Microbial Community Structure and Co-Occurrence Patterns in Closed and Open Subsidence Lake Ecosystems
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Area and Sample Collection
2.2. Physicochemical Analysis
2.3. DNA Extraction, PCR Amplification, and Sequence Analyses
2.4. Statistical Analyses
3. Results
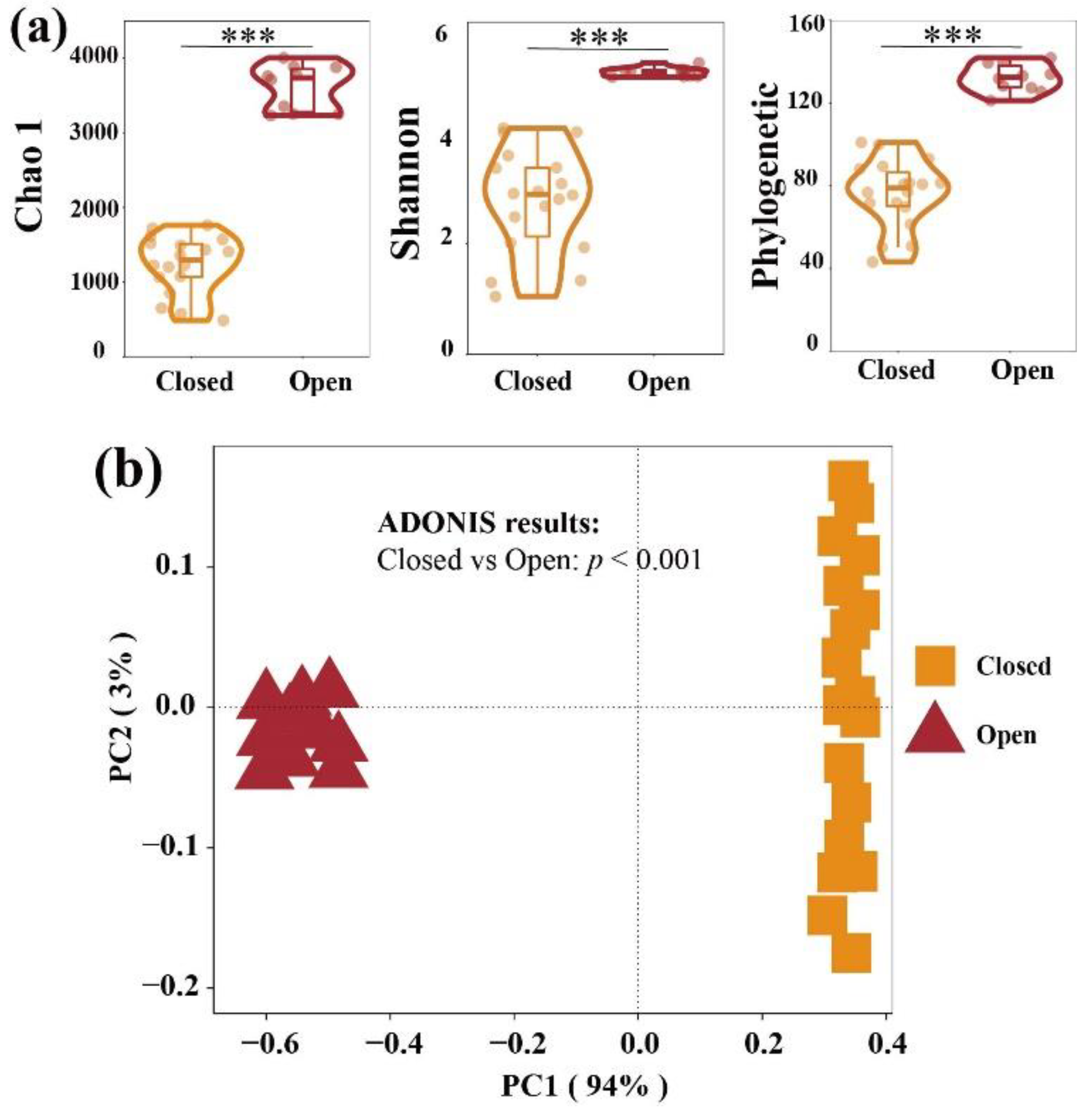
3.1. Alpha and Beta Diversity of Bacterial Communities
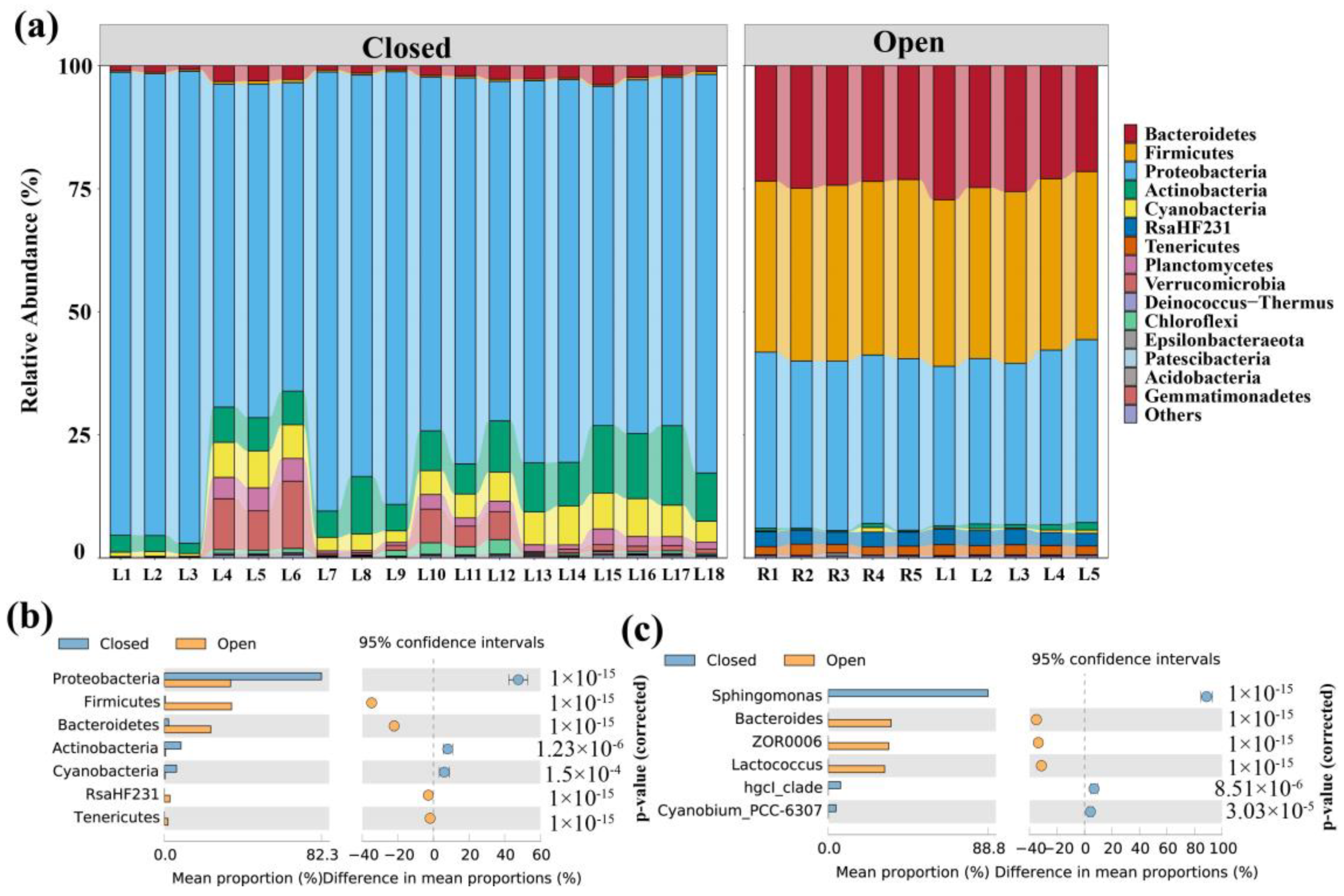
3.2. Composition of Bacterial Communities
3.3. Relationship between Environmental Factors and the Bacterial Community
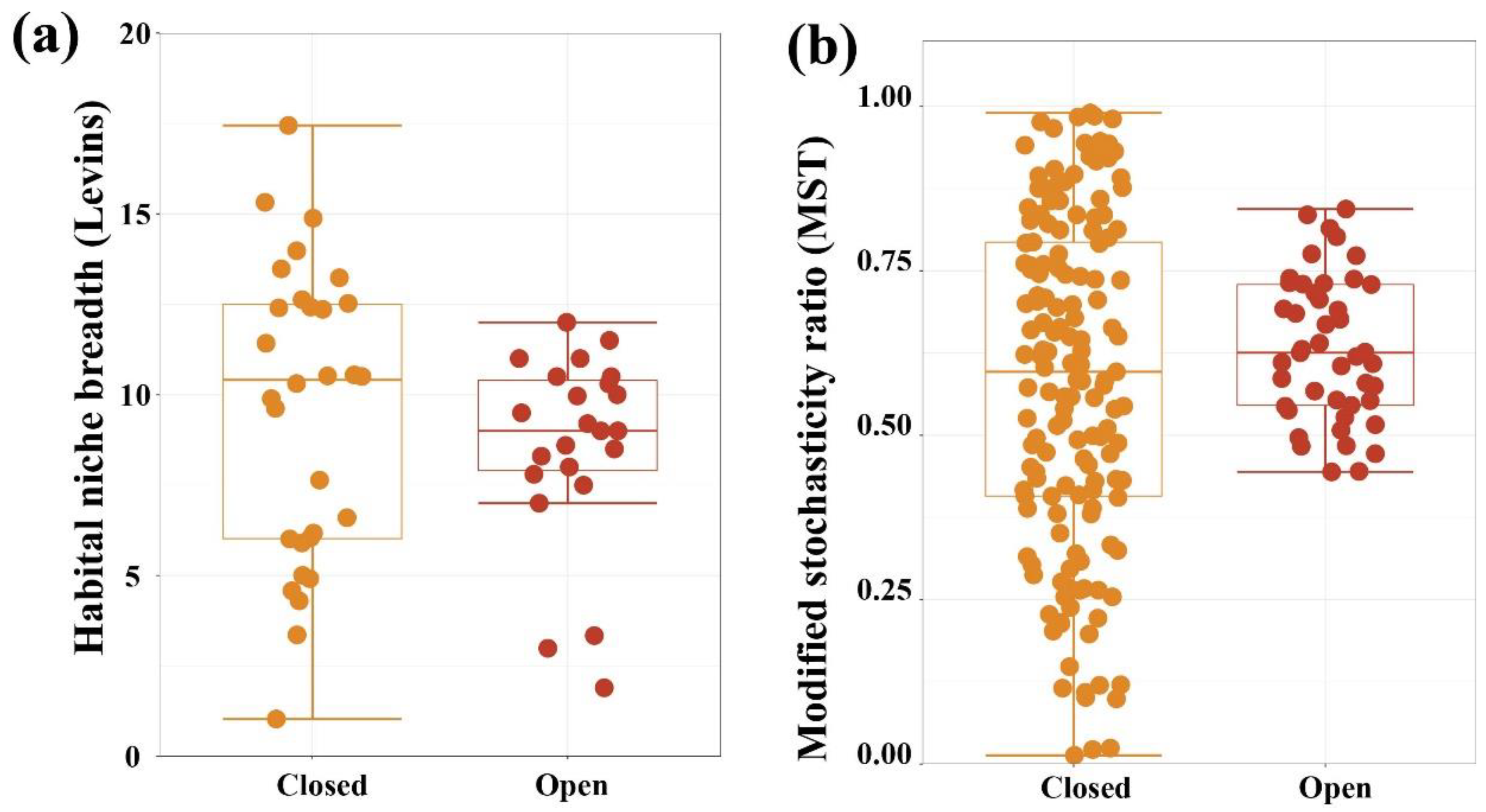
3.4. Assembly Processes of Bacterial Community
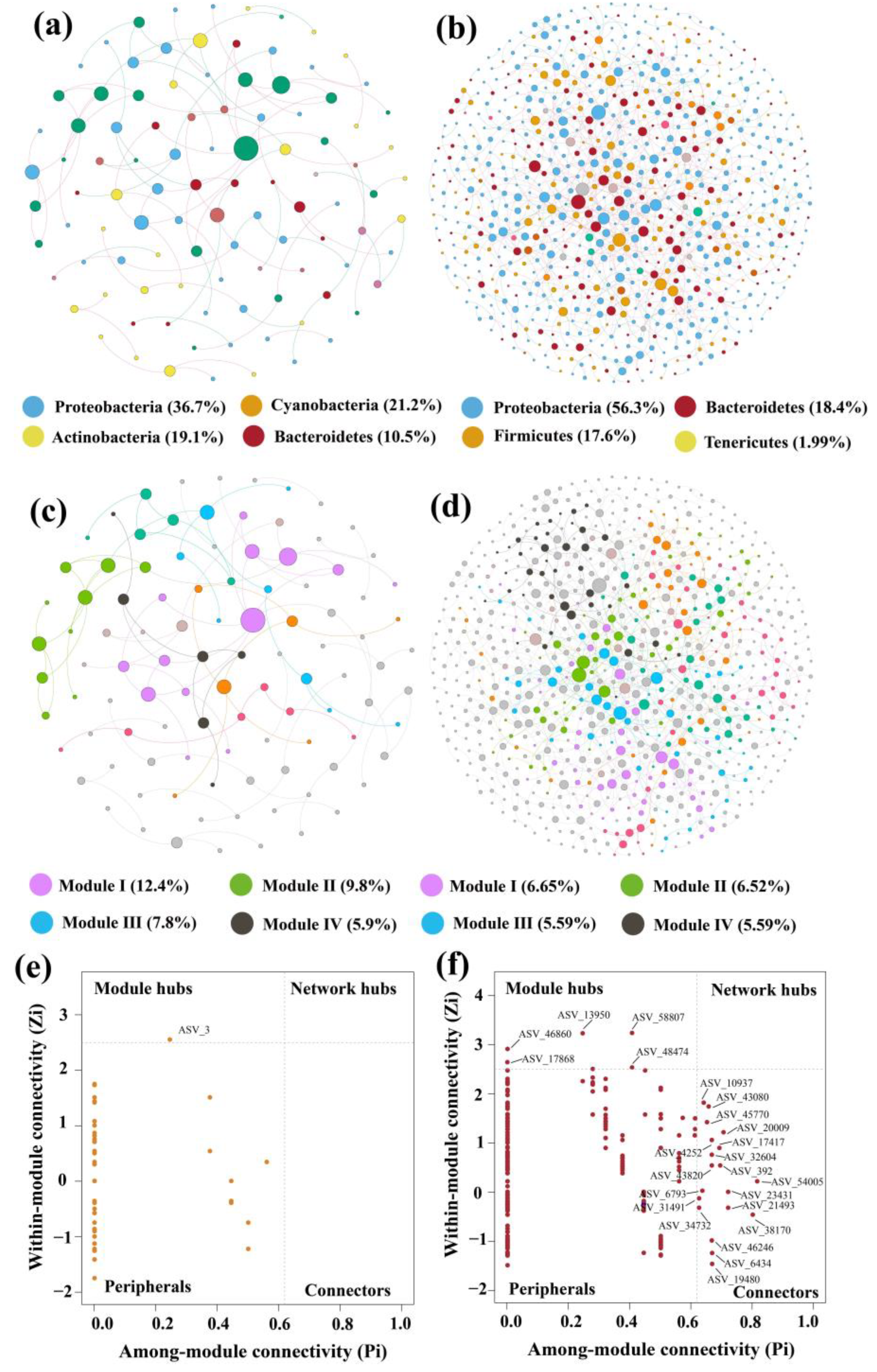
3.5. Network Analysis
4. Discussion
4.1. Community and Functional Diversity of Bacteria in Closed and Open Lakes
4.2. Differences in Bacterial Community Composition and Assembly Processes
4.3. Differences in Co-Occurrence Patterns of Bacterial Communities in Closed and Open Lakes
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Guo, G.; Wang, Y.; Ma, Z. A New Method for Ground Subsidence Control in Coal Mining. J. China Univ. Min. Technol. 2004, 33, 150–153. [Google Scholar]
- Li, X.; Zhang, Q.; Liu, Z.; Yan, J. Influence of Coal Mining on the Lower Yellow River Region and Related Management Countermeasures—Taking Heze Mining Area as an Example. In Proceedings of the MATEC Web of Conferences, Rennes, France, 8–10 September 2022; EDP Sciences: Les Ulis, France, 2022; Volume 363, p. 1023. [Google Scholar]
- Huang, J.; Tian, C.; Xing, L.; Bian, Z.; Miao, X. Green and Sustainable Mining: Underground Coal Mine Fully Mechanized Solid Dense Stowing-Mining Method. Sustainability 2017, 9, 1418. [Google Scholar] [CrossRef]
- Wang, F.; Jiang, B.; Chen, S.; Ren, M. Surface Collapse Control under Thick Unconsolidated Layers by Backfilling Strip Mining in Coal Mines. Int. J. Rock Mech. Min. Sci. 2019, 113, 268–277. [Google Scholar] [CrossRef]
- Zhang, Y.; Cao, S.; Zhang, N.; Zhao, C. The Application of Short-Wall Block Backfill Mining to Preserve Surface Water Resources in Northwest China. J. Clean. Prod. 2020, 261, 121232. [Google Scholar] [CrossRef]
- Hu, Z.; Duo, L.; Shao, F. Optimal Thickness of Soil Cover for Reclaiming Subsided Land with Yellow River Sediments. Sustainability 2018, 10, 3853. [Google Scholar] [CrossRef]
- Zhao, D.; Zeng, Y.; Wu, Q.; Mei, A.; Gao, S.; Du, X.; Yang, W. Hydrogeochemical Characterization and Suitability Assessment of Groundwater in a Typical Coal Mining Subsidence Area in China Using Self-Organizing Feature Map. Environ. Earth Sci. 2022, 81, 507. [Google Scholar] [CrossRef]
