Evolution Characteristics and Driving Factors of Cyanobacterial Blooms in Hulun Lake from 2018 to 2022
Abstract
:1. Introduction
2. Materials and Methods
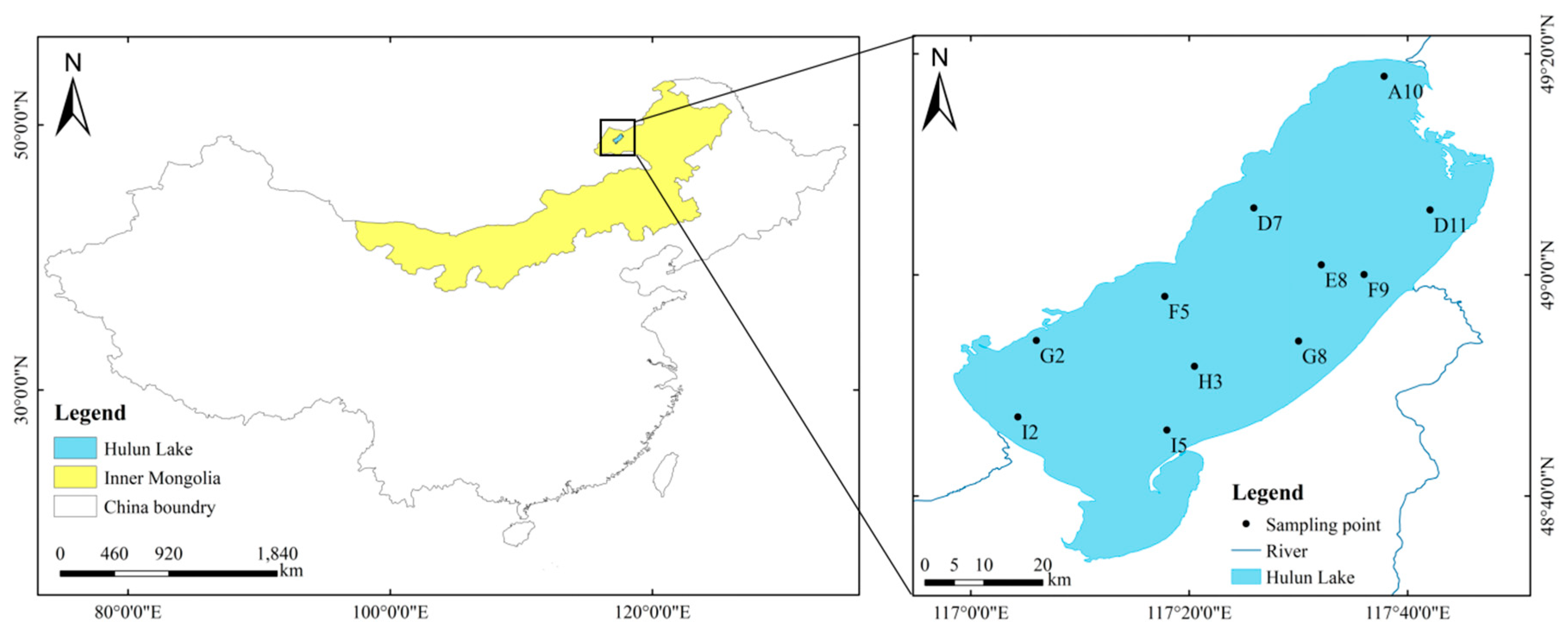
2.1. Study Area
2.2. Sample Collection and Data Source
2.2.1. Satellite Data
2.2.2. Ecological Investigation
2.2.3. Environmental Parameters
2.3. Data Processing
2.3.1. Bloom Extraction Algorithm
2.3.2. Dominant Species of Cyanobacteria
2.4. Statistical Analysis
3. Results
3.1. Change Trend of Blooms
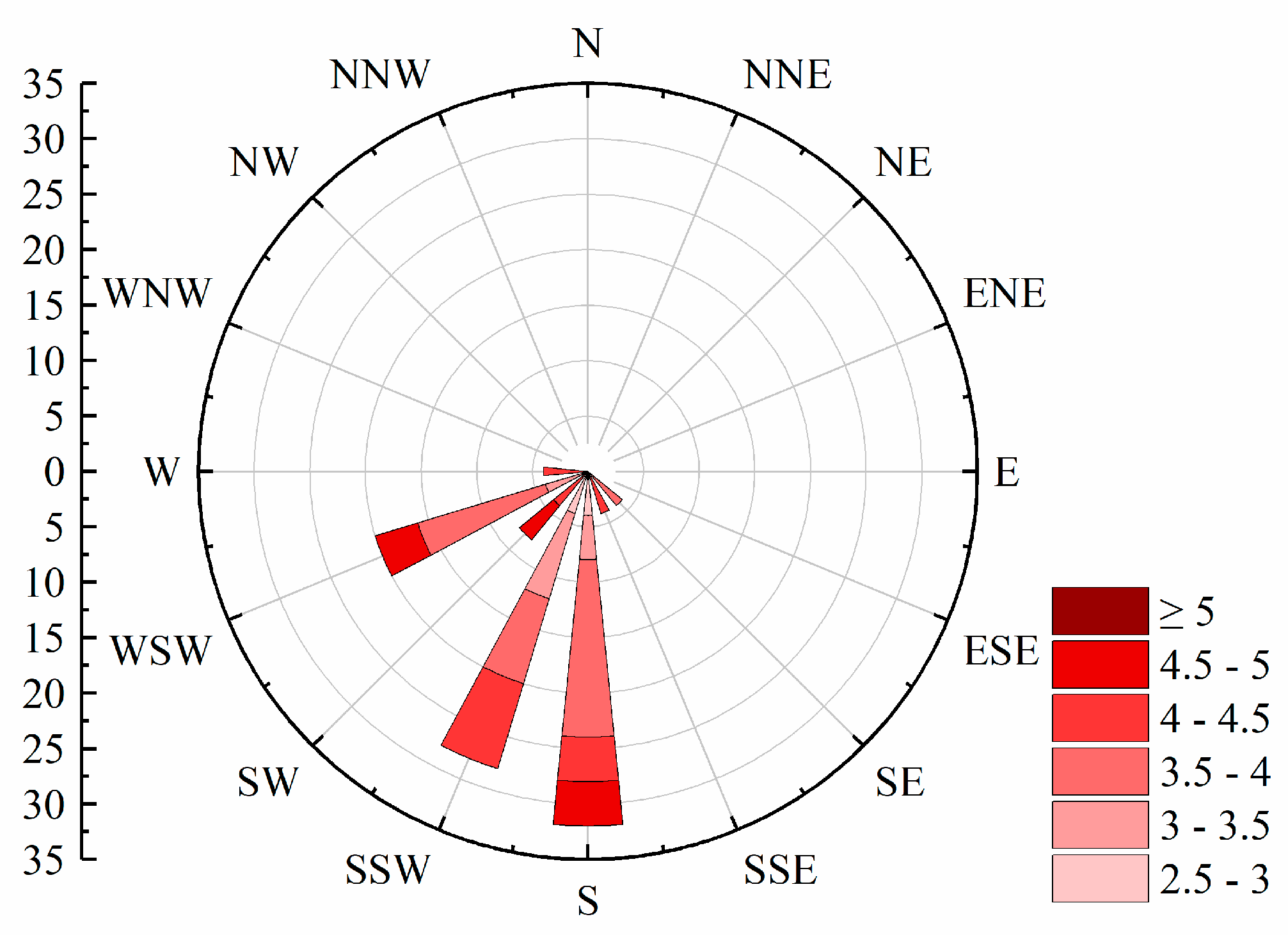
3.2. Interannual Variation of Meteorological and Environmental Factors
3.3. Driving Factors of Bloom
3.4. RDA Analysis
4. Discussion
4.1. Driving Factors of Blooms in Hulun Lake
4.2. Change Trend of Blooms in Hulun Lake
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Yuan, J.; Cao, Z.G.; Ma, J.G.; Shen, M.; Qi, T.C. Remote sensed analysis of spatial and temporal variation in phenology of algal blooms in Lake Chaohu since 1980s. J. Lake Sci. 2023, 35, 57–72. (In Chinese) [Google Scholar]
- Yan, X.C.; Xu, X.G.; Wang, M.Y.; Wang, G.X.; Wu, S.J.; Li, Z.C.; Sun, H.; Shi, A.; Yang, Y.H. Climate warming and cyanobacteria blooms: Looks at their relationships from a new perspective. Water Res. 2017, 125, 449–457. [Google Scholar] [CrossRef] [PubMed]
- Wu, R.; Liu, Y.; Zhang, S.; Shi, X.H.; Zhao, S.N.; Lu, J.P.; Kang, X.E.; Wang, S.H.; Wu, Y.; Arvola, L. Characterization of nitrogen and phosphorus at the ice-water-sediment interface and the effect of their migration on overlying water quality in Daihai Lake (China) during the freezing period. Sci. Total Environ. 2023, 164863. [Google Scholar] [CrossRef] [PubMed]
- Qin, B.Q.; Deng, J.M.; Shi, K.; Wang, J.; Brookes, J.; Zhou, J.; Zhang, Y.L.; Zhu, J.W.; Paerl, H.W.; Wu, L. Extreme Climate Anomalies Enhancing Cyanobacterial Blooms in Eutrophic Lake Taihu, China. Water Resour. Res. 2021, 57, e2020WR029371. [Google Scholar] [CrossRef]
- Wei, J.L.; Cui, Y.J.; Li, Y.Z.; Zhang, S.S.; Xu, H.Z.; Pang, Y.M.; Pei, H.Y. Distribution characteristics and environmental driving factors of cyanobacteria community in impounded Lakes and reservoirs in Shandong on the East Route of South-to-North Water diversion project. Environ. Sci. 2023, 1–15. (In Chinese) [Google Scholar] [CrossRef]
