Initial Deployment of a Mobile Sensing System for Water Quality in Urban Canals
Abstract
:1. Introduction
2. Materials and Methods
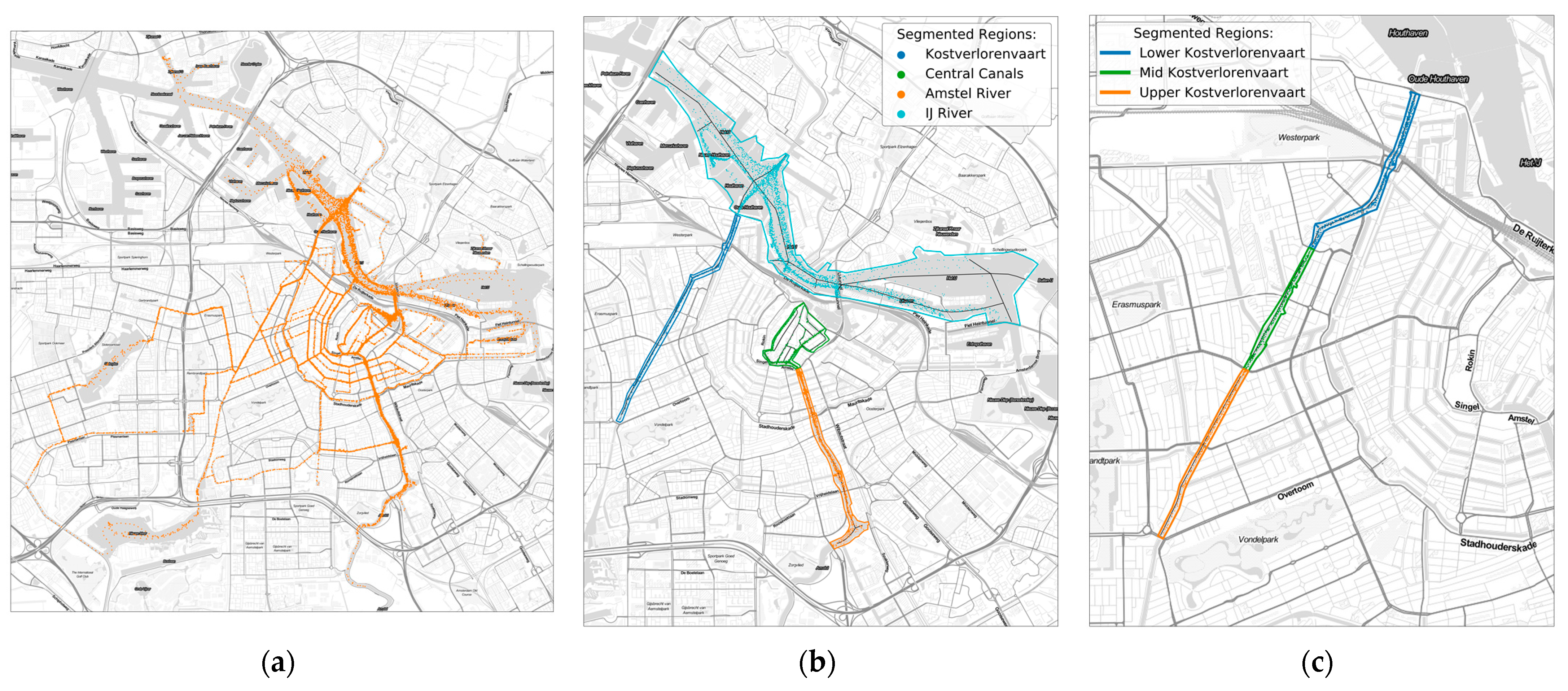
2.1. Study Area Description
2.2. Measurement Platform and Sampling Protocol
2.3. Internet of Things (IoT) Data Architecture
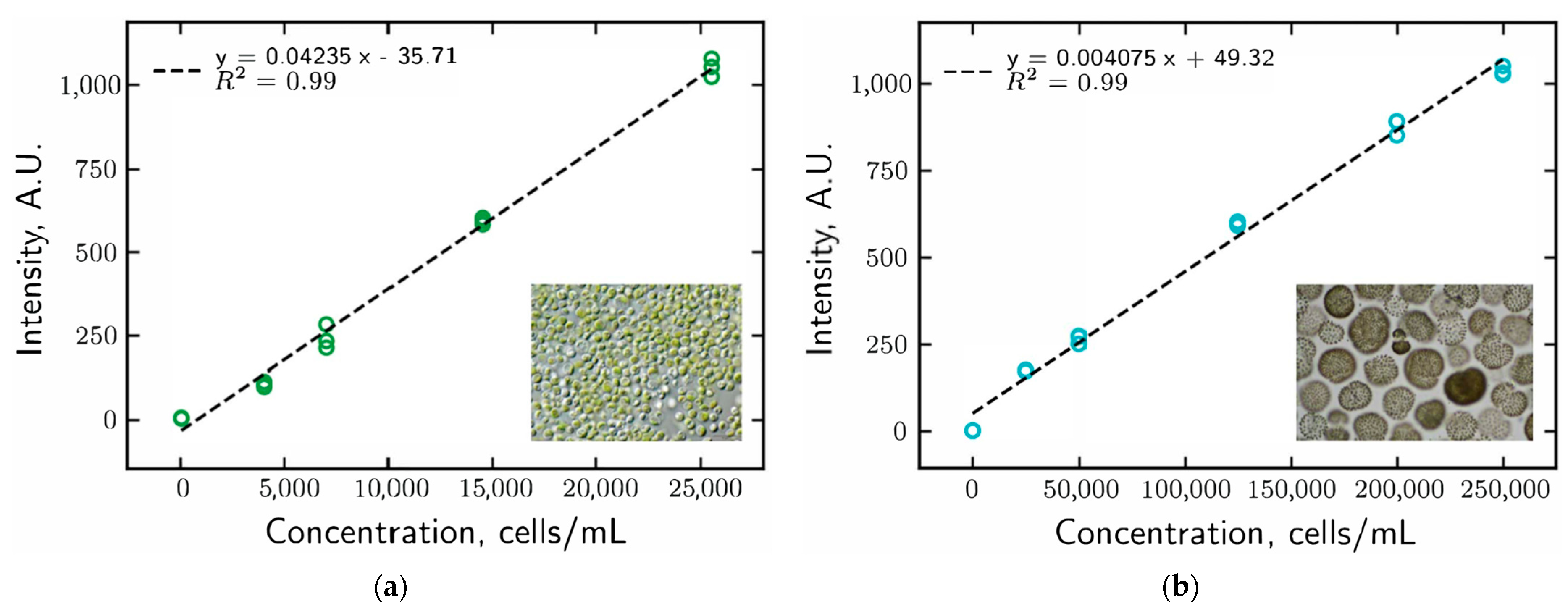
2.4. Instrument Calibrations
2.5. Spatial Segmentation
3. Results
3.1. Instrument Calibration
3.2. Spatiotemporal Sampling Coverage
3.3. Spatial Patterns of Water Quality Parameters
3.4. Deployment Challenges
4. Discussion and Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- White, M.; Smith, A.; Humphryes, K.; Pahl, S.; Snelling, D.; Depledge, M. Blue Space: The Importance of Water for Preference, Affect, and Restorativeness Ratings of Natural and Built Scenes. J. Environ. Psychol. 2010, 30, 482–493. [Google Scholar] [CrossRef]
- Schets, F.M.; van Wijnen, J.H.; Schijven, J.F.; Schoon, H.; Husman, A.M.d.R. Monitoring of Waterborne Pathogens in Surface Waters in Amsterdam, The Netherlands, and the Potential Health Risk Associated with Exposure to Cryptosporidium and Giardia in These Waters. Appl. Environ. Microbiol. 2008, 74, 2069–2078. [Google Scholar] [CrossRef] [PubMed]
- Sales-Ortells, H.; Medema, G. Screening-Level Microbial Risk Assessment of Urban Water Locations: A Tool for Prioritization. Environ. Sci. Technol. 2014, 48, 9780–9789. [Google Scholar] [CrossRef] [PubMed]
- Sales-Ortells, H.; Agostini, G.; Medema, G. Quantification of Waterborne Pathogens and Associated Health Risks in Urban Water. Environ. Sci. Technol. 2015, 49, 6943–6952. [Google Scholar] [CrossRef]
- World Health Organization. Guidelines for Safe Recreational Water Environments; Volume 1, Coastal and Fresh Waters; World Health Organization: Switzerland, Geneva, 2003. [Google Scholar]
- Hisbergues, M.; Christiansen, G.; Rouhiainen, L.; Sivonen, K.; Börner, T. PCR-Based Identification of Microcystin-Producing Genotypes of Different Cyanobacterial Genera. Arch. Microbiol. 2003, 180, 402–410. [Google Scholar] [CrossRef]
- US Environmental Protection Agency 2012 Recreational Water Quality Criteria Documents; Environmental Protection Agency: Washington, DC, USA, 2012.
