Activated Carbon Mixed with Marine Sediment is Suitable as Bioanode Material for Spartina anglica Sediment/Plant Microbial Fuel Cell: Plant Growth, Electricity Generation, and Spatial Microbial Community Diversity
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Setup
2.2. Operations
2.3. Measurements and Analysis
2.3.1. pH, Conductivity, and Acetate Analysis
2.3.2. Plant Growth Monitoring
2.3.3. DNA Analysis
2.4. Calculations
3. Results and Discussion
3.1. Mixture of Activated Carbon (AC) and Marine Sediment Effect on Plant Growth
3.2. Mixture of Marine Sediment and Activated Carbon Generating Electricity in Plant-MFCs
3.3. Diverse Microbial Communities
4. Conclusions and Outlook
5. Associated Content
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hu, S.; Niu, Z.; Chen, Y.; Li, L.; Zhang, H. Global wetlands: Potential distribution, wetland loss, and status. Sci. Total Environ. 2017, 586, 319–327. [Google Scholar] [CrossRef]
- Kingsford, R.T.; Basset, A.; Jackson, L. Wetlands: Conservation’s poor cousins. Aquat. Conserv. Mar. Freshw. Ecosyst. 2016, 26, 892–916. [Google Scholar] [CrossRef]
- Davidson, N.C.; Finlayson, C.M. Updating global coastal wetland areas presented in Davidson and Finlayson (2018). Mar. Freshw. Res. 2019, 70, 1195–1200. [Google Scholar] [CrossRef]
- Dixon, M.J.R.; Loh, J.; Davidson, N.C.; Beltrame, C.; Freeman, R.; Walpole, M. Tracking global change in ecosystem area: The Wetland Extent Trends index. Biol. Conserv. 2016, 193, 27–35. [Google Scholar] [CrossRef]
- Bai, J.; Jia, J.; Zhang, G.; Zhao, Q.; Lu, Q.; Cui, B.; Liu, X. Spatial and temporal dynamics of heavy metal pollution and source identification in sediment cores from the short-term flooding riparian wetlands in a Chinese delta. Environ. Pollut. 2016, 219, 379–388. [Google Scholar] [CrossRef]
- Varjani, S.J.; Upasani, V.N. A new look on factors affecting microbial degradation of petroleum hydrocarbon pollutants. Int. Biodeterior. Biodegrad. 2017, 120, 71–83. [Google Scholar] [CrossRef]
- Patmont, C.R.; Ghosh, U.; LaRosa, P.; Menzie, C.A.; Luthy, R.G.; Greenberg, M.S.; Cornelissen, G.; Eek, E.; Collins, J.; Hull, J.; et al. In situ sediment treatment using activated carbon: A demonstrated sediment cleanup technology. Integr. Environ. Assess. Manag. 2015, 11, 195–207. [Google Scholar] [CrossRef]
- Karanfil, T.; Kilduff, J.E. Role of Granular Activated Carbon Surface Chemistry on the Adsorption of Organic Compounds. 1. Priority Pollutants. Environ. Sci. Technol. 1999, 33, 3217–3224. [Google Scholar] [CrossRef]
- Li, L.; Quinlivan, P.A.; Knappe, D.R.U. Effects of activated carbon surface chemistry and pore structure on the adsorption of organic contaminants from aqueous solution. Carbon 2002, 40, 2085–2100. [Google Scholar] [CrossRef]
- Lee, J.; Kim, J.; Hyeon, T. Recent Progress in the Synthesis of Porous Carbon Materials. Adv. Mater. 2006, 18, 2073–2094. [Google Scholar] [CrossRef]
- Tender, L.M.; Reimers, C.E.; Stecher, H.A., III; Holmes, D.E.; Bond, D.R.; Lowy, D.A.; Pilobello, K.; Fertig, S.J.; Lovley, D.R. Harnessing microbially generated power on the seafloor. Nat. Biotechnol. 2002, 20, 821–825. [Google Scholar] [CrossRef]
- Abbas, S.Z.; Rafatullah, M.; Ismail, N.; Syakir, M.I. A review on sediment microbial fuel cells as a new source of sustainable energy and heavy metal remediation: Mechanisms and future prospective. Int. J. Energy Res. 2017, 41, 1242–1264. [Google Scholar] [CrossRef]
- Xu, P.; Xiao, E.-R.; Xu, D.; Zhou, Y.; He, F.; Liu, B.-Y.; Zeng, L.; Wu, Z.-B. Internal nitrogen removal from sediments by the hybrid system of microbial fuel cells and submerged aquatic plants. PLoS ONE 2017, 12, e0172757. [Google Scholar] [CrossRef]
- Zhou, Y.-L.; Wu, H.-F.; Yan, Z.-S.; Cai, H.-Y.; Jiang, H.-L. The enhanced survival of submerged macrophyte Potamogeton malaianus by sediment microbial fuel cells. Ecol. Eng. 2016, 87, 254–262. [Google Scholar] [CrossRef]
- Strik, D.P.B.T.B.; Hamelers (Bert), H.V.M.; Snel, J.F.H.; Buisman, C.J.N. Green electricity production with living plants and bacteria in a fuel cell. Int. J. Energy Res. 2008, 32, 870–876. [Google Scholar] [CrossRef]
- Ueoka, N.; Sese, N.; Sue, M.; Kouzuma, A.; Watanabe, K. Sizes of Anode and Cathode Affect Electricity Generation in Rice Paddy-Field Microbial Fuel Cells. J. Sustain. Bioenergy Syst. 2016, 6, 10–15. [Google Scholar] [CrossRef]
- Kaku, N.; Yonezawa, N.; Kodama, Y.; Watanabe, K. Plant/microbe cooperation for electricity generation in a rice paddy field. Appl. Microbiol. Biotechnol. 2008, 79, 43–49. [Google Scholar] [CrossRef]
