Odonata Assemblages in Anthropogenically Impacted Habitats in the Drava River—A Long-Term Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. Odonata Sampling
2.3. Environmental Variables
2.4. Data Analyses
3. Results
3.1. Environmental Variables
3.2. Odonata Assemblages
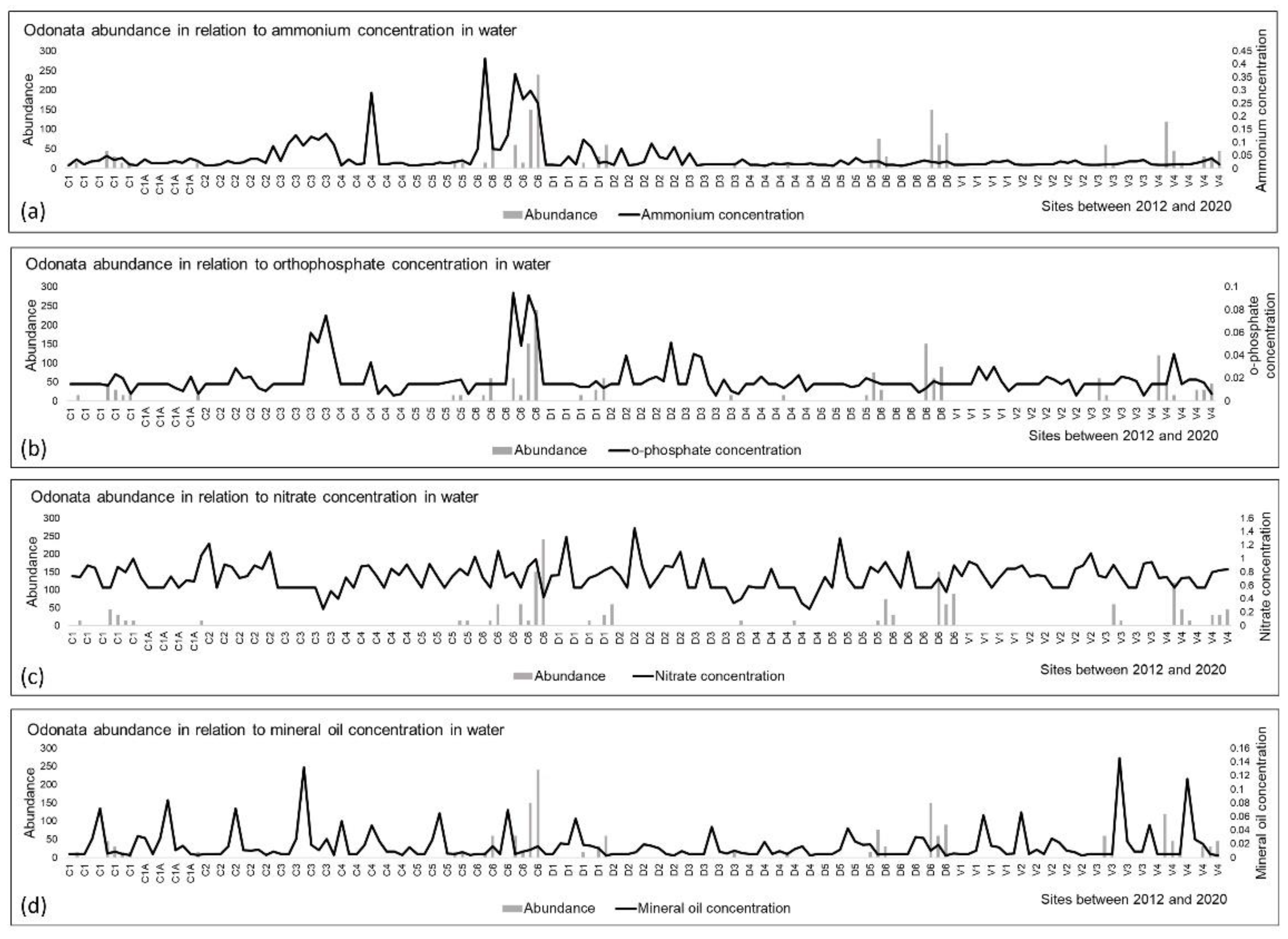
3.3. Odonata and Environmental Variables
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cazzolla Gatti, R. Freshwater biodiversity: A review of local and global threats. Int. J. Environ. Stud. 2016, 73, 887–904. [Google Scholar] [CrossRef]
- Ivković, M.; Plant, A. Aquatic insects in the Dinarides: Identifying hotspots of endemism and species richness shaped by geological and hydrological history using Empididae (Diptera). Insect Conserv. Divers. 2015, 8, 302–312. [Google Scholar] [CrossRef]
- Previšić, A.; Graf, W.; Vitecek, S.; Kučinić, M.; Bálint, M.; Keresztes, L.; Pauls, S.U.; Waringer, J. Cryptic diversity of caddisflies in the Balkans: The curious case of Ecclisopteryx species (Trichoptera: Limnephilidae). Arthropod. Syst. Phylogeny 2014, 72, 309–329. [Google Scholar] [PubMed]
- Aylward, B.; Bandyopadhyay, J.; Belausteguigotia, J.-C. Freshwater Ecosystem Services. Ecosyst. Hum. Well-Being Policy Responses 2005, 3, 213–256. [Google Scholar]
- Naiman, R.J.; Magnuson, J.J.; McKnight, D.M.; Stanford, J.A.; Karr, J.R. Freshwater ecosystems and management: A national initiative. Science 1995, 270, 584–585. [Google Scholar] [CrossRef]
- Sala, O.E.; Chapin, F.S.; Armesto, J.J.; Berlow, E.; Bloomfield, J.; Dirzo, R.; Huber-Sanwald, E.; Huenneke, L.F.; Jackson, R.B.; Kinzig, A.; et al. Global Biodiversity Scenarios for the Year 2100. Science 2000, 287, 1770–1774. [Google Scholar] [CrossRef]
- Vörösmarty, C.J.; McIntyre, P.B.; Gessner, M.O.; Dudgeon, D.; Prusevich, A.; Green, P.; Glidden, S.; Bunn, S.E.; Sullivan, C.A.; Liermann, C.R.; et al. Global threats to human water security and river biodiversity. Nature 2010, 467, 555–561. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Däll, P.; Zhang, J. Impact of climate change on freshwater ecosystems: A global-scale analysis of ecologically relevant river flow alterations. Hydrol. Earth Syst. Sci. 2010, 14, 783–799. [Google Scholar] [CrossRef] [Green Version]
- Pham, H.V.; Torresan, S.; Critto, A.; Marcomini, A. Alteration of freshwater ecosystem services under global change—A review focusing on the Po River basin (Italy) and the Red River basin (Vietnam). Sci. Total Environ. 2019, 652, 1347–1365. [Google Scholar] [CrossRef] [PubMed]
- Žganec, K.; Lajtner, J.; Ćuk, R.; Crnčan, P.; Pušić, I.; Atanacković, A.; Kralj, T.; Valić, D.; Jelić, M.; Maguire, I. Alien macroinvertebrates in Croatian freshwaters. Aquat. Invasions 2020, 15, 593–615. [Google Scholar] [CrossRef]
- Carpenter, S.R.; Stanley, E.H.; Vander Zanden, M.J. State of the world’s freshwater ecosystems: Physical, chemical, and biological changes. Annu. Rev. Environ. Resour. 2011, 36, 75–99. [Google Scholar] [CrossRef]
- Giang, P.Q.; Toshiki, K.; Kunikane, S.; Sakata, M. Climate change challenges transboundary water resources management: Drawing from the case of Vietnam. In Proceedings of the 3rd International Conference on Chemical, Biological and Environmental Sciences, Kuala Lumpur, Malaysia, 8–9 January 2013. [Google Scholar]
- Arribas, P.; Abellán, P.; Velasco, J.; Millán, A.; Sánchez-Fernández, D. Conservation of insects in the face of global climate change. In Global Climate Change and Terrestrial Invertebrates; Johnson, S.N., Jones, T.H., Eds.; Wiley-Blackwell: Chichester, UK, 2017; p. 416. [Google Scholar]
