Abstract
Membrane-based technologies have been widely used for surface water treatment. Yet, many aspects of this technology can still be improved. This study aims to develop polyethersulfone (PES)-based phase-inverted membranes to improve the morphological structure, antimicrobial properties, and performance by incorporating Poloxamer 188 and patchouli oil as the dope solution additives. The performance of the membrane was assessed for filtration of well water and by evaluating the turbidity rejection. This study used a phase inversion technique in the membrane manufacturing process with PES, PES + P188 + 1 wt% PO, PES + P188 + 3 wt% PO, and PES + P188 + 7 wt% PO. The characteristics of the obtained membranes were studied in terms of structure and morphology, microbial growth prevention, hydrophilicity, filtration flux, and ability to reduce the turbidity of well water samples. Results show that the addition of Poloxamer 188 and patchouli oil in the dope solution turned the membrane more porous (up to 73.24% increase in porosity) and more hydrophilic (the water contact angle (WCA) was lowered from 70 to 37°). The additives also increased the antibacterial properties of the membrane, as shown by up to 97.5% reducing Escherichia coli colonies on the membrane surface. Overall, the results demonstrate significant improvements in the characteristics and performance of PES membranes by incorporating Poloxamer 188 co-polymer and patchouli oil as additives in the dope solution. The modified membrane was successfully applied to remove turbidity from a water sample. The turbidity parameters in well water samples could be fully reduced in nine out of ten samples by the membrane containing 7 wt% PO additives.
1. Introduction
Water is one of humans’ basic needs as its function is very complex in various aspects of life. Efforts have been made to ensure clean water delivery of adequate quantity and quality. Natural water processed into drinking water must meet the standard quality of drinking water from the chemical, physical, and biological aspects. Meanwhile, population growth impacts the economy, technology, industrialization, and the rate of urbanization, all of which contribute to environmental pollution and lead to a decrease in the quality of surface water and groundwater. The delivery of clean water is the focus of the researchers because the challenge will escalate along with population growth.
Water purification removes impurities such as metal ions, organic compounds, suspended solids, and biological and chemical contaminants from water. Water treatment is generally carried out through coagulation, flocculation, and filtration process concluded by disinfection. The treatment can involve a more advanced process for the removal of persistent organic pollutants such as polycyclic aromatic hydrocarbons (PAHs) [1], synthetic organic chemicals such as organophosphate esters [2], and other natural organic materials that can harm human health [3]. In the last decade, membrane separation technology such as microfiltration, ultrafiltration, and nanofiltration has been established as the primary technology for water treatment [4,5,6]. In recent years, several membrane modifications have been updated to improve the quantity and quality of the water product [7,8,9]. Separating contaminants in water by membrane technology can save operational and maintenance costs because of several advantages: low area footprint, continuous operation, and simple assembly and operation [10,11].
The materials commonly used for membrane fabrication are polymers. Some polymeric materials used in the membrane manufacturing process include polyethersulfone (PES), cellulose acetate, polysulfone (PS), polyvinylidenflouride (PVDF), and polyethylenethereptalate (PET) [12,13], with PES being the most used polymer. PES membranes often suffer from the fouling rate caused by organic pollutants through the formation of an adsorption layer and/or cake on the membrane surface. Adding inorganic additives such as metal oxide nanoparticles in polymeric membranes can reduce the hydrophobic interactions between organic foulants and membrane material effectively without compromising its mechanical strength. PES-based membranes are widely used because they can be made using phase inversion and have good mechanical and chemical resistances. However, PES also has drawbacks, one of which is its hydrophobicity [14]. As demonstrated from earlier works, this deficiency can be overcome by blending some additives to improve those PES-based membrane characteristics. Additives affect the physical condition and performance of the resulting membranes [15]. Several additives have been used to increase the hydrophilicity of PES-based membranes. They include CuO nanoparticles [16], TiO2 [17,18], and polyvinyl pyrrolidone [19]. These additives can change the properties of the membrane and improve its performance [20]. In addition, an additive act as a pore-forming agent that can reduce the occurrence of fouling and increase the permeation [21]. Changes in the membrane’s properties and morphology after the additive blending have been reported. Those changes enhanced their performance in the treatment of clean water or wastewater. Most of the earlier works focused on improving the membrane properties to increase the permeability and rejection while neglecting the antimicrobial property.
This paper assesses the changes in PES membranes’ properties and morphological structure with a hybrid additive by combining a hydrophilic carrier in Poloxamer 188 (P188) and an antimicrobial properties carrier in patchouli oil (PO). P188 is a nonionic surfactant often used as a material change agent in the pharmaceutical field. It consists of a polyoxyethylene/polyoxypropylene/polyoxyethylene triblock co-polymer. Poloxamer consists of a central hydrophobic polyaxpropylene (POP) chain and two identical lateral hydrophilic polyaxtylene (POE) chains. This poloxamer co-polymer with a molecular weight of 7680–9510 g/mol has high hydrophilicity, indicated by a high Hydrophilic-Lipophilic Balance (HLB) number of 29 [22]. In this study, membrane modification was prepared using PES and P188 polymers and patchouli alcohol compounds. The latter comprises oxygenated hydrocarbon groups that possess antibacterial properties and are insoluble in water but soluble in alcohol, ether, and other organic solvents [23]. Using PO compounds as additives has many benefits, one of which is as an antibacterial property. PO has a high bacterial inhibition zone, reaching 21.5% [24]. PO can inhibit the growth of Staphylococcus aureus, MRSA, Streptococcus pyogenes, Pseudomonas aeruginosa, and Candida albicans bacteria. The chemical compounds in PO that possess antibacterial properties from patchouli plants are patchoulol, β-guaieno, gurjunene-α, α-guaiene, aromadendren, and β-Patchoulene [25]. Based on those findings, this study explored PO potential as an antimicrobial additive for PES-based phase-inverted membranes. The aim of the current study is to develop PES-based phase-inverted membranes to improve the morphological structure, antimicrobial properties, and separation performance by incorporating P188 and PO as the dope solution additives.
2. Materials and Method
2.1. Materials
The materials used in this study were polyethersulfone (PES Mw: 65 kDa, Ultrason E6020 P, BASF, Ludwigshafen, Germany), Poloxamer 188 (P-188) (99%, WAKO, Osaka, Japan) as the co-polymer, PO (Atsiri Research Center, U.S.K., Banda Aceh, Indonesia) as the additive, N-Methyl Pyrrolidone (NMP) as the solvent obtained from WAKO, Pure Chemical Industries, Ltd. (Osaka, Japan), and deionized water as the non-solvent.
2.2. Synthesis of Membrane
The dope solution was prepared by dissolving the patchouli oil additive into the NMP solvent for 20 min, after which P188 was added and stirred for another 20 min. Then, PES was added to the solution and stirred for 24 h at room temperature to form a bubble-free homogeneous solution. The dope solution consisted of a fixed PES concentration of 16 wt% with varying concentrations of additives (0, 1, 3, 5, and 7 wt%) and co-polymers P-188 of 0 and 3 wt%. The homogeneous dope solution was then cast on a glass plate using a casting applicator (Yoshimatsu, Japan) at 300 μm wet thickness. The glass plate and the casting solution were immersed in a coagulation bath containing deionized water as a non-solvent. At this stage, solidification occurred: a process that transformed the polymer from its liquid to a solid phase. Each membrane was then rinsed in deionized water to remove the residual solvent before characterization.
2.3. Morphological Structure and Surface Elements Analysis
The structure and morphology of the prepared membranes were analyzed using scanning electron microscopy (SEM) (Jeol, JSM-6360LA, Tokyo, Japan). Dry membrane samples were put directly into the SEM tube. In contrast, the membrane samples were cut while immersed in liquid nitrogen for the cross-section structural analysis. All samples were then coated with osmium powder through a coating process.
2.4. Functional Group Analysis
Functional group analysis was carried out to identify compounds and functional groups of chemical components that formed the membrane matrix. Functional group analysis was conducted using a Fourier transform infrared (FTIR) instrument (Shimadzu IRPretige-21, Shimadzu Europa GmbH, Duisburg, Germany) using infrared rays.