- Ouyang, Z.; Gao, L.; Yang, C. Distribution, Sources and Influence Factors of Polycyclic Aromatic Hydrocarbon at Different Depths of the Soil and Sediments of Two Typical Coal Mining Subsidence Areas in Huainan, China. Ecotoxicol. Environ. Saf. 2018, 163, 255–265. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; Chen, X.; Chen, Y.; Li, C.; Zheng, L. Nitrate Sources and Transformations in Surface Water of a Mining Area Due to Intensive Mining Activities: Emphasis on Effects on Distinct Subsidence Waters. J. Environ. Manag. 2021, 298, 113451. [Google Scholar] [CrossRef]
- Yi, Q.; Xu, X.; Qu, X.; Zhang, M. Characterization of Primary Productivity in the Aquatic Zones around the Huainan and Huaibei Coalmine Subsidence Areas. China Environ. Sci. 2014, 34, 2101–2110. [Google Scholar]
- Wang, X.; Zhao, Y.; Chu, Z.; Yu, X.; Fan, T.; Li, G.; Zhang, J.; Xu, X.; Zhen, Q.; Cheng, X. Influence of Solar Photovoltaic System on the Concentration and Environmental Risks of Heavy Metals in Subsidence Pond Water from Coal Mining Area: A Case Study from Huainan Subsidence Pond. Water 2022, 14, 2257. [Google Scholar] [CrossRef]
- Jiao, S.; Qi, J.; Jin, C.; Liu, Y.; Wang, Y.; Pan, H.; Chen, S.; Liang, C.; Peng, Z.; Chen, B. Core Phylotypes Enhance the Resistance of Soil Microbiome to Environmental Changes to Maintain Multifunctionality in Agricultural Ecosystems. Glob. Chang. Biol. 2022, 28, 6653–6664. [Google Scholar] [CrossRef] [PubMed]
- Wall, D.H.; Nielsen, U.N.; Six, J. Soil Biodiversity and Human Health. Nature 2015, 528, 69–76. [Google Scholar] [CrossRef] [PubMed]
- Lu, M.; Wang, X.; Li, H.; Jiao, J.J.; Luo, X.; Luo, M.; Yu, S.; Xiao, K.; Li, X.; Qiu, W. Microbial Community Assembly and Co-Occurrence Relationship in Sediments of the River-Dominated Estuary and the Adjacent Shelf in the Wet Season. Environ. Pollut. 2022, 308, 119572. [Google Scholar] [CrossRef] [PubMed]
- Rathour, R.; Gupta, J.; Mishra, A.; Rajeev, A.C.; Dupont, C.L.; Thakur, I.S. A Comparative Metagenomic Study Reveals Microbial Diversity and Their Role in the Biogeochemical Cycling of Pangong Lake. Sci. Total Environ. 2020, 731, 139074. [Google Scholar] [CrossRef]
- Yang, Y.; Chen, J.; Chen, X.; Jiang, Q.; Liu, Y.; Xie, S. Cyanobacterial Bloom Induces Structural and Functional Succession of Microbial Communities in Eutrophic Lake Sediments. Environ. Pollut. 2021, 284, 117157. [Google Scholar] [CrossRef]
- Xuan, Y.; Mai, Y.; Xu, Y.; Zheng, J.; He, Z.; Shu, L.; Cao, Y. Enhanced Microbial Nitrification-Denitrification Processes in a Subtropical Metropolitan River Network. Water Res. 2022, 222, 118857. [Google Scholar] [CrossRef]