- Takanezawa, K.; Nishio, K.; Kato, S.; Hashimoto, K.; Watanabe, K. Factors Affecting Electric Output from Rice-Paddy Microbial Fuel Cells. Biosci. Biotechnol. Biochem. 2010, 74, 1271–1273. [Google Scholar] [CrossRef]
- Kouzuma, A.; Kaku, N.; Watanabe, K. Microbial electricity generation in rice paddy fields: Recent advances and perspectives in rhizosphere microbial fuel cells. Appl. Microbiol. Biotechnol. 2014, 98, 9521–9526. [Google Scholar] [CrossRef]
- Helder, M.; Strik, D.P.B.T.B.; Hamelers, H.V.M.; Kuhn, A.J.; Blok, C.; Buisman, C.J.N. Concurrent bio-electricity and biomass production in three Plant-Microbial Fuel Cells using Spartina anglica, Arundinella anomala and Arundo donax. Bioresour. Technol. 2010, 101, 3541–3547. [Google Scholar] [CrossRef]
- Helder, M.; Strik, D.P.B.T.B.; Hamelers, H.V.M.; Kuijken, R.C.P.; Buisman, C.J.N. New plant-growth medium for increased power output of the Plant-Microbial Fuel Cell. Bioresour. Technol. 2012, 104, 417–423. [Google Scholar] [CrossRef]
- Timmers, R.A.; Strik, D.P.B.T.B.; Hamelers, H.V.M.; Buisman, C.J.N. Characterization of the internal resistance of a plant microbial fuel cell. Electrochim. Acta 2012, 72, 165–171. [Google Scholar] [CrossRef]
- Wetser, K.; Liu, J.; Buisman, C.; Strik, D. Plant microbial fuel cell applied in wetlands: Spatial, temporal and potential electricity generation of Spartina anglica salt marshes and Phragmites australis peat soils. Biomass Bioenergy 2015, 83, 543–550. [Google Scholar] [CrossRef]
- Helder, M.; Strik, D.P.; Hamelers, H.V.; Buisman, C.J. The flat-plate plant-microbial fuel cell: The effect of a new design on internal resistances. Biotechnol. Biofuels 2012, 5, 70. [Google Scholar] [CrossRef] [PubMed]
- Wetser, K.; Dieleman, K.; Buisman, C.; Strik, D. Electricity from wetlands: Tubular plant microbial fuels with silicone gas-diffusion biocathodes. Appl. Energy 2017, 185, 642–649. [Google Scholar] [CrossRef]
- Timmers, R.A.; Strik, D.P.B.T.B.; Hamelers, H.V.M.; Buisman, C.J.N. Electricity generation by a novel design tubular plant microbial fuel cell. Biomass Bioenergy 2013, 51, 60–67. [Google Scholar] [CrossRef]
- Schamphelaire, L.D.; Bossche, L.V.D.; Dang, H.S.; Höfte, M.; Boon, N.; Rabaey, K.; Verstraete, W. Microbial Fuel Cells Generating Electricity from Rhizodeposits of Rice Plants. Environ. Sci. Technol. 2008, 42, 3053–3058. [Google Scholar] [CrossRef]
- Wetser, K.; Sudirjo, E.; Buisman, C.J.N.; Strik, D.P.B.T.B. Electricity generation by a plant microbial fuel cell with an integrated oxygen reducing biocathode. Appl. Energy 2015, 137, 151–157. [Google Scholar] [CrossRef]
- Wetser, K. Electricity from Wetlands: Technology Assessment of the Tubular Plant Microbial Fuel Cell with an Integrated Biocathode. Ph.D. Thesis, Wageningen University & Research, Wageningen, The Netherlands, 2016. [Google Scholar]
- Kabutey, F.T.; Zhao, Q.; Wei, L.; Ding, J.; Antwi, P.; Quashie, F.K.; Wang, W. An overview of plant microbial fuel cells (PMFCs): Configurations and applications. Renew. Sustain. Energy Rev. 2019, 110, 402–414. [Google Scholar] [CrossRef]
- Helder, M.; Chen, W.-S.; van der Harst, E.J.M.; Strik, D.P.B.T.B.; Hamelers, H.V.M.; Buisman, C.J.N.; Potting, J. Electricity production with living plants on a green roof: Environmental performance of the plant-microbial fuel cell. Biofuels Bioprod. Biorefin. 2013, 7, 52–64. [Google Scholar] [CrossRef]
- Timmers, R.A.; Strik, D.P.B.T.B.; Hamelers, H.V.M.; Buisman, C.J.N. Long-term performance of a plant microbial fuel cell with Spartina anglica. Appl. Microbiol. Biotechnol. 2010, 86, 973–981. [Google Scholar] [CrossRef] [PubMed]
- Gray, A.J.; Marshall, D.F.; Raybould, A.F. A Century of Evolution in Spartina anglica. In Advances in Ecological Research; Begon, M., Fitter, A.H., Macfadyen, A., Eds.; Academic Press: Cambridge, MA, USA, 1991; Volume 21, pp. 1–62. ISBN 0065-2504. [Google Scholar]
- Raybould, A.F.; Gray, A.J.; Lawrence, M.J.; Marshall, D.F. The evolution of Spartina anglica C. E. Hubbard (Gramineae): Genetic variation and status of the parental species in Britain. Biol. J. Linn. Soc. 1991, 44, 369–380. [Google Scholar] [CrossRef]
- Nehring, S.; Adsersen, H. NOBANIS—Invasive Alien Species Fact Sheet—Spartina Anglica.-From: Online Database of the European Network on Invasive Alien Species-NOBANIS. 2006. Available online: https://www.nobanis.org/globalassets/speciesinfo/s/spartina-anglica/spartina_anglica.pdf (accessed on 28 January 2019).
