Real-Time Pyrolysis Dynamics of Thermally Aged Tire Microplastics by TGA-FTIR-GC/MS
Abstract
:1. Introduction
2. Materials and Methods
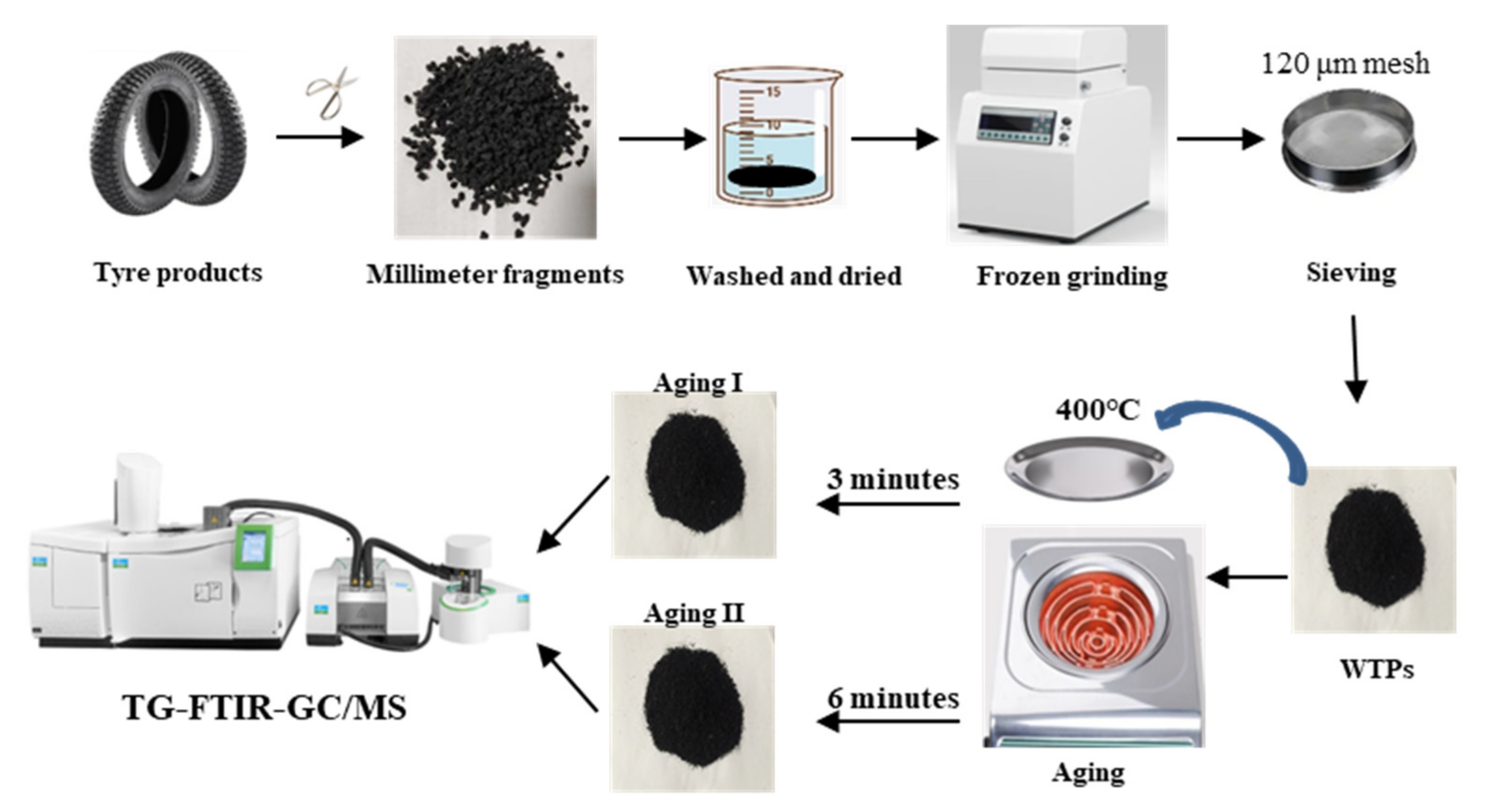
2.1. Sample Preparation and Thermal Aging
2.2. TG-FTIR-MS Experiments
2.3. TG-GC-MS Experiments
2.4. Data Process and Analysis
3. Result and Discussion
3.1. Dynamic TG Characterization of TWPs with Aging Time
3.2. Functional Groups of Pyrolytic Gases via TG-FTIR Analysis
3.2.1. TG-FTIR Characterization of Volatile Products
3.2.2. Dynamic Change in Gas Products with Pyrolysis Temperature
3.3. Identification and Dynamics on Gas Components of TWPs with Pyrolysis Temperature
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Andersson-Sköld, Y.; Johannesson, M.; Gustafsson, M.; Järlskog, I.; Lithner, D.; Polukarova, M.; Strömvall, A.-M. Microplastics from Tyre and Road Wear: A Literature Review; Swedish National Road and Transport Research Institute (VTI): Linköping, Sweden, 2020; pp. 1–133. [Google Scholar] [CrossRef]
- Bostock, J. Global Industry Tire Volume to Reach 2.7 Bilion Units by 2022; Smithers: Akron, OH, USA, 2021; pp. 1–2. [Google Scholar]
- Wagner, S.; Hüffer, T.; Klöckner, P.; Wehrhahn, M.; Hofmann, T.; Reemtsma, T. Tire wear particles in the aquatic environment—A review on generation, analysis, occurrence, fate and effects. Water Res. 2018, 139, 83–100. [Google Scholar] [CrossRef] [PubMed]
- Youn, J.S.; Kim, Y.M.; Siddiqui, M.Z.; Watanabe, A.; Han, S.; Jeong, S.; Jung, Y.-W.; Jeon, K.-J. Quantification of tire wear particles in road dust from industrial and residential areas in Seoul, Korea. Sci. Total Environ. 2021, 784, 147177. [Google Scholar] [CrossRef] [PubMed]
- Buss, A.H.; Kovaleski, J.L.; Pagani, R.N.; da Silva, V.L.; Silva, J.D.M. Proposal to reuse rubber waste from end-of-life tires using thermosetting resin. Sustainability 2019, 11, 6997. [Google Scholar] [CrossRef] [Green Version]
- Sułkowski, W.W.; Danch, A.; Moczyński, M.; Radoń, A.; Sułkowska, A.; Borek, J. Thermogravimetric study of rubber waste-polyurethane composites. J. Therm. Anal. Calorim. 2004, 78, 905–921. [Google Scholar] [CrossRef]
- Khan, F.R.; Halle, L.L.; Palmqvist, A. Acute and long-term toxicity of micronized car tire wear particles to Hyalella azteca. Aquat. Toxicol. 2019, 213, 105216. [Google Scholar] [CrossRef]
- Järlskog, I.; Strömvall, A.-M.; Magnusson, K.; Gustafsson, M.; Polukarova, M.; Galfi, H.; Aronsson, M.; Andersson-Sköld, Y. Occurrence of tire and bitumen wear microplastics on urban streets and in sweepsand and washwater. Sci. Total Environ. 2020, 729, 138950. [Google Scholar] [CrossRef]
