Index-Based Groundwater Quality Assessment of Nestos River Deltaic Aquifer System, Northeastern Greece
Abstract
1. Introduction
2. Poseidon (PoS) Index
- (i)
- The main category of the PoS index (1 to 6) and its subcategory, if any;
- (ii)
- The dominant factor (or factors with a Qfi > 20%) which controls the quality status of the sample;
- (iii)
- A final unique color characterization, which makes the data visually clearer, followed by accurate information on the level of quality degradation.
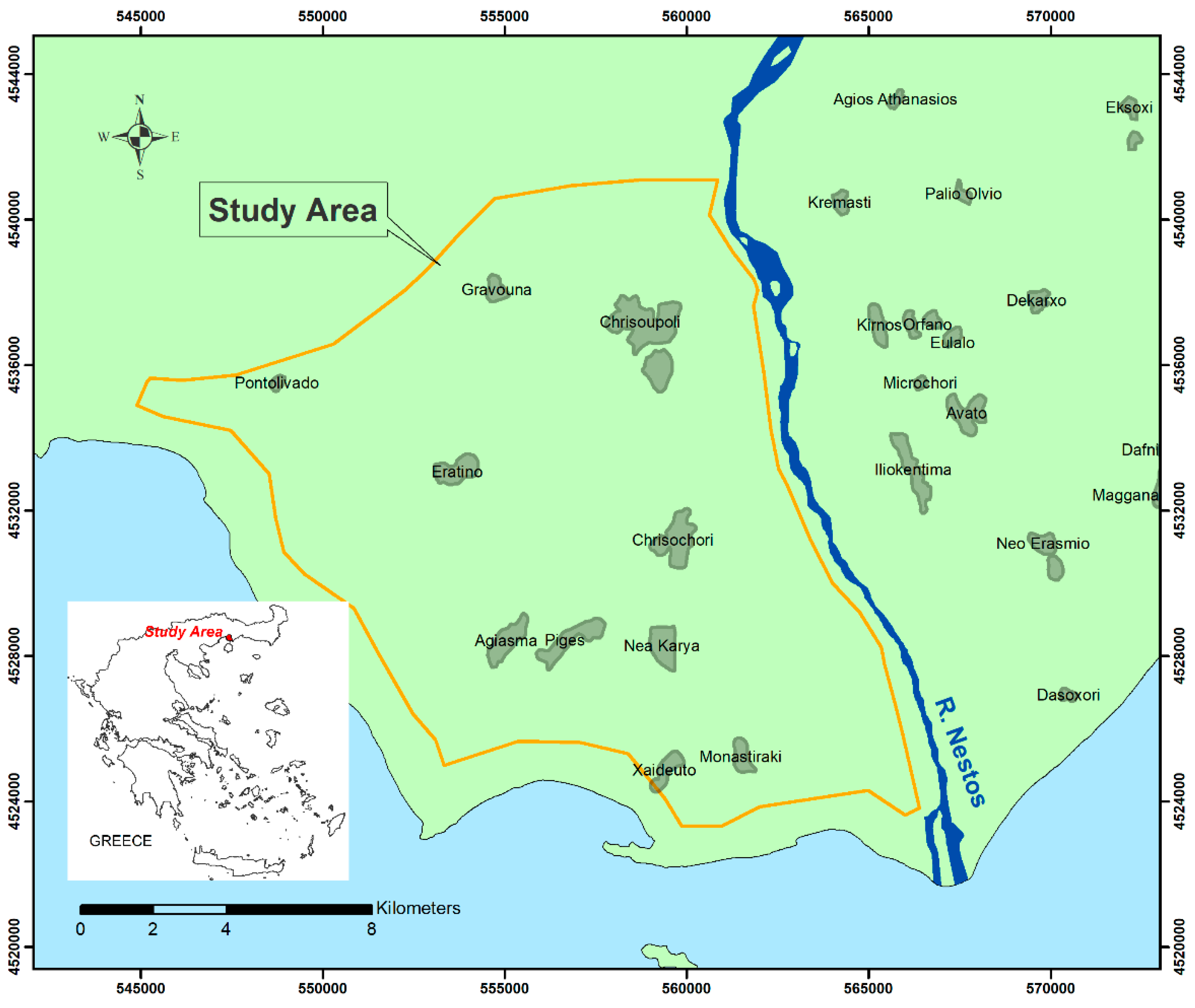
3. Case Study
4. Geo-Hydrological Setting
5. Materials and Methods
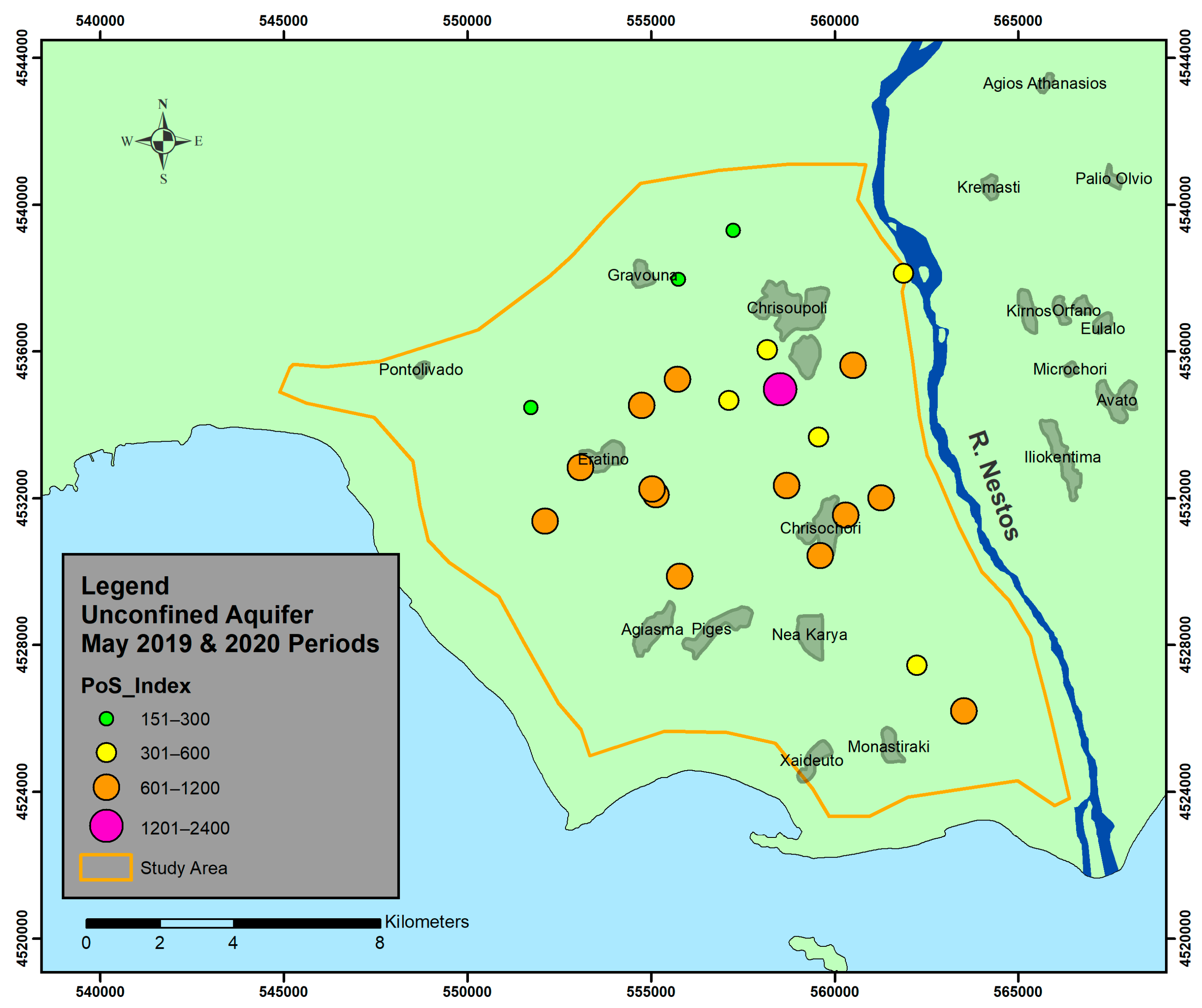
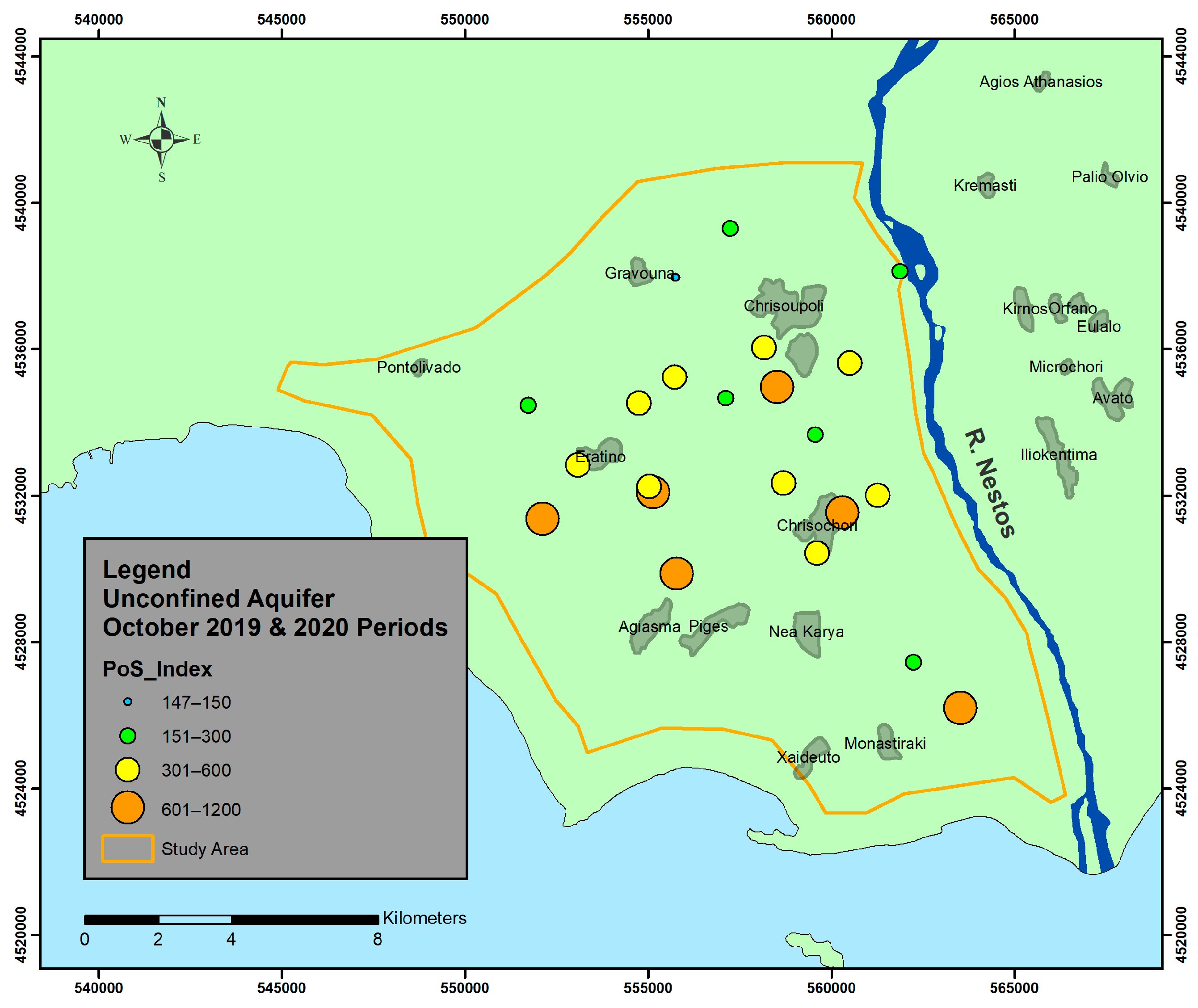
6. Results and Discussion
- Unconfined aquifer
- Confined aquifer
7. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kim, J.; Kim, R.; Lee, J.; Chang, H. Hydrogeochemical characterization of major factors affecting the quality of shallow groundwater in the coastal area at Kimje in South Korea. Environ. Geol. 2003, 44, 189–478. [Google Scholar] [CrossRef]
- Zhu, G.F.; Li, Z.Z.; Su, Y.H.; Ma, J.Z.; Zhang, Y.Y. Hydrogeochemical and isotope evidence of groundwater and recharge in Minqin Basin, Northwest China. J. Hydrol. 2007, 333, 239–251. [Google Scholar] [CrossRef]
- Sandeep, K.G.; Tziritis, E.; Sudhir, K.S.; Jayant, K.T.; Abhay, K.S. Environmental monitoring of water resources with the use of PoS index: A case study from Subarnarekha River basin, India. Environ. Earth Sci. 2018, 77, 70. [Google Scholar] [CrossRef]
- Piper, A.M. A graphic procedure in the geochemical interpretation of water analyses. Eos Trans. Am. Geophys. Union 1994, 25, 914–923. [Google Scholar]
- Durov, S.A. Natural water and graphical representation of their composition. Dokl. Akad. Nauk USSR 1948, 59, 87–90. [Google Scholar]
- Wilcox, L.V. Classification and Use of Irrigation Waters; Circular No. 969; U.S. Department of Agriculture: Washington, DC, USA, 1955; 19p. [Google Scholar]
- Revelle, R. Criteria for recognition of sea water in ground waters. Trans. Am. Geophys. Union 1941, 22, 593–597. [Google Scholar]
- Sivakumar, S.; Chandrasekaran, A.; Ravisankar, R.; Ravikumar, S.M.; Jebakumar, J.P.P.; Vijayagopal, P. Rements of natural radioactivity and evaluation of radiation hazards in coastal sediments of east coast of Tamilnadu, India using statistical approach. J. Taibah Univ. Sci. 2014, 8, 375–384. [Google Scholar] [CrossRef]
