Temporal Dynamics of CO2 Fluxes over a Non-Irrigated Vineyard
Abstract
:1. Introduction
2. Materials and Methods
2.1. Site Description
2.2. Instrumentation and Measurements
2.2.1. Eddy Covariance
2.2.2. Soil Chamber
2.2.3. Soil Properties and Meteorological Variables
2.3. Data Analysis
3. Results
3.1. Meteorological Variables
3.2. Dynamics of CO2 Fluxes
3.3. Diurnal and Seasonal Variability in Rsoil
3.4. Comparing Reco and Rsoil
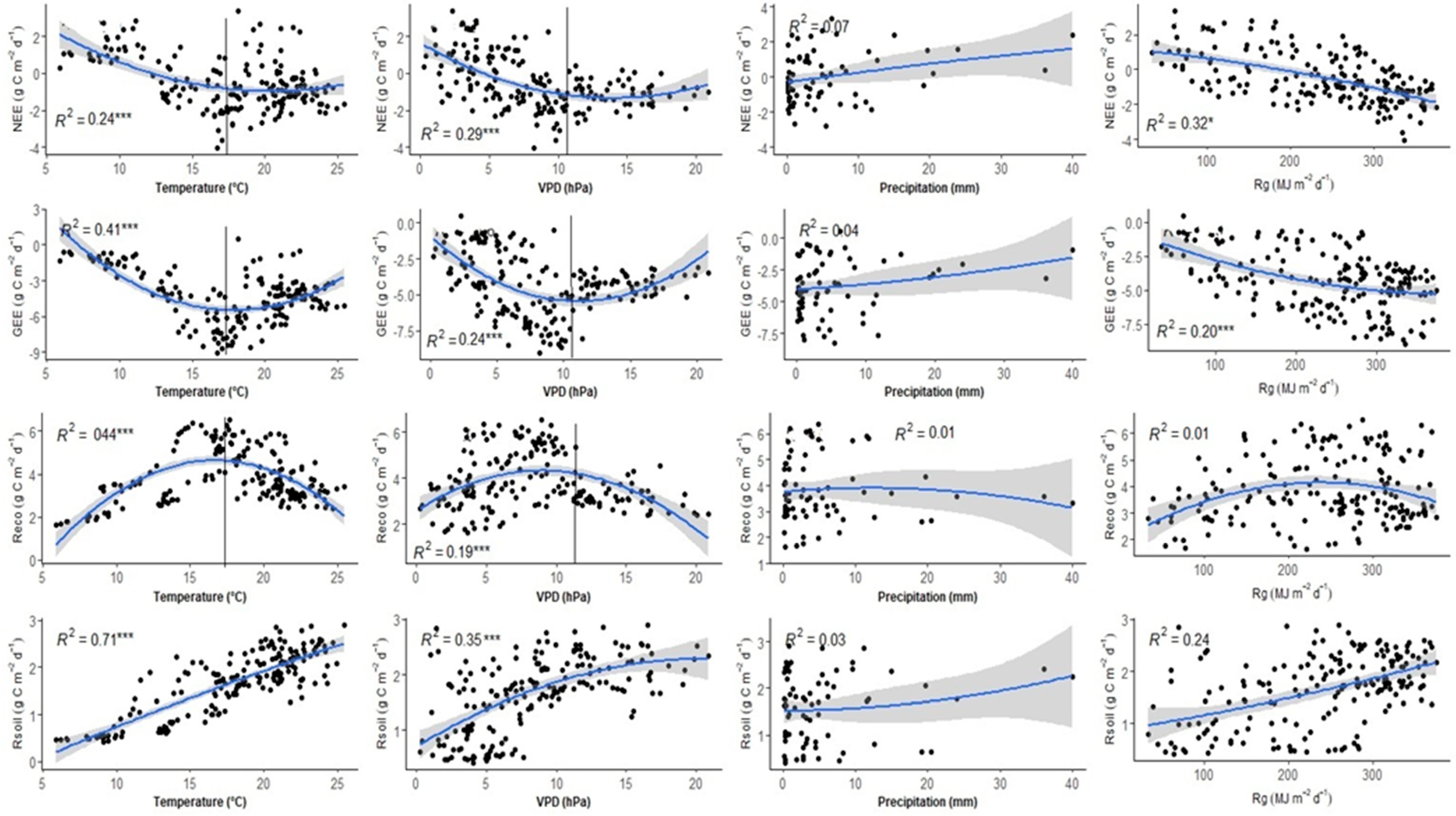
3.5. Response of CO2 Fluxes to Major Environmental Variables
4. Discussion
4.1. Variability in NEE, GEE, and Reco
4.2. CO2 Flux Response to Environmental Variables
4.3. Rsoil and Reco Comparison
4.4. Dynamics of Rsoil and Its Environmental Responses
4.5. Limitations of the Study
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Appendix A
| Soil Aggregation Size Class (mm) | <0.001 | 0.001–0.01 | 0.01–0.05 | 0.05–0.25 | >0.25 |
|---|---|---|---|---|---|
| The proportions of aggregate fractions (%) | 31 | 15 | 22 | 22 | 10 |
| April | May | June | July | August | September | |
|---|---|---|---|---|---|---|
| AIC Q10 model | 26 | 228 | 237 | 140 | 228 | 142 |
| AIC logistic model | 25 | 163 | 196 | 137 | 194 | 118 |
| R2 Q10 model | 0.83 | 0.68 | 0.80 | 0.66 | 0.57 | 0.64 |
| R2 logistic model | 0.84 | 0.80 | 0.85 | 0.69 | 0.70 | 0.73 |
| MAE Q10 model | 0.21 | 0.41 | 0.45 | 0.50 | 0.60 | 0.42 |
| MAE logistic model | 0.21 | 0.30 | 0.39 | 0.47 | 0.49 | 0.35 |
| RMSE Q10 model | 0.30 | 0.52 | 0.54 | 0.61 | 0.71 | 0.50 |
| RMSE logistic model | 0.29 | 0.41 | 0.47 | 0.59 | 0.60 | 0.44 |

References
- IPCC. Summary for Policymakers. In Climate Change 2013: The Physical Science Basis. Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; Stocker, T.F., Qin, D., Plattner, G.K., Tignor, M., Allen, S.K., Boschung, J., Nauels, A., Xia, Y., Bex, V., Midgley, P.M., Eds.; Cambridge University Press: Cambridge, UK, 2013. [Google Scholar]
- Wolff, E.; Fung, I.; Hoskins, B.; Mitchell, J.; Palmer, T.; Santer, B.; Shepherd, J.; Shine, K.; Solomon, S.; Trenberth, K.; et al. Climate Change: Evidence & Causes. An Overview from the Royal Society and the US National Academy of Sciences; The Royal Society: London, UK, 2014; p. 36. [Google Scholar]
- Hadden, D.G. Processes Controlling Carbon Fluxes in the Soil-Vegetation Atmosphere System. Ph.D. Thesis, Swedish University of Agricultural Sciences Uppsala, Uppsala, Switzerland, 2017. [Google Scholar]
- Hüblová, L.; Frouz, J. Contrasting effect of coniferous and broadleaf trees on soil carbon storage during reforestation of forest soils and afforestation of agricultural and post-mining soils. J. Environ. Manag. 2021, 290, 112567. [Google Scholar] [CrossRef]
