1. Introduction
The Ladoga and Onega lakes are Europe’s largest freshwater bodies and are unique reserves of fresh water. The management of these resources through catchments and continued efforts to maintain a high water quality is essential [
1]. Lake Onega, which is located in Karelia and is connected to Ladoga Lake by the Svir’ River, is a domestic potable and industrial water supply source, a main waterway, a commercial fishing zone, a recreation area and a reservoir of the Upper Svir’ Hydroelectric Station. Forest management, mineral mining and other agricultural activities are conducted in the Lake Onega basin. These operations, which are dependent on the lake and its watershed (especially pulp and paper mill activities), can be adversely affected due to variations in hydrological and chemical regimes that are aggravated by climatic warming [
1,
2].
The hydrological monitoring of Lake Onega and its watershed has been carried out for several decades. Various techniques, including meteorological observations, temperature measurements, measurements of the tributary runoff, monitoring the water level of Lake Onega and the mathematical simulation of the hydrological processes have been employed since the early 1960s [
3,
4,
5,
6,
7]. Clearly, the basin is experiencing significant hydrological changes, predominantly through natural processes, but the mechanisms and consequences of these changes are not yet clear [
8]. There is a lack of information about the essential water balance parameters, such as the atmospheric precipitation on the lake surface and watershed, the surface runoff (only the largest tributaries are observed), the groundwater discharge into rivers and directly into Lake Onega and evaporation. Therefore, it is difficult to develop an adequate quantitative description of the water balance through a mathematical simulation of the hydrological processes. Accordingly, modern requirements for the description of the object, which are necessary for making informed management decisions, cannot be satisfied.
This is mainly due to the deterioration of the network observation quality in the 1990s and 2000s that occurred during the period with the most significant climate changes [
8,
9,
10,
11,
12].
Moreover, the exchange time for Lake Onega was estimated by considering the water level fluctuation, Lake Bathymetry and the inflow volume [
13]. Yet, in order to acquire more reliable data, we need to take into account the direct evaporation of the water from the lake surface. If this factor is ignored, the value of the exchange time is underestimated. Meanwhile, the water balance stations that could estimate the evaporation and transpiration in the watershed and evaporation from the lake surface have not existed for 30 years. Another problem is that groundwater might seep into Lake Onega, but this hypothesis is subject to serious debate as observation data are absent. Data on stable isotopes can provide information both for quantifying the loss of water by evaporation and to diagnose the flow of groundwater into the lake.
Environmental isotopes, such as oxygen-18 (
18O) and deuterium (
2H), which are included in water molecules, are widely used as global indicators, are employed to solve water management problems and are used to carry out fundamental research [
14,
15,
16,
17,
18]. These isotopes (represented by δ
18O and δ
2H, hereinafter referred to as the isotope composition of water) are used to understand the genetic relation of surface water bodies to atmospheric precipitation and groundwater, to separate the hydrographs and to assess evaporation. Some reviews present a process-based summary of the important parameters that control δ
18O or δ
2H behavior in natural systems for use in mathematical models [
19,
20]. These studies discuss the assumptions made regarding fractionation factors and present calibration approaches, such as describing the influence of headwater conditions, the seasonality of atmospheric characteristics and specific settings such as a chain of lakes. Several studies have developed coupled hydrology and isotope mass balance models for specific lake systems [
21,
22,
23,
24,
25,
26].
Since 2009, the Northern Water Problems Institute (NWPI) at the Karelian Research Centre of RAS and Saint Petersburg State University (SPbSU) has been studying the water dynamics in the lake and the conditions of water-balance formation on the watershed by using stable isotope tracers (deuterium and oxygen-18) to better understand the processes responsible for Lake Onega’s conditions. Measurements in 2012–2017 in the Petrozavodsk Bay and its three tributaries and in the central part of Lake Onega showed that the snowmelt floods had the greatest effect on the water dynamics in the bay while evaporation had the greatest effect in the open lake [
27,
28]. Precipitation, tributaries and groundwater were sampled, and the impact of evaporation on the deuterium and oxygen-18 concentrations in the residual water was experimentally tested. A hydrograph of a small river was studied in detail, and a correlation between the precipitation events, spring flood and isotope composition of the water was obtained.
