Geochemical Response of Surface Environment to Mining of Sn-Pb-Zn Sulfide Deposits: A Case Study of Dachang Tin Polymetallic Deposit in Guangxi
Abstract
1. Introduction
2. Materials and Methods
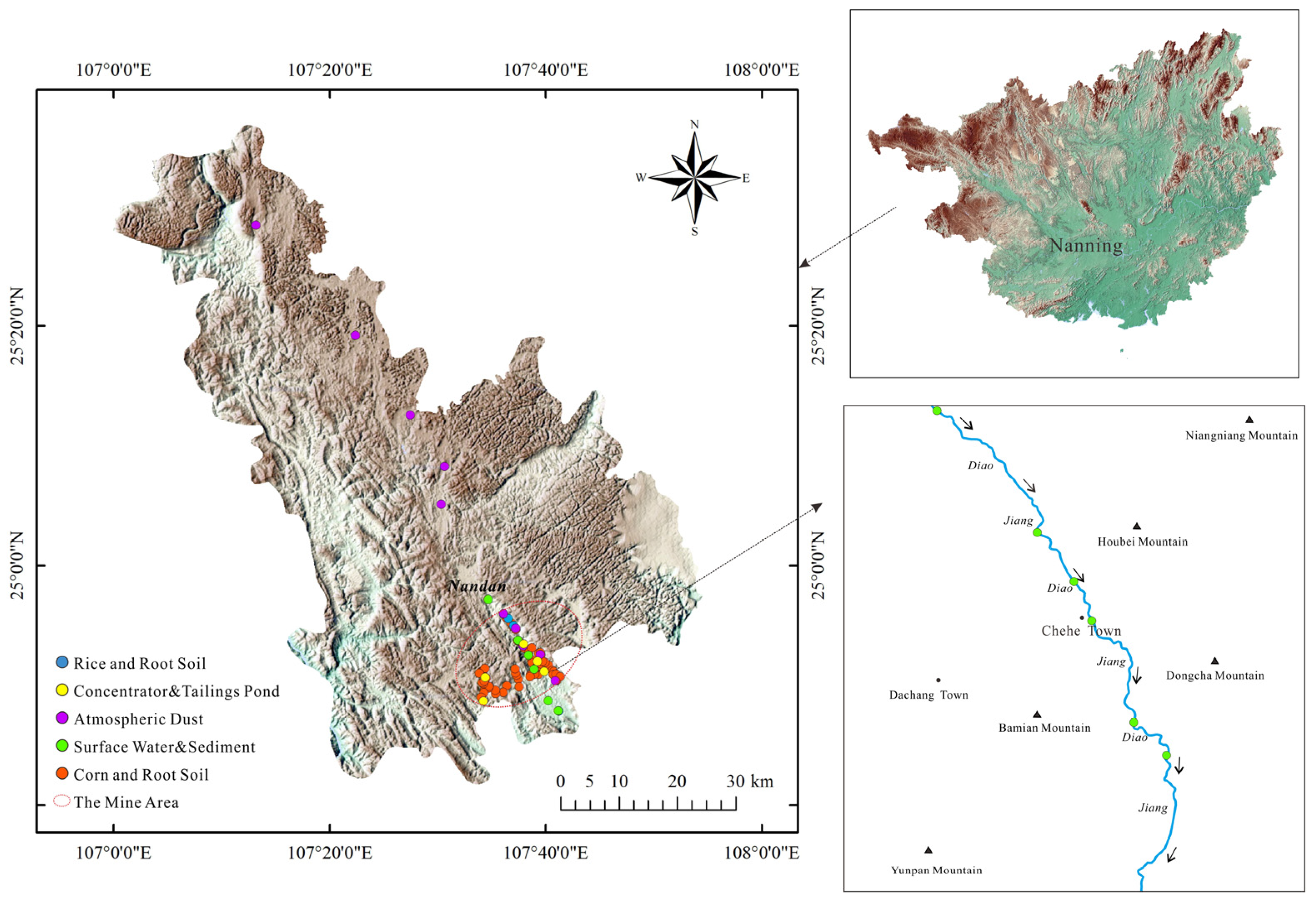
2.1. The Study Area
2.2. Sampling Methods
2.3. Analysis Methods
2.4. Data Processing
2.4.1. Deposition Ratio
2.4.2. Content Variation Coefficient