- Wang, L.; Zhang, J.; Li, H.; Yang, H.; Peng, C.; Peng, Z.; Lu, L. Shift in the Microbial Community Composition of Surface Water and Sediment along an Urban River. Sci. Total Environ. 2018, 627, 600–612. [Google Scholar] [CrossRef]
- Raven, M.R.; Keil, R.G.; Webb, S.M. Microbial Sulfate Reduction and Organic Sulfur Formation in Sinking Marine Particles. Science 2021, 371, 178–181. [Google Scholar] [CrossRef]
- Wang, L.; Liang, Z.; Guo, Z.; Cong, W.; Song, M.; Wang, Y.; Jiang, Z. Response Mechanism of Microbial Community to Seasonal Hypoxia in Marine Ranching. Sci. Total Environ. 2022, 811, 152387. [Google Scholar] [CrossRef]
- She, Z.; Pan, X.; Wang, J.; Shao, R.; Wang, G.; Wang, S.; Yue, Z. Vertical Environmental Gradient Drives Prokaryotic Microbial Community Assembly and Species Coexistence in a Stratified Acid Mine Drainage Lake. Water Res. 2021, 206, 117739. [Google Scholar] [CrossRef]
- Liu, S.; Yu, H.; Yu, Y.; Huang, J.; Zhou, Z.; Zeng, J.; Chen, P.; Xiao, F.; He, Z.; Yan, Q. Ecological Stability of Microbial Communities in Lake Donghu Regulated by Keystone Taxa. Ecol. Indic. 2022, 136, 108695. [Google Scholar] [CrossRef]
- Fang, W.; Fan, T.; Xu, L.; Wang, S.; Wang, X.; Lu, A.; Chen, Y. Seasonal Succession of Microbial Community Co-Occurrence Patterns and Community Assembly Mechanism in Coal Mining Subsidence Lakes. Front. Microbiol. 2023, 14, 202. [Google Scholar] [CrossRef] [PubMed]
- Fan, T.; Fang, W.; Zhao, Y.; Lu, A.; Wang, S.; Wang, X.; Xu, L.; Wei, X.; Zhang, L. Spatial Variations of Aquatic Bacterial Community Structure and Co-Occurrence Patterns in a Coal Mining Subsidence Lake. Diversity 2022, 14, 674. [Google Scholar] [CrossRef]
- Duan, Y.; Wang, X.; Wang, L.; Lian, J.; Wang, W.; Wu, F.; Li, Y.; Li, Y. Biogeographic Patterns of Soil Microbe Communities in the Deserts of the Hexi Corridor, Northern China. Catena 2022, 211, 106026. [Google Scholar] [CrossRef]
- Hu, X.; Liu, J.; Liang, A.; Li, L.; Yao, Q.; Yu, Z.; Li, Y.; Jin, J.; Liu, X.; Wang, G. Conventional and Conservation Tillage Practices Affect Soil Microbial Co-Occurrence Patterns and Are Associated with Crop Yields. Agric. Ecosyst. Environ. 2021, 319, 107534. [Google Scholar] [CrossRef]
- Das, A.; Lal, R.; Patel, D.P.; Idapuganti, R.G.; Layek, J.; Ngachan, S.V.; Ghosh, P.K.; Bordoloi, J.; Kumar, M. Effects of Tillage and Biomass on Soil Quality and Productivity of Lowland Rice Cultivation by Small Scale Farmers in North Eastern India. Soil Tillage Res. 2014, 143, 50–58. [Google Scholar] [CrossRef]