- Nitisoravut, R.; Regmi, R. Plant microbial fuel cells: A promising biosystems engineering. Renew. Sustain. Energy Rev. 2017, 76, 81–89. [Google Scholar] [CrossRef]
- Salinas-Juárez, M.G.; Roquero, P.; Durán-Domínguez-de-Bazúa, M.D.C. Plant and microorganisms support media for electricity generation in biological fuel cells with living hydrophytes. Bioelectrochemistry 2016, 112, 145–152. [Google Scholar] [CrossRef] [PubMed]
- Arends, J.B.A.; Speeckaert, J.; Blondeel, E.; De Vrieze, J.; Boeckx, P.; Verstraete, W.; Rabaey, K.; Boon, N. Greenhouse gas emissions from rice microcosms amended with a plant microbial fuel cell. Appl. Microbiol. Biotechnol. 2014, 98, 3205–3217. [Google Scholar] [CrossRef] [PubMed]
- Timmers, R.A.; Rothballer, M.; Strik, D.P.B.T.B.; Engel, M.; Schulz, S.; Schloter, M.; Hartmann, A.; Hamelers, B.; Buisman, C. Microbial community structure elucidates performance of Glyceria maxima plant microbial fuel cell. Appl. Microbiol. Biotechnol. 2012, 94, 537–548. [Google Scholar] [CrossRef]
- Bombelli, P.; Iyer, D.M.R.; Covshoff, S.; McCormick, A.J.; Yunus, K.; Hibberd, J.M.; Fisher, A.C.; Howe, C.J. Comparison of power output by rice (Oryza sativa) and an associated weed (Echinochloa glabrescens) in vascular plant bio-photovoltaic (VP-BPV) systems. Appl. Microbiol. Biotechnol. 2013, 97, 429–438. [Google Scholar] [CrossRef]
- Regmi, R.; Nitisoravut, R.; Charoenroongtavee, S.; Yimkhaophong, W.; Phanthurat, O. Earthen Pot–Plant Microbial Fuel Cell Powered by Vetiver for Bioelectricity Production and Wastewater Treatment. CLEAN Soil Air Water 2018, 46, 1700193. [Google Scholar] [CrossRef]
- Md Khudzari, J.; Gariépy, Y.; Kurian, J.; Tartakovsky, B.; Raghavan, G.S.V. Effects of biochar anodes in rice plant microbial fuel cells on the production of bioelectricity, biomass, and methane. Biochem. Eng. J. 2019, 141, 190–199. [Google Scholar] [CrossRef]
- Borsje, C.; Liu, D.; Sleutels, T.H.J.A.; Buisman, C.J.N.; ter Heijne, A. Performance of single carbon granules as perspective for larger scale capacitive bioanodes. J. Power Sources 2016, 325, 690–696. [Google Scholar] [CrossRef]
- Borsje, C.; Sleutels, T.; Saakes, M.; Buisman, C.J.N.; ter Heijne, A. The Granular Capacitive Moving Bed Reactor for the scale up of bioanodes. J. Chem. Technol. Biotechnol. 2019, 94, 2738–2748. [Google Scholar] [CrossRef]
- Sudirjo, E.; Buisman, C.J.N.; Strik, D.P.B.T.B. Marine Sediment Mixed with Activated Carbon Allows Electricity Production and Storage from Internal and External Energy Sources: A New Rechargeable Bio-Battery with Bi-Directional Electron Transfer Properties. Front. Microbiol. 2019, 10, 934. [Google Scholar] [CrossRef] [PubMed]
- Sudirjo, E.; Buisman, C.J.N.; Strik, D.P.B.T.B. Electricity generation from wetlands with activated carbon bioanode. IOP Conf. Ser. Earth Environ. Sci. 2018, 131, 012046. [Google Scholar] [CrossRef]
- Alslaibi, T.M.; Abustan, I.; Ahmad, M.A.; Foul, A.A. A review: Production of activated carbon from agricultural byproducts via conventional and microwave heating. J. Chem. Technol. Biotechnol. 2013, 88, 1183–1190. [Google Scholar] [CrossRef]
- Menya, E.; Olupot, P.W.; Storz, H.; Lubwama, M.; Kiros, Y. Production and performance of activated carbon from rice husks for removal of natural organic matter from water: A review. Chem. Eng. Res. Des. 2018, 129, 271–296. [Google Scholar] [CrossRef]
- Hale, S.E.; Jensen, J.; Jakob, L.; Oleszczuk, P.; Hartnik, T.; Henriksen, T.; Okkenhaug, G.; Martinsen, V.; Cornelissen, G. Short-Term Effect of the Soil Amendments Activated Carbon, Biochar, and Ferric Oxyhydroxide on Bacteria and Invertebrates. Environ. Sci. Technol. 2013, 47, 8674–8683. [Google Scholar] [CrossRef] [PubMed]
- Igalavithana, A.D.; Ok, Y.S.; Usman, A.R.A.; Al-Wabel, M.I.; Oleszczuk, P.; Lee, S.S. The Effects of Biochar Amendment on Soil Fertility. In Agricultural and Environmental Applications of Biochar: Advances and Barriers; SSSA Special Publication, Soil Science Society of America, Inc.: Madison, WI, USA, 2016; pp. 123–144. ISBN 978-0-89118-967-1. [Google Scholar]
- Bakker, J.P. Restoration of Salt Marshes. In Restoration Ecology: The New Frontier; Andel, J.V., Aronson, J., Eds.; Wiley-Blackwell: Hoboken, NJ, USA, 2012; pp. 248–262. ISBN 978-1-4443-3636-8. [Google Scholar]
- Deltares Pioneer Salt Marsh Restoration for Coastal Protection—Eastern Scheldt, NL. Available online: https://publicwiki.deltares.nl/display/BTG/Pioneer+salt+marsh+restoration+for+coastal+protection+-+Eastern+Scheldt%2C+NL (accessed on 1 July 2019).