3. Results and Discussion
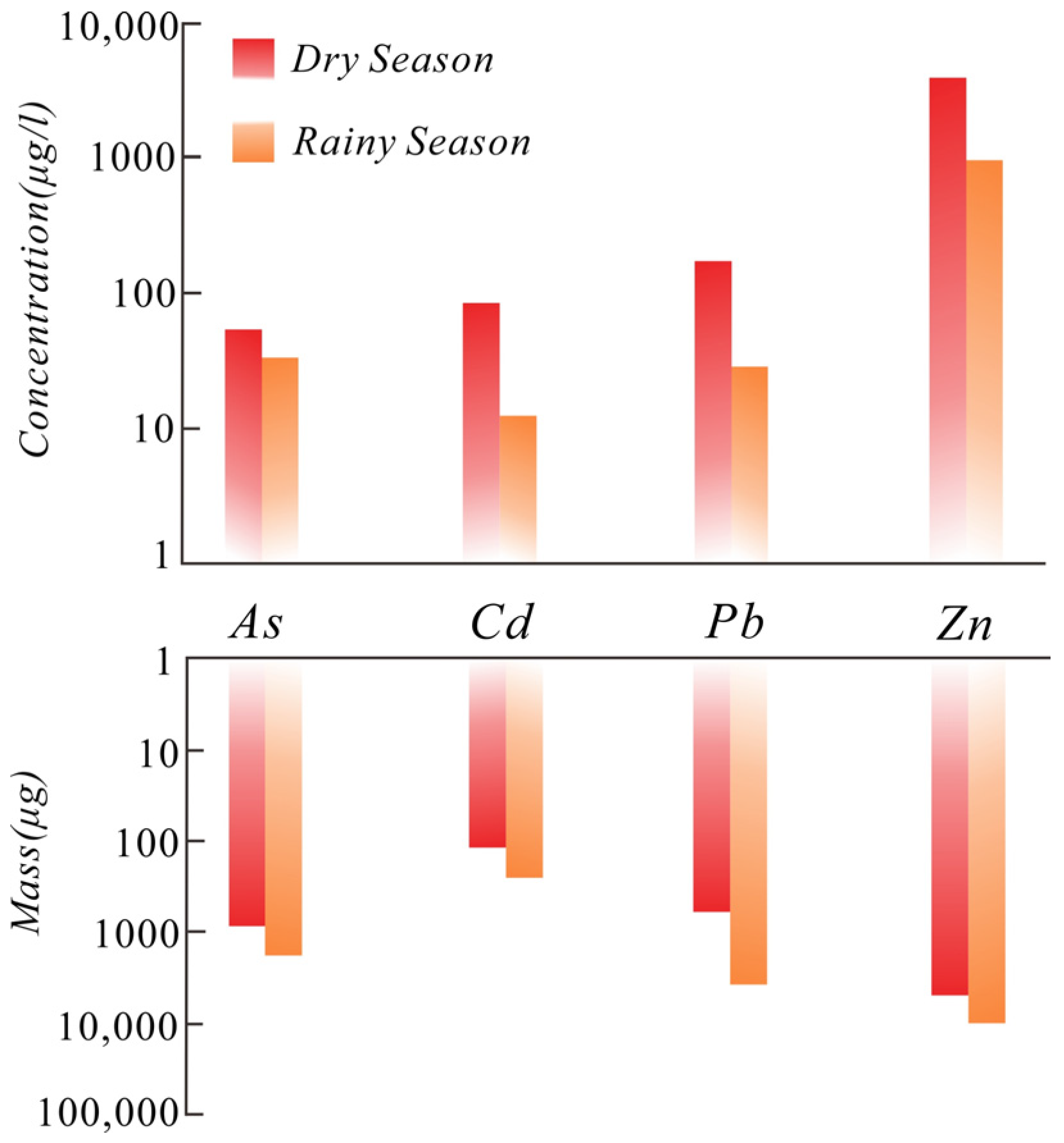
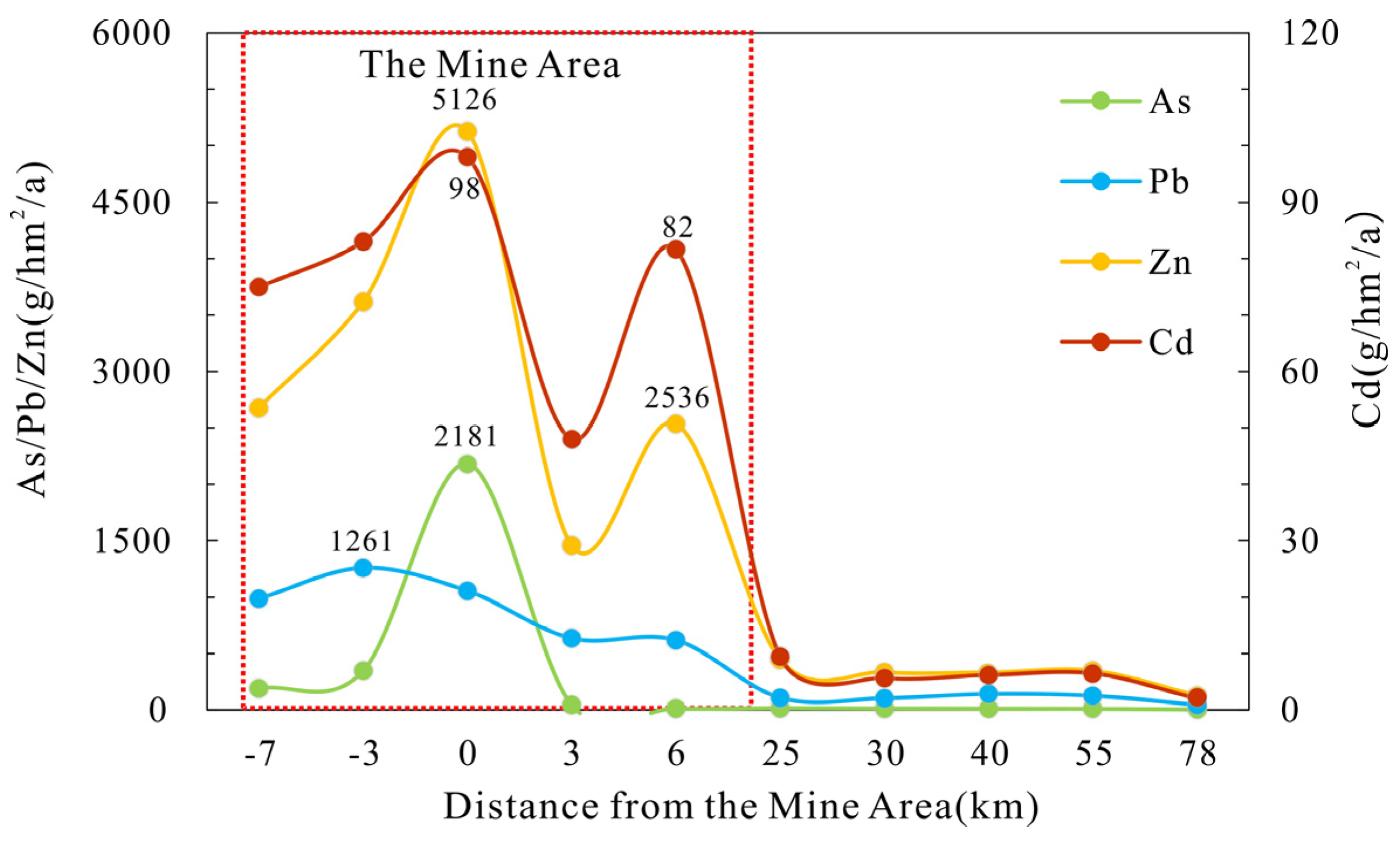
3.1. Atmospheric Dust
3.2. Mine Drainage and Surface Water
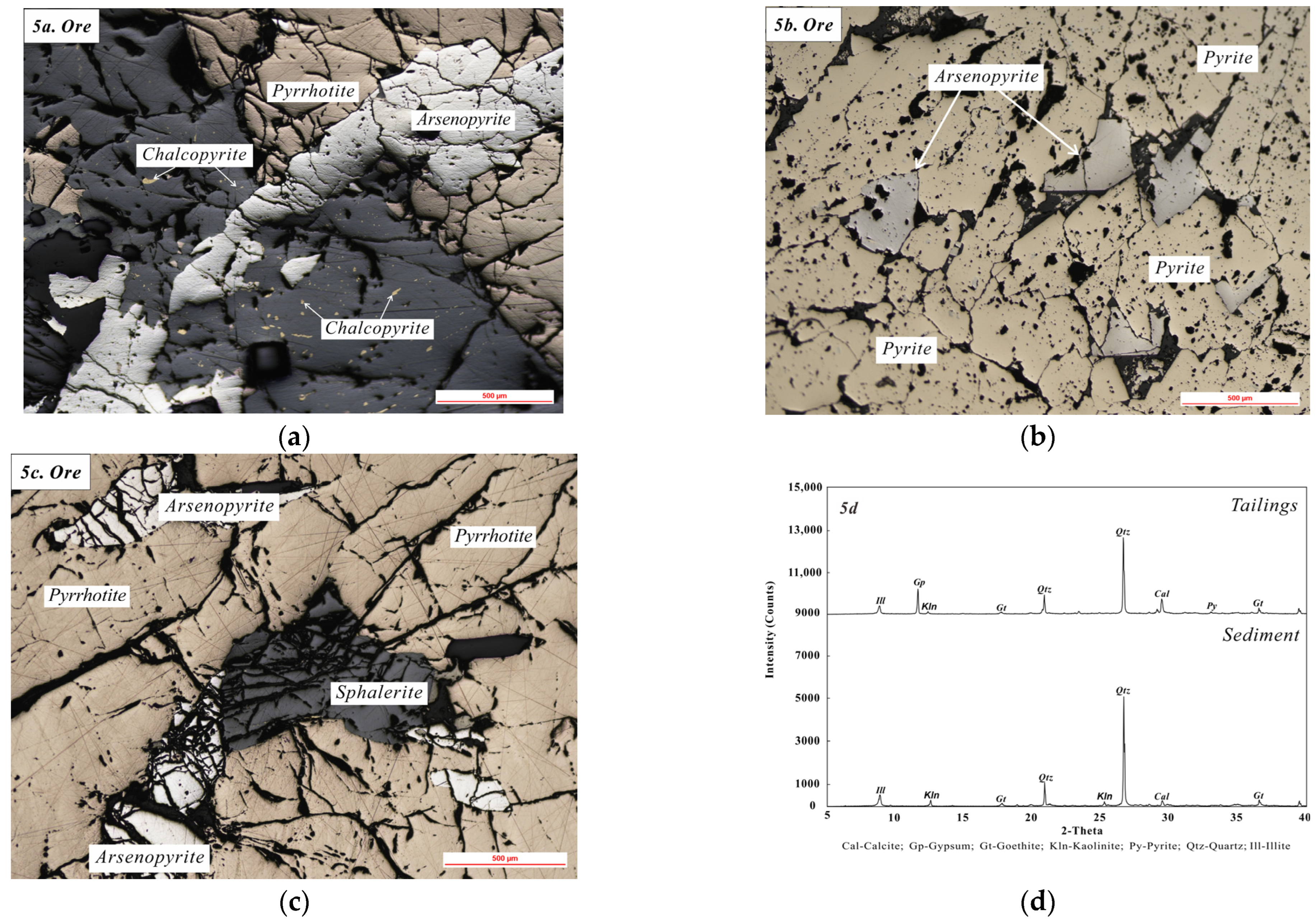
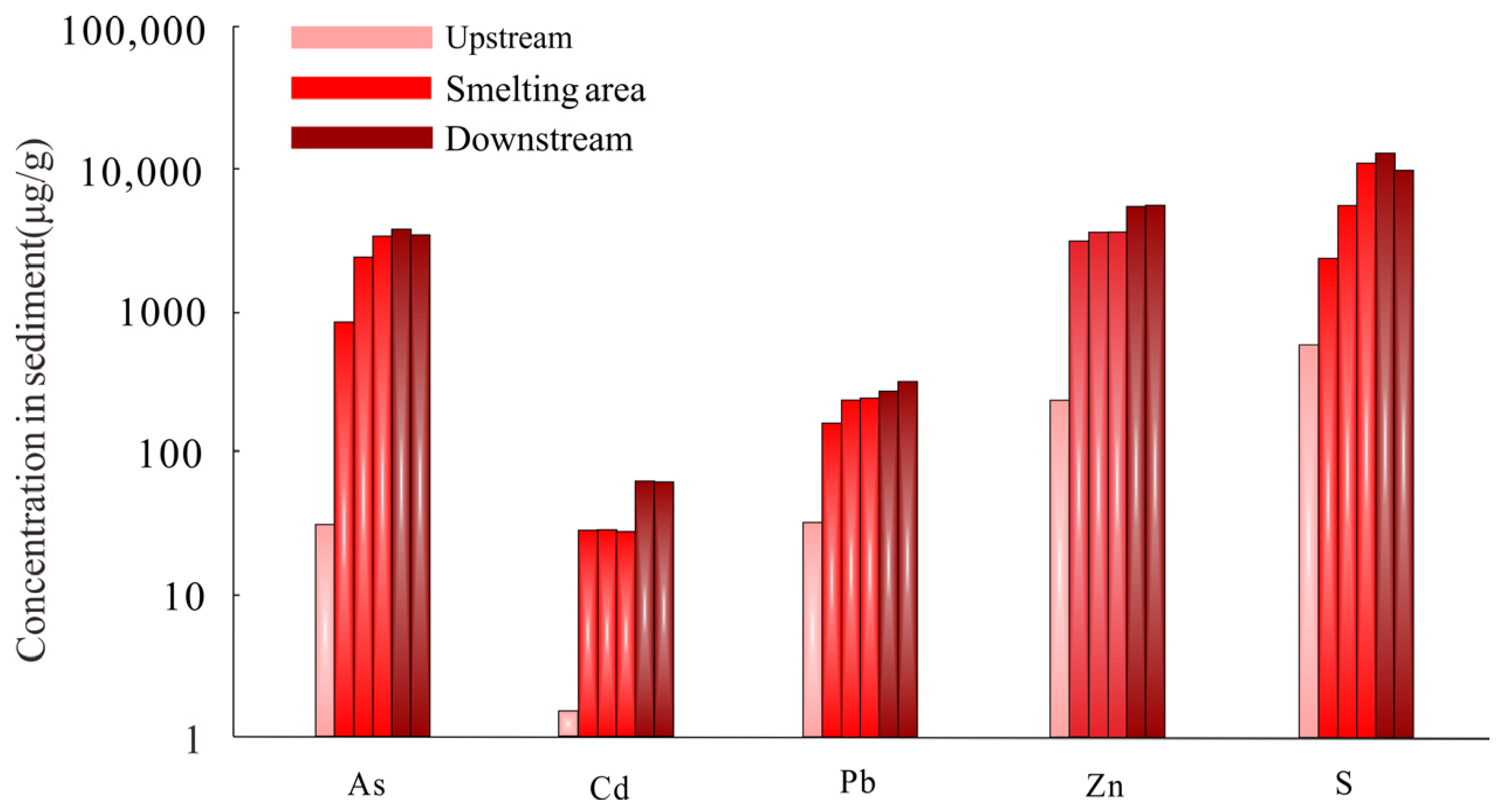
3.3. Solid Wastes
3.4. Risk Assessment for Soil and Crops in Mining Area
3.5. Comprehensive Environmental Response and Pollution Prevention Suggestions
3.5.1. Mechanism of Heavy Metal Migration and Transformation
3.5.2. Potential Risk Assessment
3.5.3. Suggestions on Prevention and Control of Pollution
- (1)
- The area of concentrated mining activities is mainly contaminated through atmospheric deposition, of which smelting emissions are the main source, supplemented by dust from transportation. Therefore, it is necessary to control atmospheric heavy metal deposition within the mine area. Rainfall has a significant impact on the concentration and flux of atmospheric deposition. Therefore, the smelting operation should be properly adjusted in the rainy season to prevent flux increases due to the driving force of rainfall, and the mining and transportation plan can be improved to minimize dust emissions.
