1. Introduction
Millions of tons of waste rock, overburden, and beneficiation wastes are produced by the global mining industry. Due to their limited economic value and the remote location of most mining settings, over 95% of these materials are disposed of, forming enormous stockpiles in the mining area [
1,
2,
3,
4]. In mining companies, the cost of waste handling and storage can represents a financial loss around 1.5–3.5% of total costs [
5]. The transformation of mining wastes is promoted to pursue a zero-waste circular model economy by evaluating solutions for their re-use [
6]. The chemical composition and geotechnical properties of the source rock determine which uses are most appropriate and whether reuse is economically feasible. Possible second life pathways of solid mining wastes include the recovery of critical raw materials, the use as backfill materials for open voids, the extraction of valuable minerals and metals from low-grade resources, their application as landscaping materials and capping materials for waste repositories, substrates for mine revegetation, and civil engineering constructions [
1,
2,
7]. Among mining materials, Fe-, Mn- and Al-rich rock wastes could be recovered as end of life products and converted into adsorbents for water treatment.
In recent years, a range of inexpensive water clean-up technologies have been developed to address the major problem of arsenic contamination in water sources. The adsorption onto filtration units filled with Fe, Mn, and Al (hydr-)oxides phases represents the prominent technological treatment [
8,
9,
10,
11,
12]. Surface complexation accounts for the high selectivity of the adsorption of arsenic onto iron, aluminum, and manganese (hydr-)oxides [
13,
14,
15,
16]. Close to point of zero charge, arsenate adsorption through anion exchange could also occur [
17]. Iron hydroxide is usually considered to be a superior arsenic adsorbent when compared to aluminum and manganese (hydr-)oxides, due to its highest efficiency at natural pH range [
8,
10,
18]. A large body of the literature is focused on As adsorption studies based on synthetic minerals, such as hematite [
19,
20,
21], magnetite [
19,
20,
21,
22], goethite [
12,
21,
22], activated alumina [
23,
24], gibbsite [
16], kaolinite and other clays [
25,
26], zeolites, and modified zeolites [
27,
28,
29,
30]. Arsenic adsorption up to 50 mg/g adsorbent were reported, with enhanced adsorption capacity, relying on the homogeneity and activity of adsorption sites [
8]. Naturally occurring minerals are more attractive for arsenic water treatment due to their large availability and cost effectiveness. Unlike synthetic iron minerals, the naturally occurring iron ores contain a variety of mineral phases and other elements. Hence, final As adsorption is expected to be lower due to the reduced number of available and accessible sorption sites and interfering and competing ions.
Nevertheless, the need for effective, robust, and low-cost devices for widespread small-scale application (i.e., at the scale of an individual household) increased the interest in testing low-cost waste materials as arsenic adsorbents [
8]. Even if their adsorption capacity can be a few mgAs/g, their performance to treat As-rich waters could be satisfactory, especially if applied to drinking water treatment targeted to groundwaters with As concentration below 200 µg/L. Nguyen et al. [
31] used a purified and enriched magnetite waste from iron ore mine to treat arsenic-rich waters. This material showed arsenic maximum adsorption capacity of 0.74 mg/g. Zhang et al. [
32] tested waste rock from natural iron ores, with hematite and goethite as prevailing mineralogical phases, and maximum adsorption capacity by Langmuir was estimated to be 0.4 mgAs/g. A low-cost material (76% pyrolusite with <10% goethite and quartz) from ferruginous manganese ore efficiently removed As at pH 2–8 from six groundwaters with As concentration in the range 40–180 µg/L [
33]. Different tools for the characterization of mining wastes are based on either conventional methods, such as X-ray diffraction and scanning electron microscopy, or advanced approaches, such as synchrotron-based microanalysis and automated mineralogy [
34].
Previous studies on mining waste reuse for arsenic removal were based on a limited number of samples with homogeneous mineral distribution. However, since rock wastes in mining stockpiles are highly heterogeneous in terms of mineralogical and chemical composition, the correct identification and selection of suitable materials for the re-use in water treatments will require cross-disciplinary approaches, primarily based on field measurements and sampling site selection.