- Wetland International Building with Nature Indonesia—Reaching Scale for Coastal Resilience. Available online: https://www.wetlands.org/publications/building-with-nature-in-indonesia/ (accessed on 1 July 2019).
- Jourdin, L.; Raes, S.M.T.; Buisman, C.J.N.; Strik, D.P.B.T.B. Critical Biofilm Growth throughout Unmodified Carbon Felts Allows Continuous Bioelectrochemical Chain Elongation from CO2 up to Caproate at High Current Density. Front. Energy Res. 2018, 6, 7. [Google Scholar] [CrossRef]
- De Smit, S.M.; de Leeuw, K.D.; Buisman, C.J.N.; Strik, D.P.B.T.B. Continuous n-valerate formation from propionate and methanol in an anaerobic chain elongation open-culture bioreactor. Biotechnol. Biofuels 2019, 12, 132. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, S.; Tomita, J.; Nishioka, K.; Hisada, T.; Nishijima, M. Development of a Prokaryotic Universal Primer for Simultaneous Analysis of Bacteria and Archaea Using Next-Generation Sequencing. PLoS ONE 2014, 9, e105592. [Google Scholar] [CrossRef]
- Edgar, R.C. Search and clustering orders of magnitude faster than BLAST. Bioinformatics 2010, 26, 2460–2461. [Google Scholar] [CrossRef]
- Quast, C.; Pruesse, E.; Yilmaz, P.; Gerken, J.; Schweer, T.; Yarza, P.; Peplies, J.; Glöckner, F.O. The SILVA ribosomal RNA gene database project: Improved data processing and web-based tools. Nucleic Acids Res. 2012, 41, D590–D596. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Garrity, G.M.; Tiedje, J.M.; Cole, J.R. Naive Bayesian classifier for rapid assignment of rRNA sequences into the new bacterial taxonomy. Appl. Environ. Microbiol. 2007, 73, 5261–5267. [Google Scholar] [CrossRef] [PubMed]
- Caporaso, J.G.; Kuczynski, J.; Stombaugh, J.; Bittinger, K.; Bushman, F.D.; Costello, E.K.; Fierer, N.; Peña, A.G.; Goodrich, J.K.; Gordon, J.I.; et al. QIIME allows analysis of high-throughput community sequencing data. Nat. Methods 2010, 7, 335–336. [Google Scholar] [CrossRef] [PubMed]
- Koo, H.; Hakim, J.A.; Morrow, C.D.; Andersen, D.T.; Bej, A.K. Chapter 9—Microbial Community Composition and Predicted Functional Attributes of Antarctic Lithobionts Using Targeted Next-Generation Sequencing and Bioinformatics Tools. In Methods in Microbiology; Gurtler, V., Trevors, J.T., Eds.; Academic Press: Cambridge, MA, USA, 2018; Volume 45, pp. 243–290. ISBN 0580-9517. [Google Scholar]
- Giusti, D.M.; Conway, R.A.; Lawson, C.T. Activated Carbon Adsorption of Petrochemicals. J. Water Pollut. Control Fed. 1974, 46, 947–965. [Google Scholar]
- Mugisidi, D.; Ranaldo, A.; Soedarsono, J.W.; Hikam, M. Modification of activated carbon using sodium acetate and its regeneration using sodium hydroxide for the adsorption of copper from aqueous solution. Carbon 2007, 45, 1081–1084. [Google Scholar] [CrossRef]
