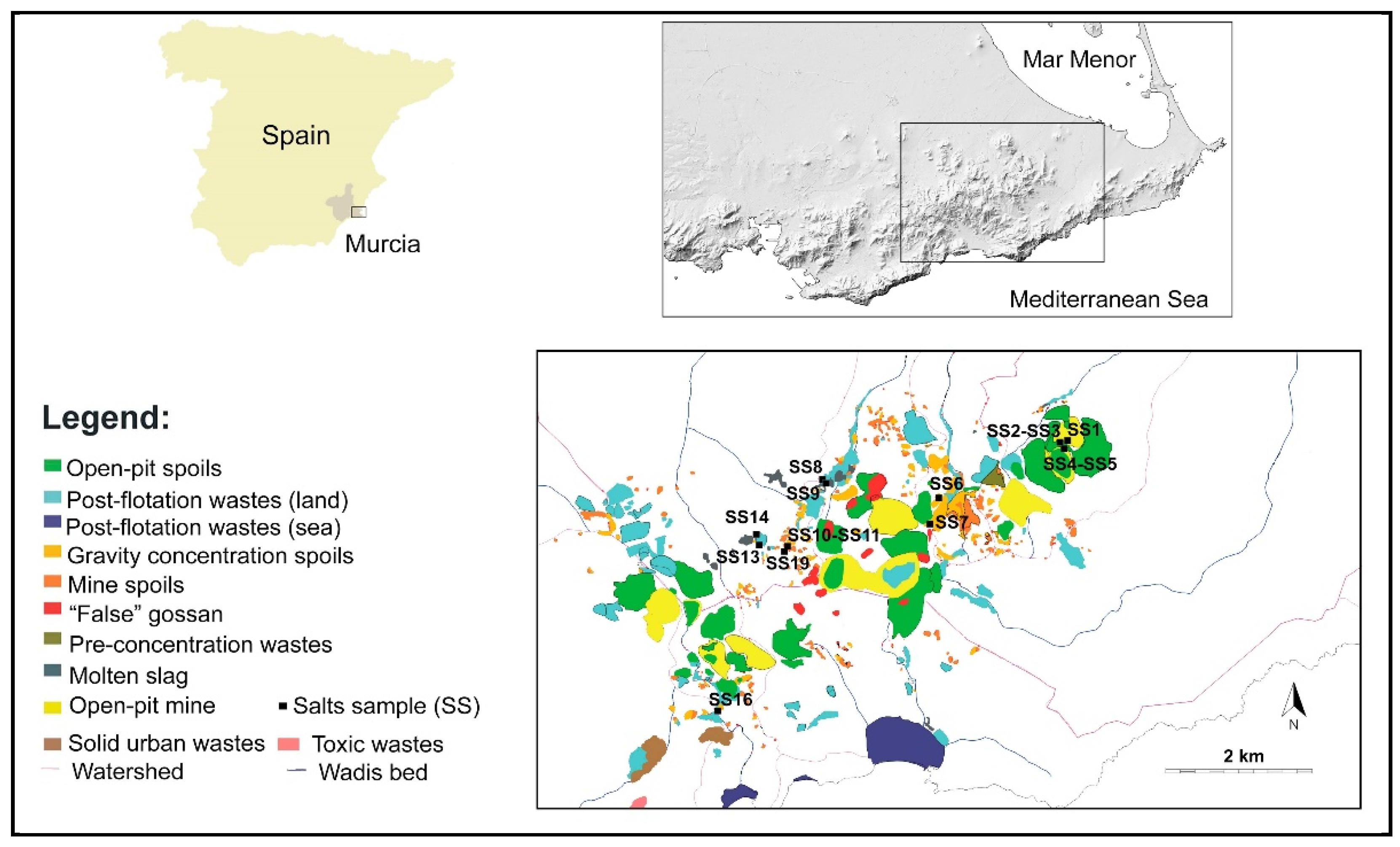
3.2. Mineralogy of the Salt Efflorescences
The semiquantitative estimations for the samples’ crystalline percentages are listed in
Table 1. Data are based on the relative intensity ratio (RIR) approach. Since many precipitates generated in this context were weakly crystalline, computed amorphousness was also provided. On the other hand, the salt efflorescences that form there are also rich in heavy metals and metalloids because the sulphide mine tailings are metalliferous. Sulphates (57.7–100%, average 85.1%), silicates (maximum value 27.2%, average 8.5%) and other binary compounds such as oxides and sulphides (up to 15.9%, average 6.5%) were also detected. The amorphousness ranged from 9.4 to 28.9%, with an average of 17.2%.
