Seawater with Added Monosodium Glutamate Residue (MSGR) Is a Promising Medium for the Cultivation of Two Commercial Marine Microalgae
Abstract
:1. Introduction
2. Materials and Methods
2.1. Microalgal Strain and Culture
2.2. Optimization of Dilution Ratio of MSGR
2.3. Experimental Design
2.4. Algal Analysis
2.4.1. Determination of the Total Chlorophyll Content
2.4.2. Measurement of Cell Biomass Concentration
2.4.3. Determination of Metabolites in Biomass
2.5. Nutrient Measurements
2.6. Statistical Analysis
3. Results and Discussion
3.1. Effects of S-MSGR on Growth
3.2. Changes to Photosynthesis in S-MSGR
3.3. Metabolite Accumulation
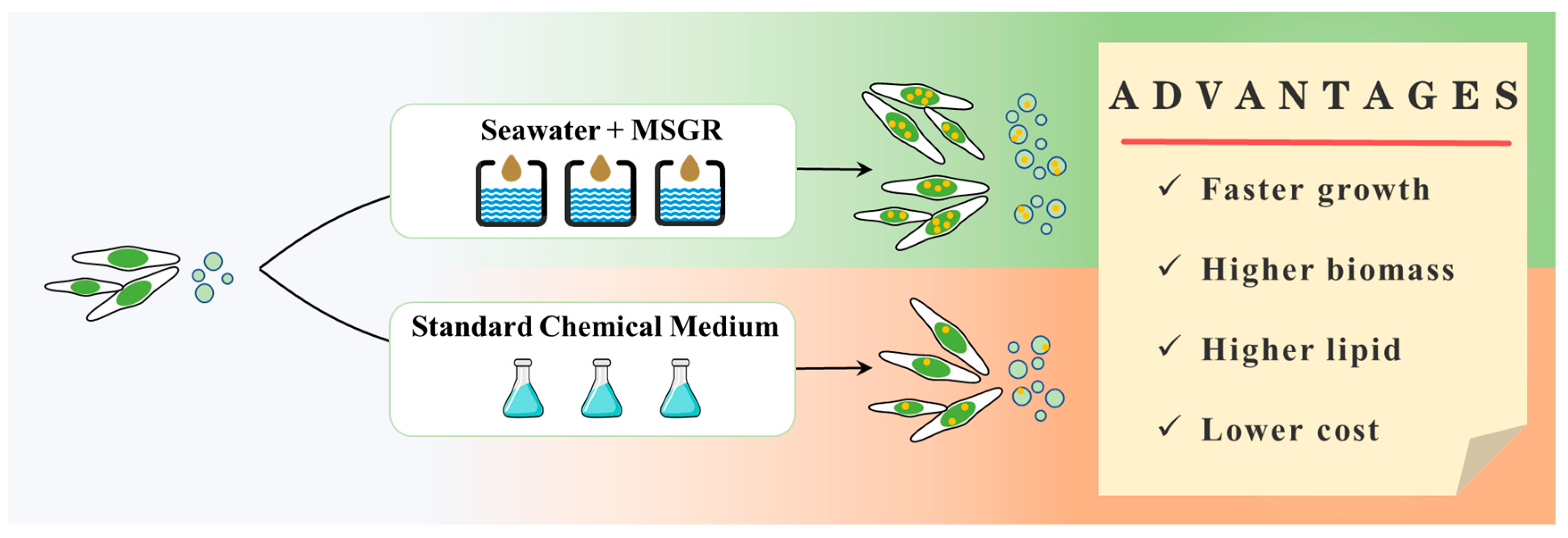
3.4. Benefit Analysis of S-MSGR for Culture of Marine Microalgae
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rizwan, M.; Mujtaba, G.; Memon, S.A.; Lee, K.; Rashid, N. Exploring the potential of microalgae for new biotechnology applications and beyond: A review. Renew. Sustain. Energy Rev. 2018, 92, 394–404. [Google Scholar] [CrossRef]
- Chew, K.W.; Yap, J.Y.; Show, P.L.; Suan, N.H.; Juan, J.C.; Ling, T.C.; Lee, D.J.; Chang, J.S. Microalgae biorefinery: High value products perspectives. Bioresour. Technol. 2017, 229, 53–62. [Google Scholar] [CrossRef] [PubMed]
- Mata, T.M.; Martins, A.A.; Caetano, N.S. Microalgae for biodiesel production and other applications: A review. Renew. Sustain. Energy Rev. 2010, 14, 217–232. [Google Scholar] [CrossRef] [Green Version]
- Su, T.; Zhao, D.; Khodadadi, M.; Len, C. Lignocellulosic biomass for bioethanol: Recent advances, technology trends, and barriers to industrial development. Curr. Opin. Green Sustain. Chem. 2020, 24, 56–60. [Google Scholar] [CrossRef]
- Rodionova, M.V.; Bozieva, A.M.; Zharmukhamedov, S.K.; Leong, Y.K.; Lan, J.C.W.; Veziroglu, A.; Veziroglu, T.N.; Tomo, T.; Chang, J.S.; Allakhverdiev, S.I. A comprehensive review on lignocellulosic biomass biorefinery for sustainable biofuel production. Int. J. Hydrog. Energy 2021, 47, 1481–1498. [Google Scholar] [CrossRef]
- Butler, T.; Kapoore, R.V.; Vaidyanathan, S. Phaeodactylum tricornutum: A diatom cell factory. Trends Biotechnol. 2020, 38, 606–622. [Google Scholar] [CrossRef] [PubMed]
- Ashour, M.; Elshobary, M.E.; El-Shenody, R.; Kamil, A.W.; Abomohra, A.E.F. Evaluation of a native oleaginous marine microalga Nannochloropsis oceanica for dual use in biodiesel production and aquaculture feed. Biomass Bioenergy 2019, 120, 439–447. [Google Scholar] [CrossRef]
- Christenson, L.; Sims, R. Production and harvesting of microalgae for wastewater treatment, biofuels, and bioproducts. Biotechnol. Adv. 2011, 29, 686–702. [Google Scholar] [CrossRef]
- Shahid, A.; Malik, S.; Zhu, H.; Xu, J.; Nawaz, M.Z.; Nawaz, S.; Alam, A.; Mehmood, M.A. Cultivating microalgae in wastewater for biomass production, pollutant removal, and atmospheric carbon mitigation; a review. Sci. Total Environ. 2020, 704, 135303. [Google Scholar] [CrossRef]
- Mohsenpour, S.F.; Hennige, S.; Willoughby, N.; Adeloye, A.; Gutierrez, T. Integrating micro-algae into wastewater treatment: A review. Sci. Total Environ. 2021, 752, 142168. [Google Scholar] [CrossRef]