- Chen, T.; Du, X.; Chen, Y.Y.; Guo, X.Y.; Xiong, W. Metabarcoding profiling of phytoplankton communities associated to Algal blooms and determining related drivers in Baiyangdian Lake. Environ. Sci. 2023, 1–12. (In Chinese) [Google Scholar]
- Peng, M.Y.; Li, C.M.; Xia, J.; Song, Z.G.; Wen, B. The dominant algal species and the controlling factors triggered 2021—2022 Huguangyan Maar Lake algal bloom event. Environ. Chem. 2023, 42, 1642–1650. (In Chinese) [Google Scholar]
- Wang, S.Q.; Zhang, X.; Wang, C.; Chen, N.C. Multivariable integrated risk assessment for cyanobacterial blooms in eutrophic lakes and its spatiotemporal characteristics. Water Res. 2023, 228, 119367. [Google Scholar] [CrossRef]
- Ho, J.C.; Michalak, A.M.; Pahlevan, N. Widespread global increase in intense lake phytoplankton blooms since the 1980s. Nature 2019, 574, 667–670. [Google Scholar] [CrossRef]
- Huang, J.C.; Zhang, Y.J.; Arhonditsis, G.B.; Cao, J.F.; Chen, Q.W.; Peng, J. The magnitude and drivers of harmful algal blooms in China’s lakes and reservoirs: A national-scale characterization. Water Res. 2020, 181, 115902. [Google Scholar] [CrossRef]
- Qi, G.H.; Ma, X.S.; He, S.Y.; Wu, P.H. Long-term spatiotemporal variation analysis and probability prediction of algal blooms in Lake Chaohu (2009–2018) based on multi-source remote sensing data. J. Lake Sci. 2021, 33, 414–427. (In Chinese) [Google Scholar]
- Dai, Y.H.; Yang, S.B.; Zhao, D.; Hu, C.M.; Xu, W.; Anderson, D.M.; Li, Y.; Song, X.P.; Boyce, D.G.; Gibson, L.; et al. Coastal phytoplankton blooms expand and intensify in the 21st century. Nature 2023, 615, 280–284. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Razzano, M.; Mou, X. Cyanobacterial blooms alter the relative importance of neutral and selective processes in assembling freshwater bacterioplankton community. Sci. Total Environ. 2020, 706, 135724. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Mou, X.Z.; Cao, H.S.; Struewing, T.; Allen, J.; Lu, J.R. Co-occurring microorganisms regulate the succession of cyanobacterial harmful algal blooms. Environ. Pollut. 2021, 288, 117682. [Google Scholar] [CrossRef] [PubMed]
