anionic/nonionic mixed surfactant. Environ. Pollut. 2007, 147, 350–357. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.-G.; Chen, B.-L.; Zhu, L.-Z. Enhanced sorption of polycyclic aromatic hydrocarbons from aqueous solution by modified pine bark. Bioresour. Technol. 2010, 101, 7307–7313. [Google Scholar] [CrossRef] [PubMed]
- Steen, W.C.; Karickhoff, S.W. Biosorption of hydrophobic organic pollutants by mixed microbial populations. Chemosphere 1984, 10, 27–32. [Google Scholar] [CrossRef]
- Chung, M.K.; Tsui, M.T.K.; Cheung, K.C.; Tam, N.F.Y.; Wong, M.H. Removal of aqueous phenanthrene by brown seaweed Sargassum hemiphyllum: Sorption-kinetic and equilibrium studies. Sep. Purif. Technol. 2007, 54, 355–362. [Google Scholar] [CrossRef]
- Yin, J.; Deng, C.-B.; Yu, Z.; Wang, X.-F.; Xu, G.-P. Effective removal of lead ions from aqueous solution using nano illite/smectite clay: Isotherm, kinetic, and thermodynamic modeling of adsorption. Water 2018, 10, 210. [Google Scholar] [CrossRef]
- Zhang, M.; Ahmad, M.; Lee, S.; Xu, L.; Ok, Y.S. Sorption of polycyclic aromatic hydrocarbons (PAHs) to lignin: Effects of hydrophobicity and temperature. Bull. Environ. Contam. Toxicol. 2014, 93, 84–88. [Google Scholar] [CrossRef] [PubMed]





| PAHs | Formula | Molecular Weight | logKow | Sw, mg·L−1 | LOD, μg·L−1 | RSD, % |
|---|---|---|---|---|---|---|
| Naphthalene (NAPH) | C10H8 | 128.2 | 3.30 | 31.0 | 12.2 | 2.25 |
| Acenaphthene (ACE) | C12H10 | 154.2 | 3.92 | 3.90 | 7.78 | 0.480 |
| Fluorene (FLU) | C13H10 | 166.2 | 4.18 | 1.69 | 8.45 | 0.640 |
| Phenanthrene (PHEN) | C14H10 | 178.3 | 4.46 | 1.18 | 3.39 | 0.230 |
| Anthracene (ANTH) | C14H10 | 178.3 | 4.45 | 0.065 | 0.408 | 0.235 |
| Pyrene (PYR) | C16H10 | 202.3 | 4.88 | 0.12 | 2.4 | 0.490 |
| Fluoranthene (FLUOR) | C16H10 | 202.3 | 5.16 | 0.26 | 0.108 | 0.157 |
| Pseudo-Second Order t/Qt = 1/(k2Qm2) + (1/Qm)t | k2, g·mg−1·min−1 | Qm, mg·g−1 | h, mg·g−1·min−1 | R2 |
|---|---|---|---|---|
| NAPH | 0.01363 | 6.128 | 0.5116 | 0.9916 |
| ACE | 0.12781 | 0.821 | 0.0862 | 0.9925 |
| FLU | 0.10715 | 0.951 | 0.0969 | 0.9956 |
| PHEN | 0.59152 | 1.418 | 1.1902 | 0.9990 |
| ANTH | 0.61155 | 0.168 | 0.0172 | 0.9906 |
| PYR | 0.22423 | 1.630 | 0.5956 | 0.9986 |
| FLUOR | 0.55333 | 0.829 | 0.3799 | 0.9984 |
| PAHs | Freundlich Model | Linear Model | |||
|---|---|---|---|---|---|
| KF, L·kg−1 | n | R2 | Kd, L·kg−1 | R2 | |
| NAPH | 289.60 | 0.8883 | 0.9516 | 363.11 ± 19.768 | 0.9711 |
| ACE | 1711.2 | 0.9234 | 0.9740 | 1719.5 ± 73.475 | 0.9820 |
| FLU | 2546.2 | 1.0164 | 0.9888 | 2515.5 ± 57.521 | 0.9948 |
| PHEN | 9369.1 | 0.8774 | 0.8859 | 7343.3 ± 641.91 | 0.9285 |
| ANTH | 7174.6 | 1.0028 | 0.9890 | 6353.4 ± 26.720 | 0.9998 |
| PYR | 123,908 | 0.7014 | 0.8702 | 22,806 ± 901.58 | 0.9846 |
| FLUOR | 18,668 | 1.0077 | 0.9967 | 19,541 ± 194.05 | 0.9990 |
| Biosorbent | Target Compound | logKow | Measured Kd, L·kg−1 | Predicted Kd, L·kg−1 | Prediction Error | Reference |
|---|---|---|---|---|---|---|
| White-rot fungi biomass: Phanerochaete hrysosporium | NAPH | 3.30 | 361.0 | 397.6 | 10.15 | [2] |
| ACE | 3.92 | 1656 | 1684 | 1.66% | ||
| FLU | 4.18 | 2890 | 3084 | 6.70% | ||
| PHEN | 4.46 | 6822 | 5918 | 13.3% | ||
| PYR | 4.88 | 24,140 | 15,732 | 34.8% | ||
| Mixed bacterial biomass | PHEN | 4.46 | 6340 | 5918 | 6.66% | [45] |
| PYR | 4.88 | 14,800 | 15,732 | 6.30% | ||
| Benzo[a]pyrene | 6.35 | 450,000 | 481,892 | 7.09% | ||
| Brown seaweed biomass: Sargassum hemiphyllum | PHEN | 4.46 | 6760 | 5918 | 14.2% | [46] |
| Fungi biomass: Rhizopus arrhizus | Lindane | 3.72 | 1057 | 1170 | 9.67% | [30] |
| Diazinon | 3.81 | 1303 | 1220 | 6.82% | ||
| Pentachlorophenol | 5.18 | 31,629 | 32,130 | 1.56% |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, D.; Lu, L.; Zhao, H.; Jin, M.; Lü, T.; Lin, J. Application of Klebsiella oxytoca Biomass in the Biosorptive Treatment of PAH-Bearing Wastewater: Effect of PAH Hydrophobicity and Implications for Prediction. Water 2018, 10, 675. https://doi.org/10.3390/w10060675
Zhang D, Lu L, Zhao H, Jin M, Lü T, Lin J. Application of Klebsiella oxytoca Biomass in the Biosorptive Treatment of PAH-Bearing Wastewater: Effect of PAH Hydrophobicity and Implications for Prediction. Water. 2018; 10(6):675. https://doi.org/10.3390/w10060675
Chicago/Turabian StyleZhang, Dong, Li Lu, Hongting Zhao, Meiqing Jin, Ting Lü, and Jun Lin. 2018. "Application of Klebsiella oxytoca Biomass in the Biosorptive Treatment of PAH-Bearing Wastewater: Effect of PAH Hydrophobicity and Implications for Prediction" Water 10, no. 6: 675. https://doi.org/10.3390/w10060675
APA StyleZhang, D., Lu, L., Zhao, H., Jin, M., Lü, T., & Lin, J. (2018). Application of Klebsiella oxytoca Biomass in the Biosorptive Treatment of PAH-Bearing Wastewater: Effect of PAH Hydrophobicity and Implications for Prediction. Water, 10(6), 675. https://doi.org/10.3390/w10060675





