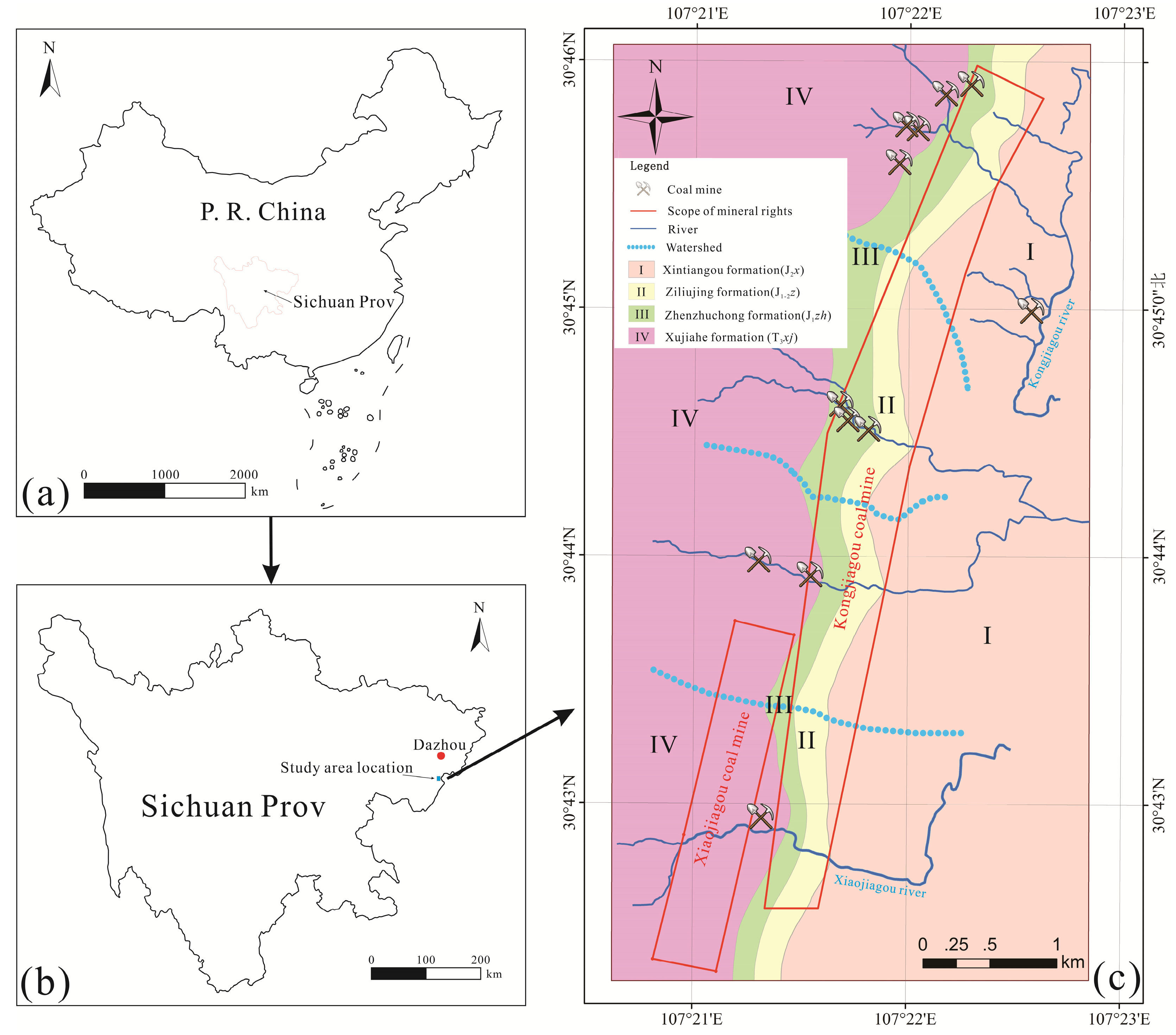
4.1. Hydrochemical Characteristics
The physicochemical and chemical properties of the water samples from the study area are summarized in
Table 1 and
Table 2. Water samples P6 and P12 were collected upstream from the Xiaojiagou and Kongjiagou coal mine drainages, respectively, to represent the two background values. The temperature difference between the groundwater and the stream was approximately 10.0 °C, which could have been affected by the temperature exceeding 22.0 °C during collection. In the dry season, the groundwater and stream samples were slightly alkaline with pH values of 7.1–8.2 and 7.6–8.1, respectively. Compared to the dry season, the pH values of groundwater were slightly acidic to alkaline (6.7–8.2) and the stream samples were alkaline (7.7–8.3) in the wet season. Most stream water samples had EC values higher than those of the groundwater samples in both the dry and wet seasons. Except for P3, which had the highest TDS value, the groundwater seemed to be more pure and contained less dissolved solids. Moreover, the groundwater and stream samples were oxidized, except for two samples (P2 and P4 in the dry season) that had negative Eh values, where more oxygen was found in the stream samples than in the groundwater samples. Furthermore, a Piper diagram [
25] was used to analyze the main ions present in the water to highlight the relative concentration of each ion in the water samples, which were divided into nine areas to show the differences in the geochemical characteristics and groundwater composition types in each area. Most groundwater and stream samples from the dry and wet seasons were dominated by Ca
2+ + Mg
2+ − HCO
3− and Ca
2+ + Mg
2+ − Cl
−, as shown in the Piper diagram (
Figure 4). The coal mine drainage samples (P2 and P3) were dominated by Ca
2+ + Mg
2+ − Cl
− + SO
42−, whereas the groundwater sample (P4) was dominated by Ca
2+ + Na
+ − HCO
3−. Stiff diagrams (
Figures S1 and S2) were used to depict the chemical state of groundwater. The high concentration of Ca
2+ and SO
42− in P3, P14, and P15 can be explained by the dissolution of the gypsum. The excess Ca
2+ and HCO
3− in P6 and P7 may be related to the dissolution of carbonate.
The Gibbs diagram further indicates the origins and influencing factors of groundwater components [
26]. As shown in
Figure 5, most samples fell within the water–rock interaction zone in both seasons, indicating that rock mineral wreathing was the major factor controlling the hydrochemicals in the study area. In both the dry and wet seasons, the Na
+/(Na
+ + Ca