- Waldo, S.; Chi, J.; Pressley, S.N.; Keeffe, P.O.; Pan, W.L.; Brooks, E.S.; Huggins, D.R.; Stöckle, C.O.; Lamb, B.K. Assessing carbon dynamics at high and low rainfall agricultural sites in the inland Pacific Northwest US using the eddy covariance method. Agric. For. Meteorol. 2016, 218–219, 25–36. [Google Scholar] [CrossRef]
- Ceschia, E.; Béziat, P.; Dejoux, J.F.; Aubinet, M.; Bernhofer, C.; Bodson, B.; Buchmann, N.; Carrara, A.; Cellier, P.; Di Tommasi, P.; et al. Management effects on net ecosystem carbon and GHG budgets at European crop sites. Agric. Ecosyst. Environ. 2010, 139, 363–383. [Google Scholar] [CrossRef]
- Gilmanov, T.G.; Aires, L.; Barcza, Z.; Baron, V.S.; Belelli, L.; Beringer, J.; Billesbach, D.; Bonal, D.; Bradford, J.; Ceschia, E.; et al. Productivity respiration, and light-response parameters of world grassland and agroecosystems derived from flux-tower measurements. Rangel. Ecol. Manag. 2010, 63, 16–39. [Google Scholar] [CrossRef]
- Kutsch, W.L.; Aubinet, M.; Buchmann, N.; Smith, P.; Osborne, B.; Eugster, W.; Wattenbach, M.; Schrumpf, M.; Schulze, E.D.; Tomelleri, E.; et al. The net biome production of full crop rotations in Europe. Agric. Ecosyst. Environ. 2010, 139, 336–345. [Google Scholar] [CrossRef]
- Brunori, E.; Farina, R.; Biasi, R. Sustainable viticulture: The carbon-sink function of the vineyard agro-ecosystem. Agric. Ecosyst. Environ. 2016, 223, 10–21. [Google Scholar] [CrossRef]
- Callesen, T.O.; Gonzalez, C.V.; Campos, F.B.; Zanotelli, D.; Tagliavini, M.; Montagnani, L. Understanding carbon sequestration, allocation, and ecosystem storage in a grassed vineyard. Geoderma Reg. 2023, 34, e00674. [Google Scholar] [CrossRef]
- Scandellari, F.; Caruso, G.; Liguori, G.; Meggio, F.; Palese, A.M.; Zanotelli, D.; Celano, G.; Gucci, R.; Inglese, P.; Pitacco, A.; et al. A survey of carbon sequestration potential of orchards and vineyards in Italy. Eur. J. Hortic. Sci. 2016, 81, 106–114. [Google Scholar] [CrossRef]
- Sharma, S.; Rana, V.S.; Prasad, H.; Lakra, J.; Sharma, U. Appraisal of Carbon Capture, Storage, and Utilization through Fruit Crops. Front. Environ. Sci. 2021, 9, 700768. [Google Scholar] [CrossRef]
- Karlsson, P. The World’s Vineyard Surface in 2020 and the Split by Country, an Analysis|Per on Forbes. BKWine Mag. 2022. Available online: https://www.bkwine.com/features/more/worlds-vineyard-surface-2020/ (accessed on 4 January 2022).
- Morandé, J.A.; Stockert, C.M.; Liles, G.C.; Williams, J.N.; Smart, D.R.; Viers, J.H. From berries to blocks: Carbon stock quantification of a California vineyard. Carbon Balance Manag. 2017, 12, 5. [Google Scholar] [CrossRef]
- Sirca, C.; Asunis, C.; Spano, D.; Arca, A.; Duce, P. Soil CO2 flux measurements in vineyard ecosystem. Acta Hortic. 2004, 664, 615–621. [Google Scholar] [CrossRef]
- Carlisle, E.; Smart, D.; Summers, M. California Vineyard Greenhouse Gas Emissions: An Assessment of the Available Literature and Determination of Research Needs; California Sustainable Winegrowing Alliance: San Francisco, CA, USA, 2009; pp. 1–48. [Google Scholar]