2.5. Hydrophicility Analysis
Hydrophilicity analysis was aimed at determining surface hydrophilicity. The membrane hydrophilicity was found by measuring the WCA on the membrane surface. Measurements were carried out by dripping water on the membrane surface, optically capturing the droplet using a smartphone camera (Samsung S22, Gyeonggi-do, Korea), and then measuring the angle.
2.6. Membrane Porosity
The porosity test was performed to determine the membrane samples’ void fraction and evaluate the additives’ effect. The membrane porosity was tested by measuring the membrane thickness and wet weight, after which they were dried in an oven at 50 °C for 3 h. The dry weight of the membrane was weighed again. The porosity of the membrane [26] was calculated using Equation (1).
where Ww is the weight of a membrane sample in a wet state (g), Wd is the weight of a dry condition of the same membrane sample (g), ρ is density (g/cm3), A is membrane surface area (cm2), and 1 represents the membrane thickness (cm).
2.7. Clean Water Flux and Rejection Test
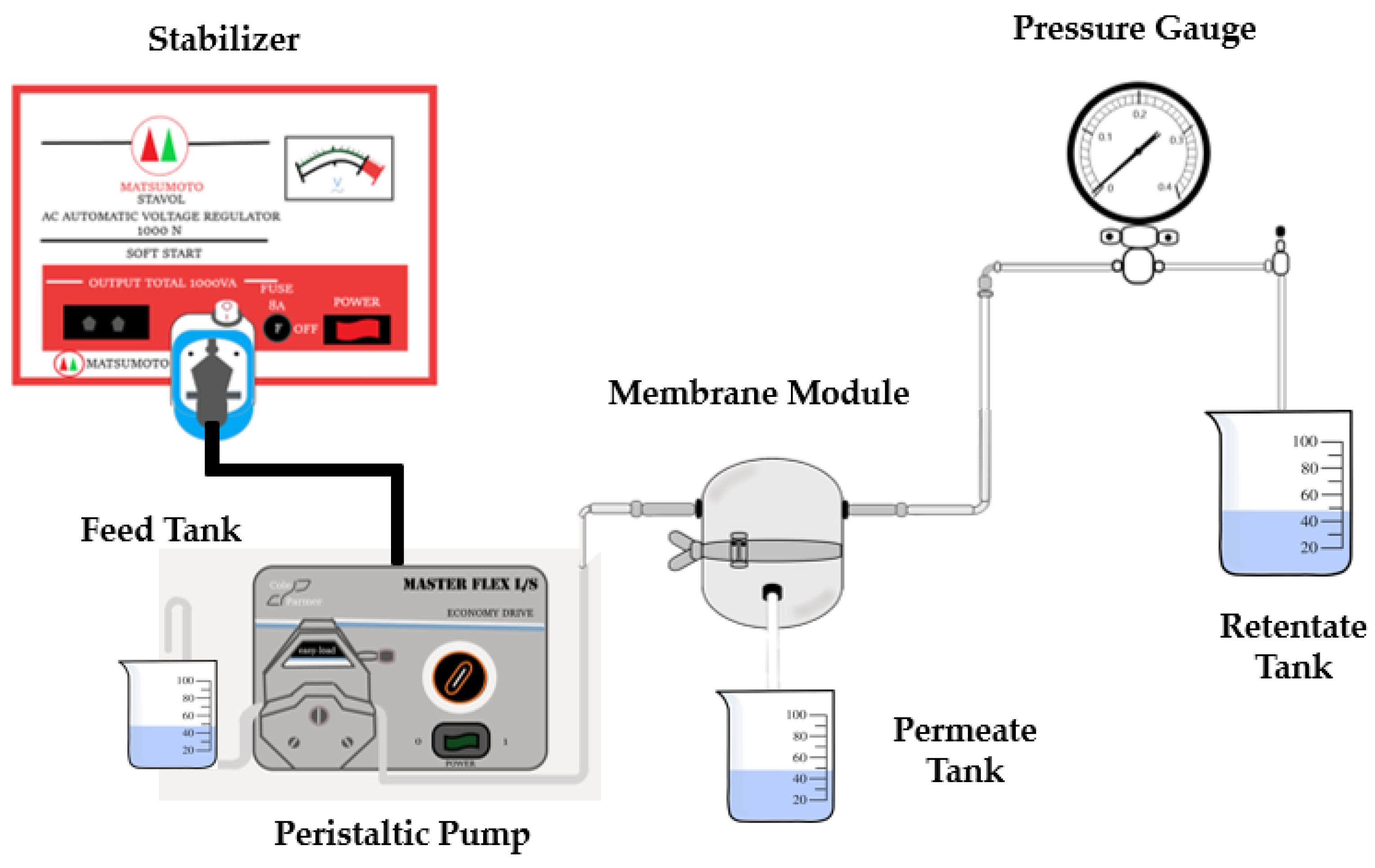
The membrane filtration performance was evaluated by measuring pure water flux and turbidity rejection to the well water sample. A crossflow filtration module measured water flux and rejection (Figure 1). Water was pumped through the filtration cell under the transmembrane pressure of 1 bar with the help of a peristaltic pump. Permeate volume was weighed every 10 min until a constant value was obtained. The test was carried out at room temperature and pressure. The membrane flux can be calculated using Equation (2).
where J is water flux (L/m2.h), dV is permeated volume (L), A is membrane surface area (m2), and Δt = time (hour).
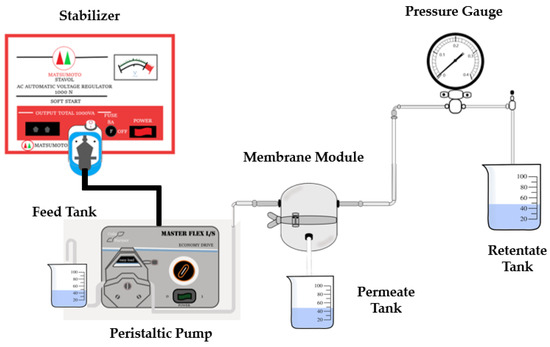
Figure 1.
Crossflow ultrafiltration module for well-water filtration.
2.8. Antibacterial Test
Another parameter in the quality of drinking water is the content of E. coli bacteria in the water. The antibacterial test was carried out using E. coli (ATCC 25922). The test was conducted using 0.5 Mac Farland of E. Coli solution smeared on Muller–Hinton agar on a petri dish. Each membrane sample was placed on the agar surface containing E. coli. The number of colonies growing on the membrane surface was counted under a magnifying glass after 24 h of incubation [27].
3. Results and Discussion
3.1. Membrane Structure and Morphology
SEM images were used to describe the structure and morphology of the membrane samples prepared by blending PES polymer with P-188 and PO compounds. The morphological structure of the membrane without and with the addition of P-188 and patchouli oil compounds could be distinguished, as shown in Figure 2.

Figure 2.
SEM images showing the effect of PO concentration on membrane morphology in cross-section (a) and enlarged cross-section near the bottom surface (b).
Figure 2 shows that all the resulting membranes were asymmetrical with a morphological structure from top to bottom. It was constituted of a thin and dense top layer supported by a finger-like structure underneath the top layer, a macro void structure near the bottom layer, and a dense structure pore on the bottom layer. Such a morphological structure is commonly found for a membrane produced through the phase inversion method by solidification in a container with a non-solvent such as water [14].
The difference in membrane morphology in all samples was in the number and size of finger-like and macro void structures and the thickness of dense structure at the bottom layer, as shown in Figure 2. The membranes with P188 and PO had a larger macro void size than the pristine PES membrane without P188 and PO (0 wt%). Increasing the PO concentration in the polymer solution resulted in membranes with a larger macro void structure. It shows that the addition of P188 and PO increased the PES membrane porosity. It was observed that the hydrophilic additives had different effects on the structure and morphology of the resulting membrane. In this study, adding P188 and PO additives can increase the macro void structure’s size and the membrane’s porosity [28].