2+) ratio of some water samples was >0.6 and beyond the boundary. This result was obtained mainly because the calculated mass concentration of Na
+ contained other undetermined cation components [
5]. The high TDS concentration suggests that there were extensively charged species in the aquifer.
The γ(Ca
2+)/γ(SO
42−) ratios of most water samples in
Figure 6a are 1:1. This finding can be explained by the complex influences of the dissolution and evaporation processes of gypsum and anhydrite (Equations (1) and (2)). Some water samples showed Ca
2+/SO
42− ratios greater than 1, indicating the addition of Ca by the weathering of Ca-rich minerals, which may involve calcite and dolomite dissolution. The coal mine drainage sample (P3) had Ca
2+/SO
42− ratios of less than 1 in both the dry and wet seasons. This characteristic may have been due to the oxidation of sulfides in the coal seams, such as pyrite (FeS
2), which produced a free acid that neutralized the carbonate minerals in the coal measures [
27]. This reaction can be explained by Equation (3).
As shown in
Figure 6b, most water samples were above the Mg
2+ + Ca
2+/HCO
3− ratio 1:1 line, indicating that excess Ca
2+ and Mg
2+ may be related to the dissolution of carbonate [
28] (Equation (4)). Carbonate minerals, such as calcite, dolomite, iron dolomite, and siderite, are usually filled in coal fractures as epigenetic minerals [
29] and are mainly formed by the reaction of CO and CO
2 produced during coalification with the fluid in coal [
30]. Sample P4 had an Mg
2+ + Ca
2+/HCO
3− ratio exceeding 1:2, indicating that more CO
2 entered the groundwater and generated a large amount of HCO
3− during the flow process [
5] (Equation (5)). In addition, the concentrations of major ions in the stream water samples were higher than those in the groundwater samples, especially in the cases of HCO
3− and Ca
2+. Owing to the enclosure of the environment, the CO
2 released by the neutralization reaction further deepened dissolution [
31], which can be explained by Equations (6) and (7).
Therefore, Ca·Mg-HCO3 groundwater was continuously discharged from the pithead, which increased the ionic concentration in the downstream drainage. It should be noted that the concentration of SO42− in the groundwater sample (P3) was 477.0 mg/L during the dry season, which exceeded the limit value of 250 mg/L in the Chinese drinking water standard; hence, it is not suitable for use as a drinking water source. However, local residents still place water pipes at the pithead to use this water for drinking, which would have a certain impact on their health.