- Oiv. International Organization of Vine and Wine World vitiviniculture situation. In Proceedings of the 38th World Congress of Vine and Wine, Mainz, Germany, 5–10 July 2015. [Google Scholar]
- Smaje, C. The Strong Perennial Vision: A Critical Review. Agroecol. Sustain. 2015, 39, 471–499. [Google Scholar] [CrossRef]
- Marras, S.; Masia, S.; Duce, P.; Spano, D.; Sirca, C. Carbon footprint assessment on a mature vineyard. Agric. For. Meteorol. 2015, 214–215, 350–356. [Google Scholar] [CrossRef]
- Tezza, L.; Meggio, F.; Vendrame, N.; Pitacco, A. Spatial and temporal variation of soil respiration in relation to environmental conditions in a vineyard of northern Italy. In Proceedings of the 19th International Meeting of Viticulture GiESCO, Trento, Italy, 9–11 June 2015. [Google Scholar]
- Smart, D.R.; Wolff, M.W.; Carlisle, E.; Del Mar Alsina Marti, M. Reducing Greenhouse Gas Emissions in the Vineyard: Advances in the Search to Develop More Sustainable Practices; Department of Viticulture & Enology University of California, Robert Mondavi Institute North: Davis, CA, USA, 2009. [Google Scholar]
- Gomiero, T.; Pimentel, D.; Paoletti, M. Is there a need for a more sustainable agriculture? Crit. Rev. Plant Sci. 2011, 30, 6–23. [Google Scholar] [CrossRef]
- Gianelle, D.; Gristina, L.; Pitacco, A.; Spano, D.; La Mantia, T.; Marras, S.; Meggio, F.; Novara, A.; Sirca, C.; Sottocornola, M. The role of vineyards in the carbon balance throughout Italy. In The Greenhouse Gas Balance of Italy, Environmental Science and Engineering; Valentini, R., Miglietta, F., Eds.; Springer: Berlin/Heidelberg, Germany, 2015; pp. 159–171. [Google Scholar]
- Scandellari, F.; Zanotelli, D.; Ceccon, C.; Bolognesi, M.; Montagnani, L.; Cassol, P.; Melo, G.W.; Tagavini, M. Enhancing prediction accuracy of soil respiration in an apple orchard by integrating photosynthetic activity into a temperature-related model. Eur. J. Soil Biol. 2015, 70, 77–87. [Google Scholar] [CrossRef]
- Nistor, E.; Dobrei, A.G.; Dobrei, A.; Camen, D.; Sala, F.; Prundeanu, H. N2O, CO2, production, and C sequestration in vineyards: A review. Water Air Soil Pollut. 2018, 229, 299. [Google Scholar] [CrossRef]
- Zhang, Z.S.; Dong, X.J.; Xu, B.X.; Chen, Y.L.; Zhao, Y.; Gao, Y.H.; Hu, Y.G.; Huang, L. Soil respiration sensitivities to water and temperature in a revegetated desert. J. Geophys. Res. Biogeosci. 2015, 120, 773–787. [Google Scholar] [CrossRef]
- Badraghi, A.; Ventura, M.; Polo, A.; Borruso, L.; Giammarchi, F.; Montagnani, L. Soil respiration variation along an altitudinal gradient in the Italian Alps: Disentangling forest structure and temperature effects. PLoS ONE 2021, 16, e0247893. [Google Scholar] [CrossRef]