- Unice, K.M.; Kreider, M.L.; Panko, J.M. Comparison of Tire and Road Wear Particle Concentrations in Sediment for Watersheds in France, Japan, and the United States by Quantitative Pyrolysis GC/MS Analysis. Environ. Sci. Technol. 2013, 47, 8138–8147. [Google Scholar] [CrossRef]
- Grigoratos, T.; Martini, G. Brake wear particle emissions: A review. Environ. Sci. Pollut. Res. Int. 2015, 22, 2491–2504. [Google Scholar] [CrossRef] [Green Version]
- Baldwin, J.M.; Bauer, D.R.; Ellwood, K.R. Rubber aging in tires. Part 1, Field results. Polym. Degrad. Stab. 2007, 92, 103–109. [Google Scholar] [CrossRef]
- Carli, L.N.; Bianchi, O.; Mauler, R.S.; Crespo, J.S. Accelerated aging of elastomeric composites with vulcanized ground scraps. J. Appl. Polym. Sci. 2012, 123, 280–285. [Google Scholar] [CrossRef]
- Wagner, S.; Klöckner, P.; Reemtsma, T. Aging of tire and road wear particles in terrestrial and freshwater environments—A review on processes, testing, analysis and impact. Chemosphere 2022, 288, 132467. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Zuo, S.; Lei, L.; Yang, X.; Wu, X. Analysis of impact factors of tire wear. J. Vib. Control 2011, 18, 833–840. [Google Scholar] [CrossRef]
- Mathissen, M.; Scheer, V.; Vogt, R.; Benter, T. Investigation on the potential generation of ultrafine particles from the tire–road interface. Atmos. Environ. 2011, 45, 6172–6179. [Google Scholar] [CrossRef]
- Fan, X.; Gan, R.; Liu, J.; Xie, Y.; Xu, D.; Xiang, Y.; Su, J.; Teng, Z.; Hou, J. Adsorption and desorption behaviors of antibiotics by tire wear particles and polyethylene microplastics with or without aging processes. Sci. Total Environ. 2021, 771, 145451. [Google Scholar] [CrossRef] [PubMed]
- Jung, U.; Choi, S.-S. Variation in Abundance Ratio of Isoprene and Dipentene Produced from Wear Particles Composed of Natural Rubber by Pyrolysis Depending on the Particle Size and Thermal Aging. Polymers 2023, 15, 929. [Google Scholar] [CrossRef]
- Kim, D.; Ahn, B.; Ryu, G.; Hwang, K.; Song, S.; Kim, W. Effect of Vinyl Group Content of the Functionalized Liquid Butadiene Rubber as a Processing Aid on the Properties of Silica Filled Rubber Compounds. Elastomers Compos. 2021, 56, 152–163. [Google Scholar] [CrossRef]
- Wang, Z.; Wei, R.; Wang, X.; He, J.; Wang, J. Pyrolysis and combustion of polyvinyl chloride (PVC) sheath for new and aged cables via thermogravimetric analysis-Fourier transform infrared (TG-FTIR) and calorimeter. Materials 2018, 11, 1997. [Google Scholar] [CrossRef] [Green Version]
- Ding, J.; Lv, M.; Zhu, D.; Leifheit, E.F.; Chen, Q.-L.; Wang, Y.-Q.; Chen, L.-X.; Rillig, M.C.; Zhu, Y.-G. Tire wear particles: An emerging threat to soil health. Crit. Rev. Environ. Sci. Technol. 2023, 53, 239–257. [Google Scholar] [CrossRef]