- Directive 2006/7/EC of the European Parliament and of the Council of 15 February 2006 Concerning the Management of Bathing Water Quality and Repealing Directive 76/160/EEC. 2006. Available online: https://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=OJ:L:2006:064:0037:0051:EN:PDF (accessed on 2 August 2022).
- Peters, S.; Ouboter, M.; Lugt, K.v.d.; Koop, S.; Leeuwen, K.v. Retrospective Analysis of Water Management in Amsterdam, The Netherlands. Water 2021, 13, 1099. [Google Scholar] [CrossRef]
- Hintaran, A.D.; Kliffen, S.J.; Lodder, W.; Pijnacker, R.; Brandwagt, D.; van der Bij, A.K.; Siedenburg, E.; Sonder, G.J.B.; Fanoy, E.B.; Joosten, R.E. Infection Risks of City Canal Swimming Events in the Netherlands in 2016. PLoS ONE 2018, 13, e0200616. [Google Scholar] [CrossRef]
- Korving, H.; Leloup, M.-J.; Ouboter, M.; Schep, S. Combined Ship-Based and Stationary Monitoring of the Amsterdam Canals. In Proceedings of the International Conference on Hydroinformatics, Hamburg, Germany, 14–18 July 2012. [Google Scholar]
- Lee, J.H.; Bang, K.W. Characterization of Urban Stormwater Runoff. Water Res. 2000, 34, 1773–1780. [Google Scholar] [CrossRef]
- Yang, Y.-Y.; Toor, G.S. Stormwater Runoff Driven Phosphorus Transport in an Urban Residential Catchment: Implications for Protecting Water Quality in Urban Watersheds. Sci. Rep. 2018, 8, 11681. [Google Scholar] [CrossRef]
- Ho, J.C.; Michalak, A.M.; Pahlevan, N. Widespread Global Increase in Intense Lake Phytoplankton Blooms since the 1980s. Nature 2019, 574, 667–670. [Google Scholar] [CrossRef]
- Gobler, C.J.; Doherty, O.M.; Hattenrath-Lehmann, T.K.; Griffith, A.W.; Kang, Y.; Litaker, R.W. Ocean Warming since 1982 Has Expanded the Niche of Toxic Algal Blooms in the North Atlantic and North Pacific Oceans. Proc. Natl. Acad. Sci. USA 2017, 114, 4975–4980. [Google Scholar] [CrossRef] [PubMed]