- Chen, S.; Zou, J.; Hu, Z.; Lu, Y. Climate and Vegetation Drivers of Terrestrial Carbon Fluxes: A Global Data Synthesis. Adv. Atmos. Sci. 2019, 36, 679–696. [Google Scholar] [CrossRef]
- Janssens, I.A.; Lankreijer, H.; Matteucci, G.; Kowalski, A.S.; Buchmann, N.; Epron, D. Productivity overshadows temperature in determining soil and ecosystem respiration across European forests. Glob. Chang. Biol. 2001, 7, 269–278. [Google Scholar] [CrossRef]
- Luo, Y.; Zhou, X. Soil Respiration and the Environment, 1st ed.; Academic Press: Sandiego, CA, USA, 2006; p. 328. [Google Scholar]
- Davidson, E.A.; Richardson, A.D.; Savage, K.E.; Hollinger, D.Y. A distinct seasonal pattern of the ratio of soil respiration to total ecosystem respiration in a sprucedominated forest. Glob. Chang. Biol. 2006, 12, 230–239. [Google Scholar] [CrossRef]
- Wang, X.; Piao, S.; Ciais, P.; Janssens, I.; Reichstein, M.; Peng, S.; Wang, T. Are ecological gradients in seasonal Q10 of soil respiration explained by climate or by vegetation seasonality? Soil Biol. Biochem. 2010, 42, 1728–1734. [Google Scholar] [CrossRef]
- Barba, J.; Cueva, A.; Bahn, M.; Barron-Gafford, G.A.; Bond-Lamberty, B.; Hanson, P.J.; Jaimes, A.; Kulmala, L.; Pumpanen, J.; Scott, R.L.; et al. Comparing ecosystem and soil respiration: Review and key challenges of tower-based and soil measurements. Agric. For. Meteorol. 2018, 249, 434–443. [Google Scholar] [CrossRef]
- Kirschbaum, M.; Eamus, D.; Gifford, R.; Roxburgh, S.; Sands, P. Definitions of some ecological terms commonly used in carbon accounting. In Proceedings of the Net Ecosystem Exchange CRC Workshop, Canberra, Australia, 18–20 April 2001; pp. 2–5. [Google Scholar]
- Reichstein, M.; Tenhunen, D.; Roupsard, O.; Ourcival, M.; Rambal, S.; Dore, S.; Valentin, R. Ecosystem respiration in two Mediterranean evergreen Holm Oak forests: Drought effects and decomposition dynamics. Funct. Ecol. 2002, 16, 27–39. [Google Scholar] [CrossRef]
- Aubinent, M.; Vesala, T.; Papale, D. Eddy Covariance: A Practical Guide to Measurement and Data Analysis; Springer Atmospheric Sciences: Dordrecht, The Netherlands, 2012; p. 438. [Google Scholar]
- Hill, T.; Chocholek, M.; Clement, R. The case for increasing the statistical power of eddy covariance ecosystem studies: Why, where and how? Glob. Chang. Biol. 2017, 23, 2154–2165. [Google Scholar] [CrossRef] [PubMed]
- Baldocchi, D.D. How eddy covariance flux measurements have contributed to our understanding of global change biology. Glob. Chang. Biol. 2020, 26, 242–260. [Google Scholar] [CrossRef]
- Guo, W.H.; Kang, S.Z.; Li, F.S.; Li, S.E. Variation of NEE and its affecting factors in a vineyard of arid region of northwest China. Atmos. Environ. 2014, 84, 349–354. [Google Scholar] [CrossRef]
- Pitacco, A.; Meggio, F. Carbon budget of the vineyard-A new feature of sustainability. In Proceedings of the 38th World Congress of Vine and Wine, Mainz, Germany, 5–10 July 2015. [Google Scholar]