- (2)
- Downstream of the mining area, the main focus should be on sediment management. Through the ore district, the river is fast flowing and downcutting the surrounding geology. The middle and lower reaches are more prone to lateral erosion and sedimentation as the flow velocity decreases and the river channel widens. Therefore, special attention should be paid to the risk of secondary pollution caused by heavy metals in sediment where the tributaries converge into the main river, river convex banks, and floodplains. There are differential hazards to corn and rice crops. Consequently, we recommend adjusting the planting structure accordingly, gradually converting paddy fields to dryland crops to reduce the risks associated with paddy soils and irrigation, and planting mainly dryland crops that are safe for consumption.
4. Conclusions
- The heavy metals pollution caused by atmospheric deposition in the mine area is relatively high. The annual deposition flux density of Cd is two-times higher than the relevant standards. Influenced by climate and topography, heavy metals contamination from atmospheric deposition migrates about 25 km along the wind direction and then decreases. About 90% of the heavy metals migrate in the form of vertical deposition in the mine area.
- Effluent drainage from the ore district contains individual heavy metals exceeding the standards, but the water quality of the river is less affected by the mining activities. After treatment, only As slightly exceeded the standard at the river bend, the pH of the water was neutral–alkaline, and the concentrations of soluble heavy metal ions were low.
- Weathering and migration of ore and tailings contribute to high concentrations of heavy metals in river sediments, mainly downstream of the mine. Risks associated with migration of sulfide particles are high, while heavy metals migrating in chemical form are more stable after co-precipitated with carbonates or adsorbed by secondary iron oxides.
- The soil in the mine area is greatly affected by mining activities. The surface soil is significantly enriched in heavy metals and greatly exceeds the standards. It is mainly influenced by atmospheric deposition. The risks of the soil environment to various crops differ. Cd in rice greatly exceeds the standard, while corn does not exhibit heavy metals exceedances and can be safely consumed.
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Li, Z.; Ma, Z.; Kuijp, T.J.v.d.; Yuan, Z.; Huang, L. A Review of Soil Heavy Metal Pollution From Mines in China: Pollution and Health Risk Assessment. Sci. Total Environ. 2014, 468–469, 843–853. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.-S.; Youm, S.-J.; Oh, C.; Cho, Y.-C.; Ahn, J.S. Geochemical and Eco-Toxicological Characteristics of Stream Water and Its Sediments Affected by Acid Mine Drainage. Catena 2017, 148, 52–59. [Google Scholar] [CrossRef]
- Tian, S.; Liang, T.; Li, K. Fine Road Dust Contamination in a Mining Area Presents a Likely Air Pollution Hotspot and Threat to Human Health. Environ. Int. 2019, 128, 201–209. [Google Scholar] [CrossRef] [PubMed]
- Hong, Y. Analysis of Environmental Impact and Prevention and Protection Measures of Small Non-Metallic Mines. Inn. Mong. Sci. Technol. Econ. 2013, 298, 50–51. [Google Scholar]
- Liu, S.; Guo, A.; Li, L. Discussion on Geological Environment Problems and Recovery of One Limestone Deposit in Guilin City of Guangxi Province. Shandong Land Resour. 2017, 33, 65–70. [Google Scholar]
- Li, B.; Yang, Z.; Ji, W.; Yu, T.; Hou, Q.; He, H.; Zhang, Q.; Wu, T.; Qin, J. Ecological Effect of A Typical Sulfide Deposit in Carbonate Area—Xijikeng Lead-Zinc Mine in Guigang, Guangxi. Geoscience 2020, 34, 957. [Google Scholar]
- Zhou, S.; Shi, Z. Study on the Environment of Metal Sulfide Mines. Acta Mineral. Sin. 2013, 33, 733–734. [Google Scholar]