- Wilson, K.; Yang, H.; Seo, C.W.; Marshall, W.E. Select metal adsorption by activated carbon made from peanut shells. Bioresour. Technol. 2006, 97, 2266–2270. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.-M.; Chung, T.-W.; Huang, C.-M.; Wu, H. Adsorption Equilibria of Acetate Compounds on Activated Carbon, Silica Gel, and 13X Zeolite. J. Chem. Eng. Data 2005, 50, 811–816. [Google Scholar] [CrossRef]
- Wang, Z.; Nie, E.; Li, J.; Yang, M.; Zhao, Y.; Luo, X.; Zheng, Z. Equilibrium and kinetics of adsorption of phosphate onto iron-doped activated carbon. Environ. Sci. Pollut. Res. 2012, 19, 2908–2917. [Google Scholar] [CrossRef] [PubMed]
- Boopathy, R.; Karthikeyan, S.; Mandal, A.B.; Sekaran, G. Adsorption of ammonium ion by coconut shell-activated carbon from aqueous solution: Kinetic, isotherm, and thermodynamic studies. Environ. Sci. Pollut. Res. 2013, 20, 533–542. [Google Scholar] [CrossRef]
- Demiral, H.; Gündüzoğlu, G. Removal of nitrate from aqueous solutions by activated carbon prepared from sugar beet bagasse. Bioresour. Technol. 2010, 101, 1675–1680. [Google Scholar] [CrossRef]
- Hong, S.; Cannon, F.S.; Hou, P.; Byrne, T.; Nieto-Delgado, C. Sulfate removal from acid mine drainage using polypyrrole-grafted granular activated carbon. Carbon 2014, 73, 51–60. [Google Scholar] [CrossRef]
- Bolan, N.S.; Elliott, J.; Gregg, P.E.H.; Weil, S. Enhanced dissolution of phosphate rocks in the rhizosphere. Biol. Fertil. Soils 1997, 24, 169–174. [Google Scholar] [CrossRef]
- Lambers, H.; Clements, J.C.; Nelson, M.N. How a phosphorus-acquisition strategy based on carboxylate exudation powers the success and agronomic potential of lupines (Lupinus, Fabaceae). Am. J. Bot. 2013, 100, 263–288. [Google Scholar] [CrossRef] [PubMed]
- Seiter, K.; Hensen, C.; Schröter, J.; Zabel, M. Organic carbon content in surface sediments—Defining regional provinces. Deep Sea Res. Part Oceanogr. Res. Pap. 2004, 51, 2001–2026. [Google Scholar] [CrossRef]
- Reish, D.J. Annelid. Encycl. Br. 2013. Available online: https://www.nobanis.org/globalassets/speciesinfo/s/spartina-anglica/spartina_anglica.pdf (accessed on 1 July 2019).
- Cunha, L.; Brown, G.G.; Stanton, D.W.G.; Da Silva, E.; Hansel, F.A.; Jorge, G.; McKey, D.; Vidal-Torrado, P.; Macedo, R.S.; Velasquez, E.; et al. Soil Animals and Pedogenesis: The Role of Earthworms in Anthropogenic Soils. Soil Sci. 2016, 181, 110–125. [Google Scholar] [CrossRef]
- Deeke, A.; Sleutels, T.H.J.A.; Hamelers, H.V.M.; Buisman, C.J.N. Capacitive Bioanodes Enable Renewable Energy Storage in Microbial Fuel Cells. Environ. Sci. Technol. 2012, 46, 3554–3560. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Ji, M.; Zhai, H. Anodic potentials, electricity generation and bacterial community as affected by plant roots in sediment microbial fuel cell: Effects of anode locations. Chemosphere 2018, 209, 739–747. [Google Scholar] [CrossRef]
