Systematic Design, Optimization, and Sustainability Assessment for Generation of Efficient Wastewater Treatment Networks
Abstract
:1. Introduction
1.1. Stages and Categories in WW Treatment
1.2. WW Treatment Technologies
1.2.1. Pretreatment Technologies
1.2.2. Primary Treatment Technologies
1.2.3. Secondary Treatment Technologies
1.2.4. Tertiary Treatment Technologies
1.3. WW Contaminants and Their Classification
1.4. WW Treatment Using Superstructure and Optimization Approach
1.5. Sustainability Assessment
2. Methodology
2.1. Model Building
2.2. Generation of Superstructure and Optimization Technique
2.3. Sustainability Assessment Using the Sustainable Process Index (SPI) Methodology
2.4. Framework for Optimal Design Evaluation
3. Results and Discussions
3.1. Municipal (Regional) Case Study
3.2. Pharmaceutical WW Case Study
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Availability of Code
References
- Crini, G.; Lichtfouse, E. Advantages and disadvantages of techniques used for wastewater treatment. Environ. Chem. Lett. 2019, 17, 145–155. [Google Scholar]
- Khor, C.S.; Chachuat, B.; Shah, N. A superstructure optimization approach for water network synthesis with membrane separation-based regenerators. Comput. Chem. Eng. 2012, 42, 48–63. [Google Scholar]
- Khor, C.S.; Foo, D.C.Y.; El-Halwagi, M.M.; Tan, R.R.; Shah, N. A Superstructure Optimization Approach for Membrane Separation-Based Water Regeneration Network Synthesis with Detailed Nonlinear Mechanistic Reverse Osmosis Model. Ind. Eng. Chem. Res. 2011, 50, 13444–13456. [Google Scholar]
- Lu, B.; Huang, S.; Grossmann, I.E. Optimal Synthesis and Operation of Wastewater Treatment Process with Dynamic Influent. Ind. Eng. Chem. Res. 2017, 56, 8663–8676. [Google Scholar]
- Zhang, W.; Wang, C.; Li, Y.; Wang, P.; Wang, Q.; Wang, D. Seeking Sustainability: Multiobjective Evolutionary Optimization for Urban Wastewater Reuse in China. Environ. Sci. Technol. 2014, 48, 1094–1102. [Google Scholar] [PubMed]
- Marcelino, R.B.P.; Leão, M.M.D.; Lago, R.M.; Amorim, C.C. Multistage ozone and biological treatment system for real wastewater containing antibiotics. J. Environ. Manag. 2017, 195, 110–116. [Google Scholar]
- Arroyo, P.; Molinos-Senante, M. Selecting appropriate wastewater treatment technologies using a choosing-by-advantages approach. Sci. Total Environ. 2018, 625, 819–827. [Google Scholar]
- Yenkie, K. Integrating the three E’s in wastewater treatment: Efficient design, economic viability, and environmental sustainability. Curr. Opin. Chem. Eng. 2019, 9, 131–138. [Google Scholar]
- Galan, B.; Grossmann, I.E. Optimal Design of Distributed Wastewater Treatment Networks. Ind. Eng. Chem. Res. 1998, 37, 4036–4048. [Google Scholar]
- Yang, Y.H.; Lou, H.H.; Huang, Y.L. Synthesis of an optimal wastewater reuse network. Waste Manag. 2000, 20, 311–319. [Google Scholar]
- Rigopoulos, S.; Linke, P. Systematic development of optimal activated sludge process designs. Comput. Chem. Eng. 2002, 26, 585–597. [Google Scholar]
- Vidal, N.; Bañares-Alcántara, R.; Rodríguez-Roda, I.; Poch, M. Design of Wastewater Treatment Plants Using a Conceptual Design Methodology. Ind. Eng. Chem. Res. 2002, 41, 4993–5005. [Google Scholar]
- Zeng, G.; Jiang, R.; Huang, G.; Xu, M.; Li, J. Optimization of wastewater treatment alternative selection by hierarchy grey relational analysis. J. Environ. Manag. 2007, 82, 250–259. [Google Scholar]
- Bozkurt, H.; Quaglia, A.; Gernaey, K.V.; Sin, G. A mathematical programming framework for early stage design of wastewater treatment plants. Environ. Model. Softw. 2015, 64, 164–176. [Google Scholar]
- Ku-Pineda, V.; Tan, R.R. Environmental performance optimization using process water integration and Sustainable Process Index. J. Clean. Prod. 2006, 14, 1586–1592. [Google Scholar]
- NPDES; USEPA. Primer for Municipal Wastewater Treatment Systems; EPA: Washington, DC, USA, 2004; p. 30.