- Rozemeijer, J.; Klein, J.; Hendriks, D.; Borren, W.; Ouboter, M.; Rip, W. Groundwater-Surface Water Relations in Regulated Lowland Catchments; Hydrological and Hydrochemical Effects of a Major Change in Surface Water Level Management. Sci. Total Environ. 2019, 660, 1317–1326. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.; Rozemeijer, J.; van Breukelen, B.M.; Ouboter, M.; van der Vlugt, C.; Broers, H.P. Groundwater Impacts on Surface Water Quality and Nutrient Loads in Lowland Polder Catchments: Monitoring the Greater Amsterdam Area. Hydrol. Earth Syst. Sci. 2018, 22, 487–508. [Google Scholar] [CrossRef]
- Yu, L.; Rozemeijer, J.C.; Broers, H.P.; van Breukelen, B.M.; Middelburg, J.J.; Ouboter, M.; van der Velde, Y. Drivers of Nitrogen and Phosphorus Dynamics in a Groundwater-Fed Urban Catchment Revealed by High-Frequency Monitoring. Hydrol. Earth Syst. Sci. 2021, 25, 69–87. [Google Scholar] [CrossRef]
- McCormick, P.V.; Cairns, J. Algae as Indicators of Environmental Change. J. Appl. Phycol. 1994, 6, 509–526. [Google Scholar] [CrossRef]
- Sieracki, C.K.; Sieracki, M.E.; Yentsch, C.S. An Imaging-in-Flow System for Automated Analysis of Marine Microplankton. Mar. Ecol. Prog. Ser. 1998, 168, 285–296. [Google Scholar] [CrossRef]
- Olson, R.J.; Sosik, H.M. A Submersible Imaging-in-Flow Instrument to Analyze Nano-and Microplankton: Imaging FlowCytobot. Limnol. Oceanogr. Methods 2007, 5, 195–203. [Google Scholar] [CrossRef]
- Göröcs, Z.; Tamamitsu, M.; Bianco, V.; Wolf, P.; Roy, S.; Shindo, K.; Yanny, K.; Wu, Y.; Koydemir, H.C.; Rivenson, Y.; et al. A Deep Learning-Enabled Portable Imaging Flow Cytometer for Cost-Effective, High-Throughput, and Label-Free Analysis of Natural Water Samples. Light Sci. Appl. 2018, 7, 66. [Google Scholar] [CrossRef]
- Beutler, M.; Wiltshire, K.H.; Meyer, B.; Moldaenke, C.; Lüring, C.; Meyerhöfer, M.; Hansen, U.P.; Dau, H. A Fluorometric Method for the Differentiation of Algal Populations In Vivo and In Situ. Photosynth. Res. 2002, 72, 39–53. [Google Scholar] [CrossRef]
- Ng, C.L.; Chen, Q.Q.; Chua, J.J.; Hemond, H.F. A Multi-Platform Optical Sensor for In Vivo and In Vitro Algae Classification. Sensors 2017, 17, 912. [Google Scholar] [CrossRef] [Green Version]
- Zieger, S.E.; Mistlberger, G.; Troi, L.; Lang, A.; Confalonieri, F.; Klimant, I. Compact and Low-Cost Fluorescence Based Flow-through Analyzer for Early-Stage Classification of Potentially Toxic Algae and in Situ Semiquantification. Environ. Sci. Technol. 2018, 52, 7399–7408. [Google Scholar] [CrossRef] [PubMed]