These data depict several groups of secondary sulphate minerals. With a maximum of 87% and an average of 47%, the hydrous metal sulphates with divalent cations (A2+SO4·nH2O, with A = Mg, Ca, Mn, Fe, Co, Ni, Cu and Zn; n = 1–7) were the most common. This group was present in every specimen examined, and their crystalline phases are as follows: hexahydrite (averaging 27%), gypsum (averaging 11.8%), starkeyite (only present in SS1 and SS6 and averaging ≤ 41%,), gunningite (averaging ≤ 7%, found exclusively in SS1, SS3 and SS15), bianchite (≤13%, found exclusively in SS1), rozenite (≤11%, found exclusively in SS6), melanterite (≤7%, found exclusively in SS13) and goslarite (≤1%, found exclusively in SS1). The halotrichite (AB2(SO4)4·22H2O, where A = Mg, Mn, FSS2+, Ni and Zn; and B = Al, Cr and FSS3+) and copiapite (A2+FSS3+4(SO4)6(OH)2·20H2O, where group A = Ca, Cu, Fe, Mg and Zn, was also found among the mixed divalent-trivalent hydrated mixed metal sulphates. The second-largest group, halotrichite, reached 100% in SS12 and had an average occurrence of 45.4% among the nine occasions on which its presence was found.
Dietrichite, apjohnite, halotrichite and pickeringite, in increasing order of abundance, represented this group. Copiapite and magnesiocopiapite are present in SS15, SS7 and SS13, respectively. These minerals, which had an overall average of 24.2%, are examples of the copiapite group. The iron hydroxysulphate mineral jarosite group [AFSS3+3(SO4)2(OH)6, where A = Na+, K+, H3O+, Ag+ and Pb2+0.5] gives the crusts their distinctive ochre hue. In this screening, an average of 9.5% of plumbojarosite, hydroniumjarosite, natrojarosite and jarosite were discovered among the six samples in which their presence was detected. Only SS11 included other sulphates as botryogen and slavikite.
The fact that metallic sulphates behave as true solid solutions is important to note since it suggests that the crystalline phases found by diffraction techniques are end-member phases. Within the range of ionic radii allowed by the crystal lattice, the actual situation involves a significant amount of cation substitution. By way of illustration, SS12 recorded 100% pickeringite, while Fe, Mn and Zn, in decreasing order, substituted for Mg at some specific atomic positions, keeping cell characteristics within the range of pickeringite.
Other substances are transported to the soil surface at the same time as the slowly emerging soluble salts. Muscovite (averaging 5.1%), greenalite (averaging 1.7%), kaolinite (with an average of 1%), hornblende (present only in SS4 and SS5 and averaging ≤ 4% in these samples) and vermiculite (present only in SS14, SS7, SS9 and SS10 and averaging ≤ 1% in these samples) served as representatives of silicates in this survey. Finally, the remaining components included oxides and sulphides. Averaging 6%, quartz reached 15.4%, while pyrite was only 3.9% present in SS6 and haematite was only present in SS8 at 5%. This wide variety in the mineralogy of salt efflorescences is consistent with the results of previous studies in other mining areas [
1,
2,
3,
4].
Regarding thermogravimetry-mass spectrometry results, the samples produced three main effluent gases during heating. Dehydration occurred in the thermal range 30–500 °C, with a thermal increase of around 10 °C/min, depending on the amount of free, constitution (or constitutional) and crystalline water, and organic matter. Carbon dioxide loss (decarbonisation) occurred in several steps over 150–650 °C in response to the occurrence of organic matter. Finally, the loss of SO
3 (desulphation) was mostly produced in the range 350–1075 °C, mainly indicating the presence of metallic sulphides and sulphates.