- Assandri, G. Anthropogenic-driven transformations of dragonfly (Insecta: Odonata) communities of low elevation mountain wetlands during the last century. Insect Conserv. Divers. 2020, 14, 26–39. [Google Scholar] [CrossRef]
- Cerini, F.; Stellati, L.; Luiselli, L.; Vignoli, L. Long-term shifts in the communities of Odonata: Effect of chance or climate change? North-West J. Zool. 2020, 16, 1–6. [Google Scholar]
- Dudgeon, D.; Arthington, A.H.; Gessner, M.O.; Kawabata, Z.; Knowler, D.; Lévêque, C.; Naiman, R.J.; Prieur-Richard, A.-H.; Soto, D.; Stiassny, M.L.J.; et al. Freshwater biodiversity: Importance, status, and conservation challenges. Biol. Rev. 2006, 81, 163–182. [Google Scholar] [CrossRef]
- Marten, M. Environmental monitoring in Baden-Württemberg with special reference to biocoenotic trend-monitoring of macrozoobenthos in rivers and methodical requirements for evaluation of long-term biocoenotic changes. Aquat. Ecol. 2001, 35, 159–171. [Google Scholar] [CrossRef]
- Outhwaite, C.L.; Gregory, R.D.; Chandler, R.E.; Collen, B.; Isaac, N.J.B. Complex long-term biodiversity change among invertebrates, bryophytes and lichens. Nat. Ecol. Evol. 2020, 4, 384–392. [Google Scholar] [CrossRef]
- Vilenica, M.; Previšić, A.; Ivković, M.; Popijač, A.; Vučković, I.; Kučinić, M.; Kerovec, M.; Gattolliat, J.-L.; Sartori, M.; Mihaljević, Z. Mayfly (Insecta: Ephemeroptera) assemblages of a regulated perennial Mediterranean river system in the Western Balkans. Biologia 2016, 71, 1038–1048. [Google Scholar] [CrossRef]
- Vilenica, M.; Kerovec, M.; Pozojević, I.; Mihaljević, Z. Odonata assemblages in anthropogenically impacted lotic habitats. J. Limnol. 2020, 80, 1–9. [Google Scholar] [CrossRef]
- Vilenica, M.; Pozojević, I.; Vučković, N.; Mihaljević, Z. How suitable are man-made water bodies as habitats for Odonata? Knowl. Manag. Aquat. Ecosyst. 2020, 421, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Franklin, J.F. Importance and Justification of Long-Term Studies in Ecology. In Long-Term Studies in Ecology; Likens, G.E., Ed.; Springer: New York, NY, USA, 1989; pp. 3–19. [Google Scholar] [CrossRef]
- Magurran, A.E.; Baillie, S.R.; Buckland, S.T.; Dick, J.M.; Elston, D.A.; Scott, E.M.; Smith, R.I.; Somerfield, P.J.; Watt, A.D. Long-term datasets in biodiversity research and monitoring: Assessing change in ecological communities through time. Trends Ecol. Evol. 2010, 25, 574–582. [Google Scholar] [CrossRef]
- Monzó, J.C.; Verdú, J.R. Effects of restoration and management of Mediterranean traditional water systems on Odonata alpha diversity: A long-term monitoring survey. Biodivers. Conserv. 2022, 31, 227–243. [Google Scholar] [CrossRef]
- Corbet, P.; Brooks, S. Dragonflies (Collins New Naturalist Library, Book 106); Collins: London, UK, 2008. [Google Scholar]
- Schmidt-Kloiber, A.; Hering, D. www.freshwaterecology.info—An online tool that unifies, standardises and codifies more than 20,000 European freshwater organisms and their ecological preferences. Ecol. Indic. 2015, 53, 271–282. [Google Scholar] [CrossRef]
- Kutcher, T.E.; Bried, J.T. Adult Odonata Conservatism as an Indicator of Freshwater Wetland Condition. Ecol. Indic. 2014, 38, 31–39. [Google Scholar] [CrossRef]