- Osorio de la Rosa, E.; Vázquez Castillo, J.; Carmona Campos, M.; Barbosa Pool, R.G.; Becerra Nuñez, G.; Castillo Atoche, A.; Ortegón Aguilar, J. Plant Microbial Fuel Cells–Based Energy Harvester System for Self-powered IoT Applications. Sensors 2019, 19, 1378. [Google Scholar] [CrossRef]
- Koop-Jakobsen, K.; Wenzhöfer, F. The Dynamics of Plant-Mediated Sediment Oxygenation in Spartina anglica Rhizospheres—A Planar Optode Study. Estuaries Coasts 2015, 38, 951–963. [Google Scholar] [CrossRef]
- Timmers, R.A.; Strik, D.P.B.T.B.; Arampatzoglou, C.; Buisman, C.J.N.; Hamelers, H.V.M. Rhizosphere anode model explains high oxygen levels during operation of a Glyceria maxima PMFC. Bioresour. Technol. 2012, 108, 60–67. [Google Scholar] [CrossRef] [PubMed]
- Arends, J.B.A.; Blondeel, E.; Tennison, S.R.; Boon, N.; Verstraete, W. Suitability of granular carbon as an anode material for sediment microbial fuel cells. J. Soils Sediments 2012, 12, 1197–1206. [Google Scholar] [CrossRef]
- Malvankar, N.S.; King, G.M.; Lovley, D.R. Centimeter-long electron transport in marine sediments via conductive minerals. ISME J. 2014, 9, 527–531. [Google Scholar] [CrossRef] [PubMed]
- Caizán-Juanarena, L.; Servin-Balderas, I.; Chen, X.; Buisman, C.J.N.; ter Heijne, A. Electrochemical and microbiological characterization of single carbon granules in a multi-anode microbial fuel cell. J. Power Sources 2019. [Google Scholar] [CrossRef]
- Fang, Z.; Cheng, S.; Wang, H.; Cao, X.; Li, X. Feasibility study of simultaneous azo dye decolorization and bioelectricity generation by microbial fuel cell-coupled constructed wetland: Substrate effects. RSC Adv. 2017, 7, 16542–16552. [Google Scholar] [CrossRef]
- Song, H.; Zhang, S.; Long, X.; Yang, X.; Li, H.; Xiang, W. Optimization of Bioelectricity Generation in Constructed Wetland-Coupled Microbial Fuel Cell Systems. Water 2017, 9, 185. [Google Scholar] [CrossRef]
- Liu, S.; Song, H.; Li, X.; Yang, F. Power Generation Enhancement by Utilizing Plant Photosynthate in Microbial Fuel Cell Coupled Constructed Wetland System. Int. J. Photoenergy 2013, 2013, 172010. [Google Scholar] [CrossRef]
- Tapia, N.F.; Rojas, C.; Bonilla, C.A.; Vargas, I.T. A New Method for Sensing Soil Water Content in Green Roofs Using Plant Microbial Fuel Cells. Sensors 2017, 18, 71. [Google Scholar] [CrossRef]
- Lasher, C.; Dyszynski, G.; Everett, K.; Edmonds, J.; Ye, W.; Sheldon, W.; Wang, S.; Joye, S.B.; Moran, M.A.; Whitman, W.B. The Diverse Bacterial Community in Intertidal, Anaerobic Sediments at Sapelo Island, Georgia. Microb. Ecol. 2009, 58, 244–261. [Google Scholar] [CrossRef]
- Wang, M.; Chen, J.-K.; Li, B. Characterization of Bacterial Community Structure and Diversity in Rhizosphere Soils of Three Plants in Rapidly Changing Salt Marshes Using 16S rDNA. Pedosphere 2007, 17, 545–556. [Google Scholar] [CrossRef]
- Wang, M.; Yang, P.; Falcão Salles, J. Distribution of Root-Associated Bacterial Communities along a Salt-Marsh Primary Succession. Front. Plant Sci. 2016, 6, 1188. [Google Scholar] [CrossRef] [PubMed]