- Zhou, T.; Li, Y.; Jiang, B.; Alatalo, J.M.; Li, C.; Ni, C. Tracking spatio-temporal dynamics of harmful algal blooms using long-term MODIS observations of Chaohu Lake in China from 2000 to 2021. Ecol. Indic. 2023, 146, 109842. [Google Scholar] [CrossRef]
- Guo, X.; Zhu, A.; Chen, R. China’s algal bloom suffocates marine life. Science 2021, 373, 751. [Google Scholar] [CrossRef]
- Wu, R.; Zhang, S.; Liu, Y.; Shi, X.H.; Zhao, S.N.; Kang, X.E.; Sun, B.; Arvola, L.; Li, G.H. Spatiotemporal variation in water quality and identification and quantification of areas sensitive to water quality in Hulun lake, China. Ecol. Indic. 2023, 149, 110176. [Google Scholar] [CrossRef]
- Jiang, Z.F.; Li, C.Y.; Zhang, S. Zooplankton in Hulun Lake and the eutrophication evaluation. J. Arid. Land Resour. Environ. 2014, 28, 158–162. (In Chinese) [Google Scholar] [CrossRef]
- Cao, M.M.; Qing, S.; Du, Y.C.Z.; Yuan, R.Q.; Shun, B.R. Remote sensing monitoring of algal blooms in Hulun Lake based on SMDPSO algorithm. J. Water Resour. Water Eng. 2021, 32, 66–72+80. (In Chinese) [Google Scholar]
- Liu, J.Q.; Shi, Q.; Song, Y.; Zou, Y.R.; Liang, C. Remote sensing monitoring and comparison of cyanobacteria in Hulun Lake based on HY-1C satellite data. Geomat. World 2022, 29, 35–38+48. (In Chinese) [Google Scholar]
- Wang, D.B.; Jun, S.; Chen, L.; Zhou, J.H.; Bai, X.Y.; Chao, L.M. Community structures of phytoplankton in the Hulun Lake during lcebound season and its relation with environmental factors. Environ. Monit. China 2019, 35, 59–66. (In Chinese) [Google Scholar] [CrossRef]
- Li, X.C.; Yu, H.X.; Dou, H.S.; Pan, H.F.; Ma, C.X. Phytoplankton functional groups and related influencing factors in Hulun Lake and adjacent waters in spring. Chin. J. Fish. 2020, 33, 31–41. (In Chinese) [Google Scholar]
- Qian, X.Y.; Li, J.B.; Ao, W.; Pang, B.; Bao, S.R.; Wang, Q.; Liu, B.; Wang, Z.L. Seasonal dynamics of phytoplankton and its relationship with environmental factors in Lake Hulun. J. Lake Sci. 2022, 34, 1814–1827. (In Chinese) [Google Scholar]
- Šimunović, M.; Kulaš, A.; Žutinić, P.; Udovič, M.G. Phytoplankton Diversity of a Natural Karst Lake Combining Morphological and Molecular Approaches. Water 2023, 15, 1379. [Google Scholar] [CrossRef]