- Simaika, J.P.; Samways, M.J. An Easy-to-Use Index of Ecological Integrity for Prioritizing Freshwater Sites and for Assessing Habitat Quality. Biodivers. Conserv. 2009, 18, 1171–1185. [Google Scholar] [CrossRef] [Green Version]
- Vilenica, M. Ecological traits of dragonfly (Odonata) assemblages along an oligotrophic Dinaric karst hydrosystem. Ann. Limnol. Int. J. Limnol. 2017, 53, 377–389. [Google Scholar] [CrossRef] [Green Version]
- Vilenica, M.; Rebrina, F.; Matoničkin Kepčija, R.; Šegota, V.; Rumišek, M.; Ružanović, L.; Brigić, A. Aquatic macrophyte vegetation promotes taxonomic and functional diversity of Odonata assemblages in intermittent karst rivers in the Mediterranean. Diversity 2022, 14, 31. [Google Scholar] [CrossRef]
- Maguire, I.; Gottstein-Matočec, S. The distribution pattern of freshwater crayfish in Croatia. Crustaceana 2004, 77, 25–47. [Google Scholar] [CrossRef]
- Previšić, A.; Mihaljević, Z.; Kerovec, M. Caddisfly (Insecta: Trichoptera) fauna of altered and man-made habitats in the Drava River, NW Croatia. Nat. Croat. 2007, 16, 181–187. [Google Scholar]
- Csiky, J.; Purger, D. Monitoring of plant species along the Drava river and in Baranja (Croatia). In Biodiversity Studies along the Drava River; Purger, J.J., Ed.; University of Pécs: Pécs, Hungary, 2008; p. 328. [Google Scholar]
- UNESCO. 2012. Available online: https://en.unesco.org/biosphere/eu-na/mura-drava-danube (accessed on 3 September 2022).
- Bonacci, O.; Tadić, Z.; Trninić, D. Effects of dams and reservoirs on the hydrological characteristics of the lower Drava River. Regul. Rivers Res. Manag. 1992, 7, 349–357. [Google Scholar] [CrossRef]
- Gerken, B.; Sternberg, K. Die Exuvien Europaïscher Libellen—The Exuviae of European Dragonflies (Insecta, Odonata); Arnika & Eisvogel/Huxaria Druckerei GmbH: Höxter/Jena, Germany, 1999; p. 354. [Google Scholar]
- Askew, R.R. The Dragonflies of Europe, 2nd ed.; Harley Books: Essex, UK, 2004; p. 308. [Google Scholar]
- Brochard, C.; Groendijk, D.; van der Ploeg, E.; Termaat, T. Fotogids Larvenhuidjes van Libellen; KNNV Publishers: Zeist, The Netherlands, 2012; p. 320. [Google Scholar]
- APHA. Standard Methods for the Examination of Water and Wastewater, 18th ed.; American Public Health Association: Washington, DC, USA, 1992. [Google Scholar]
- Clarke, K.R.; Gorley, R.N. Primer V6: User Manual/Tutorial; Plymouth: Auckland, New Zealand, 2006. [Google Scholar]
- Ter Braak, C.J.F.; Šmilauer, P. Canoco Reference Manual and User’s Guide: Software for Ordination, Version 5.0; Microcomputer Power: Ithaca, NY, USA, 2012. [Google Scholar]
- Belančić, A.; Bogdanović, T.; Franković, M.; Ljuština, M.; Mihoković, N.; Vitas, B. Red Data Book of Dragonflies of Croatia; Franković, M., Ed.; Ministry of Culture, State Institute for Nature Protection: Zagreb, Croatia, 2008. [Google Scholar]
- Kalkman, V.J.; Boudot, J.-P.; Bernard, R.; Conze, K.-J.; De Knijf, G.; Dyatlova, E.; Ferreira, S.; Jović, M.; Ott, J.; Riservato, E.; et al. European Red List of Dragonflies; Publications Office of the European Union: Luxembourg, 2010. [Google Scholar]
- Vidaković Maoduš, I.; Pozojević, I.; Vilenica, M.; Mihaljević, Z. Longitudinal dynamics of Odonata assemblages in an anthropogenically impacted lotic system. Ann. Limnol. Int. J. Lim. 2022, 58, 1–13. [Google Scholar] [CrossRef]
- Horning, C.E.; Pollard, J.E. Macroinvertebrate Sampling Techniques for Streams in Semi-Arid Regions: Comparison of the Surber Method and Unit-Effort Traveling Kick Method; Report Number EPA 600-4-78-040; US EPA: Washington, DC, USA, 1978; p. 37.
