1. Introduction
During the recent decade, the different types of nanoparticles (NPs) have extensively used worldwide [
1,
2,
3,
4,
5]. NPs are defined as materials with a size range of 1–100 nm. NP properties depend on physicochemical characteristics [
6], and some are produced in large scales due to their unique properties [
7]. The most common NPs for industrial use are based on silver particles, with around 300 different products [
8]. The production and use of silver nanoparticles (Ag-NPs) has significantly increased and will increase up to 63% until 2024 due to their efficiency [
9]. Studies have reported an antibacterial efficiency of Ag-NPs [
10,
11,
12,
13]. Ag-NPs are utilized as an antibacterial agent in different industries, but leakage of Ag-NP products during washing and trough contaminate waters can get into the environment [
14]. In other words, novel technologies provide better opportunities for human life and health, but these may cause risks for the environment. Increased use of NPs and other chemical materials has increased concerns of their exotoxin effects [
15]. Aquatic animals are exposed to Ag-NPs via food, water and sediments [
11]. Ag in ionic form induces toxicity for bacteria and can affect aquatic organisms, both freshwater and marine fish, in low concentrations [
14]. Since fertilization in fish happens in external environments, Ag-NPs may influence reproductive success [
16]. Several studies have reported accumulation of metal NPs in fish organs, followed by changes in homeostasis and reproductive performance [
17,
18,
19]. Studies have reported that accumulated Ag-NPs in tissues can induce oxidative stress [
20]. Toxicological effects of Ag-NPs on aquatic organisms were reported in
Oreochromis niloticus [
6], rainbow trout [
16], and common carp [
11]. However, information about the toxicity of several pollutants (such as pesticides and heavy metals) has limitations because their toxicity is highly dependent on the environmental conditions, species, age, size and diet of the organism [
21,
22,
23,
24,
25].
Poecilia sphenops belongs to the Poecilidae family and the Actinopterygii class. Females are bigger than males [
26].
P. sphenops produce broods of 10–140 live young, and total numbers of broods depend on the maturity and size of the female [
27]. This fish inhabits freshwater systems. The gestation period is 3–4 weeks, depending on temperature and stored sperm [
26].
Serum biochemical parameters are important biomarkers for showing the health status of fish [
28,
29,
30,
31]; in addition, hematology is a key tool for assessing the health of different species because it is a reliable and non-lethal index [
32,
33]. The LC
50 96 h test is based on studies that aim to examine the toxic effect of pollutants on aquatic organisms [
34,
35]. In the current study, we investigated the ecotoxicological effects of Ag-NPs on
P. sphenops trough examination of the survival rate of fish, as well reproductive-system and blood-biochemical parameters.
2. Materials and Methods
2.1. Ethical Standard
The procedures employed for rearing, toxicity and sampling were in agreement with protocols recommended by the ethical committee of Gorgan University of Agricultural Science and Natural Resources (Ethical number 19861). Fish were acclimated with methods for acute toxicity tests as suggested for fish, macroinvertebrates and amphibians [
36,
37,
38].
2.2. Preparation
At the beginning, 350 females and 50 males mature common molly (Poecilia sphenops) were bought from ornamental fish stores in Rasht (Guilan, Iran) and transferred to the laboratory (Faculty of Natural Resources, University of Guilan, Sowmehsara, Guilan, Iran). Male fish were just used for fertilization of the females. Females were distributed into 15 aquariums (100 cm × 40 cm × 50 cm) and acclimatized to the laboratory condition for 2 weeks. Fish were fed at the rate of 2% of body weight twice a day with a commercial diet (Ocean Nutrition Breeder, Ocean Nutrition Co., Newark, CA, US). The average weight of fish was 5 ± 0.5 g. Long-term exposure time was 62 days. Water physicochemical properties included temperature 27 ± 1 °C, pH 7–7.8, NH3 < 0.02 mg L−1, BOD 850 ± 50 mg L−1,total hardness 200 mg CaCO3 and NaCl < 1 mg L−1. Nanocid L-2000 (Nanonasb Pars Co., Tehran, Iran) was prepared as a common commercial colloidal solution of silver nanoparticles (Ag-NPs). Distilled water was used as solvent, and silver density was 1000 mg L−1. According to product specifications of Nanocid, the size of silver nanoparticles was between 20 and 30 nm.