- Catherine, A.; Escoffier, N.; Belhocine, A.; Nasri, A.B.; Hamlaoui, S.; Yéprémian, C.; Bernard, C.; Troussellier, M. On the Use of the FluoroProbe®, a Phytoplankton Quantification Method Based on Fluorescence Excitation Spectra for Large-Scale Surveys of Lakes and Reservoirs. Water Res. 2012, 46, 1771–1784. [Google Scholar] [CrossRef] [PubMed]
- Zieger, S.E.; Seoane, S.; Laza-Martínez, A.; Knaus, A.; Mistlberger, G.; Klimant, I. Spectral Characterization of Eight Marine Phytoplankton Phyla and Assessing a Pigment-Based Taxonomic Discriminant Analysis for the in Situ Classification of Phytoplankton Blooms. Environ. Sci. Technol. 2018, 52, 14266–14274. [Google Scholar] [CrossRef] [PubMed]
- Richardson, T.L.; Lawrenz, E.; Pinckney, J.L.; Guajardo, R.C.; Walker, E.A.; Paerl, H.W.; MacIntyre, H.L. Spectral Fluorometric Characterization of Phytoplankton Community Composition Using the Algae Online Analyser®. Water Res. 2010, 44, 2461–2472. [Google Scholar] [CrossRef] [PubMed]
- Macintyre, H.L.; Lawrenz, E.; Richardson, T.L. Taxonomic Discrimination of Phytoplankton by Spectral Fluorescence. In Chlorophyll a Fluorescence in Aquatic Sciences: Methods and Applications; Springer: Dordrecht, The Netherlands, 2010; ISBN 978-90-481-9268-7. [Google Scholar]
- O’Keeffe, K.P.; Anjomshoaa, A.; Strogatz, S.H.; Santi, P.; Ratti, C. Quantifying the Sensing Power of Vehicle Fleets. Proc. Natl. Acad. Sci. USA 2019, 116, 12752–12757. [Google Scholar] [CrossRef]
- Apte, J.S.; Messier, K.P.; Gani, S.; Brauer, M.; Kirchstetter, T.W.; Lunden, M.M.; Marshall, J.D.; Portier, C.J.; Vermeulen, R.C.H.; Hamburg, S.P. High-Resolution Air Pollution Mapping with Google Street View Cars: Exploiting Big Data. Environ. Sci. Technol. 2017, 51, 6999–7008. [Google Scholar] [CrossRef]
- Jaffe, J.S.; Franks, P.J.S.; Roberts, P.L.D.; Mirza, D.; Schurgers, C.; Kastner, R.; Boch, A. A Swarm of Autonomous Miniature Underwater Robot Drifters for Exploring Submesoscale Ocean Dynamics. Nat. Commun. 2017, 8, 14189. [Google Scholar] [CrossRef]