- Banerjee, S.; Walder, F.; Büchi, L.; Meyer, M.; Held, A.Y.; Gattinger, A.; Keller, T.; Charles, R.; van der Heijden, M.G.A. Agricultural Intensification Reduces Microbial Network Complexity and the Abundance of Keystone Taxa in Roots. ISME J. 2019, 13, 1722–1736. [Google Scholar] [CrossRef]
- Fuhrman, J.A. Microbial Community Structure and Its Functional Implications. Nature 2009, 459, 193. [Google Scholar] [CrossRef]
- Shi, L.; Xia, P.; Lin, T.; Li, G.; Wang, T.; Du, X. Temporal Succession of Bacterial Community Structure, Co-Occurrence Patterns, and Community Assembly Process in Epiphytic Biofilms of Submerged Plants in a Plateau Lake. Microb. Ecol. 2023, 85, 87–99. [Google Scholar] [CrossRef]
- Yan, Z.; Hao, Z.; Wu, H.; Jiang, H.; Yang, M.; Wang, C. Co-Occurrence Patterns of the Microbial Community in Polycyclic Aromatic Hydrocarbon-Contaminated Riverine Sediments. J. Hazard. Mater. 2019, 367, 99–108. [Google Scholar] [CrossRef]
- Yuan, B.; Guo, M.; Wu, W.; Zhou, X.; Li, M.; Xie, S. Spatial and Seasonal Patterns of Sediment Bacterial Communities in Large River Cascade Reservoirs: Drivers, Assembly Processes, and Co-Occurrence Relationship. Microb. Ecol. 2022, 85, 586–603. [Google Scholar] [CrossRef]
- Wang, H.; Zhang, W.; Li, Y.; Gao, Y.; Niu, L.; Zhang, H.; Wang, L. Hydrodynamics-Driven Community Coalescence Determines Ecological Assembly Processes and Shifts Bacterial Network Stability in River Bends. Sci. Total Environ. 2023, 858, 159772. [Google Scholar] [CrossRef]
- Liu, Z.; Gu, H.; Liang, A.; Li, L.; Yao, Q.; Xu, Y.; Liu, J.; Jin, J.; Liu, X.; Wang, G. Conservation Tillage Regulates Soil Bacterial Community Assemblies, Network Structures and Ecological Functions in Black Soils. Plant Soil 2021, in press. [Google Scholar] [CrossRef]
- Jiao, C.; Zhao, D.; Zeng, J.; Guo, L.; Yu, Z. Disentangling the Seasonal Co-Occurrence Patterns and Ecological Stochasticity of Planktonic and Benthic Bacterial Communities within Multiple Lakes. Sci. Total Environ. 2020, 740, 140010. [Google Scholar] [CrossRef]
- Banerjee, S.; Schlaeppi, K.; van der Heijden, M.G.A. Keystone Taxa as Drivers of Microbiome Structure and Functioning. Nat. Rev. Microbiol. 2018, 16, 567–576. [Google Scholar] [CrossRef]
- Herren, C.M.; McMahon, K.D. Keystone Taxa Predict Compositional Change in Microbial Communities. Environ. Microbiol. 2018, 20, 2207–2217. [Google Scholar] [CrossRef] [PubMed]
- Xie, K.; Xu, X.; Zhang, L. Fractions of Phosphorus and Migration in Sediments in Flooded Sinkages Around Panxie Coal Mine in Huainan. J. Ecol. Rural Environ. 2015, 31, 211–217. [Google Scholar]
- An, S.; Zhao, Q.; Jiang, C.; Wang, S.; Chen, Y.; Xie, H.; Zheng, L. Health Risk Assessment of Heavy Metals in Stagnant Water in Huainan Coal Mining Subsidence Area. China Min. Mag. 2020, 29, 88–93. [Google Scholar]