- Lindsay, M.B.J.; Moncur, M.C.; Bain, J.G.; Jambor, J.L.; Ptacek, C.J.; Blowes, D.W. Geochemical and Mineralogical Aspects of Sulfide Mine Tailings. Appl. Geochem. 2015, 57, 157–177. [Google Scholar] [CrossRef]
- Shu, X.; Zhang, Q.; Lu, G.; Yi, X.; Dang, Z. Pollution Characteristics and Assessment of Sulfide Tailings from the Dabaoshan Mine, China. Int. Biodeterior. Biodegrad. 2018, 128, 122–128. [Google Scholar] [CrossRef]
- García-Giménez, R.; Jiménez-Ballesta, R. Mine Tailings Influencing Soil Contamination by Potentially Toxic Elements. Environ. Earth Sci. 2017, 76, 51. [Google Scholar] [CrossRef]
- Lei, L.; Song, C.; Xie, X.; Li, Y.; Wang, F. Acid Mine Drainage and Heavy Metal Contamination in Groundwater of Metal Sulfide Mine at Arid Territory (BS Mine, Western Australia). Trans. Nonferrous Met. Soc. China 2010, 20, 1488–1493. [Google Scholar] [CrossRef]
- Zheng, K.; Li, H.; Wang, L.; Wen, X.; Liu, Q. Pyrite Oxidation Under Simulated Acid Rain Weathering Conditions. Environ. Sci. Pollut. Res. 2017, 24, 21710–21720. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhao, H.; Qian, L.; Sun, M.; Lv, X.; Zhang, L.; Petersen, J.; Qiu, G. A Brief Overview on the Dissolution Mechanisms of Sulfide Minerals in Acidic Sulfate Environments at Low Temperatures: Emphasis on Electrochemical Cyclic Voltammetry Analysis. Miner. Eng. 2020, 158, 106586. [Google Scholar] [CrossRef]
- Battistel, M.; Stolze, L.; Muniruzzaman, M.; Rolle, M. Arsenic Release and Transport during Oxidative Dissolution of Spatially-Distributed Sulfide Minerals. J. Hazard. Mater. 2021, 409, 124651. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Zhang, T.; Yang, W.; Zhang, Y. The Environmental Impact of Mining and Its Countermeasures. MATEC Web Conf. 2016, 63, 04010. [Google Scholar] [CrossRef]
- Wu, W.; Wang, L.; Song, Z.; Zhou, J. Discussions on the Contradiction between Development of Mineral Resources and Ecological Environment Protection in the New Era in China. China Min. Mag. 2020, 29, 6–10. [Google Scholar]
- Xu, M.; Cai, M.; Peng, Z.; Wang, X.; Chen, Y. Research on Metallogenic Zoning Characteristics and Control Mechanism of Dachang Ore Field. Miner. Resour. Geol. 2011, 25, 29–33. [Google Scholar]
- DZ/T 0295-2016; Specification of Land Quality Geochemical Assessment. Ministry of Ecology and Environment: Beijing, China, 2016.
- Tang, Q.; Yang, Z.; Zhang, B.; Feng, H.; Wang, H. A study of Elements Flux and Sources from Atmospheric Bulk Deposition in the Chengdu Economic Region. Earth Sci. Front. 2007, 14, 213–222. [Google Scholar]
- Tao, M.; Zhou, J.; Liang, J.; Cui, H.; Xu, L.; Zhu, Z. Atmospheric Deposition of Heavy Metals in Farmland Area Around a Copper Smelter. J. Argo-Environ. Sci. 2014, 33, 1328–1334. [Google Scholar]
- Ye, L.; Huang, M.; Zhong, B.; Wang, X.; Tu, Q.; Sun, H.; Wang, C.; Wu, L.; Chang, M. Wet and Dry Deposition Fluxes of Heavy Metals in Pearl River Delta Region (China): Characteristics, Ecological Risk Assessment, and Source Apportionment. J. Environ. Sci. 2018, 70, 106–123. [Google Scholar] [CrossRef]
- Chen, Q.; Ma, M.; You, Y.; Yang, D. Cadmium Input Flux in Farmland Soil Near the Dabaoshan Mining Area of Guangdong Province. Geol. Miner. Resour. South China 2020, 36, 147–152. [Google Scholar]
- Tang, J.; Li, N.; Li, H.; Bian, J.; Li, Z.; Cui, Y. Flux and Source Appointment of Heavy Metals from Atmospheric Dry and Wet Deposition in Daqing City, China. J. Jilin Univ. Earth Sci. Ed. 2012, 42, 507–513. [Google Scholar]
- Yang, Z.; Lu, W.; Long, Y. Atmospheric Dry and Wet Deposition of Heavy Metals in Changchun City, China. Res. Environ. Sci. 2009, 22, 28–34. [Google Scholar]
- GB 25466-2010; Emission Standard of Pollutants for Lead and Zinc Industry. Ministry of Ecology and Environment of the People’s Republic of China: Beijing, China, 2010.
- GB 3838-2002; Environmental Quality Standards for Surface Water. Ministry of Ecology and Environment of the People’s Republic of China: Beijing, China, 2002.