- Liu, Y.; Zhou, H.; Yan, M.; Liu, Y.; Ni, X.; Song, J.; Yi, X. Toxicity of tire wear particles and the leachates to microorganisms in marine sediments. Environ. Pollut. 2022, 309, 119744. [Google Scholar] [CrossRef]
- Veerasingam, S.; Ranjani, M.; Venkatachalapathy, R.; Bagaev, A.; Mukhanov, V.; Litvinyuk, D.; Mugilarasan, M.; Gurumoorthi, K.; Guganathan, L.; Aboobacker, V.M.; et al. Contributions of Fourier transform infrared spectroscopy in microplastic pollution research: A review. Crit. Rev. Environ. Sci. Technol. 2021, 51, 2681–2743. [Google Scholar] [CrossRef]
- Zhang, X.; Zhang, H.; Yu, K.; Li, N.; Liu, Y.; Liu, X.; Zhang, H.; Yang, B.; Wu, W.; Gao, J.; et al. Rapid Monitoring Approach for Microplastics Using Portable Pyrolysis-Mass Spectrometry. Anal. Chem. 2020, 92, 4656–4662. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Thomas, K.V.; Luo, Z.; Gowen, A.A. FTIR and Raman imaging for microplastics analysis: State of the art, challenges and prospects. TrAC Trends Anal. Chem. 2019, 119, 115629. [Google Scholar] [CrossRef]
- Kim, J.; Wi, E.; Moon, H.; Son, H.; Hong, J.; Park, E.; Kwon, J.-T.; Seo, D.Y.; Lee, H.; Kim, Y. Quantitative analysis of the concentration of nano-carbon black originating from tire-wear particles in the road dust. Sci. Total Environ. 2022, 842, 156830. [Google Scholar] [CrossRef] [PubMed]
- Thomas, J.; Cutright, T.; Pugh, C.; Soucek, M.D. Quantitative assessment of additive leachates in abiotic weathered tire cryogrinds and its application to tire wear particles in roadside soil samples. Chemosphere 2023, 311, 137132. [Google Scholar] [CrossRef]
- Luo, H.; Zhao, Y.; Li, Y.; Xiang, Y.; He, D.; Pan, X. Aging of microplastics affects their surface properties, thermal decomposition, additives leaching and interactions in simulated fluids. Sci. Total Environ. 2020, 714, 136862. [Google Scholar] [CrossRef]
- Yousef, S.; Eimontas, J.; Striūgas, N.; Mohamed, A.; Abdelnaby, M.A. Morphology, compositions, thermal behavior and kinetics of pyrolysis of lint-microfibers generated from clothes dryer. J. Anal. Appl. Pyrolysis 2021, 155, 105037. [Google Scholar] [CrossRef]
- Nel, H.A.; Chetwynd, A.J.; Kelly, C.A.; Stark, C.; Valsami-Jones, E.; Krause, S.; Lynch, I. An Untargeted Thermogravimetric Analysis-Fourier Transform Infrared-Gas Chromatography-Mass Spectrometry Approach for Plastic Polymer Identification. Environ. Sci. Technol. 2021, 55, 8721–8729. [Google Scholar] [CrossRef]
- Tang, X.; Chen, Z.; Liu, J.; Chen, Z.; Xie, W.; Evrendilek, F.; Buyukada, M. Dynamic pyrolysis behaviors, products, and mechanisms of waste rubber and polyurethane bicycle tires. J. Hazard. Mater. 2021, 402, 123516. [Google Scholar] [CrossRef]