- State Environmental Protection Administration. Methods Formonitoring and Analysis of Water and Wastewater, 4th ed.; China Environmental Science Press: Beijing, China, 2002. (In Chinese) [Google Scholar]
- Zhang, M.; Duan, H.T.; Shi, X.L.; Yu, Y.; Kong, F.X. Contributions of meteorology to the phenology of cyanobacterial blooms: Implications for future climate change. Water Res. 2012, 46, 442–452. [Google Scholar] [CrossRef]
- Liu, X.M.; Zhang, G.X. A review of studies on the impact of climate change on cyanobacteria blooms in lakes. Adv. Water Sci. 2022, 33, 316–326. (In Chinese) [Google Scholar] [CrossRef]
- Zhang, P.; Guo, C.X.; Yu, J.; Quan, Q.M.; Yao, J.L.; Wang, J.Y.; Ye, X.R.; Zhu, M.Y.; Sun, Q.L.; Zhu, G.W. Characteristics of phytoplankton community structure and its response to hydro-meteorology in summer of Qiantang River. J. Lake Sci. 2022, 34, 418–432. (In Chinese) [Google Scholar]
- Jiang, D.L. Research on temporal and spatial variation of algae blooms and its driving factors in Lake Dianchi based on GIS/RS. Master’s Thesis, Southwest University, Chongqing, China, 2015. (In Chinese). [Google Scholar]
- Sun, X.J.; Qin, B.Q.; Zhu, G.W.; Zhang, Z.P. Effect of wind-induced wave on concentration of colloidal nutrient and phytoplankton in Lake Taihu. Environ. Sci. 2007, 28, 506–511. (In Chinese) [Google Scholar] [CrossRef]
- Shi, K.; Zhang, Y.; Zhang, Y.; Li, N.; Qin, B.; Zhu, G.; Zhou, Y. Phenology of Phytoplankton Blooms in a Trophic Lake Observed from Long-Term MODIS Data. Environ. Sci. Technol. 2019, 53, 2324–2331. [Google Scholar] [CrossRef]
- Jiang, Y.J.; He, W.; Liu, W.X.; Qin, N.; Ouyang, H.L.; Wang, Q.M.; Kong, X.Z.; He, Q.S.; Yang, C.; Yang, B.; et al. The seasonal and spatial variations of phytoplankton community and their correlation with environmental factors in a large eutrophic Chinese lake (Lake Chaohu). Ecol. Indic. 2014, 40, 58–67. [Google Scholar] [CrossRef]
- Robarts, R.D.; Zohary, T. Temperature effects on photosynthetic capacity, respiration, and growth rates of bloom-forming cyanobacteria. N. Z. J. Mar. Freshw. Res. 1987, 21, 391–399. [Google Scholar] [CrossRef]