- Kumar, R.; Ghosh, A.K.; Pal, P. Synergy of biofuel production with waste remediation along with value-added co-products recovery through microalgae cultivation: A review of membrane-integrated green approach. Sci. Total Environ. 2020, 698, 134169. [Google Scholar] [CrossRef] [PubMed]
- Dębowski, M.; Zieliński, M.; Kisielewska, M.; Kazimierowicz, J.; Dudek, M.; Świca, I.; Rudnicka, A. The cultivation of lipid-rich microalgae biomass as anaerobic digestate valorization technology—A pilot-scale study. Processes 2020, 8, 517. [Google Scholar] [CrossRef]
- Kumar, R.; Chakrabortty, S.; Pal, P. Membrane-integrated physico-chemical treatment of coke-oven wastewater: Transport modelling and economic evaluation. Environ. Sci. Pollut. Res. 2015, 22, 6010–6023. [Google Scholar] [CrossRef] [PubMed]
- Jiang, L.; Sun, J.; Nie, C.; Li, Y.; Jenkins, J.; Pei, H. Filamentous cyanobacteria triples oil production in seawater-based medium supplemented with industrial waste: Monosodium glutamate residue. Biotechnol. Biofuels 2019, 12, 53. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Singh, S.; Rekha, P.D.; Arun, A.B.; Hameed, A.; Singh, S.; Shen, F.T.; Young, C.C. Glutamate wastewater as a culture medium for Azospirillum rugosum production and its impact on plant growth. Biol. Fertil. Soils 2011, 47, 419–426. [Google Scholar] [CrossRef]
- Ji, Y.; Hu, W.; Li, X.; Ma, G.; Song, M.; Pei, H. Mixotrophic growth and biochemical analysis of Chlorella vulgaris cultivated with diluted monosodium glutamate wastewater. Bioresour. Technol. 2014, 152, 471–476. [Google Scholar] [CrossRef]
- Correa, D.F.; Beyer, H.L.; Possingham, H.P.; Thomas-Hall, S.R.; Schenk, P.M. Global mapping of cost-effective microalgal biofuel production areas with minimal environmental impact. GCB Bioenergy 2019, 11, 914–929. [Google Scholar] [CrossRef] [Green Version]
- Pancha, I.; Chokshi, K.; George, B.; Ghosh, T.; Paliwal, C.; Maurya, R.; Mishra, S. Nitrogen stress triggered biochemical and morphological changes in the microalgae Scenedesmus sp. CCNM 1077. Bioresour. Technol. 2014, 156, 146–154. [Google Scholar] [CrossRef]
- Nie, C.; Jiang, L.; Yu, Z.; Yang, Z.; Hou, Q.; Pei, H. Campus sewage treatment by Golenkinia SDEC-16 and biofuel production under monochromic light. J. Chem. 2020, 2020, 5029535. [Google Scholar] [CrossRef] [Green Version]
- Jiang, L.; Pei, H.; Hu, W.; Ji, Y.; Han, L.; Ma, G. The feasibility of using complex wastewater from a monosodium glutamate factory to cultivate Spirulina subsalsa and accumulate biochemical composition. Bioresour. Technol. 2015, 180, 304–310. [Google Scholar] [CrossRef]
- Li, H.; Pei, H.; Xu, H.; Jin, Y.; Sun, J. Behavior of Cylindrospermopsis raciborskii during coagulation and sludge storage–higher potential risk of toxin release than Microcystis aeruginosa? J. Hazard. Mater. 2018, 347, 307–316. [Google Scholar] [CrossRef] [PubMed]
- Bélanger-Lépine, F.; Tremblay, A.; Huot, Y.; Barnabé, S. Cultivation of an algae-bacteria consortium in wastewater from an industrial park: Effect of environmental stress and nutrient deficiency on lipid production. Bioresource technology. 2018, 267, 657–665. [Google Scholar] [CrossRef] [PubMed]