Figure 3 depicts an illustration of decomposition in an oxidising environment.
As previously stated, it was difficult to make a detailed and precise assignment of each weight reduction with each decomposition step due to overlap between neighbouring stages, especially when organic matter was present. Data on H
2O, organic matter, SO
3 and residue content are summarised in
Table 1.
The average percentage of water percentage was 16.1%, with a range of 9.2 to 27.7%. Without taking into account the combustion of organic matter, the dehydration process can reach temperatures of 150–545 °C. This maximum level was higher in the absence of organic matter and there was more sulphate involved, as well as higher water content.
Organic matter was only found in SS4–SS7 and SS13 and varied from 1.3 to 4.3%, with an average of 3.1 % in the five samples in which it was detected. The temperature range, which was wider in an inverse proportion to the water content, was 150–670 °C. It is recognised that microbiological activity supports the biotic oxidation of sulphides and that AMD environments generally support a thriving microbiome [
8].
Sulphide oxidation occurs through an acid-producing process. For this reason, carbonates were hardly found in the salt efflorescence. Additionally, the desulphation of the jarosite mineral group occurs between 500 and 1000 °C [
44], and decomposition of the carbonates occurs during this time. If the mass spectrometry study was carried out carelessly, the results could be misinterpreted. As is common in these secondary minerals, sulphur trioxide was the component that evolved in the greatest abundance in the samples. Its content varied between 12.5 and 44.3%, with an average of 31.1%, growing proportionally to the water level, indicating the predominance of hydroxysulphates and/or hydrated sulphates. The overall decomposition temperature ranged from 350 to 1075 °C, so the 1400 °C necessary to decompose all the sulphur present in the samples was not reached. Moreover, due to the collapse of the crystal lattice, part of the SO
3 could have evolved during the final stages of dehydration and/or decarbonisation. In the end, an average SO
3 recovery value in the gaseous form of 88% was found.
The primary components of the final residue produced after burning were oxides and silicates. These fine ashes ranged from 28.1 to 75.3%, averaging 51.6%, and increased in inverse proportion to the SO3 content.
3.3. Results of the Geochemical Composition of the Salt Efflorescences
In addition, a study of the elemental composition of the salt efflorescence measured by WDXRF and TG-MS (wt.%) was carried out. The loss-on-ignition (LOI) method was used to estimate the water and carbon dioxide contents. Considering only those intensities whose values were greater than several times more than the statistical background, some interesting results have been found. According to Miller & Miller (2010) [
45], about three different fractions can be found in minerals. The major fraction contributes between 10 and 100% of the weight of the sample, the minor fraction accounts for 0.1–10% of the total, and a micro fraction has a concentration of between 1 and 1000 ppm. In the major fraction (
Table 2), elements such as S and Fe have been found with percentages of 6–19 and 0.7–23, respectively. In the minor fraction, elements such as Mg, Si, Al, Zn, Ca, Mn, Na, Pb, Cl and K were found with percentages ranging from 0.4 to 9.5, 0.1 to 9.4, 0.1 to 7.9, 0.04 to 11, 0.05 to 13, 0.05 to 3.6, 0.03 to 1.9, 0.002 to 2.5, 0.02 to 0.4 and 0.01 to 1.0, respectively. Finally, the elements in the micro fraction were As, Sr, Cd, Ba, Sb, Rb, Ag, Zr, Ti, Cu, P, Ni, Co and Cr, with an approximate concentration expressed in ppm of 1750, 451, 358, 220, 130, 110, 67, 57, 35–1610, 26–1393, 39–270, 19–179, 24–93 and 23–68, respectively. This large quantity and variety of elements, both potentially toxic and non-toxic, is common to salt efflorescences studied in other mining areas [
1,
2,
3,
4].
According to the WHO [
46], these health-relevant PTEs in potable water were a major cause of water contamination due to the high solubility of most of the salt efflorescence minerals. These PTEs include elements such as As, Cd, Ba, Sb, Cu, Ni and Cr, which were present in the micro fraction. In the same situation, elements such as Zn, Pb and Mn were present but in higher concentrations.