- Tchobanoglous, G.; Burton, F.L.; Stensel, H.D. Wastewater Engineering: Treatment and Reuse, 4th ed.; McGraw Hill Higher Education: Boston, MA, USA, 2002; ISBN 978-0-07-124140-3. [Google Scholar]
- Davis, M. Water and Wastewater Engineering; McGraw-Hill Education: Boston, MA, USA, 2010; ISBN 978-0-07-339786-3. [Google Scholar]
- Primer for Municipal Wastewater Treatment Systems. Available online: https://www3.epa.gov/npdes/pubs/primer.pdf (accessed on 5 February 2021).
- Environmental Engineering. Water, Wastewater, Soil, and Groundwater Treatment and Remediation, 6th ed.; Nemerow, N.L., Ed.; Wiley: Hoboken, NJ, USA, 2009; ISBN 978-0-470-08303-1. [Google Scholar]
- Shammas, N.K. Water Engineering; Wiley: Hoboken, NJ, USA, 2016; p. 829. [Google Scholar]
- Lee, C.S.; Robinson, J.; Chong, M.F. A review on application of flocculants in wastewater treatment. Process. Saf. Environ. Prot. 2014, 92, 489–508. [Google Scholar]
- Ødegaard, H. Optimization of flocculation/flotation in chemical wastewater treatment. Water Sci. Technol. 1995, 31, 73–82. [Google Scholar]
- De Oliveira, D.S.; Donadel, C.B. Mathematical modelling and analysis of the flocculation process in low retention time hydraulic flocculators. Water SA 2019, 45, 1–11. [Google Scholar]
- Moshiri, G.A. Constructed Wetlands for Water Quality Improvement; CRC Press: Boca Raton, FL, USA, 1993; ISBN 978-0-87371-550-8. [Google Scholar]
- Singh, R. Water and Membrane Treatment. In Membrane Technology and Engineering for Water Purification; Elsevier: Amsterdam, The Netherlands, 2015; pp. 81–178. ISBN 978-0-444-63362-0. [Google Scholar]
- Adin, A. Slow granular filtration for water reuse. Water Sci. Technol. Water Supply 2003, 3, 123–130. [Google Scholar]
- Hamoda, M.F.; Al-Ghusain, I.; Al-Jasem, D.M. Application of Granular Media Filtration in Wastewater Reclamation and Reuse. J. Environ. Sci. Health Part A 2004, 39, 385–395. [Google Scholar]
- Ho, L.; Grasset, C.; Hoefel, D.; Dixon, M.B.; Leusch, F.D.L.; Newcombe, G.; Saint, C.P.; Brookes, J.D. Assessing granular media filtration for the removal of chemical contaminants from wastewater. Water Res. 2011, 45, 3461–3472. [Google Scholar] [PubMed]
- Boutilier, L.; Jamieson, R.; Gordon, R.; Lake, C.; Hart, W. Adsorption, sedimentation, and inactivation of E. coli within wastewater treatment wetlands. Water Res. 2009, 43, 4370–4380. [Google Scholar] [CrossRef] [PubMed]
- Acher, A.; Fischer, E.; Turnheim, R.; Manor, Y. Ecologically friendly wastewater disinfection techniques. Water Res. 1997, 31, 1398–1404. [Google Scholar]
- De Sanctis, M.; Del Moro, G.; Levantesi, C.; Luprano, M.L.; Di Iaconi, C. Integration of an innovative biological treatment with physical or chemical disinfection for wastewater reuse. Sci. Total Environ. 2016, 543, 206–213. [Google Scholar]
- Von Sonntag, C. Disinfection with UV-Radiation. In Process Technologies for Water Treatment; Springer: Boston, MA, USA, 1988; pp. 159–177. ISBN 978-1-4684-8558-5. [Google Scholar]
- Blume, T. Improved wastewater disinfection by ultrasonic pre-treatment. Ultrason. Sonochem. 2004, 11, 333–336. [Google Scholar]
- Kitis, M. Disinfection of wastewater with peracetic acid: A review. Environ. Int. 2004, 30, 47–55. [Google Scholar] [PubMed]
- Dodd, M.C. Potential impacts of disinfection processes on elimination and deactivation of antibiotic resistance genes during water and wastewater treatment. J. Environ. Monit. 2012, 14, 1754–1771. [Google Scholar] [PubMed]
