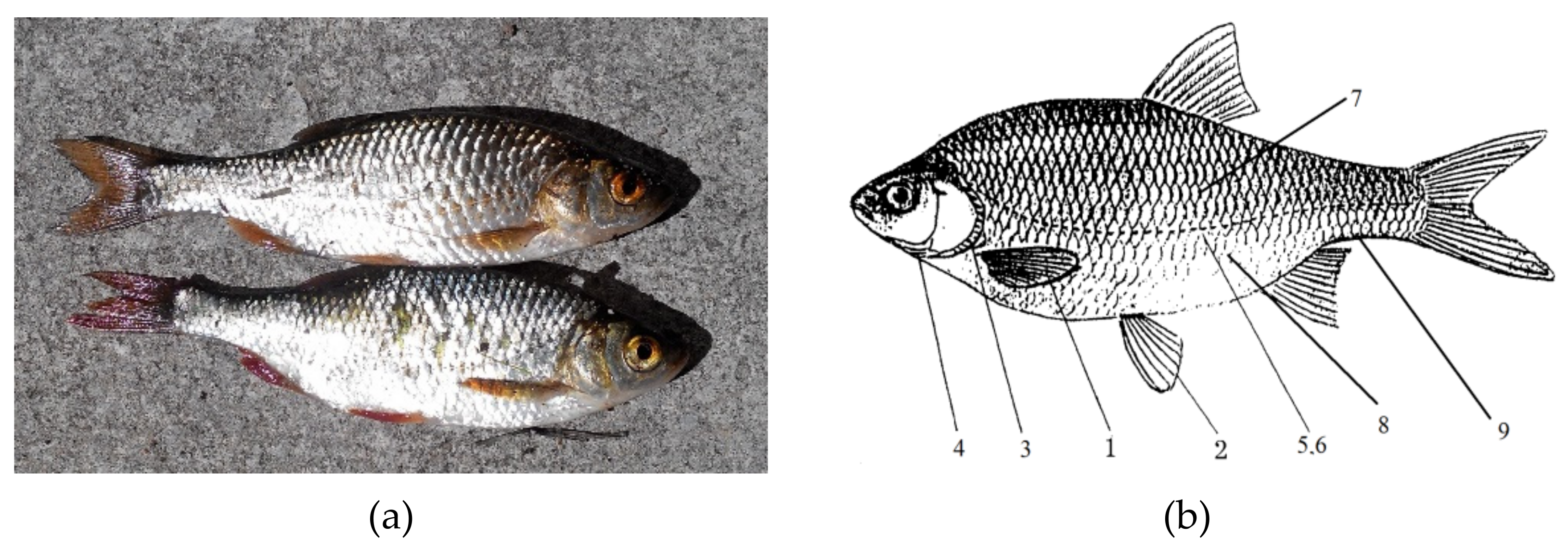
3.3. Evaluation of the Frequency of Fluctuating Asymmetry of Fish Populations in the Studied Rivers
To determine the FFA of the fish populations in this study, fish aged up to 1 year were evaluated. This was due to the fact that the active physiological development of fish occurs in the first 6 months of life. It is generally accepted that asymmetric manifestations can be caused by stress in fish. Most often, this stress originates from polluted water, i.e., water with increased concentrations of certain chemical components.
The calculated values of fluctuating asymmetry in the samples for each studied fish population in the Sluch River are shown in
Table 3 and
Table 4.
Thus, at both sampling site No. 1 and sampling site No. 2, the highest
FFA was observed in common roach (0.42 and 0.45, respectively). The lowest
FFA was found in silver crucian carp (0.13 and 0.15, respectively). The results of this study (
Table 2 and
Table 3) showed that the
FFA was different for most of the analyzed fish populations in the Sluch River. The highest
FFA for all studied fish populations was found in the gill stamens on the first gill arch (
sp.br), and the lowest
FFA was observed in the number of rays on the caudal fin (
squ.pl).
The difference in the FFA of various fish may have been related to the impact of various factors. The generally accepted theory is that changes in fish FFA are often influenced by water quality chemistry. The value of the FFA index can also be influenced by other factors that were not studied in this research (turbidity, river depth, population density (species/m3), features of the trophic chain, etc.). The level of influence of various factors on FFA may be investigated in future studies.