- Martín, R.; Maynou, X. Dragonflies (Insecta: Odonata) as indicators of habitat quality in Mediterranean streams and rivers in the province of Barcelona (Catalonia, Iberian Peninsula). Int. J. Odonatol. 2016, 19, 107–124. [Google Scholar] [CrossRef]
- Samways, M.J.; McGeoch, M.A.; New, T.R. Insect Conservation: Handbook of Approaches and Methods; Oxford University Press: Oxford, UK, 2009; p. 432. [Google Scholar]
- Raebel, E.M.; Merckx, T.; Riordan, P.; Macdonald, D.W.; Thompson, D.J. The dragonfly delusion: Why it is essential to sample exuviae to avoid biased surveys. J. Insect Conserv. 2010, 14, 523–534. [Google Scholar] [CrossRef]
- Golfieri, B.; Hardersen, S.; Maiolini, B.; Surian, N. Odonates as indicators of the ecological integrity of the river corridor: Development and application of the Odonate River Index (ORI) in northern Italy. Ecol. Indic. 2016, 61, 234–247. [Google Scholar] [CrossRef]
- Da Silva Monteiro, C., Jr.; Couceiro, S.R.M.; Hamada, N.; Juen, L. Effect of vegetation removal for road building on richness and composition of Odonata communities in Amazonia. Brazil. Int. J. Odonatol. 2013, 16, 135–144. [Google Scholar] [CrossRef]
- Pereira-Moura, L.; de Sena, W.S.; Neiss, U.G.; Marques Couceiro, S.R. Environmental integrity as a modeler of the composition of the Odonata community. Environ. Monit. Assess. 2021, 193, 160. [Google Scholar] [CrossRef] [PubMed]
- White, E.R. Minimum time required to detect population trends: The need for long-term monitoring programs. BioScience 2019, 69, 40–46. [Google Scholar] [CrossRef] [Green Version]
- Dolný, A.; Pyszko, P.; Šigutová, H. Community changes in Odonate monitoring: Why are long-term studies so relevant? Insect Conserv. Divers. 2021, 14, 597–608. [Google Scholar] [CrossRef]