- Morey, J.S.; Monroe, E.A.; Kinney, A.L.; Beal, M.; Johnson, J.G.; Hitchcock, G.L.; Dolah, F.M.V. Transcriptomic response of the red tide dinoflagellate, Karenia brevis, to nitrogen and phosphorus depletion and addition. BMC Genom. 2011, 12, 346. [Google Scholar] [CrossRef]
- Tan, X.; Shen, H.; Song, L.R. Comparative studies on physiological responses at phosphorus stress of three waterbloom-forming cyanobacteria. Acta Hydrobiol. Sin. (In Chinese). 2007, 5, 693–699. [Google Scholar]
- Zhang, Q.; Chen, Y.C.; Lin, Y.Q.; Ma, H.H.; Ding, Y.; Sun, H.; Chen, Q.W. Spatial distribution patterns of phytoplankton community during different water periods along cascade reservoirs in yhe Lancang River. Acta Sci. Circumstantiae 2022, 42, 392–401. (In Chinese) [Google Scholar] [CrossRef]
- Zhao, M.X.; Han, B.P. Analysis of factors affecting cyanobacteria bloom in a tropical reservoir (Tangxi Reservoi, China). Acta Ecol. Sin. (In Chinese). 2005, 7, 1554–1561. [Google Scholar]
- Van Dolah, F.M. Marine algal toxins: Origins, health effects, and their increased occurrence. Environ. Health Perspect. 2000, 108 (Suppl. S1), 133–141. [Google Scholar] [CrossRef]
- Tong, Y.D.; Xu, X.W.; Qi, M.; Sun, J.J.; Zhang, Y.Y.; Zhang, W.; Wang, M.Z.; Wang, X.J.; Zhang, Y. Lake warming intensifies the seasonal pattern of internal nutrient cycling in the eutrophic lake and potential impacts on algal blooms. Water Res. 2021, 188, 116570. [Google Scholar]
- Walter, J.M.; Lopes, F.A.C.; Lopes-Ferreira, M.; Vidal, L.M.; Leomil, L.; Melo, F.; Azevedo, G.S.; Oliveira, R.M.S.; Medeiros, A.J.; Melo, A.S.O.; et al. Occurrence of harmful cyanobacteria in drinking water from a severely drought-impacted semi-arid region. Front. Microbiol. 2018, 176. [Google Scholar] [CrossRef]
- Wang, C.L.; Pan, W.Y.; Han, Y.Q.; Qian, X. Effect of global climate change on cyanobacteria bloom in Taihu Lake China. Environ. Sci. 2010, 30, 822–828. (In Chinese) [Google Scholar]
- Guo, Z.Y.; Li, C.Y.; Shi, X.H.; Sun, B.; Zhao, S.N.; Quan, D.; Hou, B. Spatial and temporal distribution characteristics of chlorophyll a content and its influencing factor analysis in Hulun Lake of Cold and Dry Areas. Ecol. Environ. Sci. 2019, 28, 1434–1442. (In Chinese) [Google Scholar]