- Chen, J.H.; Chen, K.S.; Tong, L.Y. On the pyrolysis kinetics of scrap automotive tires. J. Hazard. Mater. 2001, 84, 43–55. [Google Scholar] [CrossRef]
- Idris, R.; Chong, C.T.; Asik, J.A.; Ani, F.N. Optimization studies of microwave-induced co-pyrolysis of empty fruit bunches/waste truck tire using response surface methodology. J. Clean. Prod. 2020, 244, 118649. [Google Scholar] [CrossRef]
- Gwaily, S.E.; Badawy, M.M.; Hassan, H.H.; Madani, M. Influence of thermal aging on crosslinking density of boron carbide/natural rubber composites. Polym. Test. 2003, 22, 3–7. [Google Scholar] [CrossRef]
- Liu, J.; Li, X.; Xu, L.; Zhang, P. Investigation of aging behavior and mechanism of nitrile-butadiene rubber (NBR) in the accelerated thermal aging environment. Polym. Test. 2016, 54, 59–66. [Google Scholar] [CrossRef]
- Xu, F.; Wang, B.; Yang, D.; Ming, X.; Jiang, Y.; Hao, J.; Qiao, Y.; Tian, Y. TG-FTIR and Py-GC/MS study on pyrolysis mechanism and products distribution of waste bicycle tire. Energy Convers. Manag. 2018, 175, 288–297. [Google Scholar] [CrossRef]
- Song, P.; Wu, X.; Wang, S. Effect of styrene butadiene rubber on the light pyrolysis of the natural rubber. Polym. Degrad. Stab. 2018, 147, 168–176. [Google Scholar] [CrossRef]
- Jiao, L.; Xiao, H.; Wang, Q.; Sun, J. Thermal degradation characteristics of rigid polyurethane foam and the volatile products analysis with TG-FTIR-MS. Polym. Degrad. Stab. 2013, 98, 2687–2696. [Google Scholar] [CrossRef]
- Chen, R.; Lun, L.; Cong, K.; Li, Q.; Zhang, Y. Insights into pyrolysis and co-pyrolysis of tobacco stalk and scrap tire: Thermochemical behaviors, kinetics, and evolved gas analysis. Energy 2019, 183, 25–34. [Google Scholar] [CrossRef]
- Hu, Q.; Tang, Z.; Yao, D.; Yang, H.; Shao, J.; Chen, H. Thermal behavior, kinetics and gas evolution characteristics for the co-pyrolysis of real-world plastic and tyre wastes. J. Clean. Prod. 2020, 260, 121102. [Google Scholar] [CrossRef]
- Fernández-Berridi, M.J.; González, N.; Mugica, A.; Bernicot, C. Pyrolysis-FTIR and TGA techniques as tools in the characterization of blends of natural rubber and SBR. Thermochim. Acta 2006, 444, 65–70. [Google Scholar] [CrossRef]
- Pagacz, J.; Hebda, E.; Michałowski, S.; Ozimek, J.; Sternik, D.; Pielichowski, K. Polyurethane foams chemically reinforced with POSS—Thermal degradation studies. Thermochim. Acta 2016, 642, 95–104. [Google Scholar] [CrossRef]
- Yang, R.; Wang, B.; Li, M.; Zhang, X.; Li, J. Preparation, characterization and thermal degradation behavior of rigid polyurethane foam using a malic acid based polyols. Ind. Crops Prod. 2019, 136, 121–128. [Google Scholar] [CrossRef]