- Zhang, Y.; Ma, S.; Ding, T. Research Progress of Heavy Metal Pollution in Water and Sediment in Huainan Coal Mining Subsidence Area. Agric. Technol. 2022, 42, 123. [Google Scholar]
- Jin, X.C.; Tu, Q.Y. The Standard Methods for Observation and Analysis in Lake Eutrophication; Chinese Environmental Science Press: Beijing, China, 1990; Volume 240. [Google Scholar]
- Zhang, L.; Delgado-Baquerizo, M.; Shi, Y.; Liu, X.; Yang, Y.; Chu, H. Co-Existing Water and Sediment Bacteria Are Driven by Contrasting Environmental Factors across Glacier-Fed Aquatic Systems. Water Res. 2021, 198, 117139. [Google Scholar] [CrossRef]
- Zhang, L.; Yin, W.; Wang, C.; Zhang, A.; Zhang, H.; Zhang, T.; Ju, F. Untangling Microbiota Diversity and Assembly Patterns in the World’s Largest Water Diversion Canal. Water Res. 2021, 204, 117617. [Google Scholar] [CrossRef] [PubMed]
- Borcard, D.; Gillet, F.; Legendre, P. Numerical Ecology with R; Springer: New York, NY, USA, 2011; Volume 2. [Google Scholar]
- Lai, J.; Zou, Y.; Zhang, J.; Peres-Neto, P.R. Generalizing Hierarchical and Variation Partitioning in Multiple Regression and Canonical Analyses Using the Rdacca. Hp R Package. Methods Ecol. Evol. 2022, 13, 782–788. [Google Scholar] [CrossRef]
- Louca, S.; Parfrey, L.W.; Doebeli, M. Decoupling Function and Taxonomy in the Global Ocean Microbiome. Science 2016, 353, 1272–1277. [Google Scholar] [CrossRef]
- Parks, D.H.; Tyson, G.W.; Hugenholtz, P.; Beiko, R.G. STAMP: Statistical Analysis of Taxonomic and Functional Profiles. Bioinformatics 2014, 30, 3123–3124. [Google Scholar] [CrossRef] [PubMed]
- Legendre, P.; Legendre, L. Numerical Ecology; Elsevier: Amsterdam, The Netherlands, 2012; ISBN 0444538690. [Google Scholar]
- Ning, D.; Deng, Y.; Tiedje, J.M.; Zhou, J. A General Framework for Quantitatively Assessing Ecological Stochasticity. Proc. Natl. Acad. Sci. USA 2019, 116, 16892–16898. [Google Scholar] [CrossRef]
- Barberán, A.; Bates, S.T.; Casamayor, E.O.; Fierer, N. Using Network Analysis to Explore Co-Occurrence Patterns in Soil Microbial Communities. ISME J. 2012, 6, 343–351. [Google Scholar] [CrossRef] [PubMed]
- Nemergut, D.R.; Schmidt, S.K.; Fukami, T.; O’Neill, S.P.; Bilinski, T.M.; Stanish, L.F.; Knelman, J.E.; Darcy, J.L.; Lynch, R.C.; Wickey, P. Patterns and Processes of Microbial Community Assembly. Microbiol. Mol. Biol. Rev. 2013, 77, 342–356. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.; Jiang, Y.-H.; Yang, Y.; He, Z.; Luo, F.; Zhou, J. Molecular Ecological Network Analyses. BMC Bioinform. 2012, 13, 113. [Google Scholar] [CrossRef]