Box plots are used to represent the concentration of the major elements in
Figure 7. The median is plotted near the center with log boxes of different parameters, indicating the maximum number of parameters in the normal distribution. The exceptions are the non-normal distributions of SO
42−, HCO
3−, and Ca
2+. The imbalance in SO
42− is mainly due to the large amount of AMD produced by abandoned mines in the oxidation environment, where AMD is discharged into downstream rivers. These results show that in the dry season, the concentrations of SO
4 in groundwater samples P3 and P8 are 477 mg/L and 193.9 mg/L, respectively, and the same phenomenon is observed in the wet season. The collected water samples were mainly from the coal mine drainage at the pithead and river water downstream from the drain. These water samples were collected from hills, wastelands, alluvial plains, and underground mines in the study area. Land use and land cover patterns can directly affect the water and chemical properties of the study area. Therefore, the variations of these parameters would appear not to be normally distributed, which could be caused by coal mining as well as agricultural and domestic waste [
32].
4.2. Trace Elements Concentration of the Water Samples
The concentration of 10 trace elements, including Zn, Fe, Mn, and Al in each water sample, are summarized in
Table 3. The water quality properties in the study area, according to the Chinese Standards for Drinking Water Quality (GB 5749-2006) and the WHO [
12], are also provided in
Table 3. The concentration of heavy metals (Cr, Cu, Zn, As, Cd, Hg, and Pb) in the water samples is significantly below the CNS and WHO guidelines in
Table S1; hence, it was not analyzed in this study. Although a comparison between the ion concentration of all trace elements and the above two standards is not included, this can be regarded as a general indication of the state of water quality in the study area. Iron and Mn clearly exceed the Chinese and WHO standards, whereas other trace elements are within suitable limits. The high concentrations of Mn in five water samples (P1, P2, P4, P5, and P8) during the dry season are 353.38 μg/L, 1925.14 μg/L, 132.88 μg/L, 100.86 μg/L, and 108.04 μg/L, respectively. The Mn concentration in the P2 water sample clearly exceeds the standard, reaching 1925.14 μg/L. The concentration of Fe in water samples P1, P2, P5, P7, P10, P13, and P14 also exceeds the standard value in the dry season, where the concentration of Fe in water sample P2 is 8356.48 μg/L, and those of P1 and P5 are 1563.72 μg/L and 1112.56 μg/L, respectively. The concentration of Fe in the stream water samples is approximately 300 μg/L, and the excess content is less than 100 μg/L in the dry season. Only two samples exist with excessive Mn concentration, P2 and P4, whereas there is an excessive concentration of Fe in water samples P2 and P11 in the wet season, where the concentration of Fe in P2 is 12,872.88 μg/L. The oxidation of iron sulfide and aluminosilicate minerals by acidic water is the main reason for the high concentration of Fe and Mn in coal mine drainage [
33,
34]. Thus, the reason for the high concentration of Fe and Mn in P2 is the existence of considerable amounts of iron and manganese oxides at the pithead when the mine was abandoned.
Excessive Fe and Mn content in drinking water would have adverse effects on the human body; Fe can affect the cardiovascular system, and Mn can affect the central nervous system as well as brain and reproductive functions, causing anorexia, vomiting, and diarrhea [
35]. According to the evaluation based on the Chinese standards for ground water quality (GB 14848-2017), groundwater sample P2 was classified as a class V groundwater sample because of the Fe and Mn contents exceeding 2000 μg/L and 1500 μg/L, respectively, which would not be suitable for drinking.
4.3. Variation of Downstream Concentration
The variations in the major ionic concentration from the coal mine drainage to the downstream area are depicted in
Figure 8. The drain outlets (source of contamination) of the two coal mines are located at the 0 m position. In both the dry and wet seasons, P6 and P12 were collected upstream, near the drain outlets, and their main ion concentrations (at X = 0) can be used as background values for comparison with downstream water samples. This finding shows that the concentrations of most major ions are higher than the background values. However, the concentration of HCO
3− in water sample P12 is higher than that of the downstream water sample, indicating the presence of Ca·Mg-HCO
3 in the groundwater upstream from the Kongjiagou coal mine drainage, which is enriched with HCO
3−. Compared with the background value, the Mn concentration downstream from Kongjiagou shows little change. It can also be observed that the ion concentration is highest at the beginning of the drain and gradually decreases with the downstream distance. However, downstream of the Kongjiagou drain, the concentrations of Mg, SO
42−, and HCO
3− did not decrease with distance but rather increased slightly. This may be due to the fact that Kongjiagou was a coal mine and was constantly discharging mine water into the river, resulting in the river water being in a state of ion enrichment for a long period of time. Hence, coal mine drainage from the Xiaojiagou coal mine (P8) led to the enrichment of Ca
2+, Mg
2+, SO
42−, HCO
3−, Fe, and Mn. The high concentration of these major ions indicate the impact of the coal mine drainage on the water quality of the aquifer.