- Colom, X.; Carrillo, F.; Cañavate, J. Composites reinforced with reused tyres: Surface oxidant treatment to improve the interfacial compatibility. Compos. A Appl. Sci. Manuf. 2007, 38, 44–50. [Google Scholar] [CrossRef]
- Liu, X.; Zhao, J.; Yang, R.; Iervolino, R.; Barbera, S. Effect of lubricating oil on thermal aging of nitrile rubber. Polym. Degrad. Stab. 2018, 151, 136–143. [Google Scholar] [CrossRef]
- Colom, X.; Faliq, A.; Formela, K.; Cañavate, J. FTIR spectroscopic and thermogravimetric characterization of ground tyre rubber devulcanized by microwave treatment. Polym. Test. 2016, 52, 200–208. [Google Scholar] [CrossRef]
- Lei, W.U.; Zhu, S.; University, M.T. Quantitative Study on Tire Aging Based on Characteristic Peak Fitting Method. China Rubber Ind. 2017, 8, 52–56. [Google Scholar]
- Zhang, J.; Wang, C.; Chen, F.; Zao, W.; Feng, H.; Zhao, Y. Thermal-oxidative aging behaviors of shape memory nitrile butadiene rubber composite with dual crosslinking networks. Polym. Degrad. Stab. 2020, 179, 109280. [Google Scholar] [CrossRef]
- De Monléon, Q.; Banet, P.; Chikh, L.; Fichet, O. Effects of pyromellitidiimide pattern on thermomechanical properties and thermal stability of silicone networks. Polym. Degrad. Stab. 2022, 206, 110187. [Google Scholar] [CrossRef]
- Ding, K.; Zhong, Z.; Zhang, B.; Song, Z.; Qian, X. Pyrolysis Characteristics of Waste Tire in an Analytical Pyrolyzer Coupled with Gas Chromatography/Mass Spectrometry. Energy Fuels 2015, 29, 3181–3187. [Google Scholar] [CrossRef]
- Rychlý, J.; Lattuati-Derieux, A.; Lavédrine, B.; Matisová-Rychlá, L.; Malíková, M.; Csomorová, K.; Janigová, I. Assessing the progress of degradation in polyurethanes by chemiluminescence and thermal analysis. II. Flexible polyether- and polyester-type polyurethane foams. Polym. Degrad. Stab. 2011, 96, 462–469. [Google Scholar] [CrossRef]
- Eschenbacher, A.; Varghese, R.J.; Weng, J.; Van Geem, K.M. Fast pyrolysis of polyurethanes and polyisocyanurate with and without flame retardant: Compounds of interest for chemical recycling. J. Anal. Appl. Pyrolysis 2021, 160, 105374. [Google Scholar] [CrossRef]
- Chen, X.; Huo, L.; Jiao, C.; Li, S. TG–FTIR characterization of volatile compounds from flame retardant polyurethane foams materials. J. Anal. Appl. Pyrolysis 2013, 100, 186–191. [Google Scholar] [CrossRef]
- He, J.; Jiang, L.; Sun, J.; Lo, S. Thermal degradation study of pure rigid polyurethane in oxidative and non-oxidative atmospheres. J. Anal. Appl. Pyrolysis 2016, 120, 269–283. [Google Scholar] [CrossRef]
- Tian, L.; Shen, B.; Xu, H.; Li, F.; Wang, Y.; Singh, S. Thermal behavior of waste tea pyrolysis by TG-FTIR analysis. Energy 2016, 103, 533–542. [Google Scholar] [CrossRef]