- McKenna, J.E. An enhanced cluster analysis program with bootstrap significance testing for ecological community analysis. Environ. Model. Softw. 2003, 18, 205–220. [Google Scholar] [CrossRef]
- Tziritis, E.; Panagopoulos, A.; Arampatzis, G. Development of an operational index of water quality (PoS) as a versatile tool to assist groundwater resources management and strategic planning. J. Hydrol. 2014, 517, 339–350. [Google Scholar] [CrossRef]
- Tziritis, E.; Arampatzis, G.; Hatzigiannakis, E.; Panoras, G.; Panoras, A.; Panagopoulos, A. Quality characteristics and hydrogeochemistry of irrigation waters from three major olive groves in Greece. Desalination Water Treat. 2016, 57, 11582–11591. [Google Scholar] [CrossRef]
- Tziritis, E.; Skordas, K.; Kelepertsis, A. The use of hydrochemical analyses and multivariate statistics for the characterization of groundwater resourses in the complex aquifer system. A case study in Amyros River basin, Thessaly, central Greece. Environ. Earth Sci. 2016, 75, 1–11. [Google Scholar] [CrossRef]
- Salcedo-Sánchez, E.R.; Garrido Hoyos, S.E.; Esteller Alberich, M.V.; Martínez Morales, M. Application of water quality index to evaluate groundwater quality (temporal and spatial variation) of an intensively exploited aquifer (Puebla Valley, Mexico). Environ. Monit. Assess. 2016, 188, 1–20. [Google Scholar] [CrossRef]
- Fernández, N.; Solano, F. Índices de Calidad y de Contaminación del Agua: Technical Report; Universidad de Pamplona: Norte de Santander, Colombia, 2005. (In Spanish) [Google Scholar]
- Lumb, A.; Sharma, T.C.; Bibeault, J.-F.; Klawunn, P. A Comparative Study of USA and Canadian Water Quality Index Models. Water Qual. Expo. Health 2012, 3, 203–216. [Google Scholar] [CrossRef]
- CCME. Canadian Environmental Quality Guidelines for the Protection of Aquatic Life; CCME water quality index: Technical report, 1.0; CCME: Winnipeg, MB, Canada, 2001. [Google Scholar]
- Basha, U.I.; Rajasekhar, M.; Ghosh, S. Spatial assessment of groundwater quality using CCME-WQI and hydrochemical indices: A case study from Talupula Mandal, Ananthapuramu district, South India. Appl. Water Sci. 2022, 12, 168. [Google Scholar] [CrossRef]
- Agency for Toxic Substances and Disease Registry (ATSDR). Detailed Data Table for the 2011 Priority List of Hazardous Substances. Agency for Toxic Substances and Disease Registry. 2011. Available online: https://stacks.cdc.gov/view/cdc/26622 (accessed on 20 November 2011).