- Horner-Devine, M.C.; Carney, K.M.; Bohannan, B.J.M. An Ecological Perspective on Bacterial Biodiversity. Proc. R. Soc. Lond. Ser. B Biol. Sci. 2004, 271, 113–122. [Google Scholar] [CrossRef]
- Zheng, W.; Li, R.; Qin, W.; Chen, B.; Wang, M.; Guan, W.; Zhang, X.; Yang, Q.; Zhao, M.; Ma, Z. Tidal Water Exchanges Can Shape the Phytoplankton Community Structure and Reduce the Risk of Harmful Cyanobacterial Blooms in a Semi-Closed Lake. J. Oceanol. Limnol. 2022, 40, 1868–1880. [Google Scholar] [CrossRef]
- Ruiz-González, C.; Niño-García, J.P.; Del Giorgio, P.A. Terrestrial Origin of Bacterial Communities in Complex Boreal Freshwater Networks. Ecol. Lett. 2015, 18, 1198–1206. [Google Scholar] [CrossRef]
- Yang, Y.; Gao, Y.; Huang, X.; Ni, P.; Wu, Y.; Deng, Y.; Zhan, A. Adaptive Shifts of Bacterioplankton Communities in Response to Nitrogen Enrichment in a Highly Polluted River. Environ. Pollut. 2019, 245, 290–299. [Google Scholar] [CrossRef] [PubMed]
- Tang, X.; Xie, G.; Shao, K.; Hu, Y.; Cai, J.; Bai, C.; Gong, Y.; Gao, G. Contrast Diversity Patterns and Processes of Microbial Community Assembly in a River-Lake Continuum across a Catchment Scale in Northwestern China. Environ. Microbiome 2020, 15, 10. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Deng, Y.E.; Zhang, P.; Xue, K.; Liang, Y.; Van Nostrand, J.D.; Yang, Y.; He, Z.; Wu, L.; Stahl, D.A. Stochasticity, Succession, and Environmental Perturbations in a Fluidic Ecosystem. Proc. Natl. Acad. Sci. USA 2014, 111, E836–E845. [Google Scholar] [CrossRef]
- Song, Y.; Wang, Y.; Mao, G.; Gao, G.; Wang, Y. Impact of Planktonic Low Nucleic Acid-Content Bacteria to Bacterial Community Structure and Associated Ecological Functions in a Shallow Lake. Sci. Total Environ. 2019, 658, 868–878. [Google Scholar] [CrossRef] [PubMed]
- Allers, E.; Gómez-Consarnau, L.; Pinhassi, J.; Gasol, J.M.; Šimek, K.; Pernthaler, J. Response of Alteromonadaceae and Rhodobacteriaceae to Glucose and Phosphorus Manipulation in Marine Mesocosms. Environ. Microbiol. 2007, 9, 2417–2429. [Google Scholar] [CrossRef]
- Shapovalova, A.A.; Khijniak, T.V.; Tourova, T.P.; Muyzer, G.; Sorokin, D.Y. Heterotrophic Denitrification at Extremely High Salt and PH by Haloalkaliphilic Gammaproteobacteria from Hypersaline Soda Lakes. Extremophiles 2008, 12, 619–625. [Google Scholar] [CrossRef]
- Kolvenbach, B.A.; Corvini, P.-X. The Degradation of Alkylphenols by Sphingomonas Sp. Strain TTNP3—A Review on Seven Years of Research. New Biotechnol. 2012, 30, 88–95. [Google Scholar] [CrossRef]
- Pfeifer, F. Distribution, Formation and Regulation of Gas Vesicles. Nat. Rev. Microbiol. 2012, 10, 705–715. [Google Scholar] [CrossRef]