3.2. Functional Groups of Membrane
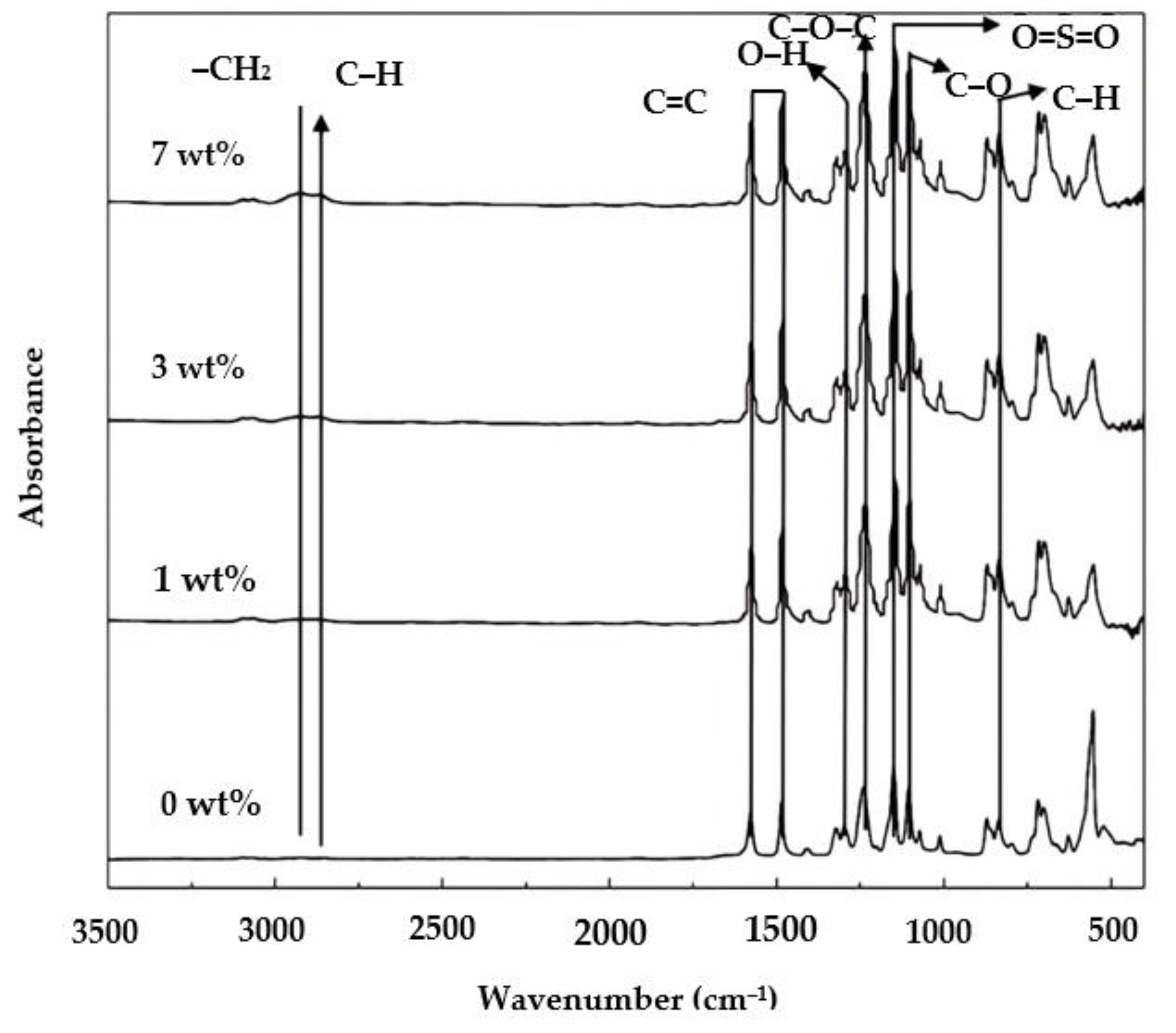
The FTIR spectra for all the resulting membranes are shown in Figure 3. FTIR Spectroscopy is usually used to analyze a chemical structure (functional group) on the surface of a sample based on a bond or molecular interaction in a material, either in the form of absorbance or transmittance of infrared light absorbed or reflected on a sample.
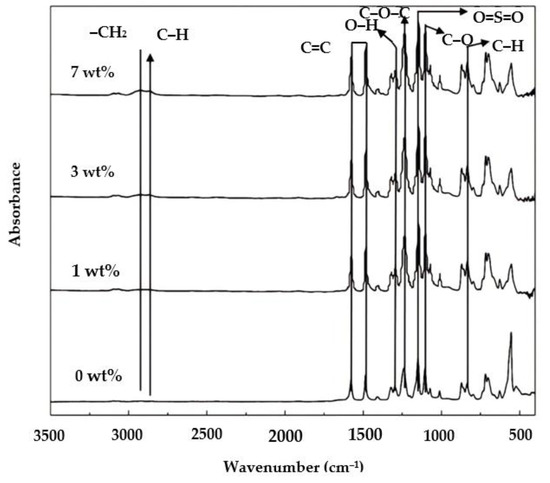
Figure 3.
FTIR spectra of the membrane samples.
PES membranes are known to have an aromatic ring, functional groups at wavenumbers 1450–1510 cm−1, ether groups at 1230–1270 cm−1, sulfone groups 1100–1200 cm−1, and aromatic C−H groups at 620–867 cm−1 [29]. In Figure 3, the resulting membranes had the appropriate functional groups representing the PES structure. The aromatic ring functional group (C=C−Ca) was found at wavenumber 1483.16 cm−1 and the sulfone group (O=S=O) at 1147.56 cm−1. Ether group (C−O−C) presented at wavenumber 1238.21 cm−1 and the aromatic C-H group at 837.05 cm−1.
The PO functional groups were identified on the modified membranes, namely, the methylene functional group (−CH2) at a wavenumber 2925.81 cm−1 and an O−H group at 1301 cm−1, as also reported earlier [30]. A small peak shows the P188 additive at wavenumber 2900 cm−1 attributed to methylene groups (−CH2) and (C−H).
3.3. Membrane Porosity
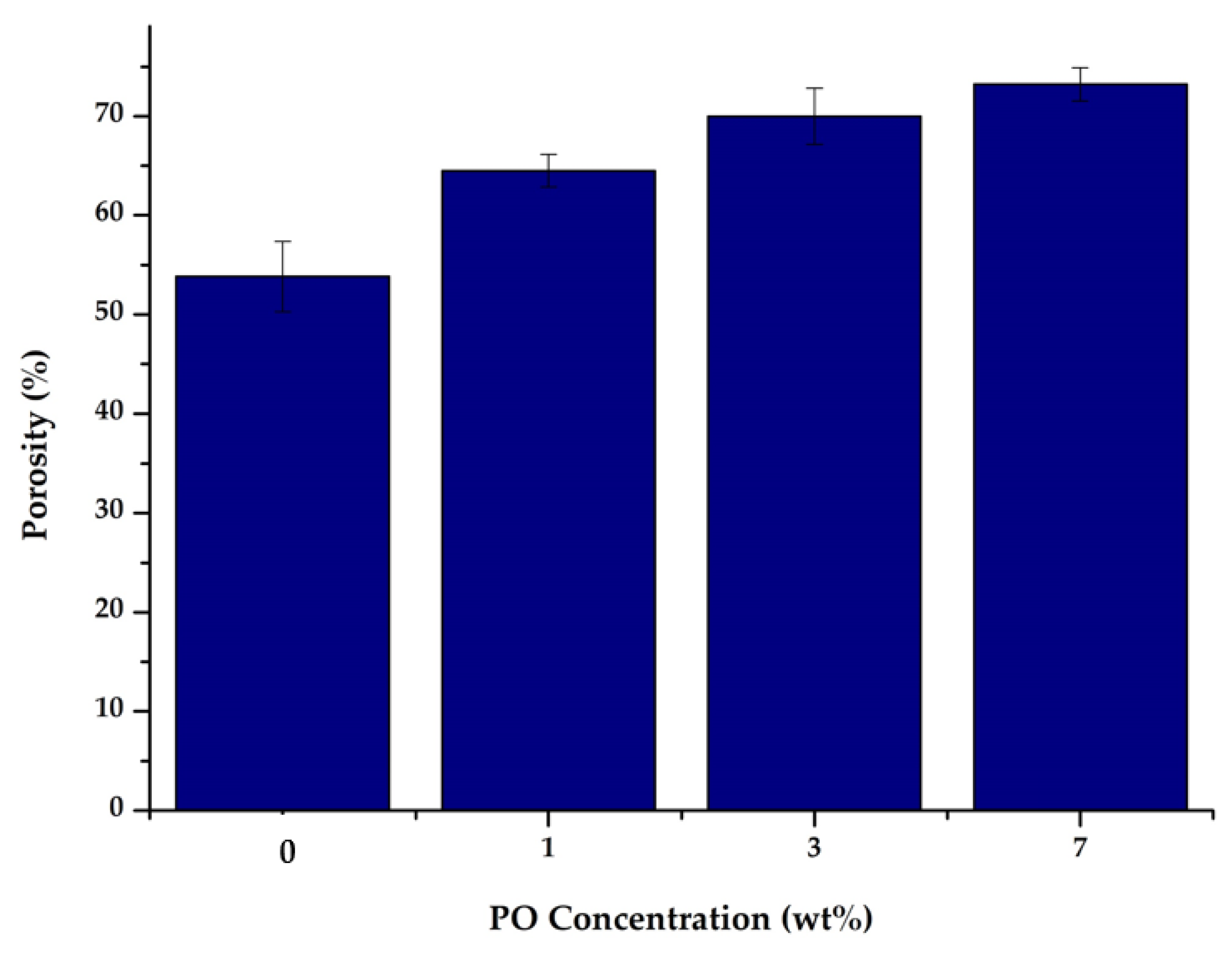
The results of the porosity measurements on each of the resulting membranes are shown in Figure 4. Membrane porosity is related to how much free space was formed during the solidification process of polymer solution between the constituent materials forming the membrane layer or mass transfer between the solvent and non-solvent phases [31]. Figure 4 shows that the PES membrane modified with P188 and PO resulted in higher porosity. The increased increment of the porosity is due to the faster rate of phase inversion due to the presence of hydroxyl groups (O−H) that attract more water [32]. This faster phase inversion resulted in the proliferation of pores in the membrane structure. Membranes with PO content of 0, 1, 3, and 7 wt% had porosities of 53.84, 64.50, 69.98, and 73.24%, respectively. The addition of PO increased the porosity, in which higher loading of PO led to more porous membranes.