- Soltani, S.; Moghaddam, A.A.; Barzegar, R.; Kazemian, N.; Tziritis, E. Hydrogeochemistry and water quality of the Kordkandi-Duzduzan plain, NW Iran: Application of multivariate statistical analysis and PoS index. Environ. Monit. Assess. 2017, 189, 455. [Google Scholar] [CrossRef] [PubMed]
- Vrouchakis, I. Hydrogeological Research Using Modern Management Tools in the Context of Qualitative and Quantitative Monitoring of the Aquifer System of the Tyrnavos Subbasin (Thessaly). Ph.D. Thesis, Athens Agronomist University, Athens, Greece, 2022. (In Greek). [Google Scholar]
- Valko, M.M.H.C.M.; Morris, H.; Cronin, M.T.D. Metals, toxicity and oxidative stress. Curr. Med. Chem. 2005, 12, 1161–1208. [Google Scholar] [CrossRef] [PubMed]
- International Toxicity Estimates for Risk (ITER). Toxicology Data Network. International Toxicity Estimates for Risk. 2011. Available online: https://pubmed.ncbi.nlm.nih.gov/15760833/ (accessed on 30 November 2012).
- World Health Organization (WHO). Guidelines for Drinking-Water Quality, 4th ed.; WHO: Geneva, Switzerland, 2011.
- Allen, M.J.; Yen, W.M. Introduction to Measurement Theory, printing ed.; Waveland Press: Long Grove, IL, USA, 2002. [Google Scholar]
- Directive 2020/2184/EC; European Directive on the Quality of Water Intended for Human Consumption. Publication Office of the European Union: Luxembourg, 2020.
- Delimani, P. Geological Changes of the Coastline in the Thrace Region and Impact on the Land Use of the Coastal Zone. Ph.D. Thesis, Department of Civil Engineering, Democritus University of Thrace, Xanthi, Greece, 2000. (In Greek). [Google Scholar]
- American Public Health Association. APHA Standard Methods for The Examination of Water and Wastewater, 23rd ed.; American Public Health Association (APHA): Washington, DC, USA, 2017. [Google Scholar]
- Novotny, V. Water Quality: Prevention, Identification and Management of Diffuse Pollution; Van Nostrand-Reinhold Publishers: New York, NY, USA, 1994; ISBN 0442005598. [Google Scholar]
- DIN EN ISO 9963-1:1996-02; Wasserbeschaffenheit—Bestimmung der Alkalinität—Teil 1: Bestimmung der Gesamten und der Zusammengesetzten Alkalinität (ISO 9963-1:1994); Deutsche Fassung EN ISO 9963-1:1995. DIN Standards Committee Water Practice (NAW): Berlin, Germany, 1995; p. 24.
- Keeney, D.R.; Nelson, D.W. Methods of Soil Analysis; Page, A.L., Ed.; Agronomy Monographs; American Society of Agronomy, Soil Science Society of America: Madison, WI, USA, 1983; ISBN 9780891189770. [Google Scholar]
- Belcher, R.; Macdonald, A.M.G.; Parry, E. On mohr’s method for the determination of chlorides. Anal. Chim. Acta 1957, 16, 524–529. [Google Scholar] [CrossRef]
- ISO 9964-3:1993; Water Quality—Determination of Sodium and Potassium—Part 3: Determination of Sodium and Potassium by Flame Emission Spectrometry. ISO: Geneva, Switzerland, 1993.
- ISO 15586:2003; Water Quality—Determination of Trace Elements Using Atomic Absorption Spectrometry with Graphite Furnace [Online]. ISO: Geneva, Switzerland, 2003. Available online: https://www.iso.org/standard/38111.html (accessed on 13 May 2018).
- Mohammed, E.; Mohammed, T.; Mohammed, A. Optimization of instrument conditions for the analysis for mercury, arsenic, antimony and selenium by atomic absorption spectroscopy. MethodsX 2018, 5, 824–833. [Google Scholar] [CrossRef]
- Nehls, G.J.; Akland, G.G. Procedures for handling aerometric data. J. Air Pollut. Control Assoc. 1973, 23, 180–184. [Google Scholar] [CrossRef]
- Clarke, J.U. Evaluation of censored data methods to allow statistical comparisons among very small samples with below detection limit observations. Environ. Sci. Technol. 1998, 32, 177–183. [Google Scholar] [CrossRef]
- Hornung, R.W.; Reed, L.D. Estimation of average concentration in the presence of nondetectable values. Appl. Occup. Environ. Hyg. 1990, 5, 46–51. [Google Scholar] [CrossRef]
- Kolios, N.; Koutsino, S.; Arvanitis, A.; Rydakis, G. Geothermal Situation in Northeastern Greece. In Proceedings of the World Geothermal Congress, Antalya, Turkey, 24–29 April 2005. [Google Scholar]
- Davraz, A.; Nalbantçılar, M.T.; Varol, S.; Önden, İ. Hydrogeochemistry and reservoir characterization of the Konya geothermal elds, Central Anatolia/Turkey. Geochemistry 2022, 82, 125867. [Google Scholar] [CrossRef]
- Morales-Simfors, N.; Bundschuh, J. Arsenic-rich geothermal fluids as environmentally hazardous materials—A global assessment. Sci. Total Environ. 2022, 817, 152669. [Google Scholar] [CrossRef] [PubMed]
- Kaasalainen, H.; Stefánsson, A.; Giroud, N.; Arnórsson, S. The geochemistry of trace elements in geothermal fluids, Iceland. Appl. Geochem. 2015, 62, 207–223. [Google Scholar] [CrossRef]
- Directive 2000/60/EC; European Framework Directive, Guidance Document 18: Guidance on Ground Water Status and Trend Assessment. European Commission: Luxembourg, 2009.
- Directive 2000/60/EC; European Directive on the Establishing a Framework for Community Action in the Field of Water Policy. European Commission: Luxembourg, 2000.
- Directive 2006/118/EC; European Directive on the Protection of Groundwater against Pollution and Deterioration. European Commission: Luxembourg, 2006.
- 1st Review of the Thracian Water District (EL 12)—National Gazette of Greece No 4680B. 2017. Available online: https://wfdver.ypeka.gr/el/consultation-gr/1revision-consultation-gr/consultation-1revision-el12-gr/ (accessed on 13 May 2018).