The
FFA results of the studied fish populations in the Ustya River are presented in
Table 5,
Table 6 and
Table 7. At sampling site No. 3, the highest
FFA values were observed in common rudd (0.40), common break (0.38), and common roach (0.36). At sampling site No. 4, the highest
FFA values were found for common roach (0.53) and common bleak (0.46). At sampling site No. 5, the highest
FFA values were determined for common bleak and common roach (0.48 and 0.46, respectively).
The analysis of the obtained data (
Table 4,
Table 5 and
Table 6) showed that the highest levels of
FFA were observed in the number of gill stamens on the first gill arch
(sp.br.) at sampling sites Nos. 3–5 for all studied fish populations, as well as the number of rays on the pectoral fin (
P) and the number of scales on the lateral line (
jj).
The third studied river was the Styr River, for which the
FFA results of the selected fish populations are presented in
Table 8,
Table 9 and
Table 10. The analysis of the data presented in these tables showed that the highest
FFA values at sampling site No. 6 were 0.39 for common bleak and 0.37 for common roach. The same pattern was observed at sampling site No. 7. At sampling site No. 8, the highest
FFA values were 0.37 for common roach and 0.33 for common bleak. For most of the analyzed fish species at all sampling sites in this river, the highest
FFA values were observed in the following features: the number of rays on the pectoral fin (
P), the number of rays on the pelvic fin (
V), and the number of gill stamens on the first gill arch (
sp.br).
After laboratory studies, six datasets were generated that correspond to each of the six studied fish populations. The datasets were formed into matrices that contained 480 rows and 15 columns (14 columns for the input variables (SS, NH4+, NO3−, NO2−, PO43−, BOD5, O2, SO42−, Cl−, Fetotal, Zn2+, Mn2+, FFA) and one column for the target output data (water quality)).
As an example, the creation and training of an ANN model for data obtained from a population of common bleak are shown. The database was split into three sets: training (60%), validation (20%), and testing (20%). The next step was training, validating, and testing the ANN models. The trial testing of the ANN models was carried out with the number of epochs equal to 20, 50, and 100. It was expected that the optimal values would be obtained at the level of 100 epochs. Increasing the number of epochs over 100 did not lead to an improvement in model performance.
Table 11 shows the parameters of the ANN models. The values of
R2 and
MAPE at 20 epochs are listed for comparison purposes.
A comparative analysis of the nine neural network models showed that the ANN 4 model had the best results (with the lowest value for the average absolute error percentage and the highest value for the coefficient of determination). This neural network used the SGD optimization algorithm and the ReLU activation function. The best MAPE and R2 values were obtained at the 100th epoch and were equal to 6.7% and 0.97187, respectively.
The following analysis refers to the ANN 4 model and presents an assessment of the adequacy of the results obtained and the prospects for the model’s use for forecasting with new datasets.
Figure 4 shows the comparison of the mean square error results for the validation and training sets. The highest MSE was obtained on the first epoch: training dataset—2.2680; validation dataset—1.3495. The lowest MSE was obtained on the 100th epoch: training dataset—0.00047; validation dataset—0.1592.
Figure 5 shows a comparison of the MAE for the validation and training sets. The highest MAE was obtained on the first epoch: training dataset—1.0000; validation dataset—0.7846. The lowest MSE was observed on the 100th epoch: training dataset—0.0125; validation dataset—0.1927.
The error histogram (
Figure 6) shows the distribution of data sampling errors.
According to the obtained histogram, we could conclude that the errors obeyed the normal distribution law, i.e., we could divide the errors into three regions (for simplicity, the distribution was considered to be normalized): +σ1 → range [0.0, 0.29]; +σ2 → range [0.3, 0.59]; +σ3 → range [0.6, 0.89]. The vast majority of errors fell within the +σ1 range, although there were also anomalous errors.
In their study, Rizal, Hayder, and Yusof aimed to develop artificial neural network models to predict six different water quality parameters. All the ANN models achieved high coefficients of determination (
R2), which ranged from 0.9906 to 0.9998 and from 0.8797 to 0.9972 for the training and testing datasets, respectively [
58].
Li et al. used a backpropagation neural network, radial basis function neural network, support vector machine, and least squares support vector machine to simulate and predict water quality parameters including dissolved oxygen, pH, ammonium-nitrogen, nitrate nitrogen, and nitrite nitrogen [
59]. The results showed that the support vector machine achieved the best prediction performance, with an accuracy of 99%. This model was recommended for simulating and predicting water quality.