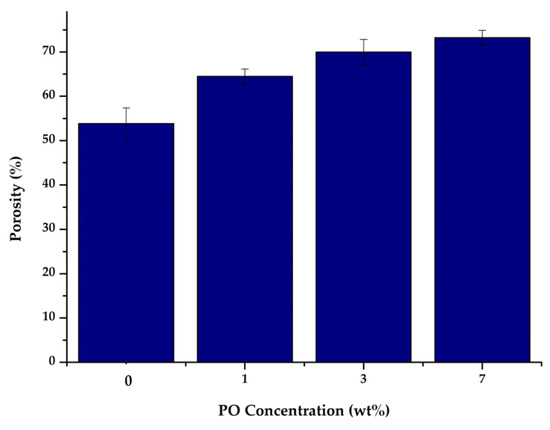
Figure 4.
Membrane porosity.
Additionally, the P-188 substance, a Pluronic surfactant polymer, can act as a membrane pore-forming agent through an irregular bonding scheme between the primary polymer and additive particles, increasing the membrane’s pore size and porosity when combined with PO. Due to the irregularity of the bonds owned, the opening of the space between the particles generates membrane pores. Multiple research studies revealing that the addition of Pluronic has affected the pore structure in polymer membranes confirms this statement [33].
On the other hand, using solvents for manufacturing membranes can also affect the membrane structure. In the phase inversion technique, the interaction of solvent and non-solvent during the membrane solidification process has a significant effect: the solubility or solubility parameters of the non-solvent affect the exchange rate of the solvent in the coagulation bath and the non-solvent in the polymer solution. Suppose the difference in solubility between solvent and non-solvent is low. In that case, non-solvent will diffuse more into the film or polymer layer, thereby accelerating the diffusion rate of solvent and non-solvent in the coagulation bath, which causes spontaneous separation (instant demixing). Thus, adding P-188 and PO results in the break of instantaneous during the phase inversion process, resulting in larger pore sizes [34].
3.4. Membrane Hydrophilicity
The membrane hydrophilicity can be determined by looking at and observing the angle of the water drop placed on the membrane surface. The contact angle of the water droplet was obtained by measuring the angle formed between the membrane surface and the bubble line of the droplet. The WCA was formed when water was dripped onto the membrane surface. The hydrophilic membrane properties can be identified by decreasing the WCA value. A high WCA value indicates that the membrane is hydrophobic [35].
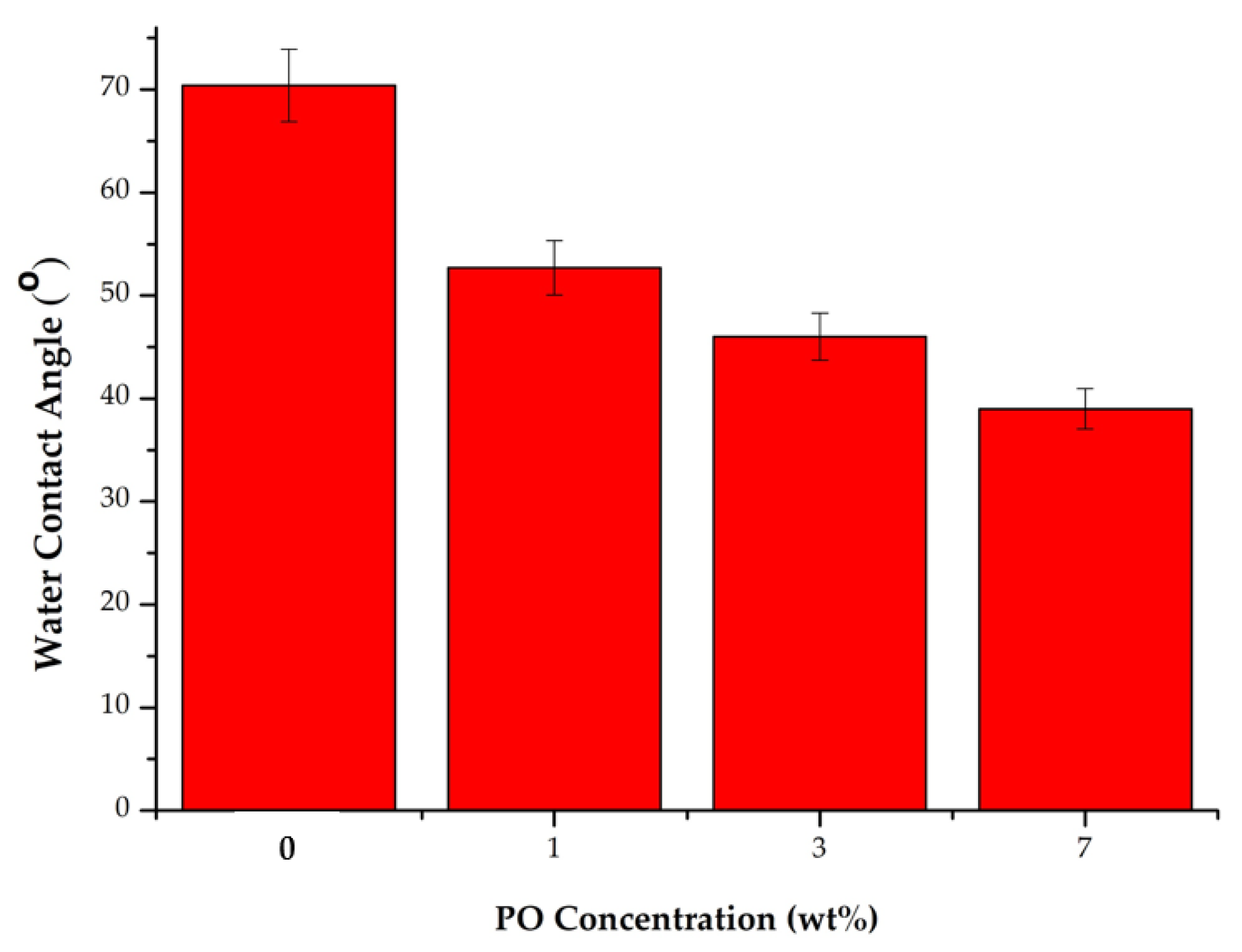
Figure 5 shows the WCA values of each type of membrane produced in this study, showing the decreasing WCA for membranes loaded with higher additive concentrations. It shows that the 0 wt% membrane had a WCA value of 70.4°, the membrane with 1 wt% PO had a WCA value of 52.7°, the 3 wt% PO membrane had a WCA value of 46°, and the 7 wt% PO membrane had a WCA value of 39.71°. The finding revealed that P188 and PO compounds played a role in increasing membrane hydrophilicity. Poloxamer 188 is a nonionic surfactant formed by polyethylene oxide (PEO) and polypropylene oxide (PPO) with the molecular structure of PEO-PPO-PEO. The dominant ethylene oxide (EO) group in poloxamer compounds is a carrier of polar hydrophilic functional groups that reduce the WCA on the membrane [9,36]. Adding PO additives also increased membrane hydrophilicity, as shown by a decreased WCA. Pure PES membrane had the highest WCA values attributed to the hydrophobicity of the PES. Meanwhile, the modified membranes had a relatively lower WCA value due to the addition of Poloxamer 188 and patchouli oil additives.