In this study, we explored the suitability of various mining rock wastes to realize water filters for As removal from contaminated waters. More specifically, we aimed (i) to evaluate if spectral information based on field measurements could help in discriminating materials with different As adsorption potential, (ii) to assess how and to what extent the mineralogical composition and element content of rock wastes can contribute to As removal processes, (iii) to elaborate a pre-screening statistical procedure to identify and select promising materials to be potentially reused in water reclamation practices.
4. Discussion
Mining activities, together with construction/demolition and manufacturing, contributed to nearly 74% of all wastes disposed of in the European Union [
39]. North America produces more than 10 times as much solid mine waste as municipal solid waste per capita [
34]. In India, more than 200 million tons of non-hazardous inorganic solid wastes are being generated every year, out of which about 80 million tons are mine tailings/ores [
40].
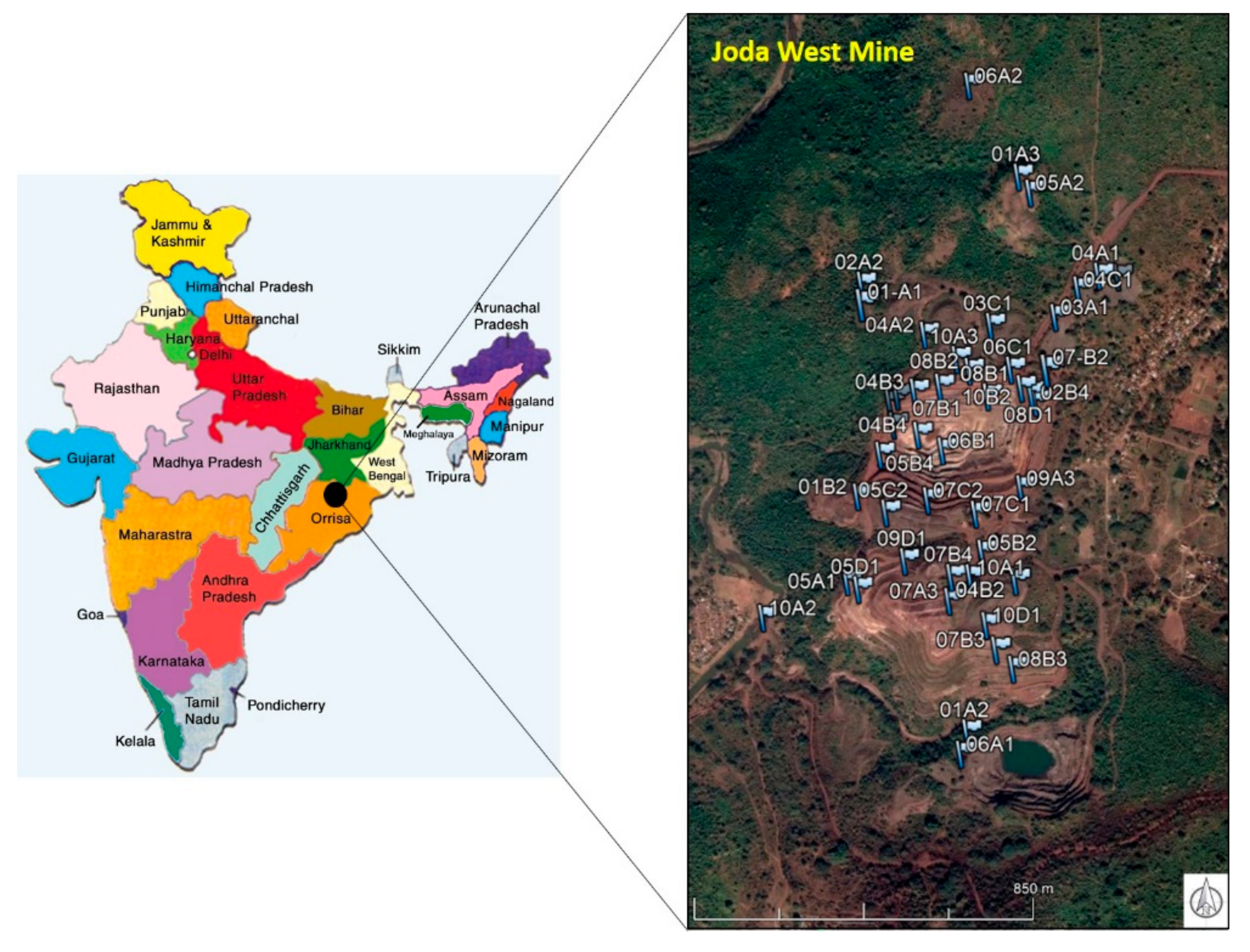
At Joda West mine, Fe, Mn and Al (hydr-)oxides, clay mineral, and quartz were the predominant mineral phases of collected rock wastes. The natural arsenic content reached up to 140 mg/kg, (higher than soil baseline concentration generally 5–10 mg/kg). In different mining environments (e.g., gold mines), arsenic content in rocks was reported in the order of g/kg, due to the presence of primary and secondary minerals [
41]. Among transition metals, Cr, Ni, Pb, and Zn showed higher concentrations than average values in soils and sediments reported by FOREGS European geochemical Atlas [
42] but much lower than their reported maximum values (
Table S3).
First exploratory adsorption tests allowed us to test and select materials that could be potentially interesting for the realization of filters for treating As-rich waters. In heterogeneous natural iron oxides, adsorption of 0.3–0.5 mg/g of arsenate were commonly found [
12,
19,