- Hassard, F.; Biddle, J.; Cartmell, E.; Jefferson, B.; Tyrrel, S.; Stephenson, T. Rotating biological contactors for wastewater treatment—A review. Process. Saf. Environ. Prot. 2015, 94, 285–306. [Google Scholar]
- Lipták, B.G.; Liu, D.H.F. Wastewater Treatment; Lewis Pub: London, UK, 2000; ISBN 978-1-56670-515-8. [Google Scholar]
- Pankhania, M.; Stephenson, T.; Semmens, M.J. Hollow fibre bioreactor for wastewater treatment using bubbleless membrane aeration. Water Res. 1994, 28, 2233–2236. [Google Scholar]
- Chang, C.-Y.; Chang, J.-S.; Vigneswaran, S.; Kandasamy, J. Pharmaceutical wastewater treatment by membrane bioreactor process—A case study in southern Taiwan. Desalination 2008, 234, 393–401. [Google Scholar]
- Radjenovic, J.; Matošić, M.; Mijatović, I.; Petrovic, M.; Barcel, D.; Radetic, M. Erratum to Membrane Bioreactor (MBR) as an Advanced Wastewater Treatment Technology; Springer: Berlin/Heidelberg, Germany, 2008; Volume 5, pp. 37–101. [Google Scholar] [CrossRef]
- Melin, T.; Jefferson, B.; Bixio, D.; Thoeye, C.; De Wilde, W.; De Koning, J.; van der Graaf, J.; Wintgens, T. Membrane bioreactor technology for wastewater treatment and reuse. Desalination 2006, 187, 271–282. [Google Scholar]
- Kivaisi, A.K. The potential for constructed wetlands for wastewater treatment and reuse in developing countries: A review. Ecol. Eng. 2001, 16, 545–560. [Google Scholar] [CrossRef]
- Saggaï, M.M.; Ainouche, A.; Nelson, M.; Cattin, F.; El Amrani, A. Long-term investigation of constructed wetland wastewater treatment and reuse: Selection of adapted plant species for metaremediation. J. Environ. Manag. 2017, 201, 120–128. [Google Scholar] [CrossRef]
- Zhang, L.; Lv, T.; Zhang, Y.; Stein, O.R.; Arias, C.A.; Brix, H.; Carvalho, P.N. Effects of constructed wetland design on ibuprofen removal—A mesocosm scale study. Sci. Total Environ. 2017, 609, 38–45. [Google Scholar] [PubMed]
- Verhoeven, J.T.A.; Meuleman, A.F.M. Wetlands for wastewater treatment: Opportunities and limitations. Ecol. Eng. 1999, 12, 5–12. [Google Scholar]
- Ismail, Z.Z.; Habeeb, A.A. Experimental and modeling study of simultaneous power generation and pharmaceutical wastewater treatment in microbial fuel cell based on mobilized biofilm bearers. Renew. Energy 2017, 101, 1256–1265. [Google Scholar] [CrossRef]
- Ortiz-Martínez, V.M.; Salar-García, M.J.; de los Ríos, A.P.; Hernández-Fernández, F.J.; Egea, J.A.; Lozano, L.J. Developments in microbial fuel cell modeling. Chem. Eng. J. 2015, 271, 50–60. [Google Scholar] [CrossRef]
- Bose, D.; Gopinath, M.; Vijay, P. Sustainable power generation from wastewater sources using Microbial Fuel Cell: Sustainable Power generation from Wastewater sources using Microbial fuel cell. Biofuels Bioprod. Bioref. 2018, 12, 559–576. [Google Scholar]
- Rajasulochana, P.; Preethy, V. Comparison on efficiency of various techniques in treatment of waste and sewage water—A comprehensive review. Resour. Effic. Technol. 2016, 2, 175–184. [Google Scholar] [CrossRef] [Green Version]
- Durai, G.; Rajasimman, M. Biological Treatment of Tannery Wastewater—A Review. J. Environ. Sci. Technol. 2010, 4, 1–17. [Google Scholar]
- Ju, F.; Zhang, T. Bacterial assembly and temporal dynamics in activated sludge of a full-scale municipal wastewater treatment plant. ISME J. 2015, 9, 683–695. [Google Scholar] [PubMed]
- Shi, X.; Leong, K.Y.; Ng, H.Y. Anaerobic treatment of pharmaceutical wastewater: A critical review. Bioresour. Technol. 2017, 245, 1238–1244. [Google Scholar] [PubMed]