- Dijkstra, K.D.B.; Lewington, R. Field Guide to the Dragonflies of Britain and Europe, 1st ed.; British Wildlife Publishing: Gillingham, UK, 2006; p. 320. [Google Scholar]
- Dijkstra, K.-D.B.; Wildermuth, H.; Martens, A. Dataset “Odonata”. The Taxa and Autecology Database for Freshwater Organisms, Version 8.0. Available online: www.freshwaterecology.info (accessed on 4 September 2022).
- Balázs, A.; Šipoš, J.; Matúšová, Z.; Hamerlík, L.; Novikmec, M.; Svitok, M. Comparison of conservation values among man-made aquatic habitats using Odonata communities in Slovakia. Biologia 2022. [Google Scholar] [CrossRef]
- Holtmann, L.; Juchem, M.; Brüggeshemke, J.; Möhlmeyer, A.; Fartmann, T. Stormwater ponds promote dragonfly (Odonata) species richness and density in urban areas. Ecol. Eng. 2018, 118, 1–11. [Google Scholar] [CrossRef]
- Šigutová, H.; Pyszko, P.; Valušák, J.; Dolný, A. Highway stormwater ponds as islands of Odonata diversity in an agricultural landscape. Sci. Total Environ. 2022, 837, 155774. [Google Scholar] [CrossRef]
- Carvalho, F.G.; Oliveira Roque, F.; Barbosa, L.; Assis Montag, L.F.; Juen, L. Oil palm plantation is not a suitable environment for most forest specialist species of Odonata in Amazonia. Anim. Conserv. 2018, 21, 526–533. [Google Scholar] [CrossRef]
- Calvão, L.B.; Juen, L.; De Oliveira Junior, J.M.B.; Batista, J.D.; Júnior, P.D.M. Land use modifies Odonata diversity in streams of the Brazilian Cerrado. J. Insect Conserv. 2018, 22, 675–685. [Google Scholar] [CrossRef]
- Oliveira, D.G.; Vargas, R.R.; Saad, A.R.; Arruda, R.O.M.; Dalmas, F.B.; Azevedo, F.D. Land use and its impacts on the water quality of the Cachoeirinha Invernada Watershed, Guarulhos (SP). Rev. Ambient. Agua 2018, 13, 1–17. [Google Scholar] [CrossRef]