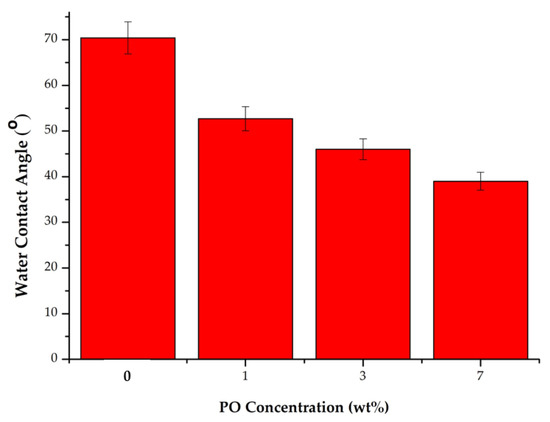
Figure 5.
WCA for each type of the membrane.
3.5. Antibacterial Properties
Antibacterial tests were carried out to determine the effect of the presence of PO in the polymer solution on the membrane’s ability to repel and alleviate bacterial growth. This study conducted the membrane antibacterial test using E. coli bacteria. The analysis was carried out by observing microbial colonies on the membrane surface through the total plate count method. At this investigation stage, five samples were prepared to determine whether the membrane’s antibacterial properties were derived from the P188 co-polymer or the PO additive. Membrane sample 1 was made from a single polymer solution (PES only), sample 2 contained PES polymer and additive P188 (3 wt%), while sample 3 contained PES and PO (3 wt%). Then, each of the samples 4 and 5 contained PES polymer + P188 (3 wt%) + PO (3 wt%) and PES + P188 (3 wt%) + PO (7 wt%).
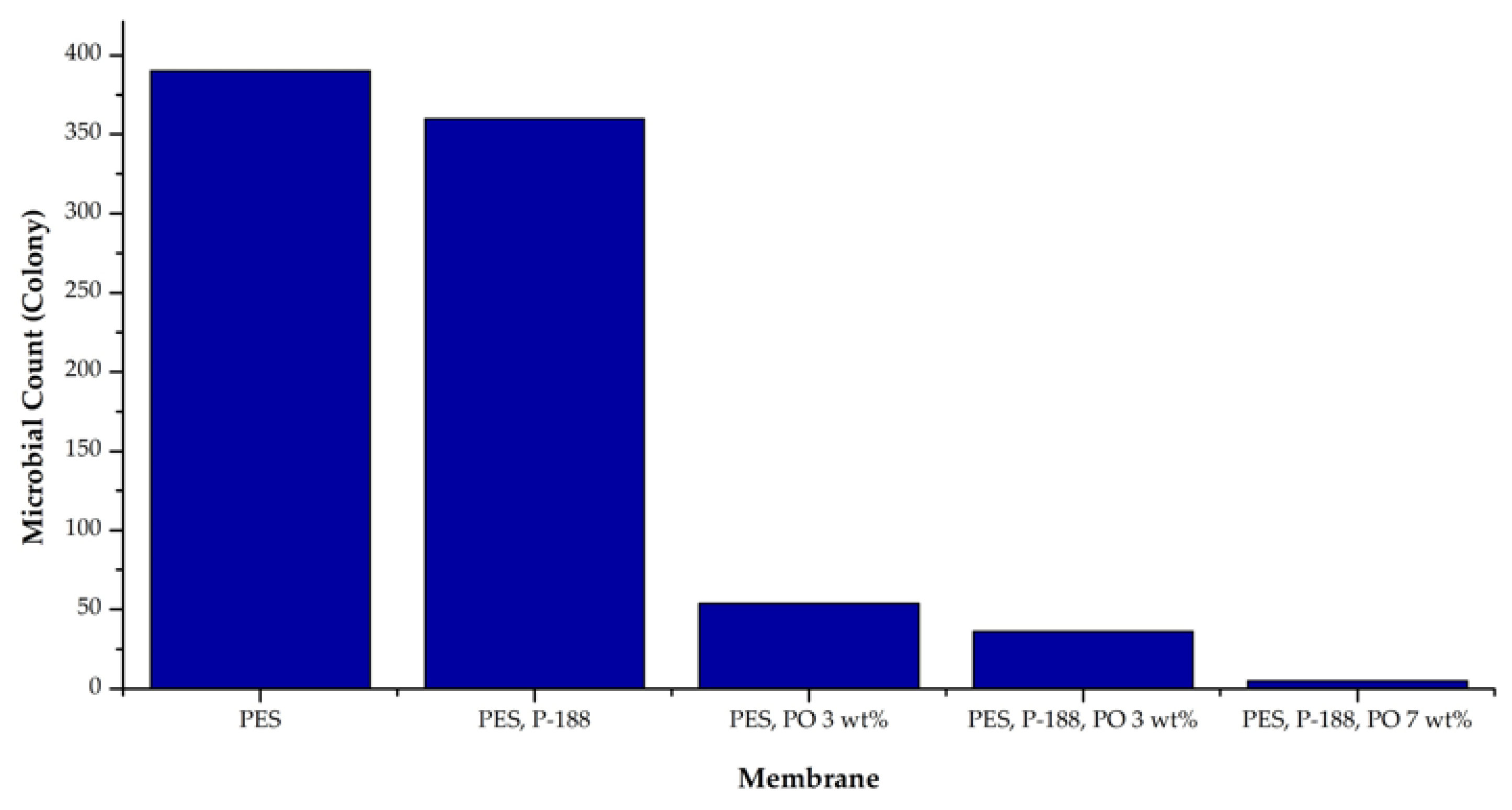
The complete antibacterial test results on all types of membranes can be seen in Figure 6. It shows that the membrane made from a dope solution containing PO produced a membrane with excellent antibacterial properties. There were 385 colonies of E. coli bacteria found on the surface of the pure PES membrane (0 wt% of PO), while only 52 bacterial colonies were found on the PO-modified PES membrane surface. P188 additive did not significantly impact the membrane antibacterial properties increment as indicated by the number of colonies on the PES + P188 membrane surface, just slightly lower than the pure PES. Further addition of PO up to 7 wt% to the polymer solution resulted in a membrane with outstanding antibacterial properties. In this case, the bacteria living on the PES/P188/PO (7 wt%) membrane surface were only four colonies.
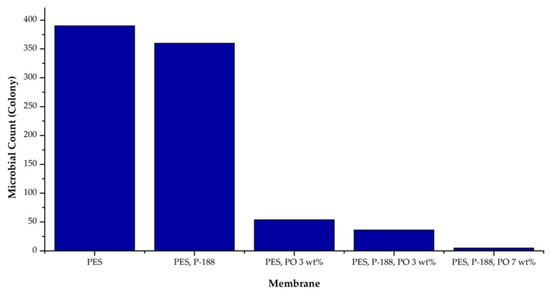
Figure 6.
The count of E. coli during the antibacterial activity test.