- Deshpande, B.D.; Agrawal, P.S.; Yenkie, M.K.N. Advanced Oxidative Degradation of Benzoic Acid and 4-Nitro Benzoic Acid–A Comparative Study; AIP Publishing LLC: Bahal, India, 2019; p. 210003. [Google Scholar] [CrossRef]
- Deng, Y.; Zhao, R. Advanced Oxidation Processes (AOPs) in Wastewater Treatment. Curr. Pollut. Rep. 2015, 1, 167–176. [Google Scholar]
- Kale, A.; Yenkie, M.K.N. A Comparative Study of Efficiency of Different AOPs for Degradation of 4-nitrophenol in Batch and Continuous Systems. J. Chem. Cheml. Sci. 2016, 9, 397–405. [Google Scholar]
- Follansbee, D.M.; Paccione, J.D.; Martin, L.L. Globally Optimal Design and Operation of a Continuous Photocatalytic Advanced Oxidation Process Featuring Moving Bed Adsorption and Draft-Tube Transport. Ind. Eng. Chem. Res. 2008, 47, 3591–3600. [Google Scholar]
- Andreozzi, R. Advanced oxidation processes (AOP) for water purification and recovery. Catal. Today 1999, 53, 51–59. [Google Scholar]
- Slater, C.S.; Uchrin, C.G.; Ahlert, R.C. Ultrafiltration processes for the characterization and separation of landfill leachates. J. Environ. Sci. Health Part A Environ. Sci. Eng. 1985, 20, 97–111. [Google Scholar]
- Ang, W.L.; Mohammad, A.W.; Hilal, N.; Leo, C.P. A review on the applicability of integrated/hybrid membrane processes in water treatment and desalination plants. Desalination 2015, 363, 2–18. [Google Scholar]
- Juang, L.-C.; Tseng, D.-H.; Lin, H.-Y. Membrane processes for water reuse from the effluent of industrial park wastewater treatment plant: A study on flux and fouling of membrane. Desalination 2007, 202, 302–309. [Google Scholar]
- Abdulgader, H.A.; Kochkodan, V.; Hilal, N. Hybrid ion exchange—Pressure driven membrane processes in water treatment: A review. Sep. Purif. Technol. 2013, 116, 253–264. [Google Scholar]
- Ashrafi, O.; Yerushalmi, L.; Haghighat, F. Wastewater treatment in the pulp-and-paper industry: A review of treatment processes and the associated greenhouse gas emission. J. Environ. Manag. 2015, 158, 146–157. [Google Scholar]
- Shawwa, A.R.; Smith, D.W.; Sego, D.C. Color and chlorinated organics removal from pulp mills wastewater using activated petroleum coke. Water Res. 2001, 35, 745–749. [Google Scholar] [PubMed]
- Bhojwani, S.; Topolski, K.; Mukherjee, R.; Sengupta, D.; El-Halwagi, M.M. Technology review and data analysis for cost assessment of water treatment systems. Sci. Total Environ. 2019, 651, 2749–2761. [Google Scholar]
- Kollmann, R.; Neugebauer, G.; Kretschmer, F.; Truger, B.; Kindermann, H.; Stoeglehner, G.; Ertl, T.; Narodoslawsky, M. Renewable energy from wastewater—Practical aspects of integrating a wastewater treatment plant into local energy supply concepts. J. Clean. Prod. 2017, 155, 119–129. [Google Scholar]
- Kulik, N.; Trapido, M.; Goi, A.; Veressinina, Y.; Munter, R. Combined chemical treatment of pharmaceutical effluents from medical ointment production. Chemosphere 2008, 70, 1525–1531. [Google Scholar] [PubMed]
- Yenkie, K.M.; Wu, W.; Clark, R.L.; Pfleger, B.F.; Root, T.W.; Maravelias, C.T. A roadmap for the synthesis of separation networks for the recovery of bio-based chemicals: Matching biological and process feasibility. Biotechnol. Adv. 2016, 34, 1362–1383. [Google Scholar]
- Yenkie, K.M.; Wu, W.; Maravelias, C.T. Synthesis and analysis of separation networks for the recovery of intracellular chemicals generated from microbial-based conversions. Biotechnol. Biofuels 2017, 10, 119. [Google Scholar]