- Pei, H.; Jiang, L.; Hou, Q.; Yu, Z. Toward facilitating microalgae cope with effluent from anaerobic digestion of kitchen waste: The art of agricultural phytohormones. Biotechnol. Biofuels 2017, 10, 76. [Google Scholar] [CrossRef] [Green Version]
- Yu, Z.; Song, M.; Pei, H.; Han, F.; Jiang, L.; Hou, Q. The growth characteristics and biodiesel production of ten algae strains cultivated in anaerobically digested effluent from kitchen waste. Algal Res. 2017, 24, 265–275. [Google Scholar] [CrossRef]
- Wang, X.W.; Liang, J.R.; Luo, C.S.; Chen, C.P.; Gao, Y.H. Biomass, total lipid production, and fatty acid composition of the marine diatom Chaetoceros muelleri in response to different CO2 levels. Bioresour. Technol. 2014, 161, 124–130. [Google Scholar] [CrossRef] [PubMed]
- Demmig-Adams, B.; Adams, W.I. Chlorophyll and carotenoid composition in leaves of Euonymus kiautschovicus acclimated to different degrees of light stress in the field. Funct. Plant Biol. 1996, 23, 649–659. [Google Scholar] [CrossRef]
- Cheah, W.Y.; Show, P.L.; Juan, J.C.; Chang, J.S.; Ling, T.C. Enhancing biomass and lipid productions of microalgae in palm oil mill effluent using carbon and nutrient supplementation. Energy Convers. Manag. 2018, 164, 188–197. [Google Scholar] [CrossRef]
- Ho, S.H.; Nakanishi, A.; Kato, Y.; Yamasaki, H.; Chang, J.S.; Misawa, N.; Hirose, Y.; Minagawa, J.; Hasunuma, T.; Kondo, A. Dynamic metabolic profiling together with transcription analysis reveals salinity-induced starch-to-lipid biosynthesis in alga Chlamydomonas sp. JSC4. Sci. Rep. 2017, 7, 45471. [Google Scholar] [CrossRef]
- Yodsuwan, N.; Sawayama, S.; Sirisansaneeyakul, S. Effect of nitrogen concentration on growth, lipid production and fatty acid profiles of the marine diatom Phaeodactylum tricornutum. Agric. Nat. Resour. 2017, 51, 190–197. [Google Scholar] [CrossRef]
- Burch, A.R.; Yothers, C.W.; Salemi, M.R.; Phinney, B.S.; Pandey, P.; Franz, A.K. Quantitative label-free proteomics and biochemical analysis of Phaeodactylum tricornutum cultivation on dairy manure wastewater. J. Appl. Phycol. 2021, 33, 2105–2121. [Google Scholar] [CrossRef]
- Wang, H.; Fu, R.; Pei, G. A study on lipid production of the mixotrophic microalgae Phaeodactylum tricornutum on various carbon sources. Afr. J. Microbiol. Res. 2012, 6, 1041–1047. [Google Scholar]
- Simonazzi, M.; Pezzolesi, L.; Guerrini, F.; Vanucci, S.; Samorì, C.; Pistocchi, R. Use of waste carbon dioxide and pre-treated liquid digestate from biogas process for Phaeodactylum tricornutum cultivation in photobioreactors and open ponds. Bioresour. Technol. 2019, 292, 121921. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.W.; Huang, L.; Ji, P.Y.; Chen, C.P.; Li, X.S.; Gao, Y.H.; Liang, J.R. Using a mixture of wastewater and seawater as the growth medium for wastewater treatment and lipid production by the marine diatom Phaeodactylum tricornutum. Bioresour. Technol. 2019, 289, 121681. [Google Scholar] [CrossRef] [PubMed]
- Su, M.; D’Imporzano, G.; Veronesi, D.; Afric, S.; Adani, F. Phaeodactylum tricornutum cultivation under mixotrophic conditions with glycerol supplied with ultrafiltered digestate: A simple biorefinery approach recovering C and N. J. Biotechnol. 2020, 323, 73–81. [Google Scholar] [CrossRef] [PubMed]