In terms of the enrichment of PTEs in the salt efflorescence, significant differences were observed between the different elements (
Table 2). The mean PTE values ranged from more than 22690 ppm (Zn) and 4200 ppm (Pb) to approximately 47 ppm (Sb), 49 ppm (Co), 52 ppm (Cr) and 68 ppm (Ni). Other intermediate values were 426 ppm (As), 230 ppm (Cu) and 143 ppm (Cd).
To estimate the enrichment factors, the average enrichment values of the soils in the area in the elements considered were needed (
Table 2). The top 60 cm of the soils’ typical local background concentrations were derived from [
47]. Analytes with EF < 10 (Co = 5, Ni = 3 and Cr = 1) were deemed to be of low enrichment, 10 < EF < 100 (As = 61, Sb = 59 and Cu = 18) suggested mild enhancement, while species with EF > 100 were thought to have a substantial enrichment (Zn = 553, Cd = 477 and Pb = 468). All of this demonstrated that these PTEs are structurally crucial in salt blooms and crusts that are associated with the presence of mining waste, such that they can act as a source of contaminants to the surrounding soil and water.
3.4. Salt Leaching Results
The findings of the leaching tests conducted on the efflorescence of salts are listed in
Table 3. As part of Council Decision 2003/33/EC [
48,
49], which defines standards and practices for the reception of residues at dumpsites (L/S = 10 L/kg; leachate extracted by DIN 38414-S4). This regulation also includes the leaching limit values (LLVs) and total dissolved solids (TDS). The pH regulation levels are given in [
38,
39], while the EC limit value is obtained from [
38]. The leaching tests were carried out at a constant room temperature (25 °C).
All samples examined did not meet the requirements for residues to be accepted at landfills. Levels of acidity, and the most mobile elements of Zn and Cd, whose mean levels surpassed the leaching limit values (LLV) for the acceptance of waste at hazardous waste landfills, were responsible for it. Taking into account the total ratios of each parameter to that of the WHO drinking water recommendations [
45,
46] for each sample, the efflorescence can be classified according to their increasing degree of harmfulness as follows: SS5 < SS10 < SS4 < SS12 < SS6 < SS9 < SS8 < SS8 < SS11 < SS7 < SS7 < SS3 < SS13 < SS2 < SS15 < SS14 < SS1. The environmental hazard level was not specifically correlated with any secondary sulphate mineral. All of them may be regarded as harmful; however, the copiapite group’s sulphates appeared to act as significant PTEs sinks.
European Council Decision 2003/33/EC [
48,
49] defines up to three sets for the reception of waste at dumpsites. Set 1 fixes the limit values for the reception of hazardous residues at hazardous waste landfills, while set 2 determines the limit values for the reception of hazardous residues at non-hazardous waste landfills and set 3 states the acceptance limits for inert waste residues landfills. As for the results of the leaching experiments, all pH values exceeded the LLVs regarded for the reception of hazardous residues at non-hazardous waste landfills, while none of them exceeded these limits for EC (
Table 3). In turn, for TDS, all values except SS4 and SS5 exceeded the LLVs for the reception of inert residues to inert waste landfills.
On the other hand, the different elements and anions considered in this study showed different behaviours depending on the set considered within Decision 2003/33/EC. The values of set 1 were exceeded for elements such as Zn (SS1-SS3, SS6-SS9 and SS13–SS15), As (SS15), Cd (SS1–SS3, SS7–SS9, SS11 and SS13–SS15), Sb (SS15) and SO42− (SS11, SS12, SS14 and SS15). Set 2 cut-off values were found to be close to or above them for Cu (SS15), Zn (SS1–SS9, SS12 and SS13–SS15), As (SS15), Cd (SS10), Sb (SS1–SS3, SS9 and SS15) and SO42− (SS1–SS3, SS6 and SS8–SS15). Finally, set 3 limits were crossed for Ni (all values except SS4, SS5 and SS10), Cu (SS2, SS6, SS8, SS9, SS14 and SS15), Zn (all values), As (SS12 and SS15), Cd (all values except SS10), Sb (SS1-SS3, SS9 and SS15), Pb (SS1, SS2, SS7 and SS13) and SO42− (all values). In addition, for Cl−, no LLV value was exceeded.