- Herna, R. Superstructure Decomposition and Parametric Optimization Approach for the Synthesis of Distributed Wastewater Treatment Networks. Ind. Eng. Chem. Res. 2004, 43, 2175–2191. [Google Scholar]
- Chea, J.D.; Lehr, A.L.; Stengel, J.P.; Savelski, M.J.; Slater, C.S.; Yenkie, K.M. Evaluation of Solvent Recovery Options for Economic Feasibility through a Superstructure-Based Optimization Framework. Ind. Eng. Chem. Res. 2020, 59, 5931–5944. [Google Scholar]
- Ahmetović, E.; Grossmann, I.E. Global superstructure optimization for the design of integrated process water networks. AIChE J. 2011, 57, 434–457. [Google Scholar]
- Garibay-Rodriguez, J.; Rico-Ramirez, V.; Ponce-Ortega, J.M. Mixed Integer Nonlinear Programming Model for Sustainable Water Management in Macroscopic Systems: Integrating Optimal Resource Management to the Synthesis of Distributed Treatment Systems. ACS Sustain. Chem. Eng. 2017, 5, 2129–2145. [Google Scholar]
- Roeleveld, P.J.; Klapwijk, A.; Eggels, P.G.; Rulkens, W.H. Sustainability of municipal waste water treatment. Water Sci. Technol. 1997, 35, 221–228. [Google Scholar]
- Muga, H.E.; Mihelcic, J.R. Sustainability of wastewater treatment technologies. J. Environ. Manag. 2008, 88, 437–447. [Google Scholar]
- Zagklis, D.P.; Arvaniti, E.C.; Papadakis, V.G.; Paraskeva, C.A. Sustainability analysis and benchmarking of olive mill wastewater treatment methods: Sustainability analysis and benchmarking of OMWW treatment methods. J. Chem. Technol. Biotechnol. 2013, 88, 742–750. [Google Scholar]
- Foley, J.; de Haas, D.; Hartley, K.; Lant, P. Comprehensive life cycle inventories of alternative wastewater treatment systems. Water Res. 2010, 44, 1654–1666. [Google Scholar]
- Corominas, L.; Foley, J.; Guest, J.S.; Hospido, A.; Larsen, H.F.; Morera, S.; Shaw, A. Life cycle assessment applied to wastewater treatment: State of the art. Water Res. 2013, 47, 5480–5492. [Google Scholar]
- Narodoslawsky, M.; Krotscheck, C. The sustainable process index (SPI): Evaluating processes according to environmental compatibility. J. Hazard. Mater. 1995, 41, 383–397. [Google Scholar]
- Krotscheck, C.; Narodoslawsky, M. The Sustainable Process Index a new dimension in ecological evaluation. Ecol. Eng. 1996, 6, 241–258. [Google Scholar]
- Shahzad, K.; Kollmann, R.; Maier, S.; Narodoslawsky, M. SPIonWEB—Ecological Process Evaluation with the Sustainable Process Index (SPI). In Computer Aided Chemical Engineering; Elsevier: Amsterdam, The Netherlands, 2014; Volume 33, pp. 487–492. ISBN 978-0-444-63434-4. [Google Scholar]
- Singh, R.K.; Murty, H.R.; Gupta, S.K.; Dikshit, A.K. An overview of sustainability assessment methodologies. Ecol. Indic. 2009, 9, 189–212. [Google Scholar]
- Deng, C.; Jiang, W.; Zhou, W.; Feng, X. New superstructure-based optimization of property-based industrial water system. J. Clean. Prod. 2018, 189, 878–886. [Google Scholar]
- Bertran, M.-O.; Frauzem, R.; Sanchez-Arcilla, A.-S.; Zhang, L.; Woodley, J.M.; Gani, R. A generic methodology for processing route synthesis and design based on superstructure optimization. Comput. Chem. Eng. 2017, 106, 892–910. [Google Scholar]
- Kılınç, M.R.; Sahinidis, N.V. Exploiting integrality in the global optimization of mixed-integer nonlinear programming problems with BARON. Optim. Methods Softw. 2018, 33, 540–562. [Google Scholar]
- Climate at a Glance | National Centers for Environmental Information (NCEI). Available online: https://www.ncdc.noaa.gov/cag/national/time-series/110/pcp/ann/1/2009-2019?base_prd=true&begbaseyear=2009&endbaseyear=2019 (accessed on 23 June 2020).