4.4. Statistical Analysis of the Water Samples Dataset
Eight variables across 15 samples were used to analyze the correlations. The correlation matrix results, including the physicochemical parameters, are summarized in
Table 4, which shows that the pH is negatively correlated with Ca
2+. The EC is positively correlated with HCO
3−, SO
42−, Mg
2+, and Ca
2+; HCO
3− is positively correlated with Cl
− and Na
+; and SO
42− is positively correlated with Ca
2+ and Mg
2+. In addition, Cl
− is positively correlated with Na
+, and Ca
2+ is positively correlated with Mg
2+. Moreover, strong positive correlations are observed between EC and SO
42−, EC and Ca
2+, EC and Mg
2+, HCO
3− and Na
+, SO
42− and Ca
2+, SO
42− and Mg
2+, Cl
− and Na
+, and Ca
2+ and Mg
2+. Some moderate and positive correlations are also observed in between HCO
3− and Cl
−.
F1 is positively correlated with the EC, SO
42−, Ca
2+, and Mg
2+ and negatively correlated with the pH (
Table 5), explaining 52% of the data variance. Moreover, F1, which is correlated with SO
42−, results in a higher overall concentration, which could be related to sulfide minerals [
36], whereas the positive correlations between F1 and both Ca
2+ and Mg
2+ also indicate that a large amount of carbonate minerals is dissolved during the process of atmospheric precipitation moving underground through the surface. Simultaneously, F1 displays a negative correlation with most of the dissolved minerals and pH. When the concentration of dissolved minerals increases, the pH decreases. These observations are primarily found in the context of AMD [
37]. Therefore, F1 can be considered an indicator of AMD. Furthermore, the F2 factor is positively correlated with HCO
3−, Cl
−, and Na
+ (
Table 5), which explains 29% of the data variance, and is correlated with the ions that characterize the carbonate minerals. The Na
+ and Cl
− ions were mainly produced by precipitation, human activities, and the dissolution of silicates [
32]. In the studied case, a strong positive correlation was observed between Na
+ and Cl
−, indicating a strong degree of dissolution evaporation in this area. Consequently, F2 can be considered as an indicator of halite dissolution. Hence, F1 can represent the high concentrations of SO
42−, Ca
2+, Mg
2+, and low pH in the abandoned coal mine drainage, and F2 can represent the relationships between the drainage and the HCO
3−, Na
+, and Cl
− concentrations in downstream rivers [
38].
4.5. Geochemistry of Stream Sediments
The concentration of metals and metalloids in the seven stream sediment samples near the mine drain were measured (
Table 6) and are displayed as box and whisker plots of the concentration of each element (
Figure 9). The concentration of Al (%), Ca (%), Mg (%), Fe (%), and Mn (ppm) in the two streams downstream of the drain are significantly different. The concentration of Ca in the stream sediments downstream of the Xiaojiagou drain is on average–3–4 times higher than that in Kongjiagou, whereas the concentration of Mg in the downstream sediments of the Kongjiagou drain is higher than that in Xiaojiagou. These findings demonstrate that large amounts of carbonate minerals are present in the stream sediments. These minerals are produced by the leaching of groundwater into a karst aquifer, which is subsequently discharged. It is worth noting that the concentrations of Fe, Mn, Cr, and Zn downstream from the Kongjiagou drain are evidently higher than those in Xiaojiagou, and the concentration of Mn is 5–6 times higher. This result clearly indicates that the Xiaojiagou coal mine is closed, whereas the Kongjiagou coal mine remains active. With mining activities, a large amount of metal minerals is discharged and deposited downstream of the coal mine drainage, affecting water and soil quality. In addition, the concentrations of Fe and Mn oxides in the anoxic zone are relatively low, whereas the concentrations of dissolved Fe and Mn in the interstitial water increase accordingly [
39].
Compared with the average background values in a low mountainous or hilly area [
40], the concentrations of Pb, P, and Ti in the seven samples were similar to the background values, whereas the concentrations of Cr, Mn, Ni, Zn, Al, Fe, and Mg in the stream sediments downstream from the Kongjiagou drain were higher than the background values, and the concentrations of Fe and Mn ions were 3–4 times higher than the background values. The samples (S1–S3) taken downstream of the Xongjiagou drain were all below the background values. Thus, with continuous mining activities, large amounts of Fe and Mn oxides were discharged into the downstream river along with coal mine drainage, resulting in the enrichment of Fe and Mn in the downstream sediments. Hence, the closure of coal mines is beneficial for the recovery of the local environment and has a positive ecological effect.