- Boeykens, S.P.; Piol, M.N.; Legal, L.S.; Saralegui, A.B.; Vázquez, C. Eutrophication decrease: Phosphate adsorption processes in presence of nitrates. J. Environ. Manag. 2017, 203, 888–895. [Google Scholar] [CrossRef]
- Clarke, E.; Baldwin, A.H. Responses of wetland plants to ammonia and water level. Ecol. Eng. 2022, 18, 257–264. [Google Scholar] [CrossRef]
- Perron, M.A.C.; Pick, F.R. Stormwater ponds as habitat for Odonata in urban areas: The importance of obligate wetland plant species. Biodivers. Conserv. 2020, 29, 913–931. [Google Scholar] [CrossRef]
- Purse, B.V.; Thompson, D.J. Oviposition Site Selection by Coenagrion mercuriale (Odonata: Coenagrionidae). Int. J. Odonatol. 2009, 12, 257–273. [Google Scholar] [CrossRef]
- Brito, J.S.; Michelan, T.S.; Juen, L. Aquatic macrophytes are important substrates for Libellulidae (Odonata) larvae and adults. Limnology 2021, 22, 139–149. [Google Scholar] [CrossRef]
- Hu, W.; Zheng, R.; Feng, X.; Kuang, F.; Chun, J.; Xu, H.; Chen, T.; Lu, J.; Li, W.; Zhang, N. Emergence inhibition, repellent activity and antifeedant responds of mineral oils against Asian citrus psyllid, Diaphorina citri (Hemiptera: Liviidae). Int. J. Pest Manag. 2020, 1–8. [Google Scholar] [CrossRef]
- Mulla, M.S.; Darwazeb, H.A.; Ede, L. Evaluation of new pyrethroids against immature mosquitoes and their effects on nontarget organisms. Mosq. News 1982, 42, 583–590. [Google Scholar]
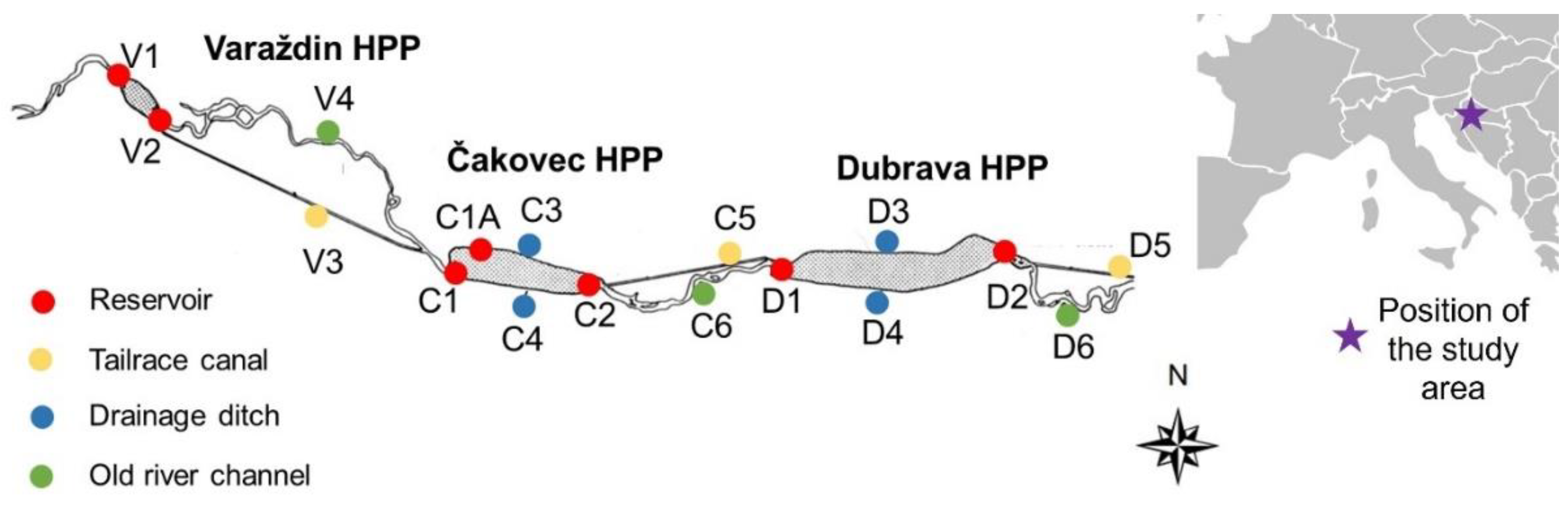
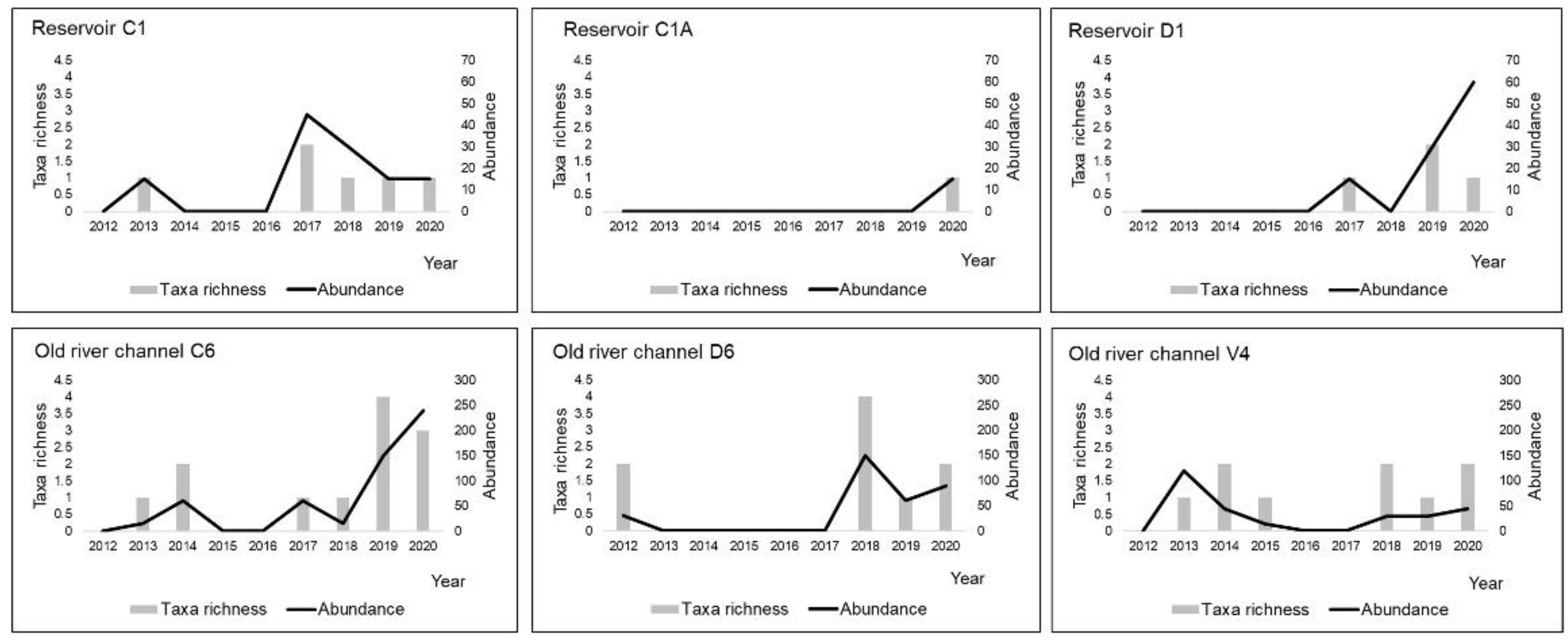
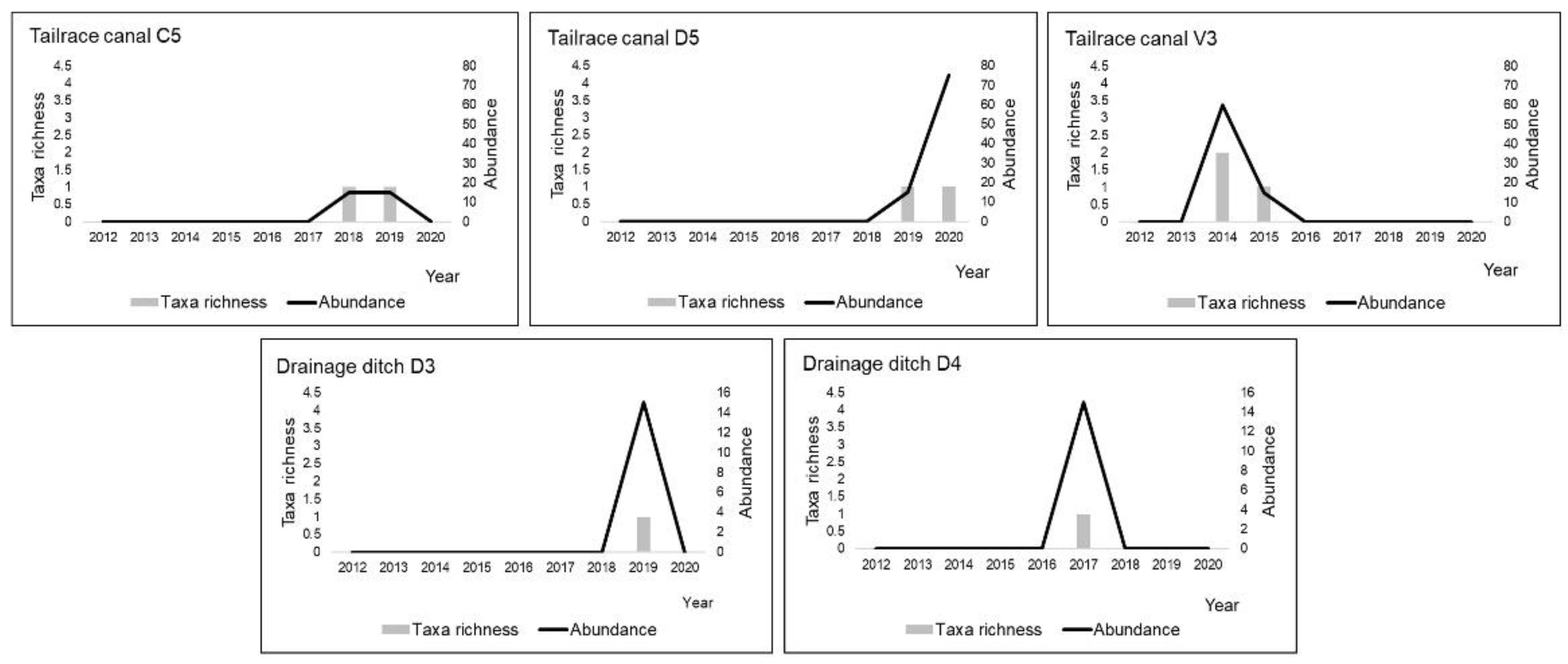


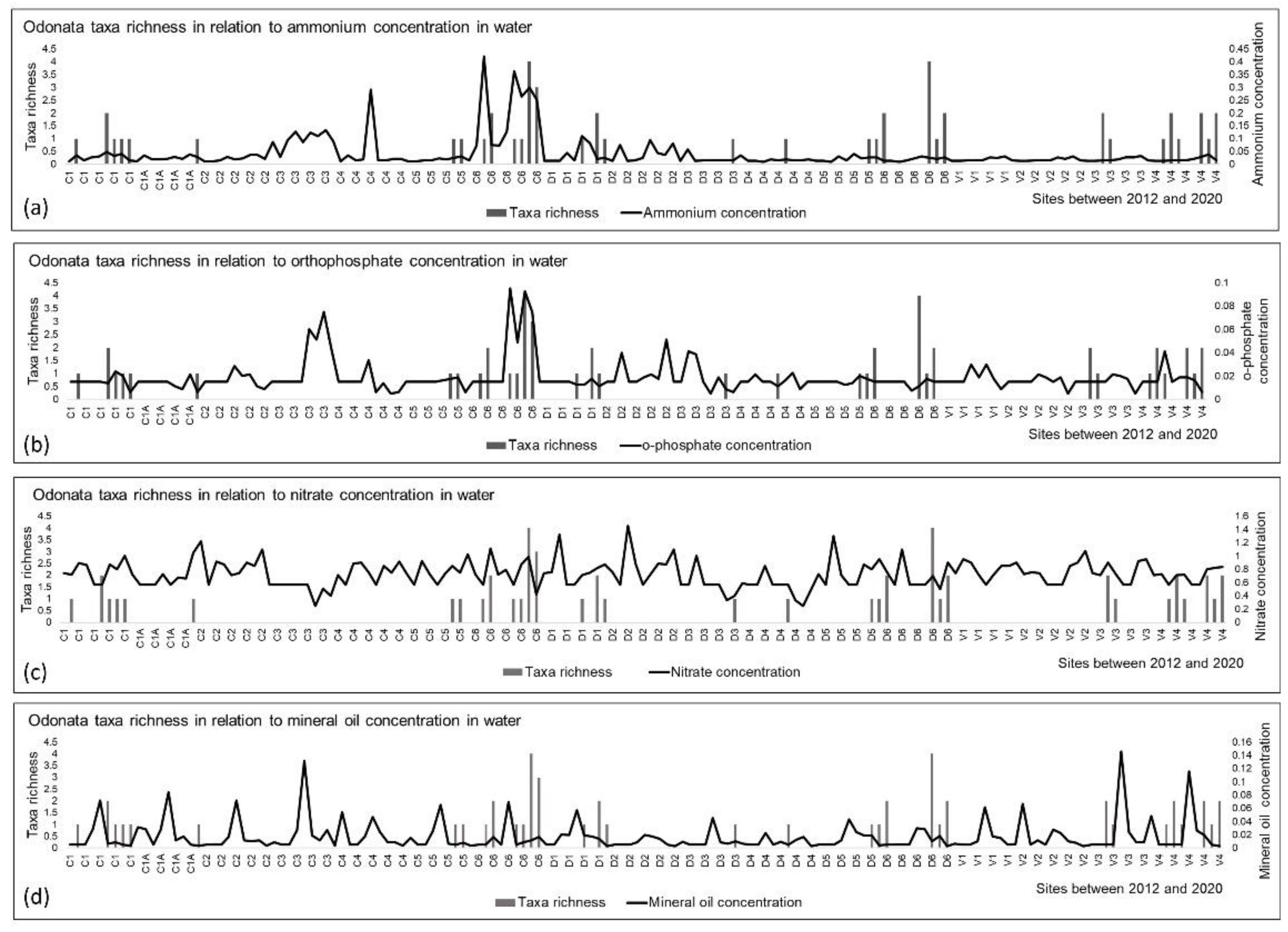
| Habitats/Environmental Parameters | Reservoirs | Drainage Ditches | Tailrace Canals | Old River Channels | ||||
|---|---|---|---|---|---|---|---|---|
| Mean | SD | Mean | SD | Mean | SD | Mean | SD | |
| Water temperature (°C) | 14.98 | 2.09 | 13.40 | 0.98 | 14.83 | 1.24 | 15.31 | 1.55 |
| pH | 8.12 | 0.16 | 7.98 | 0.17 | 8.12 | 0.15 | 8.09 | 0.16 |