- Oroza, C.; Tinka, A.; Wright, P.K.; Bayen, A.M. Design of a Network of Robotic Lagrangian Sensors for Shallow Water Environments with Case Studies for Multiple Applications. Proc. Inst. Mech. Eng. Part C J. Mech. Eng. Sci. 2013, 227, 2531–2548. [Google Scholar] [CrossRef]
- Nicholson, D.P.; Michel, A.P.M.; Wankel, S.D.; Manganini, K.; Sugrue, R.A.; Sandwith, Z.O.; Monk, S.A. Rapid Mapping of Dissolved Methane and Carbon Dioxide in Coastal Ecosystems Using the ChemYak Autonomous Surface Vehicle. Environ. Sci. Technol. 2018, 52, 13314–13324. [Google Scholar] [CrossRef]
- Tinka, A.; Rafiee, M.; Bayen, A.M. Floating Sensor Networks for River Studies. IEEE Syst. J. 2013, 7, 36–49. [Google Scholar] [CrossRef]
- Ensign, S.H.; Paerl, H.W. Development of an Unattended Estuarine Nutrient Monitoring Program Using Ferries as Data-Collection Platforms. Limnol. Oceanogr. Methods 2006, 4, 399–405. [Google Scholar] [CrossRef]
- Seppälä, J.; Ylöstalo, P.; Kaitala, S.; Hällfors, S.; Raateoja, M.; Maunula, P. Ship-of-Opportunity Based Phycocyanin Fluorescence Monitoring of the Filamentous Cyanobacteria Bloom Dynamics in the Baltic Sea. Estuar. Coast. Shelf Sci. 2007, 73, 489–500. [Google Scholar] [CrossRef]
- Anjomshoaa, A.; Duarte, F.; Rennings, D.; Matarazzo, T.J.; Desouza, P.; Ratti, C. City Scanner: Building and Scheduling a Mobile Sensing Platform for Smart City Services. IEEE Internet Things J. 2018, 5, 4567–4579. [Google Scholar] [CrossRef]
- Van Rees Vellinga, E.; Toussaint, C.G.; Wit, K.E. Water Quality and Hydrology in a Coastal Region of The Netherlands. J. Hydrol. 1981, 50, 105–127. [Google Scholar] [CrossRef]
- Rozemeijer, J.; Siderius, C.; Verheul, M.; Pomarius, H. Tracing the Spatial Propagation of River Inlet Water into an Agricultural Polder Area Using Anthropogenic Gadolinium. Hydrol. Earth Syst. Sci. 2012, 16, 2405–2415. [Google Scholar] [CrossRef]
- Kelly, M.G.; Cazaubon, A.; Coring, E.; Dell’Uomo, A.; Ector, L.; Goldsmith, B.; Guasch, H.; Hürlimann, J.; Jarlman, A.; Kawecka, B.; et al. Recommendations for the Routine Sampling of Diatoms for Water Quality Assessments in Europe. J. Appl. Phycol. 1998, 10, 215–224. [Google Scholar] [CrossRef]