- Subasankari, K.; Thanappan, V.; Jeyapragash, D.; Anantharaman, P.; Sarangi, R.K. Growth promoting studies on co-culturing Nannochloropsis oceanica with Halomonas aquamarina actively enhance the algal biomass and lipid production. Biocatal. Agric. Biotechnol. 2020, 29, 101790. [Google Scholar]
- Mitra, M.; Patidar, S.K.; George, B.; Shah, F.; Mishra, S. A euryhaline Nannochloropsis gaditana with potential for nutraceutical (EPA) and biodiesel production. Algal Res. 2015, 8, 161–167. [Google Scholar] [CrossRef]
- Ma, X.; Liu, J.; Liu, B.; Chen, T.; Yang, B.; Chen, F. Physiological and biochemical changes reveal stress-associated photosynthetic carbon partitioning into triacylglycerol in the oleaginous marine alga Nannochloropsis oculata. Algal Res. 2016, 16, 28–35. [Google Scholar] [CrossRef]
- Pal, D.; Khozin-Goldberg, I.; Didi-Cohen, S.; Solovchenko, A.; Batushansky, A.; Kaye, Y.; Sikron, N.; Samani, T.; Fait, A.; Boussiba, S. Growth, lipid production and metabolic adjustments in the euryhaline eustigmatophyte Nannochloropsis oceanica CCALA 804 in response to osmotic downshift. Appl. Microbiol. Biotechnol. 2013, 97, 8291–8306. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Cheng, J.; Xu, X.; Lu, H.; Wang, Y.; Li, X.; Yang, W. Using polyethylene glycol to promote Nannochloropsis oceanica growth with 15 vol% CO2. Sci. Total Environ. 2020, 720, 137598. [Google Scholar] [CrossRef]
- Yang, Y.H.; Du, L.; Hosokawa, M.; Miyashita, K.; Kokubun, Y.; Arai, H.; Taroda, H. Fatty acid and lipid class composition of the microalga Phaeodactylum tricornutum. J. Oleo Sci. 2017, 66, 363–368. [Google Scholar] [CrossRef] [Green Version]
- KaiXian, Q.; Borowitzka, M.A. Light and nitrogen deficiency effects on the growth and composition of Phaeodactylum tricornutum. Appl. Biochem. Biotechnol. 1993, 38, 93–103. [Google Scholar] [CrossRef]
- Sharma, N.; Fleurent, G.; Awwad, F.; Cheng, M.; Meddeb-Mouelhi, F.; Budge, S.M.; Germain, H.; Desgagné-Penix, I. Red light variation an effective alternative to regulate biomass and lipid profiles in Phaeodactylum tricornutum. Appl. Sci. 2020, 10, 2531. [Google Scholar] [CrossRef] [Green Version]
- Zhang, H.; Yin, W.; Ma, D.; Liu, X.; Xu, K.; Liu, J. Phytohormone supplementation significantly increases fatty acid content of Phaeodactylum tricornutum in two-phase culture. J. Appl. Phycol. 2021, 33, 13–23. [Google Scholar] [CrossRef]
- Wu, H.; Li, T.; Wang, G.; Dai, S.; He, H.; Xiang, W. A comparative analysis of fatty acid composition and fucoxanthin content in six Phaeodactylum tricornutum strains from different origins. Chin. J. Oceanol. Limnol. 2016, 34, 391–398. [Google Scholar] [CrossRef]
- Huete-Ortega, M.; Okurowska, K.; Kapoore, R.V.; Johnson, M.P.; Gilmour, D.J.; Vaidyanathan, S. Effect of ammonium and high light intensity on the accumulation of lipids in Nannochloropsis oceanica (CCAP 849/10) and Phaeodactylum tricornutum (CCAP 1055/1). Biotechnol. Biofuels 2018, 11, 60. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Branco-Vieira, M.; San Martin, S.; Agurto, C.; Santos, M.A.D.; Freitas, M.A.; Mata, T.M.; Martins, A.A.; Caetano, N.S. Potential of Phaeodactylum tricornutum for biodiesel production under natural conditions in Chile. Energies 2018, 11, 54. [Google Scholar] [CrossRef] [Green Version]