In the PO-containing dope solution, there were phenolic compounds, sesquiterpenes, and a study; the PO acted as an antimicrobial agent that limited the growth of E. coli agents on the surface of the PO-containing membranes. The bacterial cells disliked the presence of patchoulane and guaiane-type sesquiterpenoid compounds in PO and did not use it as a platform to form a colony to inhibit the bacterial growth [37]. PO shows an antibacterial effect. Therefore, increasing the concentration of PO depleted and hindered the growth and proliferation of E.coli [38]. The antimicrobial property was higher for membranes with higher PO loadings. A follow-up study will discuss a more comprehensive study of this antibacterial membrane in the long-term water filtration process.
3.6. Membrane Filtration Performance
In the membrane filtration process, flux is one of the critical parameters. Flux is affected by the physical and chemical properties of the membrane. In many studies, it is reported that increasing the concentration of additives in the polymer solution will cause an increase in the number and size of macro voids (large pores). However, there are also reports that the overloading dope solution with an additive would result in a membrane with a very thick, dense structure at the bottom layer.
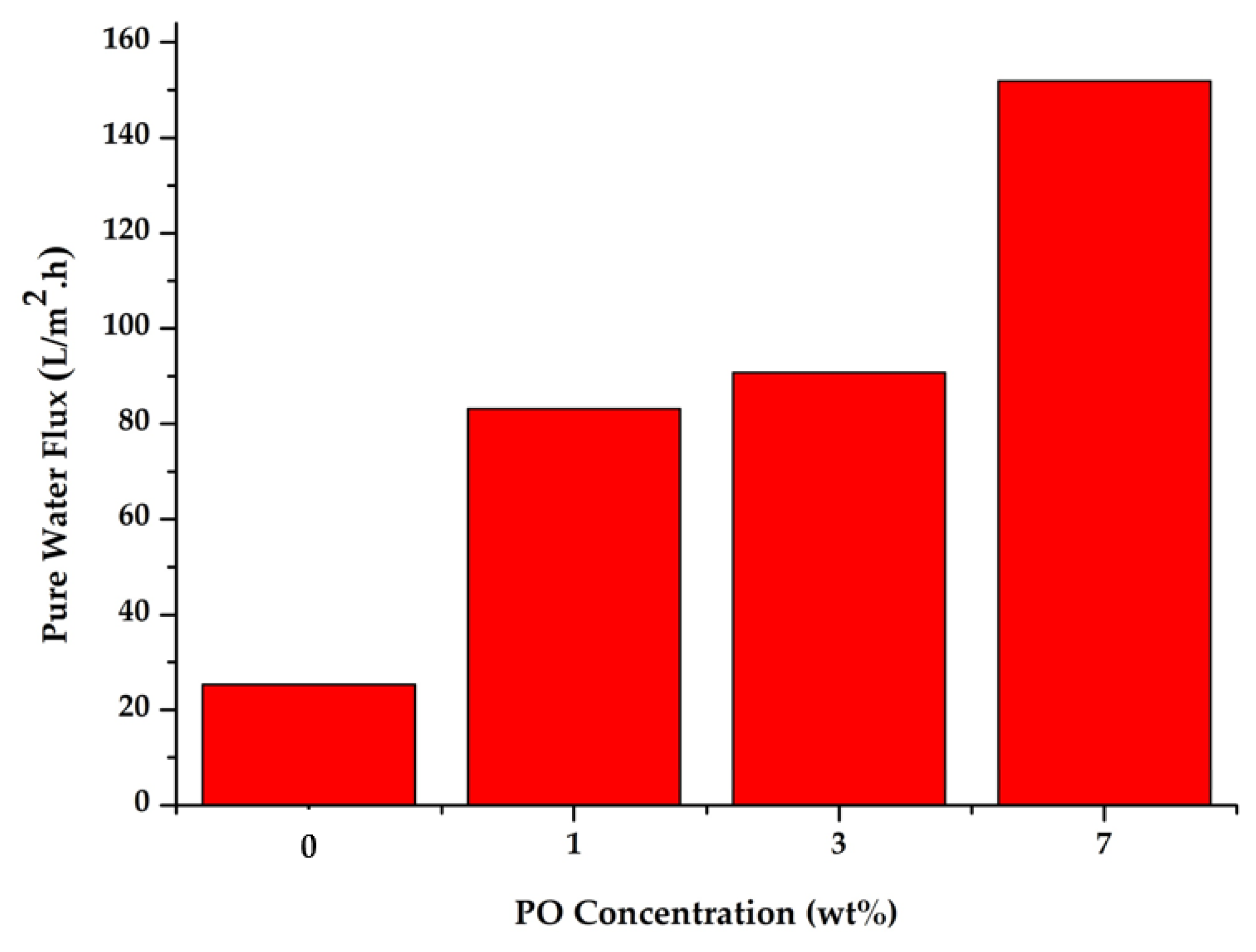
Figure 7 shows the flux data for all prepared membranes. The PES membrane without P188 or PO additives had a flux of 25.3 L/m2h. The addition of P188 and PO at 1 wt% into the dope solution increased the flux value of the resulting membrane to 83.177 L/m2h. Further addition of PO additives up to 7 wt% resulted in a membrane with a flux almost 6-fold higher than the initial flux of the pure PES membrane. This increase in flux is closely related to the morphological structure of the membrane, as shown in Figure 2. The increase in size and the change in the shape of the macro void pore structure in the membrane with additives provide a larger free space for water molecules to pass across the membrane. Therefore, in the crossflow filtration process, the membranes with P188 and PO additives would have a higher flux than the original PES membrane. The flux value is also influenced by membrane hydrophilicity that pulls more water to pass through the membrane pores [8,39]. Moreover, higher hydrophilic property of membrane surface also leads to good anti-fouling performances [40,41]. Fouling on the surface of the membrane with high hydrophilicity also sustains the filtration longer than on the hydrophobic membranes.
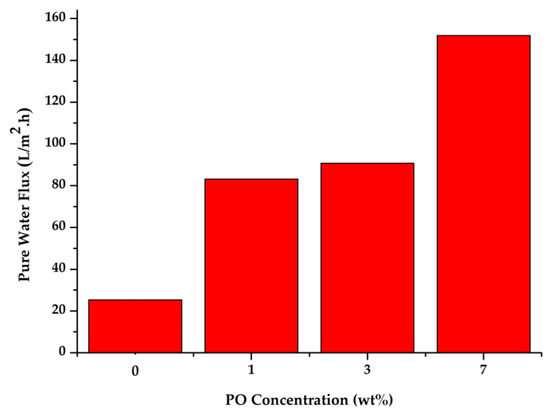
Figure 7.
Water flux of the membrane prepared with various addition of PO.
In addition to flux, another critical parameter of membrane filtration performance is the membrane’s ability to reject foulant particles in the feed solution, commonly called solute rejection. The rejection test in this study was carried out using well water samples from ten sampling points in the sub-districts of Syiah Kuala, Kuta Alam, and Kuta Raja in the City of Banda Aceh. The rejected particle parameter analyzed was turbidity. The turbidity data of water from ten wells and their corresponding permeate samples are presented in Table 1.

Table 1.
Removal efficiency of turbidity of well water after filtrated by membrane.
From the results summarized in Table 1, it is found that the turbidity values shown of all well water samples after passing through the membrane ranged from 80 to 100%. The turbidity value of the filtered drinking water must be ensured to be below the recommended value. This turbidity value of the filtrated water is far below the standard maximum turbidity in drinking water [42], indicating that the membranes could properly filter out particles of impurities found in well water. The results also showed that the membrane with the addition of 7 wt% PO had the best performance with turbidity values for all water samples with complete rejection for nine out of ten samples. The findings highlight the advantage of PO additives in simultaneously increasing the permeability and turbidity rejection.
4. Conclusions
This study reported the effectiveness of P-188 and PO as the membrane-modifying agent to enhance the performance of PES-based phase-inverted membranes. The addition of P-188 and PO in a PES polymer solution made the membrane structure more porous. The results of the morphological analysis show that the membrane produced has an asymmetric structure with porosity reaching 73.24% after being modified with 7 wt% PO. Membrane hydrophilicity measured based on WCA values showed significant increases.