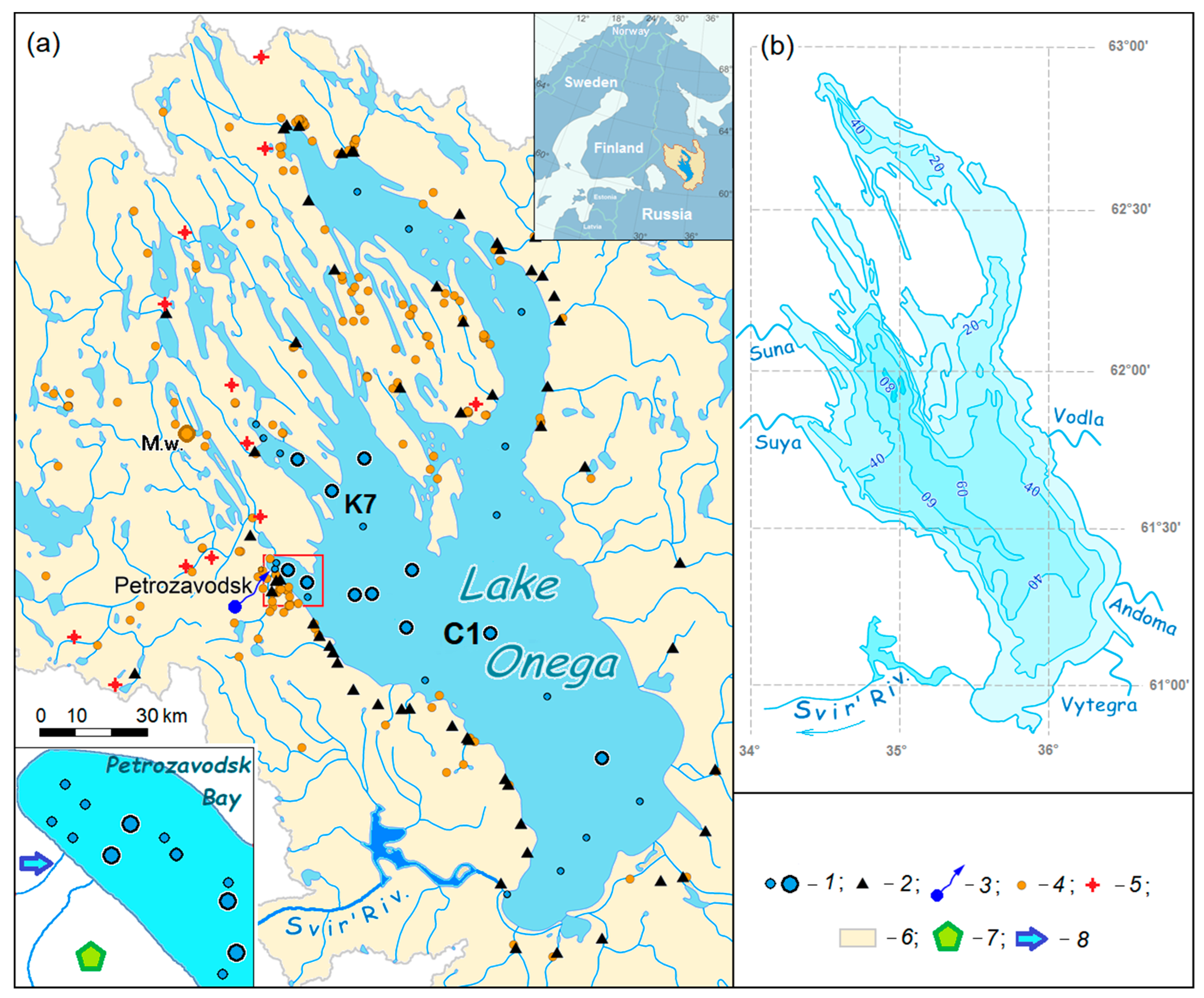
2. Site Description
The water area of Lake Onega is 9.72 × 10
3 km
2, the area of islands is 225 km
2 and the total surface of the catchment is 53.1 × 10
3 km
2 [
6]. The average depth of the lake is 30 m, and the greatest depth is 132.5 m in its central part (
Figure 1). The northern part of the lake has several large bays, which extend up to 70 km inland, with a width of up to 32 km and depths of up to 111–124 m. These depressions are elongated from northwest to southeast, and continuations of the lakes are also elongated in the northwest direction. The lowest annual water level of the lake is usually observed in April before the ice begins to float in the spring, while the highest level is observed from June to July. During the past 20 years, the largest difference between the spring and summer levels was 1.8 m. The transparency of the water reaches 8 m, and the average is about 3–4 m [
1,
29].