| Toxic Class | Substances | Points | P-Class | Partial Score (PS) | Weighting Factor (Wf) |
|---|---|---|---|---|---|
| 1 | As | 1 | VI | 10 | 0.19 |
| 2 | Pb | 10 | V | 8 | 0.16 |
| Cd | 10 | V | 8 | 0.16 | |
| 3 | NH4 | 100 | IV | 5 | 0.10 |
| NO2 | 100 | IV | 5 | 0.10 | |
| 4 | NO3 | 1000 | III | 3 | 0.06 |
| B | 5000 | II | 1.5 | 0.03 | |
| Cu | 5000 | II | 1.5 | 0.03 | |
| Mn | 5000 | II | 1.5 | 0.03 | |
| Ca | 50,000 | I | 1 | 0.02 | |
| Cl | 50,000 | I | 1 | 0.02 | |
| Fe | 50,000 | I | 1 | 0.02 | |
| K | 50,000 | I | 1 | 0.02 | |
| Mg | 50,000 | I | 1 | 0.02 | |
| Na | 50,000 | I | 1 | 0.02 | |
| SO4 | 50,000 | I | 1 | 0.02 | |
| EC | 50,000 | I | 1 | 0.02 |
| PoS Class | Range | Min | Max | Subclass Range | a | b | c | Quality Degradation Level | Color | |||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | a ≤ t | 0 | 150 | - | <t | None–Low | ||||||
| 2 | t < a < 2t | 150 | 300 | 50 | 150 | 200 | 200 | 250 | 250 | 300 | Low | |
| 3 | 2t < a < 4t | 300 | 600 | 100 | 300 | 400 | 400 | 500 | 500 | 600 | Moderate | |
| 4 | 4t < a < 8t | 600 | 1200 | 200 | 600 | 800 | 800 | 1000 | 1000 | 1200 | High | |
| 5 | 8t < a < 16t | 1200 | 2400 | 400 | 1200 | 1600 | 1600 | 2000 | 2000 | 2400 | Very High | |
| 6 | a > 16t | 2400 | - | - | - | Severe | ||||||
| Well Code | Asdis | Pbdis | Cddis | NH4+ | NO2− | NO3− | Bdis | Cudis | Mndis | Ca2+ | Cl− | Fedis | Mg2+ | Na+ | SO42− | EC |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| μg/L | μg/L | μg/L | mg/L | mg/L | mg/L | mg/L | μg/L | μg/L | mg/L | mg/L | μg/L | mg/L | mg/L | mg/L | μS/cm | |
| 25 | 1.00 | <LOD | <LOD | <LOD | 0.04 | 7.36 | 0.04 | <LOD | 844.70 | 84.40 | 10.50 | 59.18 | 11.88 | 9.05 | 22.58 | 546 |
| 26 | 25.35 | <LOD | 0.19 | 0.79 | 0.07 | 17.84 | <LOD | <LOD | 1016.10 | 76.48 | 14.88 | 6.27 | 16.53 | 16.76 | 45.04 | 549.5 |
| 27 | 6.43 | <LOD | 0.31 | 0.90 | <LOD | <LOD | <LOD | <LOD | 939.68 | 114.30 | 10.91 | 73.14 | 16.33 | 22.35 | 78.04 | 732.5 |
| 28 | 8.00 | <LOD | 0.21 | 0.06 | 0.41 | 17.34 | 0.14 | <LOD | 784.54 | 132.38 | 57.08 | 8.42 | 39.50 | 75.88 | 122.65 | 1118.5 |
| 29 | <LOD | <LOD | <LOD | 0.30 | 1.24 | 11.86 | 0.08 | <LOD | 791.35 | 81.08 | 13.28 | 156.35 | 16.38 | 23.78 | 17.85 | 635 |
| 30 | <LOD | <LOD | <LOD | <LOD | 0.25 | 5.33 | 0.11 | <LOD | 1262.35 | 80.05 | 13.73 | 128.32 | 23.60 | 38.81 | 59.76 | 722.5 |
| 31 | <LOD | <LOD | 0.46 | 0.07 | 0.23 | 8.21 | <LOD | <LOD | 1347.75 | 88.45 | 9.87 | 13.80 | 11.80 | 11.52 | 32.40 | 645.5 |
| 32 | 1.34 | <LOD | 0.23 | 0.08 | 0.25 | 9.57 | <LOD | <LOD | 797.26 | 81.23 | 7.64 | 6.48 | 8.90 | 8.16 | 3.52 | 422 |
| 33 | <LOD | 4.28 | <LOD | 4.90 | <LOD | 30.83 | <LOD | <LOD | 20.77 | 85.46 | 95.05 | 302.70 | 11.63 | 81.98 | 71.27 | 837 |
| 34 | <LOD | <LOD | <LOD | 0.11 | <LOD | 37.27 | <LOD | <LOD | 25.46 | 126.00 | 55.60 | 15.42 | 10.25 | 34.48 | 77.76 | 848 |
| 35 | 6.58 | <LOD | 0.24 | 4.11 | 0.02 | <LOD | 0.28 | <LOD | 760.88 | 64.65 | 126.29 | 26.76 | 42.13 | 127.62 | 5.97 | 1086.5 |
| 36 | 7.08 | <LOD | <LOD | 0.52 | 0.08 | 5.64 | <LOD | <LOD | 1394.25 | 95.28 | 33.44 | 32.72 | 18.58 | 28.72 | 65.04 | 717 |
| 37 | 7.98 | <LOD | 0.44 | <LOD | 0.43 | 13.17 | <LOD | <LOD | 369.54 | 55.85 | 6.56 | <LOD | 6.80 | 7.06 | <LOD | 335 |
| 38 | 8.99 | <LOD | <LOD | 0.34 | 0.02 | <LOD | 0.05 | <LOD | 1346.00 | 100.63 | 26.01 | 27.84 | 20.93 | 49.57 | 78.04 | 749 |
| 39 | 4.45 | <LOD | <LOD | <LOD | <LOD | <LOD | <LOD | <LOD | 22.67 | 106.73 | 399.11 | 19.30 | 29.10 | 182.93 | 18.88 | 1728 |
| 40 | 16.88 | <LOD | <LOD | 2.89 | <LOD | 1.22 | 0.39 | <LOD | 964.50 | 84.15 | 268.38 | 42.18 | 27.50 | 364.82 | <LOD | 2310 |
| 41 | 6.23 | <LOD | 0.19 | 5.30 | <LOD | 5.11 | 0.52 | 12.63 | 199.86 | 40.49 | 60.82 | 769.90 | 15.20 | 155.56 | <LOD | 986.5 |
| 42 | 45.58 | <LOD | <LOD | 1.66 | 0.02 | <LOD | <LOD | <LOD | 2345.60 | 67.48 | 6.89 | 102.02 | 8.48 | 10.68 | <LOD | 416 |
| 43 | 10.98 | <LOD | 0.31 | 1.37 | <LOD | <LOD | <LOD | <LOD | 1277.30 | 73.33 | 9.19 | 135.10 | 11.78 | 9.44 | <LOD | 454 |
| 44 | 3.38 | <LOD | 0.27 | 0.67 | <LOD | <LOD | 0.10 | <LOD | 981.00 | 86.83 | 25.14 | 82.75 | 19.48 | 65.79 | 74.67 | 860.5 |