Another issue to be considered is the drinking water quality. In this regard, the WHO has provided guidelines for drinking water [
46]. When comparing the values of those guidelines with the results generated in the leaching tests (
Table 3), 1 L of leachate could normally contaminate up to approximately 2000 L of potable water. In this regard, these parameters can be classified according to their contamination potential, in increasing degree, indicating the level of enrichment for each parameter compared to the WHO guidelines: Cu (39) < EC (2–12) < TDS (3–25) < Ni (2–126) < SO
42− (14–145) < Pb (365) < Zn (7–2208) < As (4092) < Cd (6290). The samples did not present any risk of pH, and Cl
− and Sb contamination. In addition to the above, another aspect considered was the enrichment in anions and metallic and metalloid elements of the salt efflorescence leachates concerning the presence of metals and other ions in the soils of the mining area [
47]. In summary, from the data collected in
Table 4, it can be concluded that the percentage of enrichment in metallic elements, metalloids, and anions of these efflorescences concerning the surrounding natural soils can be ordered as follows, in increasing sequence: SS5 < SS10 < SS4 < SS1 < SS7 < SS13 < SS9 < SS6 < SS3 < SS8 < SS8 < SS12 < SS14 < SS2 < SS11 < SS15.
In addition, in
Table 4 it can be seen how the most abundant chemical species can be displayed in increasing order as follows: Pb < Sb < As < Cu < Ni < Ni < Cd < Cl
− < Zn < SO
42−. It is noteworthy that Cd can be easily released during the solubilisation of Zn-enriched salt efflorescence. Paradoxically, although these water-soluble salts were in an area with a high presence of Pb and Zn, Pb was less of an environmental problem as it is less mobilised from salt efflorescence. These results are largely in agreement with data from previous studies [
2,
3,
4].
Complementarily, the different leachates generated by each salt efflorescence were classified using a Ficklin diagram [
38,
39,
40] (
Figure 4).
For this purpose, both the pH values and the sum of metal(loid)s generated in the efflorescence leachates carried out according to the DIN 38414-S4 leaching test were considered. This resulted in a total of four categories of the diagram out of a possible 12. In this respect, four of them (SS4, SS7, SS9 and SS10) were considered as acid low-metal, five of them (SS5, SS6, SS8, SS11 and SS12) were considered as high-acid low-metal, four of them (SS1, SS2, SS3 and SS13) were considered as acid high-metal, while the remaining two (SS14 and SS15) were classified as high-acid high-metal. In summary, about two-thirds were considered low-metal solutions, while the rest of the leachates were categorised as high-metal solutions. Consequently, these leachates can have a significant impact on soil and water quality. In addition, the risk level calculated by adding the proportions of each individual to the WHO guideline limits [
46] was found to be consistent with this Ficklin plot.
3.5. Statistical Results
Pearson’s correlation test and PCA were used in this statistical study. Using the Kolmogorov–Smirnov test, the hypothesis of the normality of the datasets has been validated.
The Pearson product-moment correlation coefficient was entered for each pair of variables to create the correlation matrix. A two-tailed significance test revealed that the correlations were significant. Usually, there are intricate relationships between various aspects [
50]. Data normalisation (Z-score) and Pearson’s correlation matrix were used to eliminate multivariate problems and increase the relevance of mineral data [
51].
This statistical study took into account 15 salt efflorescences (E) and their leachates (L). Solids-related parameters included TG-MS data (H
2O and organic matter levels), as well as potentially toxic elements from WDXRF analysis and the total of these elements. The main sulphate groups measured by XRD were considered; namely, divalent metal(loid) sulphate minerals with the general formula A
2+SO
4-nH
2O. Minerals from the copiapite, halotrichite and jarosite groups were considered. Finally, several variables related to saline efflorescence leachates were also considered, including pH, EC, the soluble fraction of the above PTE as determined by ICP-MS, and Cl
− and SO
42− ion chromatography measurements.
Table 5 shows in bold the significant correlations at the 0.05 level between the values (2-tailed).