- Ng, C.L.; Senft-Grupp, S.; Hemond, H.F. A Multi-Platform Optical Sensor for In Situ Sensing of Water Chemistry. Limnol. Oceanogr. Methods 2012, 10, 978–990. [Google Scholar] [CrossRef]
- Wang, W.; Shan, T.; Leoni, P.; Fernández-Gutiérrez, D.; Meyers, D.; Ratti, C.; Rus, D. Roboat II: A Novel Autonomous Surface Vessel for Urban Environments. In Proceedings of the 2020 IEEE/RSJ International Conference on Intelligent Robots and Systems (IROS), Las Vegas, NV, USA, 22–25 October 2020; pp. 1740–1747. [Google Scholar]
- Chang, H.-C.; Hsu, Y.-L.; Hung, S.-S.; Ou, G.-R.; Wu, J.-R.; Hsu, C. Autonomous Water Quality Monitoring and Water Surface Cleaning for Unmanned Surface Vehicle. Sensors 2021, 21, 1102. [Google Scholar] [CrossRef]
- Boss, E.; Ackleson, S.; Balch, B.; Chase, A.; Dall’olmo, G.; Freeman, S.; Haëntjens, N.; Loftin, J.; Neary, W.; Nelson, N.; et al. Inherent Optical Property Measurements and Protocols: Best Practices for the Collection and Processing of Ship-Based Underway Flow-Through Optical Data; IOCCG: Dartmouth, NS, Canada, 2019. [Google Scholar]








Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Meyers, D.; Zheng, Q.; Duarte, F.; Ratti, C.; Hemond, H.F.; van der Blom, M.; van der Helm, A.W.C.; Whittle, A.J. Initial Deployment of a Mobile Sensing System for Water Quality in Urban Canals. Water 2022, 14, 2834. https://doi.org/10.3390/w14182834
Meyers D, Zheng Q, Duarte F, Ratti C, Hemond HF, van der Blom M, van der Helm AWC, Whittle AJ. Initial Deployment of a Mobile Sensing System for Water Quality in Urban Canals. Water. 2022; 14(18):2834. https://doi.org/10.3390/w14182834
Chicago/Turabian StyleMeyers, Drew, Qinmin Zheng, Fábio Duarte, Carlo Ratti, Harold F. Hemond, Marcel van der Blom, Alex W.C. van der Helm, and Andrew J. Whittle. 2022. "Initial Deployment of a Mobile Sensing System for Water Quality in Urban Canals" Water 14, no. 18: 2834. https://doi.org/10.3390/w14182834
APA StyleMeyers, D., Zheng, Q., Duarte, F., Ratti, C., Hemond, H. F., van der Blom, M., van der Helm, A. W. C., & Whittle, A. J. (2022). Initial Deployment of a Mobile Sensing System for Water Quality in Urban Canals. Water, 14(18), 2834. https://doi.org/10.3390/w14182834