- Gadipelly, C.; Pérez-González, A.; Yadav, G.D.; Ortiz, I.; Ibáñez, R.; Rathod, V.K.; Marathe, K.V. Pharmaceutical Industry Wastewater: Review of the Technologies for Water Treatment and Reuse. Ind. Eng. Chem. Res. 2014, 53, 11571–11592. [Google Scholar]
- Deblonde, T.; Cossu-Leguille, C.; Hartemann, P. Emerging pollutants in wastewater: A review of the literature. Int. J. Hyg. Environ. Health 2011, 214, 442–448. [Google Scholar] [PubMed]
- Shariati, F.P.; Mehrnia, M.R.; Salmasi, B.M.; Heran, M.; Wisniewski, C.; Sarrafzadeh, M.H. Membrane bioreactor for treatment of pharmaceutical wastewater containing acetaminophen. Desalination 2010, 250, 798–800. [Google Scholar]






| Contaminant | Concentration | Units |
|---|---|---|
| Acids/Chlorides | 5 | mg/L |
| COD | 68–272 | mg/L |
| BOD | 100–400 | mg/L |
| Settable Solids | 250–450 | mg/L |
| Lead | 30–80 | mg/L |
| Zinc | 1 | mg/L |
| Nickel | 0.04 | mg/L |
| Copper | 40–100 | mg/L |
| Specialized Chemicals | <0.5 | µg/L |
| Contaminants | Inlet Concentrations (g/m3) | Outlet Specifications (mg/m3) |
|---|---|---|
| Solids (settleable) | 200 | ≤2 |
| Metals (Pb, Cu, Zn, Ni) | 0.1 | ≤0.005 |
| Chemicals (acids, chlorides, organics, and inorganics) | 1 | ≤0.001 |
| Model Statistics | Values |