| 45 | 10.09 | <LOD | <LOD | 0.78 | 0.06 | 12.50 | 0.42 | <LOD | 966.90 | 105.40 | 55.60 | 12.13 | 38.45 | 134.16 | 132.18 | 1206 |
| 46 | 4.94 | <LOD | 0.19 | 0.61 | 0.26 | 12.02 | 0.07 | <LOD | 1314.25 | 116.45 | 24.99 | 22.30 | 30.85 | 43.48 | 109.64 | 816 |
| Well Code | Asdis | Pbdis | Cddis | NH4+ | NO2− | NO3− | Bdis | Cudis | Mndis | Ca2+ | Cl− | Fedis | Mg2+ | Na+ | SO42− | EC |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| μg/L | μg/L | μg/L | mg/L | mg/L | mg/L | mg/L | μg/L | μg/L | mg/L | mg/L | μg/L | mg/L | mg/L | mg/L | μS/cm | |
| 25 | 5.11 | <LOD | <LOD | 0.17 | <LOD | 5.09 | <LOD | <LOD | 266.00 | 97.63 | 9.31 | 11.68 | 11.30 | 9.10 | 26.31 | 553 |
| 26 | 23.00 | <LOD | <LOD | 0.96 | 0.06 | 14.76 | 0.08 | <LOD | 387.50 | 79.48 | 13.52 | 9.58 | 14.48 | 15.21 | 44.85 | 571.5 |
| 27 | 6.21 | <LOD | <LOD | 0.74 | <LOD | <LOD | <LOD | <LOD | 709.50 | 109.60 | 9.25 | 15.39 | 13.95 | 17.29 | 51.22 | 653 |
| 28 | 12.74 | <LOD | <LOD | 0.07 | 0.15 | 34.41 | 0.13 | <LOD | 315.76 | 80.43 | 36.55 | 4.85 | 22.25 | 66.11 | 87.22 | 884 |
| 29 | 4.37 | <LOD | <LOD | 0.38 | 0.03 | 6.57 | 0.05 | <LOD | 263.46 | 78.58 | 10.56 | 14.82 | 12.53 | 20.08 | 24.04 | 541.5 |
| 30 | 2.94 | <LOD | <LOD | 0.14 | 0.13 | 11.21 | 0.10 | <LOD | 896.75 | 98.95 | 14.62 | 29.91 | 24.65 | 41.75 | 64.67 | 756 |
| 31 | 3.05 | <LOD | <LOD | <LOD | 0.31 | 12.62 | <LOD | <LOD | 712.00 | 121.25 | 10.59 | 11.00 | 10.90 | 10.39 | 42.13 | 673.5 |
| 32 | 2.29 | <LOD | <LOD | 0.12 | 0.20 | 7.29 | <LOD | <LOD | 300.06 | 89.58 | 11.56 | 8.12 | 8.88 | 8.90 | 32.49 | 516 |
| 33 | 1.73 | <LOD | <LOD | 10.12 | 0.06 | <LOD | 0.04 | <LOD | 17.72 | 33.64 | 85.81 | 201.24 | 12.88 | 57.43 | 83.40 | 728 |
| 34 | 3.10 | <LOD | 0.27 | <LOD | <LOD | 31.82 | <LOD | <LOD | 7.00 | 141.23 | 39.58 | 30.42 | 9.38 | 37.85 | 65.95 | 830 |
| 35 | 8.39 | <LOD | <LOD | 1.28 | <LOD | <LOD | 0.74 | <LOD | 403.88 | 73.53 | 22.51 | 38.67 | 31.90 | 43.49 | 68.04 | 704.5 |
| 36 | 2.84 | <LOD | <LOD | 0.37 | 0.06 | 6.14 | <LOD | <LOD | 466.75 | 100.75 | 11.22 | 10.65 | 18.90 | 29.46 | 65.40 | 675.5 |
| 37 | 9.91 | <LOD | <LOD | 0.17 | <LOD | 6.30 | <LOD | <LOD | 138.53 | 46.85 | 5.37 | 35.85 | 5.65 | 6.87 | 8.31 | 317 |
| 38 | 9.19 | <LOD | <LOD | 0.34 | <LOD | <LOD | 0.10 | <LOD | 848.13 | 91.50 | 15.01 | 10.73 | 22.10 | 86.87 | 67.90 | 771 |
| 39 | 4.38 | <LOD | <LOD | <LOD | <LOD | <LOD | 0.09 | <LOD | 9.87 | 154.43 | 261.27 | 32.97 | 41.15 | 268.76 | 38.67 | 2295 |
| 40 | 21.89 | <LOD | <LOD | 2.04 | 0.02 | 3.11 | 0.27 | <LOD | 404.76 | 64.55 | 268.38 | 27.87 | 20.28 | 238.58 | 5.97 | 1617 |
| 41 | 8.20 | <LOD | <LOD | 2.92 | <LOD | 1.15 | 0.28 | <LOD | 180.81 | 48.78 | 26.12 | 187.57 | 11.88 | 66.45 | <LOD | 626.5 |
| 42 | 23.26 | <LOD | <LOD | 1.41 | <LOD | <LOD | 0.06 | <LOD | 732.31 | 63.60 | 6.68 | 119.71 | 7.85 | 11.11 | <LOD | 390.5 |
| 43 | 8.12 | <LOD | <LOD | 1.60 | <LOD | <LOD | 0.04 | <LOD | 500.75 | 73.88 | 5.72 | 49.50 | 11.93 | 1.00 | <LOD | 469 |
| 44 | 4.14 | <LOD | <LOD | 0.77 | <LOD | <LOD | 0.14 | <LOD | 939.80 | 110.18 | 18.86 | 65.01 | 21.85 | 72.49 | 74.95 | 892.5 |
| 45 | 6.95 | <LOD | <LOD | 0.11 | <LOD | 6.89 | 0.46 | <LOD | 313.25 | 100.58 | 130.79 | 18.01 | 38.83 | 132.40 | 97.59 | 1235 |
| 46 | 4.03 | <LOD | <LOD | 0.23 | 0.09 | 16.18 | 0.04 | <LOD | 760.38 | 116.45 | 19.66 | 38.20 | 29.53 | 42.98 | 120.55 | 802.5 |
| Well code | Asdis | Pbdis | Cddis | NH4+ | NO2− | NO3- | Bdis | Cudis | Mndis | Ca2+ | Cl− | Fedis | Mg2+ | Na+ | SO42− | EC |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| μg/L | μg/L | μg/L | mg/L | mg/L | mg/L | mg/L | μg/L | μg/L | mg/L | mg/L | μg/L | mg/L | mg/L | mg/L | μS/cm | |
| 1 | 9.72 | <LOD | <LOD | 0.12 | <LOD | <LOD | 0.50 | <LOD | 12.95 | 189.63 | 164.25 | 189.63 | 2.41 | 253.19 | 52.95 | 1234.5 |
| 2 | 99.03 | <LOD | <LOD | 2.84 | <LOD | <LOD | 0.20 | <LOD | 756.85 | 226.48 | 42.87 | 226.48 | 18.40 | 75.26 | <LOD | 741.5 |
| 3 | 4.80 | <LOD | <LOD | 0.77 | <LOD | <LOD | 0.16 | <LOD | 662.69 | 27.46 | 23.65 | 27.46 | 16.70 | 95.96 | <LOD | 705.0 |
| 4 | 1.40 | <LOD | <LOD | 0.50 | <LOD | <LOD | <LOD | <LOD | 650.76 | 49.07 | 52.98 | 49.07 | 10.55 | 36.59 | 2.79 | 637.0 |