| Conductivity (µS/cm) | 283 | 25 | 339 | 19 | 284 | 21 | 307 | 29 |
| Dissolved oxygen concentration (O2 mg/L) | 9.84 | 0.62 | 6.47 | 0.71 | 9.64 | 0.49 | 9.39 | 0.90 |
| Oxygen saturation (%) | 101.56 | 5.93 | 64.90 | 7.27 | 97.59 | 3.84 | 95.91 | 9.49 |
| Biological oxygen demand (O2 mg/L) | 1.59 | 0.86 | 1.09 | 0.91 | 1.42 | 0.84 | 1.48 | 0.90 |
| Chemical oxygen demand (O2 mg/L) | 6.33 | 5.90 | 5.87 | 5.30 | 5.60 | 5.49 | 5.50 | 5.27 |
| Ammonium concentration (N mg/L) | 0.028 | 0.021 | 0.046 | 0.057 | 0.020 | 0.008 | 0.085 | 0.120 |
| Nitrite concentration (N mg/L) | 0.038 | 0.142 | 0.046 | 0.147 | 0.045 | 0.170 | 0.044 | 0.138 |
| Nitrate concentration (N mg/L) | 0.793 | 0.192 | 0.595 | 0.178 | 0.769 | 0.175 | 0.753 | 0.213 |
| Total nitrogen concentration (N mg/L) | 1.217 | 0.422 | 0.829 | 0.447 | 1.105 | 0.273 | 1.114 | 0.352 |
| Total phosphorous concentration (P mg/L) | 0.062 | 0.050 | 0.044 | 0.048 | 0.046 | 0.032 | 0.054 | 0.040 |
| Orthophosphate concentration (P mg/L) | 0.017 | 0.007 | 0.020 | 0.016 | 0.015 | 0.003 | 0.025 | 0.024 |
| Phenol concentration (mg/L) | 0.0061 | 0.0045 | 0.0053 | 0.0030 | 0.0073 | 0.0062 | 0.0082 | 0.0104 |
| Pesticide concentration (µg/L) | 0.0033 | 0.0024 | 0.0031 | 0.0024 | 0.0031 | 0.0025 | 0.0032 | 0.0026 |
| Mineral oil concentration (mg/L) | 0.016 | 0.019 | 0.017 | 0.023 | 0.020 | 0.029 | 0.016 | 0.024 |
| Faecal coliform bacteria (nr/1000 mL) | 4610 | 5922 | 20,728 | 116,604 | 2975 | 4459 | 14,449 | 37,149 |
| Habitat Type | Reservoirs | Drainage Ditches | Tailrace Canals | Old River Channels | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Site | C1 | C1A | C2 | D1 | D2 | V1 | V2 | C3 | C4 | D3 | D4 | C5 | D5 | V3 | C6 | D6 | V4 |
| Platycnemis pennipes (Pallas, 1771) | 6.7 | 1.7 | 3.3 | 3.3 | 6.7 | 30.0 | 15.6 | 1.7 | |||||||||