The filtration test showed that the membrane from the dope solution containing 7% wt of PO had superior performance in terms of pure water flux reaching 152 L/m2h. Turbidity tests are also carried out to see the filtration performance of the membrane. Turbidity parameters in well water samples could be reduced to below the allowed turbidity of drinking water after the filtration process with all membranes produced in this study. Turbidity in seven well water samples could be reduced to zero after filtration with a PES-based membrane containing 7 wt% PO as an additive in its dope solution. The addition of P188 and PO also significantly increased the antibacterial properties, as indicated by the number of bacteria colony reduction by 97.5%. In short, this study demonstrated the improvement of membrane characteristics and performance by incorporating P-188 and PO as additives in the dope solution.
Author Contributions
Conceptualization, N.A.; data curation, J.J.; formal analysis, J.J.; investigation, J.J., W.A.D. and N.H.; project administration, S.S.; resources, S.S. and A.F.; supervision, N.A. and M.R.B.; writing—original draft, W.A.D. and N.H.; writing—review and editing, N.A., M.P.A. and A.F. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Data Availability Statement
Not applicable.
Acknowledgments
The authors would like to gratefully thank the Universitas Syiah Kuala for financial support of the research under the Research Professor with Research Contract Number (116/UN11/SPK/PNBP/2021).
Conflicts of Interest
The authors declare no conflict of interest.
References
- Gutierrez-Urbano, I.; Villen-Guzman, M.; Perez-Recuerda, R.; Rodriguez-Maroto, J.M. Removal of Polycyclic Aromatic Hydrocarbons (PAHs) in Conventional Drinking Water Treatment Processes. J. Contam. Hydrol. 2021, 243, 103888. [Google Scholar] [CrossRef] [PubMed]
- Choo, G.; Oh, J.E. Seasonal Occurrence and Removal of Organophosphate Esters in Conventional and Advanced Drinking Water Treatment Plants. Water Res. 2020, 186, 116359. [Google Scholar] [CrossRef] [PubMed]
- He, H.; Li, T.; He, C.; Chen, J.; Chu, H.; Dong, B. Removal of Natural Organic Matter in Full-Scale Conventional and Advanced Water Treatment Plants: Assimilable Organic Carbon and Its Precursors. Chem. Eng. J. Adv. 2021, 8, 100183. [Google Scholar] [CrossRef]
- Chang, H.; Zhu, Y.; Yu, H.; Qu, F.; Zhou, Z.; Li, X.; Yang, Y.; Tang, X.; Liang, H. Long-Term Operation of Ultrafiltration Membrane in Full-Scale Drinking Water Treatment Plants in China: Characteristics of Membrane Performance. Desalination 2022, 543, 116122. [Google Scholar] [CrossRef]
- Qiu, Z.; Chen, J.; Dai, R.; Wang, Z. Modification of Ultrafiltration Membrane with Antibacterial Agent Intercalated Layered Nanosheets: Toward Superior Antibiofouling Performance for Water Treatment. Water Res. 2022, 219, 118539. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; He, Z.-H.; Wang, K.-P.; Wang, X.-M.; Xie, Y.-F.F.; Hou, L. Loose Nanofiltration Membranes for Selective Rejection of Natural Organic Matter and Mineral Salts in Drinking Water Treatment. J. Memb. Sci. 2022, 662, 120970. [Google Scholar] [CrossRef]
- Jebur, M.; Chiao, Y.H.; Thomas, K.; Patra, T.; Cao, Y.; Lee, K.; Gleason, N.; Qian, X.; Hu, Y.; Malmali, M.; et al. Combined Electrocoagulation-Microfiltration-Membrane Distillation for Treatment of Hydraulic Fracturing Produced Water. Desalination 2021, 500, 114886. [Google Scholar] [CrossRef]
- Purnima, M.; Paul, T.; Pakshirajan, K.; Pugazhenthi, G. Onshore Oilfield Produced Water Treatment by Hybrid Microfiltration-Biological Process Using Kaolin Based Ceramic Membrane and Oleaginous Rhodococcus opacus. Chem. Eng. J. 2023, 453, 139850. [Google Scholar] [CrossRef]
- Mulyati, S.; Muchtar, S.; Arahman, N.; Syamsuddin, Y.; Mat Nawi, N.I.; Harun, N.Y.; Bilad, M.R.; Firdaus, Y.; Takagi, R.; Matsuyama, H. Two-Step Dopamine-to-Polydopamine Modification of Polyethersulfone Ultrafiltration Membrane for Enhancing Anti-Fouling and Ultraviolet Resistant Properties. Polymers 2020, 12, 2051. [Google Scholar] [CrossRef]
- Muchtar, S.; Wahab, M.Y.; Fang, L.F.; Jeon, S.; Rajabzadeh, S.; Takagi, R.; Mulyati, S.; Arahman, N.; Riza, M.; Matsuyama, H. Polydopamine-Coated Poly(Vinylidene Fluoride) Membranes with High Ultraviolet Resistance and Antifouling Properties for a Photocatalytic Membrane Reactor. J. Appl. Polym. Sci. 2019, 136, 47312. [Google Scholar] [CrossRef]
- Aghapour Aktij, S.; Zirehpour, A.; Mollahosseini, A.; Taherzadeh, M.J.; Tiraferri, A.; Rahimpour, A. Feasibility of Membrane Processes for the Recovery and Purification of Bio-Based Volatile Fatty Acids: A Comprehensive Review. J. Ind. Eng. Chem. 2020, 81, 24–40. [Google Scholar] [CrossRef]
- Ekambaram, K.; Doraisamy, M. Surface Modification of PVDF Nanofiltration Membrane Using Carboxymethylchitosan-Zinc Oxide Bionanocomposite for the Removal of Inorganic Salts and Humic Acid. Colloids Surf. A Physicochem. Eng. Asp. 2017, 525, 49–63. [Google Scholar] [CrossRef]
- Guo, J.; Khan, S.; Cho, S.H.; Kim, J. ZnS Nanoparticles as New Additive for Polyethersulfone Membrane in Humic Acid Filtration. J. Ind. Eng. Chem. 2019, 79, 71–78. [Google Scholar] [CrossRef]
- Arahman, N.; Maruyama, T.; Sotani, T.; Matsuyama, H. Effect of Hypochlorite Treatment on Performance of Hollow Fiber Membrane Prepared from Polyethersulfone/N-Methyl-2-Pyrrolidone/Tetronic 1307 Solution. J. Appl. Polym. Sci. 2008, 110, 687–694. [Google Scholar] [CrossRef]
- Amiyati, D.R.; Indarti, D.; Muflihah, Y.M. Pengaruh Variasi Waktu Penguapan Terhadap Kinerja Membran Selulosa Asetat Pada Proses Ultrafiltrasi. Berk. Sainstek 2017, 5, 7. [Google Scholar] [CrossRef][Green Version]
- Nasrollahi, N.; Aber, S.; Vatanpour, V.; Mahmoodi, N.M. Development of Hydrophilic Microporous PES Ultrafiltration Membrane Containing CuO Nanoparticles with Improved Antifouling and Separation Performance. Mater. Chem. Phys. 2019, 222, 338–350. [Google Scholar] [CrossRef]
- Safarpour, M.; Vatanpour, V.; Khataee, A. Preparation and Characterization of Graphene Oxide/TiO2 Blended PES Nanofiltration Membrane with Improved Antifouling and Separation Performance. Desalination 2016, 393, 65–78. [Google Scholar] [CrossRef]
- Bae, J.; Baek, I.; Choi, H. Mechanically Enhanced PES Electrospun Nanofiber Membranes (ENMs) for Microfiltration: The Effects of ENM Properties on Membrane Performance. Water Res. 2016, 105, 406–412. [Google Scholar] [CrossRef]