In sum, Lake Onega contains 1152 river tributaries, but only 52 are over 10 km long [
6]. Rivers make up 76% of the water balance of Lake Onega (17.1 km
3/year), and 24% of atmospheric precipitation occurs directly over the lake water area. The groundwater inflow is affected by the rivers and is quantitatively taken into account when measuring the hydrograph separation of the annual runoff. The direct groundwater discharge in the lake (bypassing the river network) is less than 1% [
30,
31]. The Svir’ River connects the Onega and Ladoga lakes and carries about 84% (18.8 km
3/year) of the runoff from Lake Onega, while 16% is consumed by evaporation. The water regime of the Svir’ River is completely regulated by the dam in the upper part of the river.
Lake Onega has a dimictic water circulation scheme, which is affected by the wind trajectories and the water inflow from the tributaries. In the winter, the lake is covered with a continuous ice cover. Currents in the lake remain constant only at the mouths of the rivers flowing into the lake and against the mouth of the Svir’ River. A thermobar (frontal zone) is formed in the spring–summer season and divides the lake into two areas: (a) stratified coastal and (b) homothermal deep water. A thermobar begins to form after the ice thaws when the near-shore shallow-water regions are heated to a temperature of maximal density (4 °C) more rapidly than the volume of the deep water. Long-term data show that the thermobar disappears in late June. Direct thermal stratification is then established and a summer heating period begins [
1,
6].
By the time of the June sampling, the water in the bays had already been warmed. The temperature of the surface layer at some stations was up to 10 °C, and the bottom temperature was 3.6–6.0 °C. In the open lake water, the temperature in June did not exceed 4 °C (minimum 2.5 °C). The sampling in late summer and early autumn overlapped with the onset of homeothermy in the lake at a mean water temperature of 6–8 °C. In this period, the water temperature started to decrease. The upper part of the water column, which reaches up to 20 m, was still warm (with temperatures up to +9.5 °C), while the lower part was colder (maximum +6.6 °C).
3. Materials and Methods
Lake water area. Water samples were collected from the “Ecologist” Research Vessel using equipment from the Core Facility of the Karelian Research Centre of the Russian Academy of Sciences (Petrozavodsk city, Russia) at monitoring stations located in the bays and in the open part of the lake (
Figure 1). Fieldwork mainly took place in the summer and autumn from 2012–2017. In March 2015, 2016 and 2017, water samples were taken from the ice at Petrozavodsk Bay (stations with index “P”,
Figure 1) and in the central part of the lake (stations “C”). Seven ice samples were also taken in Petrozavodsk Bay. At most stations, the water samples were collected with a bathometer from the surface layer (1 m) and the near-bottom layer. At some stations, the water body was sampled in more detail (by 4–8 different intervals at depth). The samples were filtered, poured into 20–40 mL plastic vials, hermetically sealed and stored in a refrigerator at 4 degrees, usually in an intermediate container, to avoid the influence of evaporation. About 415 water samples were analyzed in total.
Tributaries. From 2014–2018, the largest tributaries in the lower reach of the rivers were sampled in different seasons (
Figure 1). In 2008–2018 and in 2012–2017, runoff monitoring (flow volume, temperature, electrical conductivity, δ
2H and δ
18O analyses) was carried out in a small river called the Neglinka River (with a watershed area of 17 km
2). Samples from the Neglinka River were collected weekly during low water periods and daily during spring flooding. A total of 150 samples from 53 of the largest tributaries and about 420 samples from the Neglinka River were analyzed.
Groundwater. The basin of Lake Onega is located at the junction of the Baltic shield and the Russian platform. Water from the sedimentary complexes and crystalline rocks was analyzed. Samples were taken only from flowing wells or operating wells with an electrical pump and from ascending springs. This method allows for the exclusion of the influence of human, seasonal and other random factors. About 280 water samples were collected.