| 5 | 15.34 | <LOD | <LOD | 0.75 | <LOD | <LOD | <LOD | 5.55 | 830.11 | 87.55 | 8.61 | 87.55 | 8.48 | 17.45 | <LOD | 412.0 |
| 6 | 71.51 | <LOD | <LOD | 1.90 | <LOD | <LOD | 0.10 | <LOD | 1346.95 | 129.01 | 8.45 | 129.01 | 17.55 | 46.30 | <LOD | 511.0 |
| 7 | 30.82 | <LOD | <LOD | 3.02 | <LOD | <LOD | <LOD | <LOD | 814.25 | 6.30 | 33.07 | 6.30 | 5.08 | 22.68 | <LOD | 553.5 |
| 8 | 0.97 | <LOD | <LOD | 0.09 | <LOD | <LOD | 0.12 | <LOD | 177.75 | <LOD | 7.47 | <LOD | 18.30 | 44.00 | 3.15 | 446.5 |
| 9 | 29.00 | <LOD | <LOD | 0.93 | 0.02 | <LOD | 0.18 | <LOD | 838.20 | 7.20 | 4.27 | 7.20 | 15.73 | 31.65 | <LOD | 436.5 |
| 10 | 19.03 | <LOD | <LOD | 0.09 | <LOD | <LOD | 0.52 | <LOD | 6.07 | 116.21 | 97.27 | 116.21 | 1.61 | 232.38 | 41.76 | 1035.0 |
| 11 | 14.72 | <LOD | 0.19 | 0.08 | <LOD | <LOD | 0.38 | 13.40 | 19.70 | 70.89 | 164.66 | 70.89 | 4.00 | 249.67 | 34.58 | 1172.5 |
| 12 | 3.56 | <LOD | 0.19 | <LOD | <LOD | <LOD | 0.16 | <LOD | 22.43 | 52.64 | 102.64 | 52.64 | 5.05 | 184.17 | 30.13 | 910.0 |
| 13 | 26.10 | <LOD | <LOD | 0.74 | <LOD | <LOD | 0.09 | <LOD | 481.71 | 373.85 | 46.75 | 373.85 | 10.43 | 48.93 | 2.70 | 700.5 |
| 14 | 38.68 | <LOD | 0.18 | 3.61 | <LOD | 1.07 | 0.18 | <LOD | 611.16 | 202.35 | 98.87 | 202.35 | 18.58 | 142.08 | <LOD | 987.5 |
| 15 | 95.70 | <LOD | 0.23 | 4.59 | 0.38 | <LOD | 0.57 | <LOD | 694.81 | 30.53 | 107.19 | 30.53 | 32.40 | 232.79 | <LOD | 1424.5 |
| 16 | 11.63 | <LOD | 0.20 | 0.21 | <LOD | <LOD | 0.39 | <LOD | 18.35 | 17.17 | 113.85 | 17.17 | 2.25 | 219.66 | 4.52 | 1004.0 |
| 17 | 3.98 | <LOD | 0.18 | <LOD | 0.03 | 5.99 | <LOD | <LOD | 465.93 | 6.41 | 13.13 | 6.41 | 11.03 | 18.20 | 9.40 | 435.0 |
| 18 | 14.62 | <LOD | 0.17 | 1.96 | <LOD | <LOD | 0.12 | <LOD | 782.25 | 307.94 | 7.20 | 307.94 | 9.60 | 35.84 | 3.79 | 464.5 |
| 19 | 2.72 | <LOD | <LOD | 0.17 | <LOD | <LOD | 0.41 | <LOD | 110.59 | 27.00 | 456.34 | 27.00 | 10.38 | 399.61 | 28.49 | 2127.5 |
| 20 | 16.19 | <LOD | 0.19 | 0.05 | <LOD | <LOD | 0.24 | <LOD | 27.00 | 21.69 | 24.50 | 21.69 | 1.14 | 160.95 | <LOD | 683.5 |
| 21 | 15.83 | <LOD | 0.42 | <LOD | <LOD | <LOD | 0.47 | <LOD | 19.03 | <LOD | 115.05 | <LOD | 2.08 | 222.89 | 15.95 | 1046.0 |
| 22 | < LOD | <LOD | 0.21 | <LOD | <LOD | 142.83 | <LOD | <LOD | 10.03 | 17.24 | 45.39 | 17.24 | 21.03 | 64.48 | 51.79 | 1166.0 |
| 23 | 9.72 | <LOD | 0.20 | 0.76 | <LOD | 55.69 | <LOD | <LOD | 40.81 | 55.26 | 133.55 | 55.26 | 14.08 | 121.27 | 71.49 | 1085.0 |
| 24 | < LOD | <LOD | 0.34 | <LOD | 0.02 | 28.72 | 0.19 | <LOD | 624.44 | 68.30 | 825.41 | 68.30 | 53.18 | 321.27 | 102.22 | 1915.5 |
| Well Code | Asdis | Pbdis | Cddis | NH4+ | NO2- | NO3- | Bdis | Cudis | Mndis | Ca2+ | Cl- | Fedis | Mg2+ | Na+ | SO42- | EC |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| μg/L | μg/L | μg/L | mg/L | mg/L | mg/L | mg/L | μg/l | μg/L | mg/L | mg/L | μg/L | mg/L | mg/L | mg/L | μS/cm | |
| 1 | 8.34 | <LOD | <LOD | <LOD | <LOD | <LOD | 0.37 | <LOD | 8.24 | 43.73 | 93.30 | 43.73 | 3.17 | 188.91 | 33.67 | 977.0 |
| 2 | 63.52 | <LOD | <LOD | 2.67 | <LOD | <LOD | 0.21 | <LOD | 567.50 | 77.36 | 45.76 | 77.36 | 18.55 | 79.05 | <LOD | 747.0 |
| 3 | 6.08 | <LOD | <LOD | 0.75 | <LOD | 2.84 | 0.16 | <LOD | 499.25 | 11.24 | 21.78 | 11.24 | 15.35 | 87.08 | <LOD | 679.0 |
| 4 | 2.44 | <LOD | <LOD | 0.61 | <LOD | <LOD | <LOD | <LOD | 295.92 | 14.85 | 51.35 | 14.85 | 9.25 | 44.25 | 7.06 | 638.0 |
| 5 | 12.99 | <LOD | <LOD | 1.23 | <LOD | 1.88 | <LOD | <LOD | 400.24 | 5.03 | 7.64 | 5.03 | 7.55 | 15.01 | <LOD | 425.5 |
| 6 | 2.42 | <LOD | <LOD | 0.65 | <LOD | 1.46 | 0.05 | <LOD | 1006.25 | 10.92 | 7.67 | 10.92 | 15.93 | 48.22 | <LOD | 477.0 |
| 7 | 21.11 | <LOD | <LOD | 2.83 | <LOD | 3.60 | <LOD | <LOD | 314.57 | 41.91 | 32.28 | 41.91 | 8.85 | 24.86 | <LOD | 547.5 |
| 8 | 1.38 | <LOD | <LOD | 0.08 | <LOD | <LOD | 0.14 | <LOD | 70.16 | 3.50 | 7.61 | 3.50 | 16.03 | 41.26 | <LOD | 447.0 |
| 9 | 26.09 | <LOD | <LOD | 1.01 | <LOD | <LOD | 0.10 | <LOD | 656.75 | 21.60 | 4.25 | 21.60 | 14.30 | 36.29 | <LOD | 434.0 |
| 10 | 16.85 | <LOD | <LOD | 0.12 | <LOD | <LOD | 0.50 | <LOD | <LOD | 50.67 | 92.73 | 50.67 | 1.34 | 213.76 | 42.31 | 1024.0 |