|---|---|
| Equations | 425 |
| Variables | 312 |
| Discrete Variable | 14 |
| Relative Gap | 0.0001 |
| Solution Time | 0.188 s |
| Solution | USD 1.52 million/y |
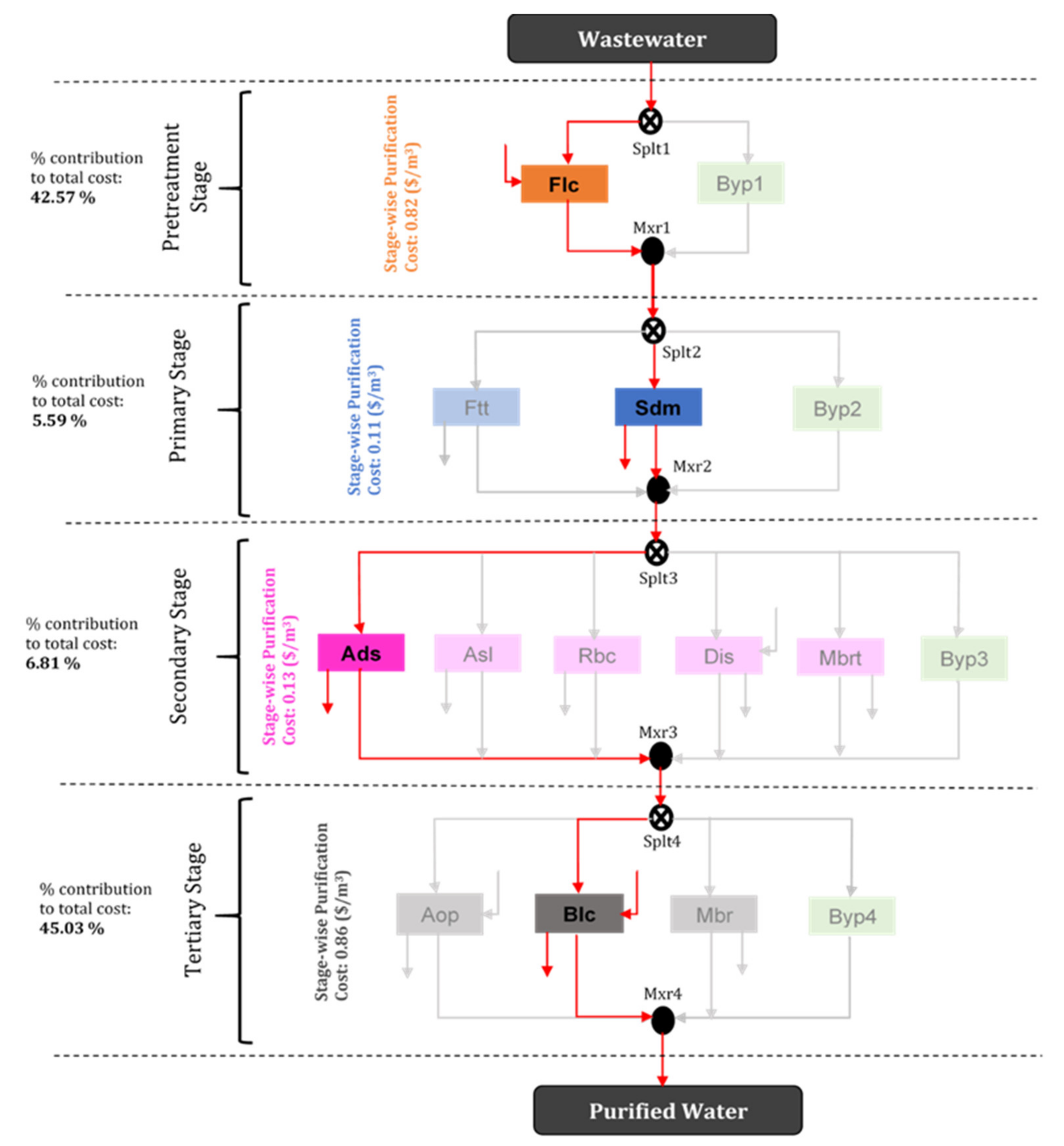
| Treatment | Treatment Network Pathway | Cost (USD/m3 WW) |
|---|---|---|
| First best (optimal treatment option) | Flc–Sdm–Ads–Blc | 1.92 |
| Second best treatment network | Flc–Sdm–Dis–Blc | 5.89 |
| Third best treatment network | Flc–Ftt–Dis–Blc | 8.56 |
| Contaminants | Concentration (mg/L) |
|---|---|
| Acetaminophen | 32.5 |
| Dextromethorphan HBr | 1.0 |
| Guaifenesin | 20.0 |
| Phenylephrine HCl | 0.5 |
| Contaminants | Entering Stream (g/m3) | Purity Specifications (mg/m3) |
|---|---|---|
| Solids | 10 | ≤2 |
| Metals | 0.01 | ≤0.005 |
| Chemicals | 44 | ≤5 |
| Pharmaceutical (APIs) | 0.4 | ≤0.02 |
| Model Statistics | Values |
|---|---|
| Equations | 503 |
| Variables | 358 |
| Discrete Variable | 15 |
| Relative Gap | 0.0001 |
| Solution Time | 0.125 s |
| Solution | USD 2.72 million/y |
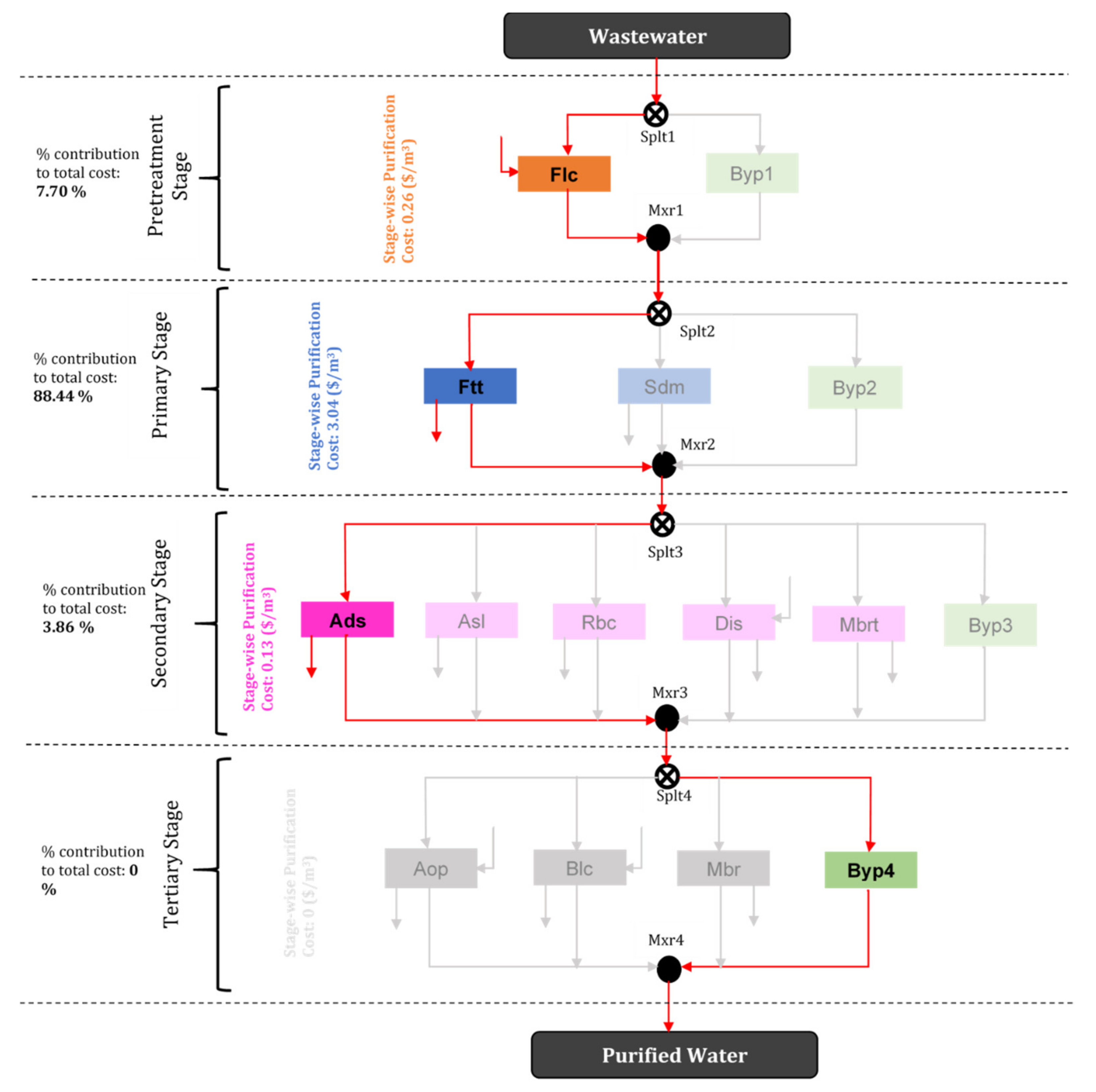
| Treatment | Treatment Network Pathway | Cost (USD/m3 WW) |
|---|---|---|
| First best (optimal treatment option) | Flc–Ftt–Ads–Byp4 | 3.44 |
| Second best treatment network | Flc–Ftt–Dis–Byp4 | 7.45 |