Atmospheric precipitation. The collection of the atmospheric precipitation was carried out in Petrozavodsk city on the roof of the IWPN building (61°47′ N, 34°21′ E, altitude 94 m) in 2009–2018. Samples were taken based on a weekly average and after periods of massive precipitation. Depending on the weather conditions, the number of samples per month varied from 2 to 12. Rainfall was collected by a sampler consisting of a receiving funnel and a 1-liter storage tank, to which synthetic oil was added to prevent evaporation. Snow was collected in a plastic bag and placed in a rain gauge. The snow was then melted at room temperature, and the volume of the water was measured. A total of 413 samples were analyzed. The isotope composition of the snow cover was studied using a submeridional and two sublatitudinal profiles in March 2016. Forty-five sites were sampled, and the results are described in Borodulina et al. [
32].
Evaporation experiments. In 2014–2016, a total of 28 experiments were carried out to estimate the influence of the disequilibrium isotopic fractionation during evaporation on the deuterium and oxygen-18 concentrations in the residual water. Atmospheric precipitation with an initial volume of 3 L, which was poured into the glass chemical crystallizer, was used for the experiment, and every one-to-two days, a 2 mL sample was taken from the evaporator. At the same time, the temperature and volume of the residual water were measured (the air temperature was taken from the weather station). During the experiment, the isotopic composition of the residual water became less depleted, which was clearly seen during the analysis. The approximation lines on the deuterium vs. oxygen-18 diagram for the evaporation process in April–May and the first half of June were δ2H = 4.69 × δ18O − 33.0 and Δ(δ18O) = 25.9 × ξ, and those for July–September were δ2H = 5.59 × δ18O − 18.5 and Δ(δ18O) = 17.4 × ξ, where ξ is the amount of evaporated water and Δ(δ18O) is the shift in the isotope composition of oxygen.
Analytical method. The deuterium and oxygen-18 concentrations in the water were measured using the Picarro L-2120-i laser infrared analyzer from the Center of X-ray Diffraction Studies at the Research Park of St. Petersburg State University (St. Petersburg, Russia). All the results are presented as parts per thousand relative to the composition of the average oceanic water (SMOW). The International Atomic Energy Agency standards V-SMOW-2, GISP and SLAP and the United States Geological Survey standards USGS-45 and USGS-46 were used. The uncertainty of the measurements was ±0.1‰ for oxygen-18 and ±1‰ for deuterium.
5. Discussion
Generally, the seasonal variations in the isotope composition of the lake water at the deep stations were less than those at the surface layer (
Figure 5) due to the mixing and the longer residence time of the water in the deep parts of the lake [
29,
33]. In most cases, in the spring, the isotope composition of the surface layer of the lake water was more depleted of deuterium and oxygen-18, and it ranged from −11.6 to −9.3‰ for δ
18O and from −85 to −77‰ for δ
2H.
In autumn, the isotope composition of the surface layer of the lake water varied from −10.3 to −9.3‰ for δ
18O and from −80 to −71‰ for δ
2H (
Figure 5). In the bottom layer, similar changes in the isotopic composition of the water were observed, but the scatter of the average values between spring and autumn was somewhat smaller relative to the surface layer.
On the δ
2H vs. δ
18O diagram, the isotope composition of the lake water in March was approximated by the equation δ
2H = 2.91 × δ
18O − 48 with R
2 = 0.27 (
Figure 6a). This angle coefficient is not similar to the typical coefficient for the evaporation line. This was probably due to the combined effect of the ice cover and the spring flood, which was provoked by the snow melt on the watershed. Firstly, ice greatly decreases the wind-induced mixing of the water in the lake. Secondly, the snowmelt water inflow, which begins somewhat earlier than the complete disappearance of the lake ice, is responsible for the local isotope depletion of the lake water near the tributary mouths [
34].