20]. Some of the tested samples showed a satisfactory As adsorption capacity (>0.25 mgAs/g at Ci = 1 mgAs/L). At this concentration, treated magnetite waste from iron ore showed adsorption below 0.2 mg/g [
31]. Chakravarty et al. [
33] tested a ferruginous manganese ore material, mainly constituted by pyrolusite and goethite, and concentration in condition similar to this study resulted in 0.2 mgAs/g adsorption according to Langmuir As(V) equation. The As adsorption capacity of three hematite-rich iron ore samples was 0.17–0.48 mgAs/g [
32]. The presence of quartzite and less reactive clays at high crystallinity (e.g., muscovite) were the major limiting factors for adsorption.
Since the reflectance spectrum of rocks depends on their mineralogical composition [
43,
44], we noted that the reflectance in red band range, measured by hyperspectral field radiometer on undisturbed rock wastes, provided valuable indications for the on-site pre-selection of materials with lower As removal potential. The rock wastes with a limited availability of adsorption sites could be discarded, with no need to carry out further measurements. A variety of reflectance spectroscopy-based applications, relying on the spectral signatures of minerals able to bind/sorb metal(loids), have been developed to promote indirect detection and avoid expensive laboratory measurements. Pallottino et al. [
45] realized a predictive model for As contamination in calcareous soil surrounding thermal springs based on the diffuse VIS-NIR spectral reflectance. In that study, As content was largely associated (>46%) with the sole CaCO
3 phase. According to our outcomes, a first pre-screening step could be used to exclude spotted materials containing high amounts of quartzite and muscovite, but the presence of dark-red minerals (amorphous and crystalline) cannot be directly discriminated through the spectral signals in the red band, given the observed mineralogical complexity and heterogeneity of As adsorption phases. Appropriate spectral information should be collected by better refining band selection in order to exclude the less adsorptive materials (i.e., quartz and muscovite) and to identify good adsorbents (i.e., kaolinite).