- Zhou, Y.; Song, S.; Zhang, C.; Yang, X.; Liu, C. Water Environmental Geochemical Response of Rivers to Mines and Mining Activity—A Case Study of the Diaojiang River Drainage System, Guangxi, China. Geol. Bull. China 2005, 24, 940–944. [Google Scholar]
- Moncur, M.C.; Jambor, J.L.; Ptacek, C.J.; Blowes, D.W. Mine Drainage from the Weathering of Sulfide Minerals and Magnetite. Appl. Geochem. 2009, 24, 2362–2373. [Google Scholar] [CrossRef]
- Zheng, K.; Li, H.; Wang, L.; Wen, X.; Liu, Q. Galena Weathering Under Simulated Acid Rain Conditions: Electrochemical Processes and Environmental Assessments. Environ. Sci. Process Impacts 2018, 20, 822–832. [Google Scholar] [CrossRef]
- McKibben, M.A.; Tallant, B.A.; del Angel, J.K. Kinetics of Inorganic Arsenopyrite Oxidation in Acidic Aqueous Solutions. Appl. Geochem. 2008, 23, 121–135. [Google Scholar] [CrossRef]
- Wu, P.; Liu, C.; Yang, Y.; Zhang, G. Release and Transport of (Heavy) Metals and Their Environmental Effect in Mining Activities. Acta Mineral. Sin. 2001, 21, 213–218. [Google Scholar]
- Wu, Z.; Song, Y.; Hou, Z.; Liu, Y.; Zhuang, L. The World-Class Huoshaoyun Nonsulfide Zinc-Lead Deposit, Xinjiang, NW China: Formation by Supergene Oxidization of a Mississippi Valley-Type Deposit. Earth Sci. 2019, 44, 1987–1997. [Google Scholar]
- Jian, L. Research on Spatial Distribution and Formation Mecharism of Heavy Metals and Arsenic Species in Guangxi Diaojiang River. Master’s Thesis, Northwest A&F University, Xianyang, China, 2010. [Google Scholar]
- GB 15618-2018; Soil Environmental Quality Risk Control Standard for Soil Contamination on Agricultural Land. Ministry of Ecology and Environment of the People’s Republic of China: Beijing, China, 2018.
- GB 2762-2017; National Standard for Food Safety Maximum Levels of Contaminants in Foods. China’s National Health Commission (NHC): Beijing, China, 2017.
- Lu, S.; Zhang, Y.; Yu, Y.; Zhong, X.; Tian, M.; Huang, Y.; Song, B. Characteristics of Heavy Metal Accumulation in Soil-Corn System Contents and Their Health Risks in Nandan, Guangxi. J. Ecol. Rural. Environ. 2017, 33, 706–714. [Google Scholar]
- Berger, A.C.; Bethke, C.M.; Krumhansl, J.L. A process model of natural attenuation in drainage from a historic mining district. Appl. Geochem. 2000, 15, 655–666. [Google Scholar] [CrossRef]
- Huang, C.-C.; Liang, C.-M.; Yang, T.-I.; Chen, J.-L.; Wang, W.-K. Shift of bacterial communities in heavy metal contaminated agricultural land during a remediation process. PLoS ONE 2021, 16, e0255137. [Google Scholar] [CrossRef] [PubMed]
- Fiordelisi, A.; Piscitelli, P.; Trimarco, B.; Coscioni, E.; Iaccarino, G.; Sorriento, D. The mechanisms of air pollution and particulate matter in cardiovascular diseases. Heart Fail. Rev. 2017, 22, 337–347. [Google Scholar] [CrossRef] [PubMed]

| Dry Season | Rainy Season | |||||||
|---|---|---|---|---|---|---|---|---|
| As | Cd | Pb | Zn | As | Cd | Pb | Zn | |
| Min | 1.27 | 0.78 | 22.55 | 52.12 | 4.65 | 1.24 | 24.08 | 74.75 |
| Max | 552.15 | 58.70 | 197.71 | 2632.15 | 1628.54 | 76.19 | 1161.17 | 3173.91 |
| Mean | 89.18 | 13.68 | 87.95 | 577.33 | 196.22 | 27.86 | 421.26 | 1123.85 |
| Annual deposition flux density of heavy metals in the atmosphere of different regions [19,20,21,22,23,24] | ||||||||
| Nandan | Guixi | Dinghu Mountain | Dabaoshan | Changchun | Daqing | Chengdu | ||
| As | 285 | - | - | - | 47.9 | 8.1 | 27.7 | |
| Cd | 41.55 | 65.6 | 5.7 | 1.97 | 2.5 | 1.7 | 17.7 | |
| Pb | 509 | 700 | 428.4 | - | 123.1 | 157.1 | 459.5 | |
| Zn | 1701 | 2250 | 1857.8 | - | 481.5 | 788.1 | 1478.3 | |