- Leloup, J.; Fossing, H.; Kohls, K.; Holmkvist, L.; Borowski, C.; Jørgensen, B.B. Sulfate-reducing bacteria in marine sediment (Aarhus Bay, Denmark): Abundance and diversity related to geochemical zonation. Environ. Microbiol. 2009, 11, 1278–1291. [Google Scholar] [CrossRef] [PubMed]
- Burdorf, L.D.W.; Tramper, A.; Seitaj, D.; Meire, L.; Hidalgo-Martinez, S.; Zetsche, E.-M.; Boschker, H.T.S.; Meysman, F.J.R. Long-distance electron transport occurs globally in marine sediments. Biogeosciences 2017, 14, 683–701. [Google Scholar] [CrossRef]
- Larsen, S.; Nielsen, L.P.; Schramm, A. Cable bacteria associated with long-distance electron transport in New England salt marsh sediment. Environ. Microbiol. Rep. 2015, 7, 175–179. [Google Scholar] [CrossRef] [PubMed]
- Barton, L.L.; Fauque, G.D. Chapter 2 Biochemistry, Physiology and Biotechnology of Sulfate-Reducing Bacteria. In Advances in Applied Microbiology; Academic Press: Cambridge, MA, USA, 2009; Volume 68, pp. 41–98. ISBN 0065-2164. [Google Scholar]
- Thomas, F.; Giblin, A.E.; Cardon, Z.G.; Sievert, S.M. Rhizosphere heterogeneity shapes abundance and activity of sulfur-oxidizing bacteria in vegetated salt marsh sediments. Front. Microbiol. 2014, 5, 309. [Google Scholar] [CrossRef]






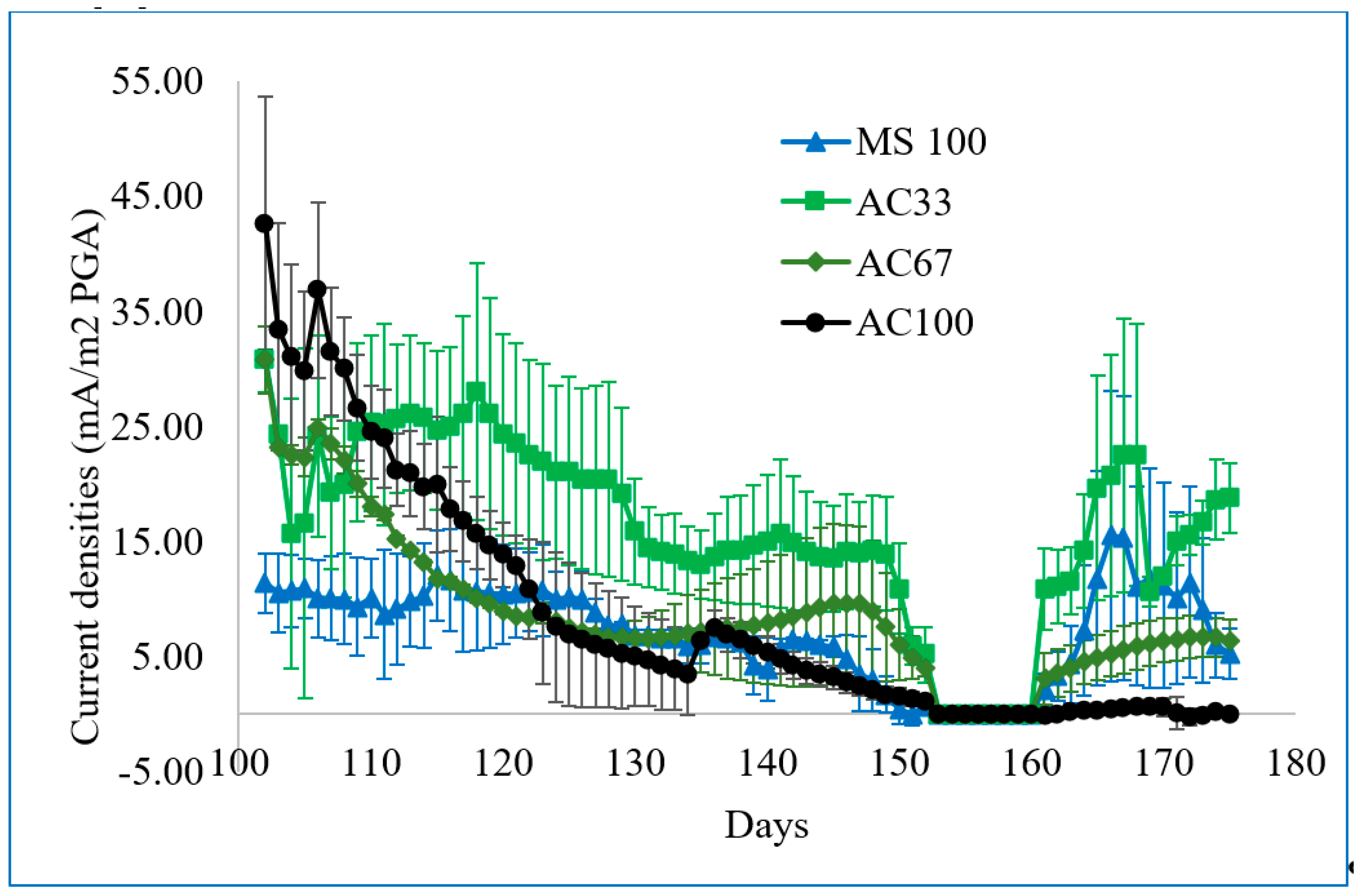

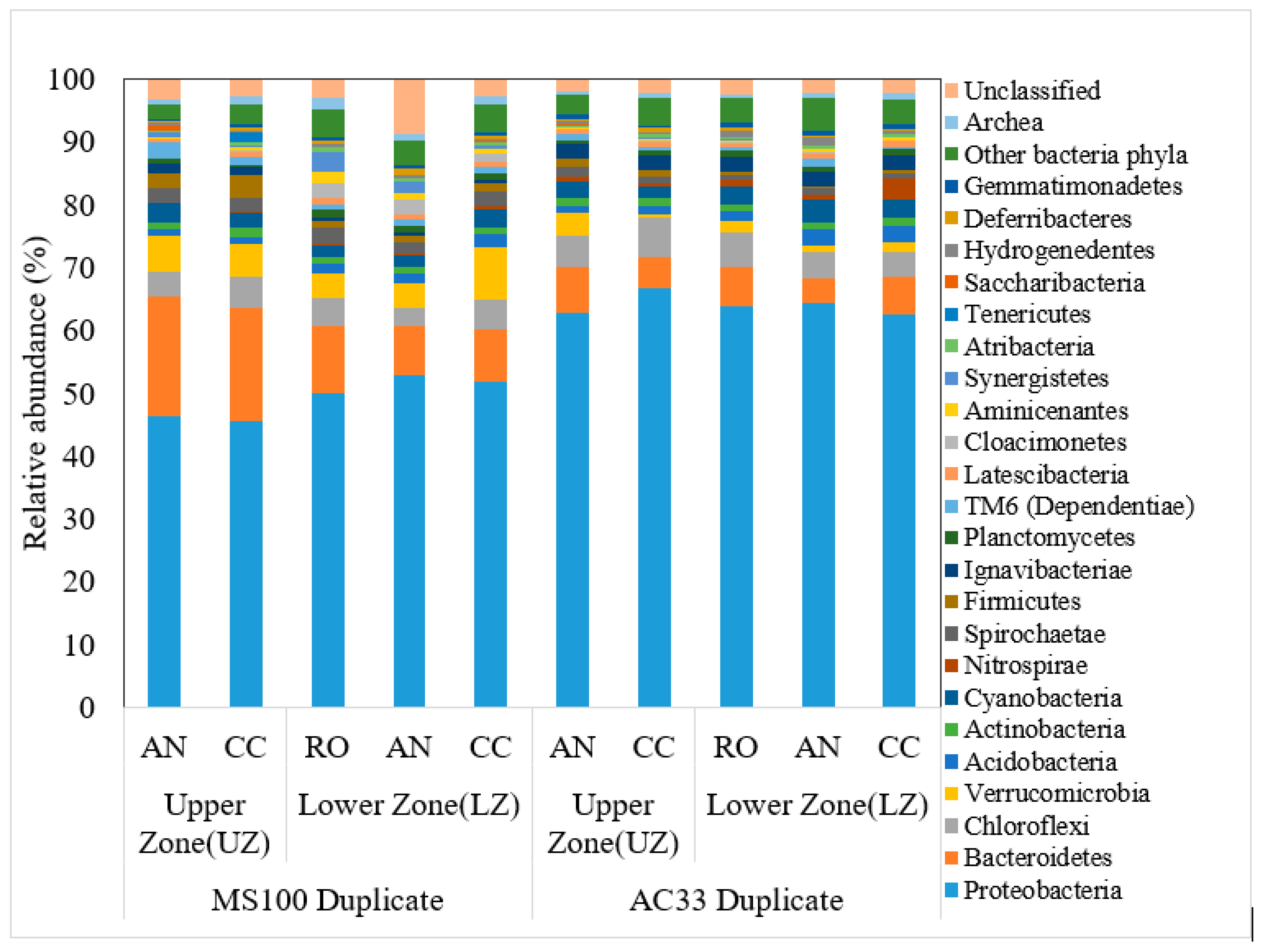
| Reactor Type/Plant Species | Anode/Current Collector | Cathode | Current Density (mA/m2 PGA) | Power Density (mW/m2 PGA) | Method | Ref. | ||
|---|---|---|---|---|---|---|---|---|
| Av. | Max | Av. | Max | |||||
| Flat plate Spartina anglica | Marine sediment/small graphite rod (MS100) | Graphite felt, air cathode | 9.01 | 15.49 | 0.37 | 0.91 | A | This study |
| (2 weeks) | 45 * | (2 weeks) | 8 * | |||||
| - | (0.3 A/m3) * | - | (46 mW/m3) * | D | ||||
| Flat plate Spartina anglica | 33% AC + 67% marine sediment/small graphite rod (AC33) | Graphite felt, air cathode | 16.01 | 22.53 | 1.04 | 1.93 | A | This study |
| (2 weeks) | 819 * | (2 weeks) | 148* | |||||
| - | (4.8 A/m3) * | - | (863 mW/m3) * | D | ||||
| Flat plate Spartina anglica | 67% AC + 33% marine sediment/small graphite rod (AC67) | Graphite felt, air cathode | 5.46 | 8.42 | 0.12 | 0.27 | A | This study |
| (2 weeks) | 12,496 * | (2 weeks) | 2249* | |||||
| - | (73 A/m3) * | - | (13,150 mW/m3) * | D | ||||
| Flat plate Spartina anglica | AC/small graphite rod (AC100) | Graphite felt, air cathode | 0.2 | 1.58 | 0.00 | 0.00 | A | This study |
| (2 weeks) | 19,752 * | (2 weeks) | 3555 * | |||||
| - | 11.5 A/m3 * | - | (20,786 mW/m3) * | D | ||||
| Flat-plate Spartina anglica | Graphite felt/gold wire | Graphite felt, Ferric cyanide cathode | 74–384 (4 weeks) | 469 | 47–155 (4 weeks) | 211 | A | [21] |
| Cylindrical Spartina anglica | Graphite grain/Graphite rod | Graphite felt/gold wire, Potassium ferric cyanide | - | - | 21 | - | A | [20] |