2.3. Acute Toxicity Test for Adult Fish
Acute toxicity was conducted as reported by previous studies [
4,
8]. Female fish were divided into eight treatments (0, 5, 10, 15, 25, 35, 45 and 60 mg L
−1 of Ag-NPs) with 3 replications. Fish were maintained in 24 aquariums (100 cm × 40 cm × 50 cm) filled with 80 L water. There were 7 fish in each aquarium. The mortality rate for mature fish was calculated at interval times of 24, 48, 72 and 96 h after addition of Ag-NPs [
39,
40]. Before the addition of Ag-NPs, water physicochemical properties were controlled. Nominal concentrations of Ag-NP suspension were dissolved in distilled water and added.
2.4. Reproductive Parameters
Reproductive parameters were investigated as reported by previous studies [
8]. In summary, 20 pairs of adult fish were divided into five groups (0, 3, 5, 10 and 15 mg L
−1 of Ag-NPs) and four replications for 62 days. The concentrations were selected based on LC
50 for 96 h. We expected that concentrations would not cause mortality but induce physiological effects in the test time [
41]. The water source was replaced daily, and water physicochemical properties were constantly checked. Female fish were kept under a lighting regime of 16 h light and 8 h dark [
42]. Reproductive success was considered as the number of viable larvae calculated for each pair until the last larva. The survival rate for larvae was calculated at time intervals of 24, 48, 72 and 96 h after birth of the first larvae. Parturition time of fish was considered from the start of the 62-day period to birth of the last larvae. We used 14 cameras (Axton Wi-Fi camera Model M9021W, North Salt Lake, Utah, US) to record behaviors and movement of fish in each group, and the obtained data were collected daily and reviewed by Adobe After Effect software (AAE CS6).
2.5. Acute Toxicity Test for Larvae
Following determination of LC50 96 h of Ag-NPs for mature individuals, 105 larvae (12 h after birth) were submitted to different concentrations of Ag-NPs. One-day larvae were divided into five groups. Nominal concentrations of Ag-NPs (0, 3, 5, 10 and 15 mg L−1) were selected based on the acute toxicity of Ag-NPs for mature fish, in agreement with THE mortality rate of mature fish. Mortality rate was investigated in time intervals of 24 h from 24–96 h after the addition of Ag-NPs. Water physicochemical properties were checked.
2.6. Blood Parameters
On day 63 and after one day of harvesting, blood samples were collected from each group (five female fish) using a syringe and via the stem vein. Blood samples were centrifuged at 3000 rpm for 6 min, and sera were investigated for total protein, albumin, cholesterol and triglyceride using commercial kits of Pars Azmoon (Pars Azmoon corp, Tehran, Iran) with the help of an auto-analyzer (Eppendorf, EPOS, Hamburg, Germany). Glucose was measured by the glucose oxidase methods as reported by previous studies [
43]. Parts of blood samples were transferred into heparinized vials for assessment of red blood cell (RBC) count, hematocrit and white blood cell (WBC) count, as reported by previous studies [
44].
2.7. Data Analysis
Lethal concentrations (in 50% of the population) in time intervals of 24 h were investigated by using a probit analysis with a 95% confidence rate. Data for normality were investigated by Kolmogorov-Smirnov test. Since reproductive and blood parameters were normally distributed, the data were analyzed by one-way ANOVA following post-hoc LSD test. Data were reported as mean ± SD, and all analyses were run using SPSS software (version 20).