The halotrichite group minerals showed a substantial positive association with water content because they have the highest amount of crystalline water (22 molecules). On the other hand, minerals from the group of the divalent metal sulphate, with up to seven water molecules in its composition, showed a negative correlation with water. In addition, there were remarkable positive relationships between EC, Ni (L) and SO42− concentrations and water content. On the other hand, there were negative significant correlations between organic matter and H2O, SO42− and EC.
Positive and significant correlations were found for Zn, Cd, Ni and Cu, with the sum of the set of elements studied (∑M), both in solid and leached form, i.e., all PTEs except those of low leachability (Pb and As). Similarly, Zn, Cd and Ni, and the sum of the PTEs appeared significantly and favourably correlated among themselves. Other significant favourable correlations were found in the copiapite group with Cu in its solid form of sulphate salt efflorescence samples (E) and As in its form of leachates of sulphate salt efflorescence (L), among other correlations. On the other hand, the halotrichite group showed positive correlations with Ni (L) and SO42−, as did the jarosite group with Pb (E). The pH also appeared positively correlated with the divalent metal(loid) sulphate minerals group and with Pb (L), as did EC and Ni (L) with SO42−, Cu (L) with As (L) and SO42−, among others. Interestingly, the Cl− concentration did not correlate with the remaining parameters. Additionally, the divalent metal(loid) sulphate minerals group showed significant negative correlations with the halotrichite group, Ni (L) and SO42−.
Although in previous studies Pb, Cd and Ni had a negative correlation trend with pH [
41], in the sense that at low pH values the mobility of these metal ions increases [
5], in this case, there is either no significant correlation or it is positive, as in the case of Pb, which may be due to the low pH value in all the samples analysed. On the other hand, although organic matter does not present positive correlations with most of the variables considered, the vast majority of these correlations, especially the significant ones, turn out to be negative, which coincides with what has been indicated by other authors [
41].
Significant correlations between minerals and their major (Zn, Pb, etc.) and trace (Cd, As, etc.) elements, such as those found in this research, have been demonstrated by several studies [
52,
53]. However, in many other aspects, the behaviour of these salt efflorescences differs, at least in part, from that shown by the soils and mining tailings described by other authors. Thus, some significant correlations have been found for primary and secondary minerals, such as those described between Fe-Al oxides with Ni and As [
54], between Ni, SO
4, TDS, pH and EC with each other [
55] or between pH and SO
4 [
56], have not been significant for the supergene minerals considered in this study. However, some described correlations between EC, pH and texture of mining waste sediments that seem to determine the mineralogical composition, dissolution and oxidation processes and their influence on the physicochemical properties and mobility of potentially toxic elements associated with metallic mining waste, may be key in the formation of saline efflorescences from these mining tailings [
1,
6,
9,
10,
53,
57,
58]. Similarly, different types of parent materials can generate soils and residues enriched in Ni and Cr [
59], while a distinctly clayey and Fe-enriched texture can act to enrich soils in As [
60]; something that could explain the presence of these elements in salt crusts present in these environments.
Additionally, a PCA analysis was conducted to quantify all of the mineralogical processes in the research area and to determine the link between the variables in the salt efflorescence. The proportional contribution of each mineral source to the salt crust was determined using this technique.
This statistical procedure is often used to identify and understand the hidden complex and causal relationships between the different properties of the dataset. This is achieved by examining the structure of the data at a smaller scale size and keeping as much variability of the data as possible. It is necessary to estimate the number of principal components in the data to achieve this. The eigenvectors formed by the decomposition of a correlation matrix between pairs of parameters are arranged in order of their associated values. PCA is a mathematical procedure that entails auto-scaling values to create new parameters, computing the covariance matrix and removing elements that only add a small amount of variation to the datasets [
8].