In addition to the inflow process, other factors also influenced the variation in the stable isotopes. In March, part of the samples from the surface layer of the water demonstrated isotope fractionation, which shifted the figurative points on the δ
2H vs. δ
18O diagram to the left from the LMWL (
Figure 6a). This could have been a result of the new ice formation when the residual water became isotopically depleted in comparison to the initial composition. The isotopic fractionation that occurred during the water freezing could be indirectly demonstrated by the ice composition of Lake Onega. Seven ice samples were taken simultaneously during the testing campaign at the end of winter. The ice cores were divided into upper and bottom layers, where the lower layer was composed of a mass of transparent ice without the bubbly air-saturated and milky interlayers. This massive ice had a composition of δ
18O = −8.4‰ and δ
2H = −62‰ on average as a result of the disequilibrium isotope fractionation when the water froze, and its figurative points were shifted to the right from the LMWL in the δ
2H vs. δ
18O diagram. The upper layer had an ice isotope composition of δ
18O = −9.5‰ and δ
2H = −71‰ on average, which is more isotopically depleted than the ice mass due to snow accumulation. The isotope effects are in good agreement with the databank in terms of the chemical composition of the ice, the snow on the ice and the under-ice water of Lake Onega [
35,
36] and also with the general tendencies of the ice-cover formations on lakes [
37].
In June, evaporation is predominantly active. For the June samples (
Figure 6a), the approximation line on the δ
2H vs. δ
18O diagram had an equation of δ
2H = 5.39 × δ
18O − 23 and the slope coefficient approached to the standard for the evaporation line. At this time, the strength of the winds and water mixing caused them to drop significantly; additionally, the amount of precipitation was still small and the temperature of the air was not high.
By the end of the summer, the range of the δ
18O and δ
2H variation decreased, and the isotope composition of the water became heavier than in the spring (
Figure 6b). This was attributed to the wind mixing of the isotopically heavy summer precipitations and the impact of evaporation. The approximation equation had an angle coefficient of 1.73 for July–August and of 2.54 for the September–October data (
Figure 6b). This was most likely because the isotope composition of the lake water in late summer was the homogenized value due to wind mixing [
3]; therefore, it led to a very large error in the angle parameter assessment by the linear approximation. In particular, the approximation accuracy (R
2) was 0.65 for the June data and only 0.06 for the July–August data. The trend of the isotope composition of the lake water in September–October for this parameter was 0.21.
During July and August, small lakes and swamps on the Onega Lake catchments are warm. The water vapor is transported by the air mass from the catchment on the Onega Lake water area. Here, it condensates from the air due to the lower temperature of the water in Onega Lake [
13] and affects the isotope composition of the surface water of the lake. In this case, the figurative points should also move to the left from the LMWL on the δ
2H vs. δ
18O diagram (
Figure 4b). It is currently impossible to estimate the quantitative contribution of condensation to the water balance of the lake.
Evaporation of the lake water is clearly indicated by the shift of the figurative points to the right from the LMWL in the δ
2H vs. δ
18O diagram (inset in
Figure 2 and
Figure 6), which is the result of the disequilibrium isotope fractionation. Calculations using the experimental relationship between the shift in the oxygen isotopes (Δ(δ
18O)) and the amount of evaporated water (ξ) showed that 4–8% of the water loss due to evaporation occurred in the spring and 4–10% occurred in early autumn, averaging about 9%. These values were somewhat smaller than those obtained by the hydrological calculations. This could be the consequence of the nonuniform sampling network on the lake water area during the isotope study. An additional factor may be that the transpiration account was absent in the standard hydrological calculations, as it was implicitly included in the evaporation [
38].
For the different parts of Lake Onega, a fine structure of the water dynamics was noted in varying depths (see stations C1 and K7 in
Figure 7 and the location of stations in
Figure 1). The Kondopoga Bay (station K7) is one of the large deep-water bays of Lake Onega. The Suna River, which flows in this bay, is the third largest tributary of Lake Onega, and therefore the calculated exchange time of the water in the bay is small and is not more than a year. As result, the seasonal variation in the isotope composition of the water in the bay depends entirely on the composition of the water in the Suna River. Actually, the range of the δ
2H and δ
18O variation in the bay (station K7 in
Figure 7) was lower than that in the open lake (station C1 in
Figure 7) because the evaporation and direct rainfall strongly affected the lake water at end of the summer.