Khoi et al. used a case study to evaluate the performance of twelve machine learning models in estimating surface water quality [
60]. The water quality index was calculated based on water quality data at four monitoring stations for the period 2010–2017. The prediction performance of the machine learning models was evaluated using two efficiency indicators (
R2 and RMSE). The results indicated that all twelve ML models had good performance in predicting the water quality index, but that extreme gradient boosting achieved the best performance (
R2 = 0.989 and RMSE = 0.107).
Thus, by comparing the error indicator and R2 values of the ANN model developed in the current study (MAPE = 6.7%; R2 = 0.97187) with those of models from other studies, we can state that the level of agreement between the predicted and target data was satisfactory.
Various recent studies have shown that fish can be used as bioindicators [
46,
53]. The analysis of the health status of fish, their morphological traits, and other indicators is often a good monitoring tool, especially for assessing the physical and chemical pollution of surface water bodies [
39,
54]. At the same time, researchers have distinguished between highly sensitive fish (salmon, trout, common roach, zander, char, common bleak, and ninnow); medium-sensitivity fish (common bream, European perch, and common rudd); and low-sensitivity fish (silver crucian carp and common carp) [
61,
62,
63,
64].
A study of selected fish populations in the Sluch, Ustya, and Styr rivers showed that the highest FFA values corresponded to common roach and common bleak, and the lowest to silver crucian carp. If we compare the FFA values of these fish in the studied rivers, then common roach and common bleak had the highest values in sampling sites 3–5 of the Ustya River. This could be explained by the fact that the water quality in these sections of the river was the worst of all the studied sections. The chemical indicators of water quality that significantly exceeded their allowable levels were SS, NO2−, NH4+, BOD5, Fetotal, Zn2+, and Mn2+. The excessive levels of the above indicators in the Ustya River in the period June–August suggests an active decay processes at the bottom of the reservoir. Rotting activity is associated with increased water and air temperatures in the summer. The period under study included the months, especially August, when the processes of vegetation by micro- and macrophytes are almost complete. At this time, plants cease to actively consume metals such as Fetotal, Zn2+, and Mn2+, and so their concentrations become significantly increased.
The increased content of NO2− and NH4+ and the pH value of 8 indicated that the process of denitrification was taking place in the water. During the period of this study, the active growth of macrophytes was observed, causing the presence of soluble oxygen in the water. The analysis of the chemical composition of the water in the Ustya River showed that the concentration of soluble oxygen varied from 7.5 to 10.2 mgO2/dm3. It is known that at such oxygen concentrations, the process of denitrification occurs rather slowly. Thus, the intensification of the denitrification process should not be encouraged, since a decrease in the oxygen concentration can lead to the death of all aerobic organisms.
Fish are usually found at the highest trophic level in freshwater habitats and absorb pollution from the environment they live in [
65,
66]. Eating contaminated fish can be a public health risk.
Töre et al. determined the probable public health risks posed by heavy metals (Mn, Cd, Fe, Cu, Ni, Zn, Co, Pb, Cr, and As) in 72 muscle and gill samples from six fish species. The highest muscle tissue levels for eight of the ten heavy metals measured were found in the C. macrostomus species. The results showed that the intake of the analyzed fish species might present a toxicological hazard and threaten community health [
67]. The daily consumption of such fish could have harmful effects on human health. Therefore, the assessment of the
FA level of the most popular fish species in the studied rivers is of great importance. The fish with the most pronounced
FFA could serve as a biological indicator of the local situation.
In regard to the level of pollution of the Ustya River as a whole, it can be noted that at this moment the pollution is not at a critical level. However, given that this situation may change over time, it is necessary to apply the right usage policies to the river and regularly monitor its pollution parameters. The studied rivers are mainly of aesthetic and recreational value, and they are also actively used by local residents for amateur fishing.
However, for the effective management of water resources, i.e., to ensure the health of fish populations and maintain the marginal concentrations of biogenic elements and heavy metals, it is advisable to use established technological methods. In many countries, natural limestone is applied for this purpose. Adding limestone to natural surface waters can slow down the process of decay at the bottom of the reservoir. As evidenced by studies of this practice, in summer it is able to reduce the intensity of eutrophication, i.e., the concentration of PO
43− [
68] and heavy metals [
69]. During the dissociation of limestone, the pH value of the water increases, which makes it possible to slow down the processes of decay in the bottom of the reservoir and thus increase the aesthetic and recreational value of the river.