| 11 | 13.18 | <LOD | <LOD | 0.06 | <LOD | <LOD | 0.38 | <LOD | 13.74 | 22.86 | 159.86 | 22.86 | 4.40 | 223.71 | 32.95 | 1199.5 |
| 12 | 3.14 | <LOD | <LOD | <LOD | <LOD | <LOD | 0.15 | <LOD | <LOD | 20.89 | 100.49 | 20.89 | 4.40 | 159.09 | 28.58 | 908.0 |
| 13 | 12.37 | <LOD | <LOD | 0.50 | <LOD | <LOD | 0.04 | <LOD | 105.70 | 35.47 | 40.86 | 35.47 | 9.48 | 63.96 | 7.06 | 704.0 |
| 14 | 29.25 | <LOD | <LOD | 1.93 | <LOD | <LOD | 0.38 | <LOD | 425.38 | 435.60 | 94.67 | 435.60 | 16.30 | 154.93 | 5.40 | 998.5 |
| 15 | 56.23 | <LOD | <LOD | 5.11 | <LOD | 1.06 | 0.76 | <LOD | 323.16 | 37.83 | 233.51 | 37.83 | 38.10 | 318.16 | <LOD | 1437.5 |
| 16 | 11.78 | <LOD | <LOD | <LOD | <LOD | <LOD | 0.46 | <LOD | 12.86 | 345.00 | 103.05 | 345.00 | 17.80 | 208.99 | 31.40 | 1021.0 |
| 17 | 3.75 | <LOD | <LOD | 0.05 | 0.03 | 2.75 | <LOD | <LOD | 128.83 | 24.96 | 6.43 | 24.96 | 7.73 | 11.45 | 11.13 | 362.7 |
| 18 | 10.36 | <LOD | <LOD | 1.83 | <LOD | <LOD | 0.14 | <LOD | 421.88 | 72.95 | 6.48 | 72.95 | 8.93 | 37.85 | <LOD | 464.5 |
| 19 | 2.02 | <LOD | <LOD | 0.11 | <LOD | <LOD | 0.41 | <LOD | 26.66 | 20.68 | 337.70 | 20.68 | 9.60 | 434.88 | 45.67 | 2200.0 |
| 20 | 14.91 | 2.60 | <LOD | <LOD | <LOD | <LOD | 0.25 | <LOD | 6.13 | 35.77 | 20.48 | 35.77 | 0.74 | 147.41 | <LOD | 666.0 |
| 21 | ||||||||||||||||
| 22 | 1.30 | <LOD | 0.21 | <LOD | <LOD | 133.65 | <LOD | <LOD | 13.86 | 15.85 | 52.94 | 15.85 | 20.63 | 51.99 | 95.49 | 1182.5 |
| 23 | 9.00 | <LOD | <LOD | <LOD | <LOD | 57.49 | <LOD | <LOD | 34.00 | 30.27 | 101.25 | 30.27 | 13.50 | 129.40 | 85.49 | 1141.5 |
| 24 | 1.50 | <LOD | <LOD | 0.09 | 0.09 | 26.20 | 0.20 | <LOD | 411.38 | 23.30 | 590.33 | 23.30 | 51.88 | 374.84 | 125.22 | 2820.0 |
| Well Code | PoS Index | Sample Classification | Quality Degradation Level | |
|---|---|---|---|---|
| Class | Color | |||
| 25 | 546 | 3c/Mndis | Moderate | |
| 26 | 1152 | 4c/Asdis, Mndis | High | |
| 27 | 736 | 4a/Mndis | High | |
| 28 | 711 | 4a/Asdis, Mndis | High | |
| 29 | 541 | 3c/Mndis | Moderate | |
| 30 | 790 | 4a/Mndis | High | |
| 31 | 839 | 4b/Mndis | High | |
| 32 | 532 | 3c/Mndis | Moderate | |
| 33 | 236 | 2b/NH4+ | Low | |
| 34 | 98 | 1/NO3- | None–Low | |
| 35 | 722 | 4a/Mndis | High | |
| 36 | 1002 | 4c/Mndis | High | |
| 37 | 420 | 3b/Mndis | Moderate | |
| 38 | 1005 | 4c/Mndis | High | |
| 39 | 186 | 2a/Asdis | Low | |
| 40 | 1054 | 4c/Mndis | High | |
| 41 | 454 | 3b/Asdis, NH4+, Mndis | Moderate | |
| 42 | 2305 | 5c/Asdis, Mndis | Very High | |
| 43 | 1019 | 4c/Asdis, Mndis | High | |
| 44 | 704 | 4a/Mndis | High | |
| 45 | 865 | 4b/Asdis, Mndis | High | |
| 46 | 946 | 4b/Mndis | High | |
| Well Code | PoS Index | Sample Classification | Quality Degradation Level | |
|---|---|---|---|---|
| Class | Color | |||
| 25 | 286 | 2c/Asdis, Mndis | Low | |
| 26 | 736 | 4a/Asdis, Mndis | High | |
| 27 | 577 | 3c/Asdis, Mndis | Moderate | |
| 28 | 519 | 3c/Asdis, Mndis | Moderate | |
| 29 | 278 | 2c/Asdis, Mndis | Low | |
| 30 | 638 | 4a/Mndis | High | |
| 31 | 522 | 3c/Mndis | Moderate | |
| 32 | 255 | 2c/Mndis | Low | |
| 33 | 288 | 2c/NH4+ | Low | |
| 34 | 147 | 1/As, NO3- | None–Low | |
| 35 | 483 | 3b/Asdis, Mndis | Moderate | |
| 36 | 374 | 3a/Mndis | Moderate | |
| 37 | 297 | 2c/Asdis, Mndis | Low | |
| 38 | 721 | 4a/Asdis, Mndis | High | |
| 39 | 195 | 2a/Asdis | Low | |
| 40 | 785 | 4a/Asdis, Mndis | High | |
| 41 | 365 | 3a/Asdis, Mndis | Moderate | |
| 42 | 929 | 4b/Asdis, Mndis | High | |
| 43 | 501 | 3c/Asdis, Mndis | Moderate | |
| 44 | 691 | 4a/Mndis | High | |
| 45 | 407 | 3b/Asdis, Mndis | Moderate | |
| 46 | 594 | 3c/Mndis | Moderate | |
| Sample Code | PoS Index | Sample Classification | Quality Degradation Level | |
|---|---|---|---|---|
| Class | Color | |||
| 1 | 278 | 2c/Asdis | Low | |
| 2 | 2468 | 6/Asdis | Severe | |
| 3 | 526 | 3c/Mndis | Moderate | |
| 4 | 448 | 3b/Mndis | Moderate | |
| 5 | 816 | 4b/Asdis, Mndis | High | |
| 6 | 2242 | 5c/Asdis, Mndis | Very High | |
| 7 | 1156 | 4c/Asdis, Mndis | High | |
| 8 | 154 | 2a/Mndis | Low | |
| 9 | 1092 | 4c/Asdis, Mndis | High | |
| 10 | 440 | 3b/Asdis | Moderate | |
| 11 | 371 | 3a/Asdis | Moderate | |