| (8 weeks) | ||||||||
| - | - | - | 222 | C | ||||
| Flat plate with two cathode compartments Spartina anglica | Graphite felt/golden wire | Graphite felt/golden wire, oxygen reducing biocathode | - | - | - | 679 | C | [28] |
| 240 | ||||||||
| - | - | (2 weeks) | - | B | ||||
| Polyacrylic plastic cylinder Ipomoea aquatica | Granular activated carbon/stainless steel mesh | Granular activated carbon/stainless steel mesh, air cathode | - | 0.66 A/m3 | - | 274 mW/m3 | C | [83] |
| Organic glass pipe Phragmites australis | Activated granular carbon/stainless steel mesh | Activated granular carbon, air cathode | - | 0.49 A/m3 | - | 4.5 (200 mW/m3) | C | [84] |
| Polycarbonate plastic cylinder Ipomoea aquatica | Thick granular activated carbon/titanium wire | Stainless steel mesh, air cathode | - | - | - | 12.42 | C | [85] |
| Glass cylinder Spartina anglica | Graphite granules/golden wire | Graphite felt, air cathode | - | - | - | 79 | C | [32] |
| Modular Sedum species | Carbon felt | AC/graphite rood, air cathode | - | 5 | - | 114.6 (μW/m2) | C | [86] |
| Perspex tubes Rice(Oryza sativa) | Graphite granule/vermiculite/carbon rod | Graphite felt interwoven carbon rod, air cathode | - | 580 | - | 72 | D | [38] |
| Circular graphite felt electrode in a rice paddy field Rice (Oryza sativa) | Graphite felt connected via epoxy encapsulated wires | Graphite felt with platinum catalyst connected via epoxy encapsulated wires, air cathode | - | - | - | 140 | D | [16] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sudirjo, E.; Buisman, C.J.N.; Strik, D.P.B.T.B. Activated Carbon Mixed with Marine Sediment is Suitable as Bioanode Material for Spartina anglica Sediment/Plant Microbial Fuel Cell: Plant Growth, Electricity Generation, and Spatial Microbial Community Diversity. Water 2019, 11, 1810. https://doi.org/10.3390/w11091810
Sudirjo E, Buisman CJN, Strik DPBTB. Activated Carbon Mixed with Marine Sediment is Suitable as Bioanode Material for Spartina anglica Sediment/Plant Microbial Fuel Cell: Plant Growth, Electricity Generation, and Spatial Microbial Community Diversity. Water. 2019; 11(9):1810. https://doi.org/10.3390/w11091810
Chicago/Turabian StyleSudirjo, Emilius, Cees J.N. Buisman, and David P.B.T.B. Strik. 2019. "Activated Carbon Mixed with Marine Sediment is Suitable as Bioanode Material for Spartina anglica Sediment/Plant Microbial Fuel Cell: Plant Growth, Electricity Generation, and Spatial Microbial Community Diversity" Water 11, no. 9: 1810. https://doi.org/10.3390/w11091810
APA StyleSudirjo, E., Buisman, C. J. N., & Strik, D. P. B. T. B. (2019). Activated Carbon Mixed with Marine Sediment is Suitable as Bioanode Material for Spartina anglica Sediment/Plant Microbial Fuel Cell: Plant Growth, Electricity Generation, and Spatial Microbial Community Diversity. Water, 11(9), 1810. https://doi.org/10.3390/w11091810