| Calopteryx splendens (Harris, 1782) | 1.7 | ||||||||||||||||
| Calopteryx virgo (Linnaeus, 1758) | 1.7 | 3.3 | |||||||||||||||
| Coenagrionidae non det. | 6.7 | 1.7 | |||||||||||||||
| Ischnura elegans Vander Linden, 1820 | 2.8 | 1.7 | |||||||||||||||
| Erythromma najas Hansemann, 1823 | 1.7 | ||||||||||||||||
| Erythromma lindenii (Selys, 1840) | 11.7 | ||||||||||||||||
| Enallagma cyathigerum (Charpentier, 1840) | 11.7 | ||||||||||||||||
| Gomphus vulgatissimus (Linnaeus, 1758) | 1.7 | ||||||||||||||||
| Onychogomphus forcipatus (Linnaeus, 1758) | 5.0 | 1.7 | 5.0 | 6.7 | 10.0 | 15.0 | |||||||||||
| Orthetrum brunneum (Fonscolombe, 1837) | 1.7 | ||||||||||||||||
| Orthetrum cancellatum (Linnaeus, 1758) | 6.7 | ||||||||||||||||
| Taxa richness | 3 | 1 | 0 | 3 | 0 | 0 | 0 | 0 | 0 | 1 | 1 | 1 | 1 | 2 | 4 | 5 | 5 |
| Abundance | 13.4 | 1.7 | 0.0 | 11.7 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 1.7 | 1.7 | 3.3 | 6.7 | 8.3 | 60.1 | 36.8 | 21.8 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vilenica, M.; Mihaljević, Z. Odonata Assemblages in Anthropogenically Impacted Habitats in the Drava River—A Long-Term Study. Water 2022, 14, 3119. https://doi.org/10.3390/w14193119
Vilenica M, Mihaljević Z. Odonata Assemblages in Anthropogenically Impacted Habitats in the Drava River—A Long-Term Study. Water. 2022; 14(19):3119. https://doi.org/10.3390/w14193119
Chicago/Turabian StyleVilenica, Marina, and Zlatko Mihaljević. 2022. "Odonata Assemblages in Anthropogenically Impacted Habitats in the Drava River—A Long-Term Study" Water 14, no. 19: 3119. https://doi.org/10.3390/w14193119
APA StyleVilenica, M., & Mihaljević, Z. (2022). Odonata Assemblages in Anthropogenically Impacted Habitats in the Drava River—A Long-Term Study. Water, 14(19), 3119. https://doi.org/10.3390/w14193119