The presence of iron minerals (goethite and hematite), together with Al-rich kaolinite, contributed the most to As adsorption. At natural pH range of 6–8, the adsorption onto iron (hydr-)oxides is the most competitive, since Fe-based materials have a favorable surface charge (pHpzc 7-9) for oxyanion adsorption, while Mn oxides are mostly negatively charged (pHpzc 2–3). Adsorption of aluminum (hydr-)oxides is known to be maximum at pH 4–5 [
10]. On the contrary, Fe-Al binary oxides showed to be attractive adsorbents for both As(V) and As(III) removal from contaminated waters [
46]. The ability of Mn dioxides to sorb As(III) and As(V) appeared to be related also to materials with highly ordered pyrolusite having low specific surface (7.9 m
2/g). Conversely, poorly crystalline birnessite has higher specific surface area of 27.7 m
2/g [
47].
The presence of Mn minerals, either amorphous or crystalline, lowered As adsorption. Arsenic adsorption onto hydroxides was correlated to Fe and amorphous phases, characterized by edge structures more efficient in hosting arsenate ions than crystalline minerals. For example, the transformation of amorphous FeOOH to crystalline FeOOH would reduce sorption sites and surface area, thus lowering the number of ions that can be adsorbed [
15,
41,
48]. We found that amorphous fraction of Fe and Al were important in promoting As adsorption. Pedersen et al. [
49] observed a decrease in adsorbed arsenic clearly correlated with the transformation of ferrihydrite and lepidocrocite into more crystalline phases as goethite and hematite. Pigna et al. [
50] showed better As removal capacity of non-crystalline Al(OH)x than gibbsite. Fine-grained and poorly crystalline Mn oxides showed good adsorption properties, even if in Mn-ores pyrolusite (most stable and abundant) and birnessite minerals are often encountered [
51]. Fe, Al, and Mn minerals with medium grade crystallinity are responsible for As adsorption, since crystallization process kinetic and environmental conditions might induce defects in crystalline structures, which are suitable as adsorption sites.
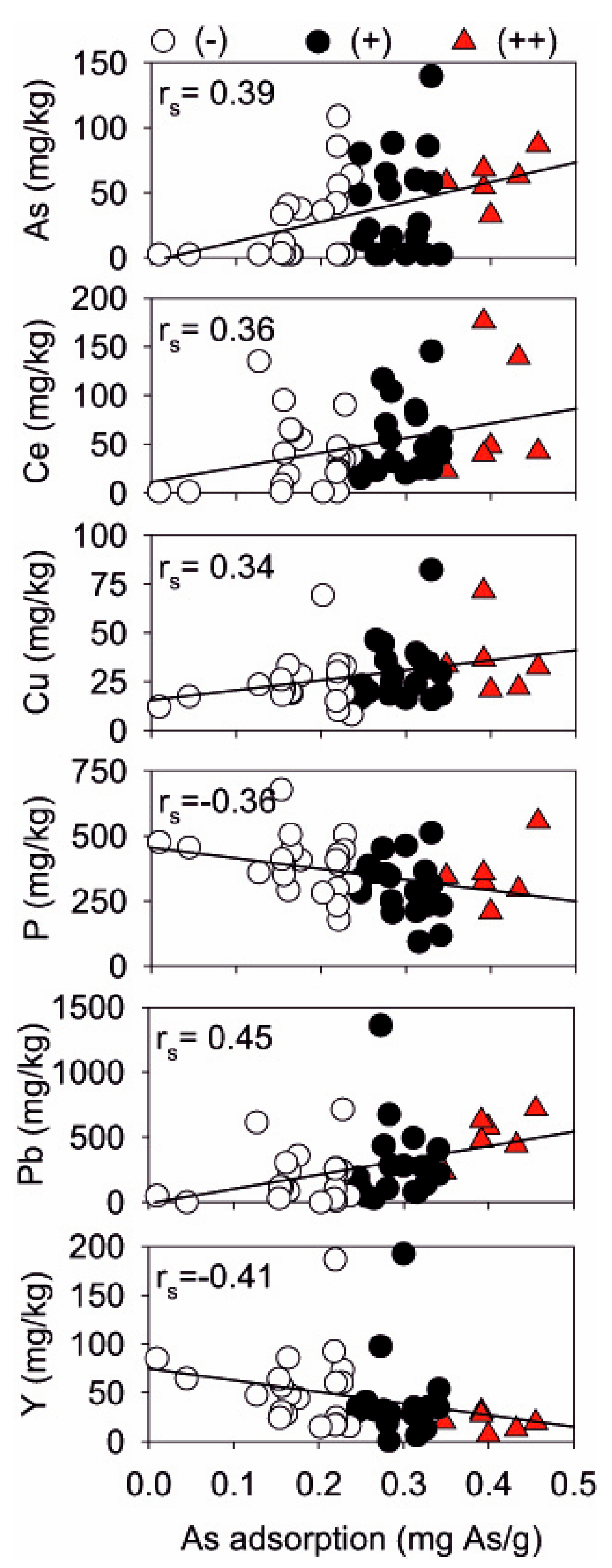
In line with literature reports, the correlation of As adsorption with the presence of rare earth elements (Ce and La) and transition metals (Pb, Cu) suggested that their variation mostly explained changes in As adsorption capacity [
10,
52,
53,
54,
55]. These elements could be incorporated in mineral structures or adsorbed on specific sites. The adsorption of heavy metals onto clay and oxides surface might cause a pHpzc shift towards higher values, thus rendering surfaces more positive at higher pH and promoting adsorption of oxyanions [
8,
56,
57,
58]. Fe (hydr-)oxides structure may incorporate metals cations and adsorb As-ions more effectively, due to a better matching of ion size and orientation, also by shortening the atom-to-atom distances between adsorbent and adsorbate [
59]. Mohapatra et al. [