- Kwon, E.; Castaldi, M.J. Fundamental understanding of the thermal degradation mechanisms of waste tires and their air pollutant generation in a N2 atmosphere. Environ. Sci. Technol. 2009, 43, 5996–6002. [Google Scholar] [CrossRef] [PubMed]
- Khaleel, M.; Soykan, U.; Çetin, S. Influences of turkey feather fiber loading on significant characteristics of rigid polyurethane foam: Thermal degradation, heat insulation, acoustic performance, air permeability and cellular structure. Constr. Build. Mater. 2021, 308, 125014. [Google Scholar] [CrossRef]
- He, Y.; Chen, X.; Tang, X.; Chen, S.; Evrendilek, F.; Chen, T.; Dai, W.; Liu, J. Co-combustion dynamics and products of textile dyeing sludge with waste rubber versus polyurethane tires of shared bikes. J. Environ. Chem. Eng. 2023, 11, 109196. [Google Scholar] [CrossRef]
- Šourková, M.; Adamcová, D.; Vaverková, M.D. The influence of microplastics from ground tyres on the acute, subchronical toxicity and microbial respiration of soil. Environments 2021, 8, 128. [Google Scholar] [CrossRef]
- Arockiasamy, A.; Toghiani, H.; Oglesby, D.; Horstemeyer, M.F.; Bouvard, J.-L.; King, R.L. TG–DSC–FTIR–MS study of gaseous compounds evolved during thermal decomposition of styrene-butadiene rubber. J. Therm. Anal. Calorim. 2013, 111, 535–542. [Google Scholar] [CrossRef]





| Chemical | Compounds Name | Atomic | Onset/°C | Peak/°C | ||||
|---|---|---|---|---|---|---|---|---|
| Formula | Mass | Origin | AgingI | AgingII | Origin | AgingI | AgingII | |
| C7H8 | Methylbenzene | 92 | 346 | 335 | 340 | 476 | 470 | 470 |
| C8H10 | m-Xylene | 106 | 373 | 373 | 373 | 470 | 464 | 460 |
| C8H8 | Styrene | 104 | 381 | 371 | 356 | 476 | 437 | 460 |
| C10H16 | D-Limonene | 136 | 366 | 366 | 366 | 409 | 409 | 403 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bai, G.; Fu, J.; Zhou, Q.; Pan, X. Real-Time Pyrolysis Dynamics of Thermally Aged Tire Microplastics by TGA-FTIR-GC/MS. Water 2023, 15, 1944. https://doi.org/10.3390/w15101944
Bai G, Fu J, Zhou Q, Pan X. Real-Time Pyrolysis Dynamics of Thermally Aged Tire Microplastics by TGA-FTIR-GC/MS. Water. 2023; 15(10):1944. https://doi.org/10.3390/w15101944
Chicago/Turabian StyleBai, Guangteng, Juyang Fu, Qian Zhou, and Xiangliang Pan. 2023. "Real-Time Pyrolysis Dynamics of Thermally Aged Tire Microplastics by TGA-FTIR-GC/MS" Water 15, no. 10: 1944. https://doi.org/10.3390/w15101944
APA StyleBai, G., Fu, J., Zhou, Q., & Pan, X. (2023). Real-Time Pyrolysis Dynamics of Thermally Aged Tire Microplastics by TGA-FTIR-GC/MS. Water, 15(10), 1944. https://doi.org/10.3390/w15101944