4. Discussion
Aquatic environments (rivers, lakes, seas) are often the last destination of many pollutants, among them, Ag-NPs [
45]. Several studies have reported that Ag-NPs cause mortality in adult fish and their larvae [
46,
47,
48]. Sohn et al. [
49] suggested that 1.8 mg L
−1 can be used as a mean value for lethal concentrations of Ag-NPs for Japanese rice fish (
Oryzias latipes) as a model of freshwater fish. The reported concentration was lower than values obtained in the current study (26.85 mg L
−1 and 6.22 mg L
−1 for adult fish and larvae of common molly, respectively). The reason for this difference could be the dissimilarity of the test parameters. Toxicity of Ag-NPs may be dependent on the environmental or test conditions, species, age, size and diet of the organism [
21,
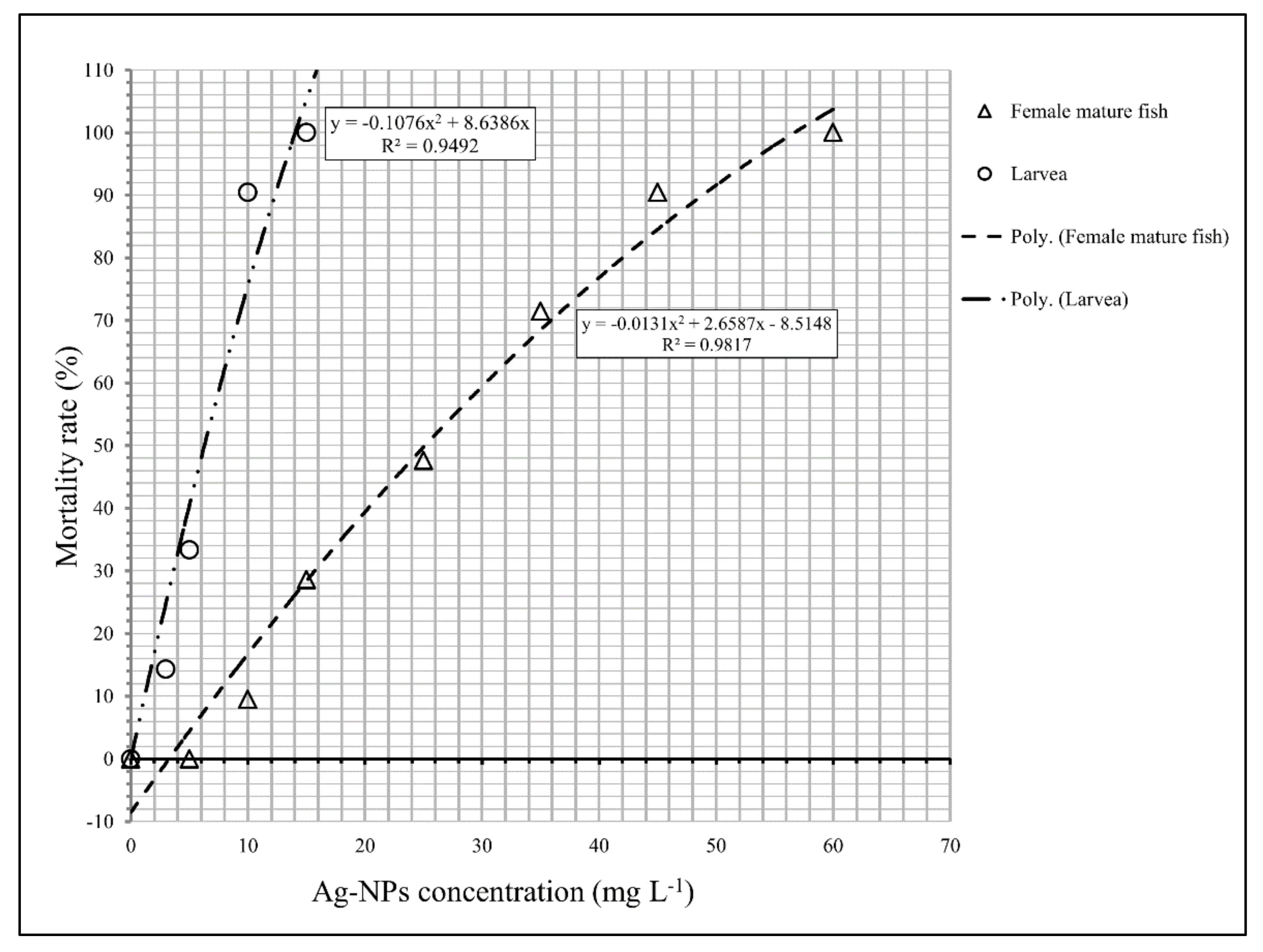
23]. Furthermore, other reasons of this difference could be solvent of commercial suspension used in the present study (Ag-NPs, Nanocid L-2000, Nanonasb Pars Co., Tehran, Iran), which, according product specifications, was distilled water. Current study results show that Ag-NPs had a slight toxicity effect to the female fish and a moderate toxicity effect on the larvae (
Table 6). Based on these findings, larvae were more susceptible to Ag-NPs, which is in agreement with results reported by a previous study on CuO-NPs [
50]. Massarsky et al. [
51] stated that Ag-NPs display an ability to accumulate in some tissues and induce oxidative stress, which can result in increased mortality. Authors have also shown increased intercellular production of reactive oxygen species (ROS), as well as the destruction of DNA structure after submission to Ag-NPs. Ag-NPs can decrease WBCs and affect immune-system responses.