Variations in the isotope composition of the water in Petrozavodsk Bay are shown in
Figure 8 (see the location at the inset in
Figure 1). In June, after the meltwater flood, the lake water displayed the highest degree of depletion in the isotope composition, which was closest to the weighted mean annual composition of the precipitation. In the previous hydrological period (March in
Figure 8), the ice cover was not completely thawed, and the isotope composition of the water was slightly heavier than in June (the points on the δ
2H vs. δ
18O diagram shifted more to the right of the LMWL). This was the result of mixing the water with the heavy isotope composition from the previous year (see August in
Figure 8) and the winter river runoff with the depleted isotope composition (
Figure 4). At the end of the summer (August in
Figure 8), the isotope composition of the water was the heaviest of the year.
The river water with an average composition of δ
18O ≈ −13.6‰ and δ
2H ≈ −98‰ was much more isotopically depleted than the mean annual composition of the precipitation and lake water due to the discharge of the groundwater (
Figure 4). In the northwestern regions of Russia, the isotope depletion of the groundwater was the result of the groundwater recharge mainly during the spring snowmelt flood. Additionally, in the last two decades, the highly depleted isotopic composition of the river water in the winter could also be associated with the more frequent winter thaws when the isotopically light snowmelt water is added to the groundwater discharge in the river.
For the major Karelian rivers, the seasonal dependence of the isotope composition of the water is not as strong as that for minor streams (see the above example for the Suna River). This is probably because with the large watershed, the seasonal precipitations are mixed, and the river runoff redistributes this water on an interannual scale, as it does for the other great watersheds [
39,
40]. In the western part of Lake Onega’s catchment, the large lakes and wetlands are absent. Consequently, here the minor rivers display a supply dominated by groundwater throughout the year, as indicated by the more depleted isotope composition of the water even in the summer, whereby δ
18O < −12‰ and δ
2H < −95‰ (
Figure 4). In some cases, e.g., the small rivers in the east part of the lake catchment, the recharge of the saline chloride sodium groundwater, which has an exotic depleted isotopic composition (
Figure 4), is felt. Naturally, such a small addition to the river runoff is not reflected in the isotope composition of the lake water.
Brackish and saline water with a chloride sodium composition has a significantly depleted isotope composition that is close to the average isotope composition of modern snow (
Table 1). The latter seems to indicate that the groundwater is old and could have formed during the last climatic cooling event [
41]. A similar situation with the presence of old water occurred in the Karelian Isthmus in the Leningrad Region, which is similar to South Karelia in terms of the hydrological conditions. While sampling the Riphean sandstone in the Ladoga graben at a depth of about 250 m, high-pressure water with a mineralization of about 0.65 g/L, δ
18O = −16.6‰ and δ
2H = −119‰ was revealed [
42,
43]. This suggests that the water, which was formed from precipitation during a cold climatic period, could make up a large area in the stagnant hydrodynamic zone of this region. At present, these small-volume waters are apparently discharged into Lake Ladoga [
43]. Whether there is a discharge of brackish water into Lake Onega is not yet clear.
The runoff of Neglinka River, with a catchment square of about 17 km
2 and whose mouth is located in the upper part of Petrozavodsk Bay (
Figure 1), was monitored in 2011–2016 (
Figure 9). The chronological graph shows not only the regular seasonal variations but also well-defined anomalies in the isotope composition of the river water, which were provoked by the spring flood and the heavy atmospheric depositions [
44]. These features of stable isotope variations can be used to subdivide the hydrograph and construct a small watershed model [
45,
46].
During the summer, evaporation has a strong effect on the isotope composition of the tributaries in the Lake Onega watershed (on the δ
2H vs. δ
18O diagram, part of the figurative points were shifted to the right of the LMWL). For the major rivers, the isotope shift was evident not only in the summer but also in the winter. The water loss due to evaporation from the major rivers was calculated following the experimental findings of the dependence of the isotope shift Δ(δ
18O) in the residual water on the amount of evaporated water ξ. The estimated loss of water in the tributaries in the summer was 11–22%. The enrichment of the tributary water by the heavy isotopes was directly proportional to the area of the lakes and wetlands on the drainage surface, but the influence of the lakes was greater than that of the wetlands (
Figure 10). Losses of water by evaporation can be reflected in the chemical composition of the river runoff [
47].