- Huisman, J.; Codd, G.A.; Paerl, H.W.; Ibelings, B.W.; Verspagen, J.M.H.; Visser, P.M. Cyanobacterial Blooms. Nat. Rev. Microbiol. 2018, 16, 471–483. [Google Scholar] [CrossRef]
- Ahmed, W.; Yusuf, R.; Hasan, I.; Goonetilleke, A.; Gardner, T. Quantitative PCR Assay of Sewage-Associated Bacteroides Markers to Assess Sewage Pollution in an Urban Lake in Dhaka, Bangladesh. Can. J. Microbiol. 2010, 56, 838–845. [Google Scholar] [CrossRef] [PubMed]
- Ge, Y.; Lou, Y.; Xu, M.; Wu, C.; Meng, J.; Shi, L.; Xia, F.; Xu, Y. Spatial Distribution and Influencing Factors on the Variation of Bacterial Communities in an Urban River Sediment. Environ. Pollut. 2021, 272, 115984. [Google Scholar] [CrossRef]
- Shi, X.; Ng, K.K.; Li, X.-R.; Ng, H.Y. Investigation of Intertidal Wetland Sediment as a Novel Inoculation Source for Anaerobic Saline Wastewater Treatment. Environ. Sci. Technol. 2015, 49, 6231–6239. [Google Scholar] [CrossRef]
- Kuang, T.; He, A.; Lin, Y.; Huang, X.; Liu, L.; Zhou, L. Comparative Analysis of Microbial Communities Associated with the Gill, Gut, and Habitat of Two Filter-Feeding Fish. Aquac. Rep. 2020, 18, 100501. [Google Scholar] [CrossRef]
- Zhu, C.; Li, D.; Chen, W.; Ban, S.; Liu, T.; Wen, H.; Jiang, M. Effects of Dietary Host-Associated Lactococcus Lactis on Growth Performance, Disease Resistance, Intestinal Morphology and Intestinal Microbiota of Mandarin Fish (Siniperca chuatsi). Aquaculture 2021, 540, 736702. [Google Scholar] [CrossRef]
- Van der Gucht, K.; Cottenie, K.; Muylaert, K.; Vloemans, N.; Cousin, S.; Declerck, S.; Jeppesen, E.; Conde-Porcuna, J.-M.; Schwenk, K.; Zwart, G. The Power of Species Sorting: Local Factors Drive Bacterial Community Composition over a Wide Range of Spatial Scales. Proc. Natl. Acad. Sci. USA 2007, 104, 20404–20409. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.; Gao, J.; Wu, Q.; Ai, Y.; Huang, Y.; Wei, W.; Sun, S.; Weng, Q. Co-Occurrence Pattern and Function Prediction of Bacterial Community in Karst Cave. BMC Microbiol. 2020, 20, 137. [Google Scholar] [CrossRef] [PubMed]
- Newton, R.J.; Jones, S.E.; Eiler, A.; McMahon, K.D.; Bertilsson, S. A Guide to the Natural History of Freshwater Lake Bacteria. Microbiol. Mol. Biol. Rev. 2011, 75, 14–49. [Google Scholar] [CrossRef]
- Ruprecht, J.E.; Birrer, S.C.; Dafforn, K.A.; Mitrovic, S.M.; Crane, S.L.; Johnston, E.L.; Wemheuer, F.; Navarro, A.; Harrison, A.J.; Turner, I.L. Wastewater Effluents Cause Microbial Community Shifts and Change Trophic Status. Water Res. 2021, 200, 117206. [Google Scholar] [CrossRef]