60] modified goethite surface by doping Cu(II), Ni(II), or Co(II) to enhance arsenate uptake capacities. Lu et al. [
61] observed that the presence of Pb during the process of ferrihydrite transformation to hematite induced the formation of nanoparticles with a loose and porous structure in comparison with the compact structure of pure hematite nanoparticles.
The presence of bivalent cations (namely, Ni(II), Co(II), Mg(II)) were reported to enhance As adsorption capacity [
33]. Natural and modified enriched clays with exchangeable cations and anions have been widely tested as adsorbents for water treatment [
62,
63,
64,
65]. In pillared or intercalated clays with transition positively charged metals, clay sheets increase each other’s distances. Adsorption increased due to change in surface area and charge, with positively charged surface enhancing penetrability of As oxyanions [
66]. Na et al. [
67] demonstrated that Ti-pillared montmorillonite was an efficient material for the removal of arsenate and arsenite from aqueous solutions. Doušová et al. [
68] proved that pre-treatment of low-grade clay materials Fe (Al, Mn) salts can significantly improve their sorption affinity to As oxyanions. A simplified multiple linear model was proposed, based on XRF measurements, as a predictive tool to guide mining wastes selection to realize removal filters for As contaminated water. As predicted = 0.241 + 0.00929[As] + 0.000424[La] + 0.000139[Pb] − 0.00022[P]. This model applicability is site-specific and strictly related to the mineralogical, geological and chemical context encountered at JWIMM. The natural presence of arsenic in the sampled materials was one of the best predictors for As adsorption, thus indicating that rock wastes kept their original and natural As-adsorbing affinity. On the contrary, higher concentrations of Y and Rb were found at low As adsorption levels only. These two elements are found in association with phosphate rocks [
69,
70]. Due to chemical similarity, phosphate is also known to be a competitor for the As adsorption sites and, together with silicate, a major interfering ion for As removal processes onto oxides [
71,
72,
73,
74]. Furthermore, waste materials active for arsenate could be also successfully tested for phosphate removal to reduce P-load from surface water and promote its recovery as critical raw material [
75,
76].
Overall, due to the elevated presence of iron oxides and the co-occurring kaolinite in most suitable samples, the factors that turned out to be more significant to differentiate potential adsorption capacity were elements adsorbed onto major phases, which were able to positively modify surrounding adsorption site structure. The possible release of As together with other potentially toxic metals from reused rock wastes should be consciously investigated in the long term to promote safer applications, especially if intended for human consumption purposes.