Furthermore, the concentrations of Co, Cu, Sr, and Ca in all the samples exceeded the background values. The concentration of Cu in sample S3 was as high as 85.3 ppm, which may have been caused by local human activities (domestic waste and fertilization) [
32], and the concentrations of Ca in samples S1 and S2 downstream from the Xongjiagou drain were much higher than those in the other samples, indicating that carbonate dissolution took place in the Xiaojiagou coal mine. These phenomena can be explained by the dissolution and precipitation of minerals (calcite and dolomite) during stream sedimentation and mine drainage [
5]. Hence, according to the standard environmental quality evaluation for soil analysis (GB 15618-1995), all seven stream sediment samples had good-quality soil, reflecting the secondary standard of environmental quality. This type of soil is suitable for planting and animal husbandry, and the soil quality does not cause harm or pollution to the environment.
4.6. Statistical Analysis of the Stream Sediments Samples Dataset
The correlation matrix of the stream sediment samples (
Table 7) highlights that Cr has strong positive correlations with Zn, Rb, Pb, Ti, Al, Fe, and Mg and moderately positive correlations with Ni and P. This may be because some chromium minerals, such as picotite (Fe (Cr, Al)
2O
4), enter the stream with groundwater during mining [
41]. Moreover, Mn has a strong positive correlation with Mg and moderately positive correlations with Ti, Al, and Fe. Co has a strong negative correlation with Sb and moderately negative correlations with Sr and Ca. High positive loadings of these elements indicate a strong connection with mining. Coal mine drainage causes minerals rich in these elements to enter into the river, leading to an increase in these elements in stream sediments, which subsequently affects the stream water [
42]. Furthermore, Ni has strong positive correlations with Zn and Al and moderately positive correlations with Rb, Pb, Ti, Fe, and Mg. In addition, Zn exhibits strong positive correlations with Rb, Pb, Ti, Al, Fe, and Mg. The relationships between Ni and Zn indicate the effect of local human activities (domestic waste and fertilization) and lithology (weathering of parent rocks) [
32]. Al has strong positive correlations with Fe and Mg, whereas Fe has a strong positive correlation with Mg. Hence, the stream sediment correlations reflect the adsorption capacities of Fe, Mn, and Al oxides in metals and metalloids within the surface environment [
43]. This finding also highlights the influence of coal mine drainage, because the concentrations of Fe, Zn, Mn, Cr, Al, and Mg in the stream sediments directly obtained from the coal mine drainage are the highest (
Table 6).
4.7. Correlations with Coal Mine Drainage
Based on the data analysis results summarized in
Table 1,
Table 2 and
Table 6, the closure of the Xiaojiagou coal mine caused groundwater to remain in the abandoned mine channel longer than in the Kongjiagou coal mine. In addition, the groundwater samples have a weakly acidic environment, which is conducive to the dissolution of carbonate minerals. Therefore, the concentrations of Ca
2+ and Mg
2+ in the water samples downstream from Xiaojiagou (P8–P11) are significantly higher than those in downstream water samples P12–P15. This phenomenon directly increases the Ca
2+ content in the downstream sediments to far higher than the background value, and the concentrations of Ca in Xiaojiagou are higher than those in Kongjiagou. During the mining process, many minerals containing Fe and Mn are dissolved or suspended in the groundwater along with the discharged mine water inflow, forming downstream stream sediments [
44]. Consequently, the Fe and Mn contents in the stream sediments (S4–S7) in the lower reaches of Kongjiagou are much higher than the background values.
An XRD analysis was performed on two stream sediment samples (S1 and S4). Mineral phases such as quartz and cuprite are observed in both samples (
Figure 10). Magnetite and delafossite, which are characteristics of weathered mining waste, indicate the possible presence of various mineral phases of environmental interest [
45]. Muscovite and pyrite are evident in each sample, indicating that the coal mine drainage transported these minerals from the strata into the stream. Calcite is the major mineral phase in S1 and belongs to the waste-rock deposit of the Xiaojiagou coal mine. In addition, a large amount of quartz in the Xujiahe Triassic Formation entered the river with the coal mine drainage (
Figure 11).
Generally, the concentration of metals and metalloids in the stream sediments were affected by the coal mine drainage and were generally enriched in Cr, Mn, Zn, Sr, Co, Ni, Cu, Al, Ca, Fe, and Mg, and the concentrations of Mn, Ca, and Fe were significantly increased. These findings demonstrate that strong positive correlations exist between the concentrations of trace elements in coal mine drainage and stream sediments.