- Cienfuegos-Martínez, K.; Monroy-Dosta, M.d.C.; Hamdan-Partida, A.; Hernández-Vergara, M.P.; Aguirre-Garrido, J.F.; Bustos-Martínez, J. Effect of the Probiotic Lactococcus Lactis on the Microbial Composition in the Water and the Gut of Freshwater Prawn (Macrobrachium rosenbergii) Cultivate in Biofloc. Aquac. Res. 2022, 53, 3877–3889. [Google Scholar] [CrossRef]
- Huang, W.; Chen, X.; Wang, K.; Chen, J.; Zheng, B.; Jiang, X. Comparison among the Microbial Communities in the Lake, Lake Wetland, and Estuary Sediments of a Plain River Network. Microbiologyopen 2019, 8, e00644. [Google Scholar] [CrossRef] [PubMed]
- Ramírez, C.; Coronado, J.; Silva, A.; Romero, J. Cetobacterium Is a Major Component of the Microbiome of Giant Amazonian Fish (Arapaima gigas) in Ecuador. Animals 2018, 8, 189. [Google Scholar] [CrossRef] [PubMed]
- Chhetri, G.; Kim, I.; Kim, J.; So, Y.; Seo, T. Chryseobacterium tagetis Sp. Nov., a Plant Growth Promoting Bacterium with an Antimicrobial Activity Isolated from the Roots of Medicinal Plant (Tagetes patula). J. Antibiot. 2022, 75, 312–320. [Google Scholar] [CrossRef] [PubMed]
- Breton-Deval, L.; Sanchez-Flores, A.; Juárez, K.; Vera-Estrella, R. Integrative Study of Microbial Community Dynamics and Water Quality along The Apatlaco River. Environ. Pollut. 2019, 255, 113158. [Google Scholar] [CrossRef] [PubMed]
- Reis, M.P.; Suhadolnik, M.L.S.; Dias, M.F.; Ávila, M.P.; Motta, A.M.; Barbosa, F.A.R.; Nascimento, A.M.A. Characterizing a Riverine Microbiome Impacted by Extreme Disturbance Caused by a Mining Sludge Tsunami. Chemosphere 2020, 253, 126584. [Google Scholar] [CrossRef] [PubMed]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, S.; Hu, Y.; Fan, T.; Fang, W.; Liu, X.; Xu, L.; Li, B.; Wei, X. Microbial Community Structure and Co-Occurrence Patterns in Closed and Open Subsidence Lake Ecosystems. Water 2023, 15, 1829. https://doi.org/10.3390/w15101829
Wang S, Hu Y, Fan T, Fang W, Liu X, Xu L, Li B, Wei X. Microbial Community Structure and Co-Occurrence Patterns in Closed and Open Subsidence Lake Ecosystems. Water. 2023; 15(10):1829. https://doi.org/10.3390/w15101829
Chicago/Turabian StyleWang, Shun, Youbiao Hu, Tingyu Fan, Wangkai Fang, Xiaopeng Liu, Liangji Xu, Bing Li, and Xiangping Wei. 2023. "Microbial Community Structure and Co-Occurrence Patterns in Closed and Open Subsidence Lake Ecosystems" Water 15, no. 10: 1829. https://doi.org/10.3390/w15101829
APA StyleWang, S., Hu, Y., Fan, T., Fang, W., Liu, X., Xu, L., Li, B., & Wei, X. (2023). Microbial Community Structure and Co-Occurrence Patterns in Closed and Open Subsidence Lake Ecosystems. Water, 15(10), 1829. https://doi.org/10.3390/w15101829