When conducting a PCA, the adequacy of sampling must be verified, which requires that the Kaiser–Meyer–Olkin (KMO) test is equal to or greater than 0.50 for the factor arrangement [
43]. Furthermore, it is required that the probability related to Bartlett’s test of sphericity is not as large as the level of noteworthiness, which translates into the prerequisite that the significance level of Bartlett’s test is <0.05 being fulfilled [
43]. After the verification of these two requirements, and therefore being relevant to perform the PCA, the extraction values in the communalities table should be checked in such a way that only those parameters that contribute more than 50% of their variance in the dataset are considered for the PCA test. Varimax with Kaiser normalisation was applied as a rotation method, to ensure that each parameter had a maximum correlation with a single principal component while correlating close to zero with the other elements.
After checking the values of the communalities, it was finally decided to analyse 10 variables with extraction values of the communalities above 0.75, except for one value of 0.66. These 10 variables include the six PTEs considered, in their solid salt form (Ni, Cu, Zn, As, Cd and Pb), and the four groups of supergene minerals considered (divalent metal(loid) sulphate minerals, halotrichite group minerals, copiapite group minerals and jarosite group minerals). These analysed variables should make it possible to unravel the interactions between the different elements under consideration and the secondary minerals.
Table 6 shows the PCA results for the variables mentioned above for the salt crust samples studied.
The KMO test measures, among other things, the adequacy of sampling, while Bartlett’s test of sphericity indicates the strength of correlation between variables. In this study, the KMO test result met its minimum requirement of being at least equal to 0.5, while Bartlett’s test with a significance = 0.000 showed that effective reduction and transformation of the dimensions of the standardised variables can be achieved through PCA. Because of the above, the PCA test with orthogonal varimax rotation was carried out. This methodology has previously been used for similar research purposes by numerous authors [
41,
42,
43,
61].
Four correlated components were extracted by PCA with a total explained variance of more than 87% (
Table 6). When all this information is transferred to a graph (
Figure 5), it can be determined that component 1 showed a close correlation between metals such as Zn, Cd and Ni. On the other hand, component 2 corresponds to the Cu content which revealed strong relationships with the copiapite group minerals. In turn, component 3 includes the Pb and As contents, which showed a moderate interrelationship with the jarosite group minerals. And, finally, component 4 revealed a strong negative relationship with halotrichite group minerals, but a positive one with divalent metal(loid) sulphate minerals.
Zn, Cd and Ni contents of the salt efflorescences were identified as a component explaining one-third of the total observed variance. In turn, halotrichite minerals and divalent metal(loid) sulphate minerals appeared strongly associated with another component, albeit with different signs.
Copiapites were an important component for Cu and responsible for a high percentage of the variance, which is consistent with the results observed in the correlation matrix studied above (
Table 5). These revealing data suggest that the variant of the copiapite mineral group occurring in the study area may be cuprocopiapite. (Cu
2+Fe
3+4(SO
4)
6(OH)
2·20H
2O). This type of copiapite has traditionally been associated with copper-enriched Chilean mining areas [
62]. However, although it is not easy to detect because it usually requires sophisticated techniques such as Raman spectroscopy, it has also been detected in other mining areas in the same mountain range of the study area [
12,
63]. Consequently, this finding can be considered of great interest and should be taken into account for future mineralogical studies in this area to help confirm this issue.
In addition, jarosite efflorescence shared a major component with Pb and As, results that contrast with previous studies insofar as they related this group of minerals to other distinct elements, such as Fe and Cu [
42]. This rare association of jarosite with Pb and As has already been described as a Pb-As-jarosite ((H
3O)
0.68Pb
0.32Fe
2.86(SO
4)
1.69(AsO
4)
0.31(OH)
5.59(H
2O)
0.41) and considered as an analogue of beudantite [
64]. The most common version of this mineral is Pb-jarosite, widely described in different environmental conditions [
7,
65]. In this sense, although this variant of jarosite has never been described for this study area, it has already been mentioned in other mining areas of the same geological complex [
66]. Therefore, this finding can also be considered of great interest and should also be taken into account for future mineralogical studies of this mining area.
In addition, the data of this study are in agreement with observations made by other authors, which indicate that during the neoformation of supergene minerals, they trap elements—Pb, Zn, Cd, As and Cu among others—, as well as SO
42− which are available in mine tailings during dry periods and discharged by dissolution during precipitation events [
1,
6,
9,
10,
42,
53,
58,
67,
68].