- Wan, C.; Bai, F.W.; Zhao, X.Q. Effects of nitrogen concentration and media replacement on cell growth and lipid production of oleaginous marine microalga Nannochloropsis oceanica DUT01. Biochem. Eng. J. 2013, 78, 32–38. [Google Scholar] [CrossRef]
- Wang, Z.; Cheng, J.; Li, K.; Zhu, Y.; Liu, J.; Yang, W.; Xu, J.; Park, J.Y. Comparison of photosynthetic carbon fixation of Nannochloropsis oceanica cultivated with carbon suppliers: CO2, NaHCO3 and CH3OH. J. CO2 Util. 2020, 41, 101235. [Google Scholar] [CrossRef]
- Mitra, M.; Mishra, S. Effect of glucose on growth and fatty acid composition of an euryhaline eustigmatophyte Nannochloropsis oceanica under mixotrophic culture condition. Bioresour. Technol. Rep. 2018, 3, 147–153. [Google Scholar] [CrossRef]
- Touliabah, H.E.S.; Almutairi, A.W. Effect of phytohormones supplementation under nitrogen depletion on biomass and lipid production of Nannochloropsis oceanica for integrated application in nutrition and biodiesel. Sustainability 2021, 13, 592. [Google Scholar] [CrossRef]
- Li, T.; Chen, Z.; Wu, J.; Wu, H.; Yang, B.; Dai, L.; Wu, H.; Xiang, W. The potential productivity of the microalga, Nannochloropsis oceanica SCS-1981, in a solar powered outdoor open pond as an aquaculture feed. Algal Res. 2020, 46, 101793. [Google Scholar] [CrossRef]
- Savvidou, M.G.; Sotiroudis, T.G.; Kolisis, F.N. Cell surface and cellular debris-associated heat-stable lipolytic enzyme activities of the marine alga Nannochloropsis oceanica. Biocatal. Biotransform. 2016, 34, 24–32. [Google Scholar] [CrossRef]
- Ra, C.H.; Sirisuk, P.; Jung, J.H.; Jeong, G.T.; Kim, S.K. Effects of light-emitting diode (LED) with a mixture of wavelengths on the growth and lipid content of microalgae. Bioprocess Biosyst. Eng. 2018, 41, 457–465. [Google Scholar] [CrossRef] [PubMed]
- Yuan, W.; Ma, Y.; Wei, W.; Liu, W.; Ding, Y.; Balamurugan, S. Sequential treatment with bicarbonate and low-temperature to potentiate both biomass and lipid productivity in Nannochloropsis oceanica. J. Chem. Technol. Biotechnol. 2019, 94, 3413–3419. [Google Scholar] [CrossRef]





Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yu, S.; Yu, Z.; Hou, Q.; Pei, H. Seawater with Added Monosodium Glutamate Residue (MSGR) Is a Promising Medium for the Cultivation of Two Commercial Marine Microalgae. Water 2022, 14, 975. https://doi.org/10.3390/w14060975
Yu S, Yu Z, Hou Q, Pei H. Seawater with Added Monosodium Glutamate Residue (MSGR) Is a Promising Medium for the Cultivation of Two Commercial Marine Microalgae. Water. 2022; 14(6):975. https://doi.org/10.3390/w14060975
Chicago/Turabian StyleYu, Siteng, Ze Yu, Qingjie Hou, and Haiyan Pei. 2022. "Seawater with Added Monosodium Glutamate Residue (MSGR) Is a Promising Medium for the Cultivation of Two Commercial Marine Microalgae" Water 14, no. 6: 975. https://doi.org/10.3390/w14060975