- Li, J.L.; Luo, C.L.; Lv, H.; Xu, J.F.; Luo, L.C.; Pan, M.; He, F.; Man, X.M.; Zhang, R.F.; Gong, F.L.; et al. Spatio-temporal variation and driving factors of algal bloom at Lake Dianchi during 2002–2018. Acta Ecol. Sin. 2023, 43, 878–891. (In Chinese) [Google Scholar]
- Zhang, C.; Lai, S.Y.; Gao, X.P.; Liu, H.A. A review of the potential impacts of climate change on water environment in lakes and reservoirs. J. Lake Sci. 2016, 28, 691–700. (In Chinese) [Google Scholar]




| TN | TP | WT | pH | DO | AT | PP | WD | WS | |
|---|---|---|---|---|---|---|---|---|---|
| Area | −0.277 | 0.005 | 0.102 | 0.254 | 0.068 | 0.085 | −0.008 | −0.004 | 0.455 * |
| Number | Dominant Species | Dominance (y) | |
|---|---|---|---|
| August | November | ||
| (Summer) | (Autumn) | ||
| 1 | Microcystis sp. | 0.491 | 0.208 |
| 2 | Planktolyngbya circumcreta | 0.126 | |
| 3 | Planktothrix agardhii | 0.024 | |
| 4 | Planktothrix spiroides | 0.03 | |
| 5 | Pseudanabaena sp. | 0.058 | |
| 6 | Dolichospermum flos-aquae | 0.25 | |
| 7 | Aphanizomenon sp. | 0.022 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, X.; Liu, Y.; Zhang, S.; Li, G.; Tao, Y.; Wang, S.; Yu, H.; Shi, X.; Zhao, S. Evolution Characteristics and Driving Factors of Cyanobacterial Blooms in Hulun Lake from 2018 to 2022. Water 2023, 15, 3765. https://doi.org/10.3390/w15213765
Li X, Liu Y, Zhang S, Li G, Tao Y, Wang S, Yu H, Shi X, Zhao S. Evolution Characteristics and Driving Factors of Cyanobacterial Blooms in Hulun Lake from 2018 to 2022. Water. 2023; 15(21):3765. https://doi.org/10.3390/w15213765
Chicago/Turabian StyleLi, Xingyue, Yu Liu, Sheng Zhang, Guohua Li, Yulong Tao, Shihuan Wang, Haifeng Yu, Xiaohong Shi, and Shengnan Zhao. 2023. "Evolution Characteristics and Driving Factors of Cyanobacterial Blooms in Hulun Lake from 2018 to 2022" Water 15, no. 21: 3765. https://doi.org/10.3390/w15213765
APA StyleLi, X., Liu, Y., Zhang, S., Li, G., Tao, Y., Wang, S., Yu, H., Shi, X., & Zhao, S. (2023). Evolution Characteristics and Driving Factors of Cyanobacterial Blooms in Hulun Lake from 2018 to 2022. Water, 15(21), 3765. https://doi.org/10.3390/w15213765