- Kourde-Hanafi, Y.; Loulergue, P.; Szymczyk, A.; Van der Bruggen, B.; Nachtnebel, M.; Rabiller-Baudry, M.; Audic, J.L.; Pölt, P.; Baddari, K. Influence of PVP Content on Degradation of PES/PVP Membranes: Insights from Characterization of Membranes with Controlled Composition. J. Memb. Sci. 2017, 533, 261–269. [Google Scholar] [CrossRef]
- Supriyadi, J.; Hakika, D.C.; Kusworo, T.D. Peningkatan kinerja membran selulosa asetat untuk pengolahan air payau dengan modifikasi penambahan aditif dan pemanasan. J. Tekonologi Kim. Ind. 2013, 2, 96–108. [Google Scholar]
- Bahremand, A.H.; Mousavi, S.M.; Ahmadpour, A.; Taherian, M. Biodegradable Blend Membranes of Poly(Butylene Succinate)/Cellulose Acetate/Dextran: Preparation, Characterization and Performance. Carbohydr. Polym. 2017, 173, 497–507. [Google Scholar] [CrossRef]
- Fernandes, A.V.; Pydi, C.R.; Verma, R.; Jose, J.; Kumar, L. Design, Preparation and in Vitro Characterizations of Fluconazole Loaded Nanostructured Lipid Carriers. Braz. J. Pharm. Sci. 2020, 56, 1–14. [Google Scholar] [CrossRef]
- Heptiana, E. Analisis Komparasi Tempat Tumbuh dan Jenis Alat Suling Terhadap Rendemen dan Mutu Minyak Nilam (Pogestemon cablin Benth.). SYLVA 2019, 8, 46–52. [Google Scholar] [CrossRef]
- Widowati, R.; Handayani, S.; Lasdi, I. Aktivitas antibakteri minyak nilam (Pogostemon cablin) terhadap beberapa spesies bakteri uji. Pro-Life 2019, 6, 244. [Google Scholar]
- Adhayani, L.; Ramadhani, R.; Ristianti, R. Kapasitas Daya Hambat Antibakteri Minyak Atsiri Nilam Aceh (Pogostemon cablin Benth.) Terhadap Methicillin-Resistant Staphylococcus Aureus (MRSA). PHARMACY J. Farm. Indones. (Pharm. J. Indones.) 2021, 18, 130. [Google Scholar] [CrossRef]
- Sari, M.; Tamrin; Kaban, J.; Alfian, Z. A Novel Composite Membrane Pectin from Cyclea Barbata Miers Blend with Chitosan for Accelerated Wound Healing. Polym. Test 2021, 99, 107207. [Google Scholar] [CrossRef]
- Arahman, N.; Rosnelly, C.M.; Windana, D.S.; Fahrina, A.; Silmina, S.; Maimun, T.; Mulyati, S.; Fathanah, U.; Aprilia, S.; Bilad, M.R.; et al. Antimicrobial Hydrophilic Membrane Formed by Incorporation of Polymeric Surfactant and Patchouli Oil. Polymers 2021, 13, 3872. [Google Scholar] [CrossRef] [PubMed]
- Mokarizadeh, H.; Raisi, A. Industrial Wastewater Treatment Using PES UF Membranes Containing Hydrophilic Additives: Experimental and Modeling of Fouling Mechanism. Environ. Technol. Innov. 2021, 23, 101701. [Google Scholar] [CrossRef]
- White, J.L. Interpretation of Infrared Spectra of Soil Minerals. Soil Sci. 1971, 112, 22–31. [Google Scholar] [CrossRef]
- Fahmi, Z.; Mudasir, M.; Rohman, A. Attenuated Total Reflectance-FTIR Spectra Combined with Multivariate Calibration and Discrimination Analysis for Analysis of Patchouli Oil Adulteration. Indones. J. Chem. 2020, 20, 1–8. [Google Scholar] [CrossRef]
- Koyuncu, I.; Yavuzturk Gul, B.; Esmaeili, M.S.; Pekgenc, E.; Orhun Teber, O.; Tuncay, G.; Karimi, H.; Parvaz, S.; Maleki, A.; Vatanpour, V. Modification of PVDF Membranes by Incorporation Fe3O4@Xanthan Gum to Improve Anti-Fouling, Anti-Bacterial, and Separation Performance. J. Environ. Chem. Eng. 2022, 10, 107784. [Google Scholar] [CrossRef]
- Fahrina, A.; Arahman, N.; Aprilia, S.; Bilad, M.R.; Silmina, S.; Sari, W.P.; Sari, I.M.; Gunawan, P.; Pasaoglu, M.E.; Vatanpour, V.; et al. Functionalization of PEG-AgNPs Hybrid Material to Alleviate Biofouling Tendency of Polyethersulfone Membrane. Polymers 2022, 14, 1908. [Google Scholar] [CrossRef] [PubMed]
- Sadrzadeh, M.; Bhattacharjee, S. Rational Design of Phase Inversion Membranes by Tailoring Thermodynamics and Kinetics of Casting Solution Using Polymer Additives. J. Memb. Sci. 2013, 441, 31–44. [Google Scholar] [CrossRef]
- Fathanah, U.; Meilina, H. Karakterisasi Dan Kinerja Membran Polyethersulfone Termodifikasi Aditif Anorganik secara Blending Polimer. J. Serambi Eng. 2021, 6, 2407–2414. [Google Scholar] [CrossRef]
- Wenten, I.G.; Khoiruddin, K.; Wardani, A.K.; Aryanti, P.T.P.; Astuti, D.I.; Komaladewi, A.A.I.A.S. Preparation of Antifouling Polypropylene/ZnO Composite Hollow Fiber Membrane by Dip-Coating Method for Peat Water Treatment. J. Water Process Eng. 2020, 34, 101158. [Google Scholar] [CrossRef]
- Le, A.N.M.; Tran, N.M.P.; Phan, T.B.; Tran, P.A.; Tran, L.D.; Nguyen, T.H. Poloxamer Additive as Luminal Surface Modification to Modulate Wettability and Bioactivities of Small-Diameter Polyurethane/Polycaprolactone Electrospun Hollow Tube for Vascular Prosthesis Applications. Mater. Today Commun. 2021, 26, 101771. [Google Scholar] [CrossRef]
- Srivastava, S.; Lal, R.K.; Singh, V.R.; Rout, P.K.; Padalia, R.C.; Yadav, A.K.; Bawitlung, L.; Bhatt, D.; Maurya, A.K.; Pal, A.; et al. Chemical Investigation and Biological Activities of Patchouli (Pogostemon cablin (Blanco) Benth) Essential Oil. Ind. Crops Prod. 2022, 188, 115504. [Google Scholar] [CrossRef]
- Lukitawesa; Safarudin, A.; Millati, R.; Taherzadeh, M.J.; Niklasson, C. Inhibition of Patchouli Oil for Anaerobic Digestion and Enhancement in Methane Production Using Reverse Membrane Bioreactors. Renew. Energy 2018, 129, 748–753. [Google Scholar] [CrossRef]
- Jin, P.; Yuan, S.; Zhang, G.; Zhu, J.; Zheng, J.; Luis, P.; van der Bruggen, B. Polyarylene Thioether Sulfone/Sulfonated Sulfone Nanofiltration Membrane with Enhancement of Rejection and Permeability via Molecular Design☆. J. Memb. Sci. 2020, 608, 118241. [Google Scholar] [CrossRef]
- Fahrina, A.; Yusuf, M.; Muchtar, S.; Fitriani, F.; Mulyati, S.; Aprilia, S.; Rosnelly, C.M.; Bilad, M.R.; Ismail, A.F.; Takagi, R.; et al. Development of Anti-Microbial Polyvinylidene Fluoride (PVDF) Membrane Using Bio-Based Ginger Extract-Silica Nanoparticles (GE-SiNPs) for Bovine Serum Albumin (BSA) Filtration. J. Taiwan Inst. Chem. Eng. 2021, 123, 323–331. [Google Scholar] [CrossRef]
- Wang, S.Y.; Rolly Gonzales, R.; Zhang, P.; Istirokhatun, T.; Takagi, R.; Motoyama, A.; Fang, L.F.; Matsuyama, H. Surface Charge Control of Poly(Methyl Methacrylate-Co-Dimethyl Aminoethyl Methacrylate)-Based Membrane for Improved Fouling Resistance. Sep. Purif. Technol. 2021, 279, 119778. [Google Scholar] [CrossRef]
- Park, W.-I.; Jeong, S.; Im, S.-J.; Jang, A. High Turbidity Water Treatment by Ceramic Microfiltration Membrane: Fouling Identification and Process Optimization. Environ. Technol. Innov. 2020, 17, 100578. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).