Our findings show that Ag-NPs prolonged parturition time and reduced reproductive success and survival rate. Several studies have reported that accumulation of metal NPs induces changes in reproductive performance [
17,
18,
19]. Johari [
16] reported that Ag-NPs decrease fertilization and reproduction success of rainbow trout, while another study showed that Ag-NPs did not have significant effects on fertilization capability of sea urchin sperm, although they caused morphological changes in their larvae [
52]. Exposure of fish to NPs can have various negative effects on survival rate [
50,
53], such as inducing oxidative stress, apoptosis, inflammation and genotoxicity [
54]. Seemingly, Ag-NPs are able to induce toxic changes in common molly with negative effects on the reproductive system.
Hematological parameters (RBC, WBC and hematocrit) were significantly reduced in fish exposed to Ag-NPs, especially at higher concentrations. Results for negative effects of Ag-NPs on hematology parameters are in agreement with previous studies [
55]. RBCs have an important role in supplying required oxygen to the body’s cells, and hematocrit is known as the volumetric percentage of RBCs in the blood that is influenced by the number and size of RBCs. Ag-NPs enter into the bloodstream via the gastrointestinal tract or gill membrane and can cause accumulation of RBCs [
56], leading to oxidative stress [
57]. Studies have reported toxicological effects of Ag-NPs on hematological parameters via increased peroxidation [
58], induction of damage in hematopoietic tissues [
59] and morphological changes in RBCs [
60]. With regards to negative effects of Ag-NPs on WBC, it was reported that Ag-NPs induced intracellular cytotoxicity via the release of toxic Ag
+ into immune cells [
61], as well as by induction of depolarization in the mitochondrial membrane and inactivation of enzymes [
62].
Results from this study show that Ag-NPs lowered the serum concentration of glucose while increasing the serum concentrations of glucose, triglycerides, albumin and total protein. Similar results were reported by a previous study [
11], while in another study, conflicting results were observed [
6]. The serum concentrations of total protein, albumin and glucose are good markers for the immunological and nutritional condition of fish. Albumins are the most abundant proteins in blood that regulate osmotic pressure of blood, also acting as a carrier for external compounds. Blood glucose is severely affected by stressors. Changes in serum concentrations of protein might be a good index for immunity status and liver health [
63,
64,
65]. As mentioned, Ag-NPs in organisms induce stress conditions and increase the levels of reactive oxygen species. During stress, the body consumes glucose and releases proteins and lipids into blood as energy sources. Stress increases the secretion of cortisol hormones that increase glucose production from liver glycogen [
11] that might be consumed and destroyed in blood flow. The increased concentration of serum triglycerides, cholesterol and protein in fish submitted to Ag-NPs might be attributed to the use of these compounds for energy supply in stressed fish [
66,
67].
Even though LC50 at 96 h was around 25 mg/L, our results show that much lower concentrations can have profound negative impacts on the reproductive and blood parameters of fish. However, concentrations used were still lower in order to estimate concentrations of total silver in water in natural environments, ranging from 0.09 to 320 μg L
−1 [
68]. Due to the uncertainty of these estimates and the widespread use of nanoparticles in various industries, concerns about their further spread in aquatic environments are increasing [
69] because, as mentioned, aquatic environments are the last destination of pollutants [
45].