| Third best treatment network | Flc–Ftt–Byp3–Blc | 20.80 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aboagye, E.A.; Burnham, S.M.; Dailey, J.; Zia, R.; Tran, C.; Desai, M.; Yenkie, K.M. Systematic Design, Optimization, and Sustainability Assessment for Generation of Efficient Wastewater Treatment Networks. Water 2021, 13, 1326. https://doi.org/10.3390/w13091326
Aboagye EA, Burnham SM, Dailey J, Zia R, Tran C, Desai M, Yenkie KM. Systematic Design, Optimization, and Sustainability Assessment for Generation of Efficient Wastewater Treatment Networks. Water. 2021; 13(9):1326. https://doi.org/10.3390/w13091326
Chicago/Turabian StyleAboagye, Emmanuel A., Sean M. Burnham, James Dailey, Rohan Zia, Carley Tran, Maya Desai, and Kirti M. Yenkie. 2021. "Systematic Design, Optimization, and Sustainability Assessment for Generation of Efficient Wastewater Treatment Networks" Water 13, no. 9: 1326. https://doi.org/10.3390/w13091326
APA StyleAboagye, E. A., Burnham, S. M., Dailey, J., Zia, R., Tran, C., Desai, M., & Yenkie, K. M. (2021). Systematic Design, Optimization, and Sustainability Assessment for Generation of Efficient Wastewater Treatment Networks. Water, 13(9), 1326. https://doi.org/10.3390/w13091326