| Medium | Sampling Site | As | Cd | Pb | Zn | SO42− | pH |
| Mine Sewage (n = 3) | Tailings pond/dam | 0.33 | 0.0005 | 0.0007 | 0.037 | 1372 | 6.61 |
| 0.007 | 0.019 | 0.0007 | 2.45 | 215 | 7.29 | ||
| 1.61 | 0.016 | 0.04 | 0.079 | 1472 | 7.28 | ||
| GB 25466-2010 | Direct Discharge | 0.3 | 0.05 | 0.5 | 1.5 | - | 6–9 |
| Indirect Discharge | 0.3 | 0.05 | 0.5 | 1.5 | - | 6–9 | |
| Concentration | As | Cd | Pb | Zn | SO42− | pH | |
| Surface Water (n = 6) | Range | 0.018–0.18 | 0.0001–0.0032 | 0.0006–0.0007 | 0.0095–0.047 | 87.4–250 | 7.75–8.06 |
| Mean | 0.07 | 0.0011 | 0.0007 | 0.0218 | 155 | - | |
| GB 3838-2002 | Class III | 0.05 | 0.005 | 0.05 | 1.0 | 250 | 6–9 |
| Fe2O3 | S | As | Cd | Pb | Zn | pH | ||
|---|---|---|---|---|---|---|---|---|
| % | % | μg/g | μg/g | μg/g | μg/g | - | ||
| Tailings (n = 2) | Min | 3.57 | 2.51 | 5484 | 22.6 | 328 | 2833 | 7.71 |
| Mean | 6.64 | 3.45 | 5908 | 47 | 2203 | 6397 | - | |
| Max | 9.70 | 4.39 | 6331 | 71 | 4078 | 9960 | 7.85 | |
| Ore (n = 4) | Min | 21.42 | 21.87 | 32,440 | 85.90 | 3395 | 10,310 | 3.20 |
| Mean | 21.52 | 24.57 | 65,073 | 91.98 | 3759 | 10,663 | - | |
| Max | 21.69 | 27.14 | 99,080 | 99.30 | 4010 | 11,210 | 4.88 | |
| Sediment (n = 6) | Min | 5.00 | 0.06 | 32.60 | 1.60 | 34 | 243 | 7.32 |
| Mean | 7.98 | 0.73 | 2321 | 35.70 | 218 | 3710 | - | |
| Max | 10.86 | 1.34 | 3788 | 64.70 | 334 | 5850 | 8.80 | |
| ΔCsediment-tailings | - | 20.25% | −78.77% | −60.71% | −23.72% | −90.10% | −42.01% | - |
| ΔCsediment-ore | - | −62.93% | −97.02% | −96.43% | −61.19% | −94.20% | −65.21% | - |
| Paddy Field (n = 3) | As | Cd | Pb | Zn | pH |
| Min | 49.3 | 1.92 | 61.5 | 222 | 5.68 |
| Mean | 61 | 4.04 | 138.50 | 420 | - |
| Max | 67.2 | 5.41 | 196 | 548 | 6.2 |
| Exceedance rate | 100% | 100% | 66.67% | 100% | - |
| Dryland (n = 62) | As | Cd | Pb | Zn | pH |
| Min | 23.3 | 0.15 | 28 | 67.9 | 4.47 |
| Mean | 685 | 12.53 | 569 | 1562 | - |
| Max | 5273 | 108 | 5003 | 14,715 | 8.48 |
| Exceedance rate | 90.32% | 98.39% | 70.97% | 93.55% | - |
| Rice (n = 3) | As | Cd | Pb | Zn | Corn (n = 62) | As | Cd | Pb | Zn |
|---|---|---|---|---|---|---|---|---|---|
| Min | 0.25 | 0.26 | 0.078 | 19.1 | Min | 0.03 | 0.0075 | 0.07 | 19.9 |
| Mean | 0.27 | 0.60 | 0.09 | 21.03 | Mean | 0.10 | 0.02 | 0.09 | 31.10 |
| Max | 0.3 | 1.13 | 0.1 | 24.8 | Max | 0.32 | 0.085 | 0.2 | 46.2 |
| Exceedance rate | - | 100% | 0% | - | Exceedance rate | 0% | 0% | 0% | - |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, B.; Yu, T.; Ji, W.; Liu, X.; Lin, K.; Li, C.; Ma, X.; Yang, Z. Geochemical Response of Surface Environment to Mining of Sn-Pb-Zn Sulfide Deposits: A Case Study of Dachang Tin Polymetallic Deposit in Guangxi. Water 2023, 15, 1550. https://doi.org/10.3390/w15081550
Li B, Yu T, Ji W, Liu X, Lin K, Li C, Ma X, Yang Z. Geochemical Response of Surface Environment to Mining of Sn-Pb-Zn Sulfide Deposits: A Case Study of Dachang Tin Polymetallic Deposit in Guangxi. Water. 2023; 15(8):1550. https://doi.org/10.3390/w15081550
Chicago/Turabian StyleLi, Bo, Tao Yu, Wenbing Ji, Xu Liu, Kun Lin, Cheng Li, Xudong Ma, and Zhongfang Yang. 2023. "Geochemical Response of Surface Environment to Mining of Sn-Pb-Zn Sulfide Deposits: A Case Study of Dachang Tin Polymetallic Deposit in Guangxi" Water 15, no. 8: 1550. https://doi.org/10.3390/w15081550
APA StyleLi, B., Yu, T., Ji, W., Liu, X., Lin, K., Li, C., Ma, X., & Yang, Z. (2023). Geochemical Response of Surface Environment to Mining of Sn-Pb-Zn Sulfide Deposits: A Case Study of Dachang Tin Polymetallic Deposit in Guangxi. Water, 15(8), 1550. https://doi.org/10.3390/w15081550