| 12 | 134 | 1/Asdis | None–Low | |
| 13 | 852 | 4b/Asdis, Mndis | High | |
| 14 | 1243 | 5a/Asdis, Mndis | Very High | |
| 15 | 2443 | 6/Asdis | Severe | |
| 16 | 299 | 2a/Asdis | Low | |
| 17 | 380 | 3a/Asdis, Mndis | Moderate | |
| 18 | 824 | 4b/Asdis, Mndis | High | |
| 19 | 234 | 2b/Asdis, Mndis | Low | |
| 20 | 369 | 3a/Asdis | Moderate | |
| 21 | 387 | 3a/Asdis | Moderate | |
| 22 | 221 | 2b/NO3- | Low | |
| 23 | 351 | 3a/Asdis | Moderate | |
| 24 | 564 | 3c/Mndis | Moderate | |
| Sample Code | PoS Index | Sample Classification | Quality Degradation Level | |
|---|---|---|---|---|
| Class | Color | |||
| 1 | 218 | 2b/Asdis | Low | |
| 2 | 1657 | 5b/Asdis | Very high | |
| 3 | 457 | 3b/Asdis, Mndis | Moderate | |
| 4 | 258 | 2c/Mndis | Low | |
| 5 | 526 | 3c/Asdis, Mndis | Moderate | |
| 6 | 668 | 4a/Mndis | High | |
| 7 | 675 | 4a/Asdis, Mndis | High | |
| 8 | 90 | 1/Asdis, Mndis | None–low | |
| 9 | 929 | 4b/Asdis, Mndis | High | |
| 10 | 387 | 3a/Asdis | Moderate | |
| 11 | 326 | 3a/Asdis | Moderate | |
| 12 | 296 | 2c/Asdis | Low | |
| 13 | 343 | 3a/Asdis | Moderate | |
| 14 | 934 | 4b/Asdis, Mndis | High | |
| 15 | 1484 | 5a/Asdis | Very high | |
| 16 | 318 | 3a/Asdis | Moderate | |
| 17 | 168 | 2a/Asdis, Mndis | Low | |
| 18 | 508 | 3c/Asdis, Mndis | Moderate | |
| 19 | 165 | 2a/Asdis, Na+ | Low | |
| 20 | 344 | 3a/Asdis | Moderate | |
| 21 | ||||
| 22 | 247 | 2b/NO3- | Low | |
| 23 | 316 | 3a/Asdis | Moderate | |
| 24 | 458 | 3b/Mndis | Moderate | |
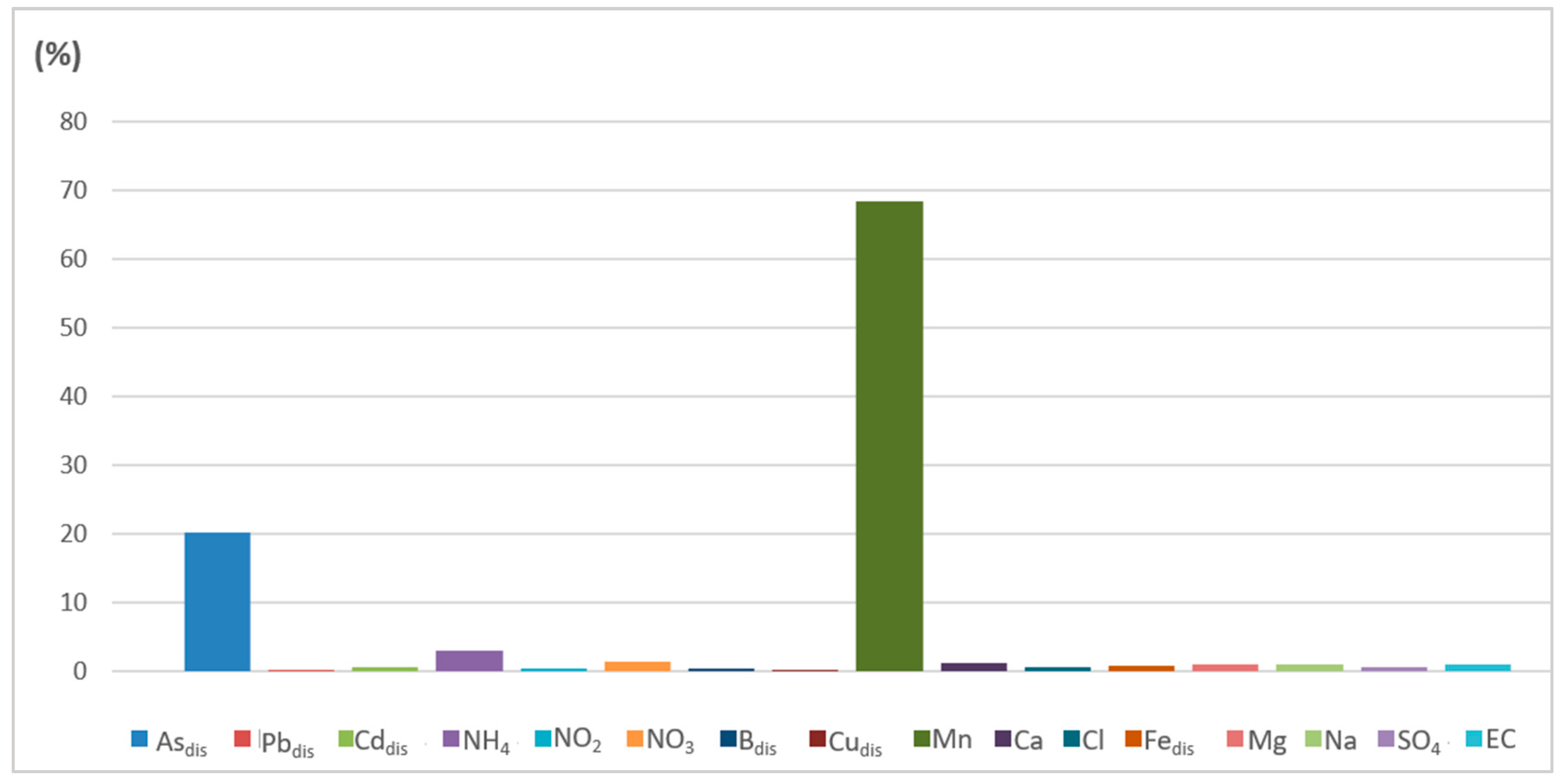
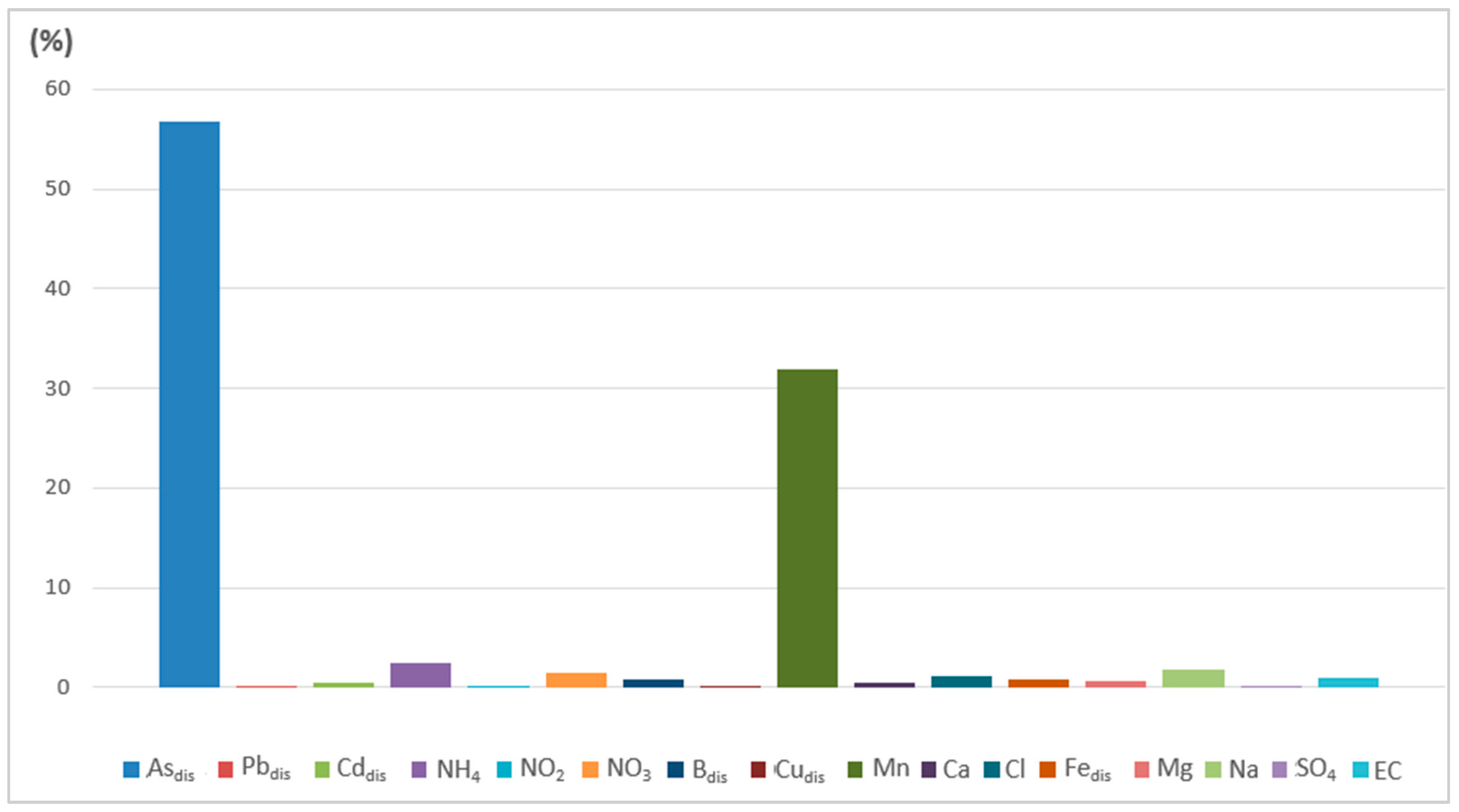
| Wet Period (May 2019 and 2020) | ||||||
|---|---|---|---|---|---|---|
| PoS Index Class | 1 | 2 | 3 | 4 | 5 | 6 |
| No. of samples | 1 | 2 | 5 | 13 | 1 | 0 |
| % (percentage) | 4.55 | 9.09 | 22.73 | 59.09 | 4.55 | 0.00 |
| Dominant factors (Qfs) | NO3- | NH4+ | Mndis | Mndis | Mndis | |
| Asdis | Asdis | Asdis | Asdis | |||
| NH4+ | ||||||
| Dry Period (October 2019 and 2020) | ||||||
| PoS Index class | 1 | 2 | 3 | 4 | 5 | 6 |
| No. of samples | 2 | 6 | 9 | 5 | 0 | 0 |
| % (percentage) | 9.09 | 27.27 | 40.91 | 22.73 | 0.00 | 0.00 |
| Dominant factors (Qfs) | NO3- | Asdis | Asdis | Asdis | ||
| Asdis | Mndis | Mndis | Mndis | |||
| NH4+ | ||||||
| Wet Period (May 2019 and 2020) | ||||||
|---|---|---|---|---|---|---|
| PoS Index Class | 1 | 2 | 3 | 4 | 5 | 6 |
| No. of samples | 1 | 6 | 9 | 3 | 2 | 2 |
| % (percentage) | 4.17 | 25.00 | 37.50 | 12.50 | 8.33 | 8.33 |
| Dominant factors (Qfs) | Asdis | Asdis | Asdis | Asdis | Asdis | Asdis |
| Mndis | Mndis | Mndis | Mndis | |||
| NH4+ | ||||||
| NO3- | ||||||
| Dry Period (October 2019 and 2020) | ||||||
| PoS Index class | 1 | 2 | 3 | 4 | 5 | 6 |
| No. of samples | 1 | 6 | 10 | 4 | 2 | 0 |
| % (percentage) | 4.35 | 26.09 | 43.48 | 17.39 | 8.70 | 0.00 |
| Dominant factors (Qfs) | Asdis | Asdis | Asdis | Asdis | Asdis | |
| Mndis | Mndis | Mndis | Mndis | |||
| Na+ | ||||||
| NO3- | ||||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kampas, G.; Panagopoulos, A.; Gkiougkis, I.; Pouliaris, C.; Pliakas, F.-K.; Kinigopoulou, V.; Diamantis, I. Index-Based Groundwater Quality Assessment of Nestos River Deltaic Aquifer System, Northeastern Greece. Water 2024, 16, 352. https://doi.org/10.3390/w16020352
Kampas G, Panagopoulos A, Gkiougkis I, Pouliaris C, Pliakas F-K, Kinigopoulou V, Diamantis I. Index-Based Groundwater Quality Assessment of Nestos River Deltaic Aquifer System, Northeastern Greece. Water. 2024; 16(2):352. https://doi.org/10.3390/w16020352
Chicago/Turabian StyleKampas, George, Andreas Panagopoulos, Ioannis Gkiougkis, Christos Pouliaris, Fotios-Konstantinos Pliakas, Vasiliki Kinigopoulou, and Ioannis Diamantis. 2024. "Index-Based Groundwater Quality Assessment of Nestos River Deltaic Aquifer System, Northeastern Greece" Water 16, no. 2: 352. https://doi.org/10.3390/w16020352
APA StyleKampas, G., Panagopoulos, A., Gkiougkis, I., Pouliaris, C., Pliakas, F.-K., Kinigopoulou, V., & Diamantis, I. (2024). Index-Based Groundwater Quality Assessment of Nestos River Deltaic Aquifer System, Northeastern Greece. Water, 16(2), 352. https://doi.org/10.3390/w16020352