- Chiriaco, M.V.; Belli, C.; Chiti, T.; Trotta, C.; Sabbatini, S. The potential carbon neutrality of sustainable viticulture showed through a comprehensive assessment of the greenhouse gas (GHG) budget of wine production. J. Clean. Prod. 2019, 225, 435–450. [Google Scholar] [CrossRef]
- Vendrame, N.; Tezza, L.; Pitacco, A. Study of the Carbon Budget of a Temperate-Climate Vineyard: Inter-Annual Variability of CO2 Flux. Am. J. Enol. Vitic. 2019, 70, 34–41. [Google Scholar] [CrossRef]
- Lazíková, J.; Rumanovská, Ľ.; Takáč, I.; Prus, P.; Fehér, A. Regional differences of agricultural land market in Slovakia: A challenge for sustainable agriculture. Agriculture 2021, 11, 353. [Google Scholar] [CrossRef]
- Nevidzany Village. The Nevidzany Economic and Social Development Programme. 2008. Available online: https://www.obecnevidzany.sk/files/2021-09-30-115322-PHSR_Nevidzany.pdf (accessed on 29 August 2023).
- Knauer, J.; El-Madany, T.S.; Zaehle, S.; Migliavacca, M. Bigleaf-An R package for the calculation of physical and physiological ecosystem properties from eddy covariance data. PLoS ONE 2018, 13, e0201114. [Google Scholar] [CrossRef]
- Wutzler, T.; Moffat, A.; Migliavacca, M.; Knauer, J.; Sickel, K.; Šigut, L.; Menzer, O.; Reichstein, M. Basic and extensible post-processing of eddy covariance flux data with REddyProc. Biogeosciences 2018, 15, 5015–5030. [Google Scholar] [CrossRef]
- Papale, D.; Reichstein, M.; Aubinet, M.; Canfora, E.; Bernhofer, C.; Kutsch, W.; Longdoz, B.; Rambal, S.; Valentini, R.; Vesala, T.; et al. Towards a standardized processing of Net Ecosystem Exchange measured with eddy covariance technique: Algorithms and uncertainty estimation. Biogeosciences 2006, 3, 571–583. [Google Scholar] [CrossRef]
- Goulden, M.; Munger, J.; Fan, S.M.; Daube, B.; Wofsy, S. Measurements of Carbon Sequestration by Long-Term Eddy Covariance: Methods and a Critical Evaluation of Accuracy. Glob. Chang. Biol. 1996, 2, 169–182. [Google Scholar] [CrossRef]
- Reichstein, M.; Falge, E.; Baldocchi, D.; Papale, D.; Aubinet, M.; Berbigier, P.; Bernhofer, C.; Buchmann, N.; Gilmanov, T.; Granier, A.; et al. On the separation of net ecosystem exchange into assimilation and ecosystem respiration: Review and improved algorithm. Glob. Chang. Biol. 2005, 11, 1424–1439. [Google Scholar] [CrossRef]
- Wilczak, J.M.; Oncley, S.P.; Stage, S.A. Sonic anemometer tilt correction algorithms. Bound.-Layer Meteorol. 2001, 99, 127–150. [Google Scholar] [CrossRef]
- Falge, E.; Baldocchi, D.; Olson, R.; Anthoni, P.; Aubinet, M.; Bernhofer, C.; Burba, G.; Ceulemans, R.; Clement, R.; Dolman, H.; et al. Gap filling strategies for defensible annual sums of net ecosystem exchange. Agric. For. Meteorol. 2001, 107, 43–69. [Google Scholar] [CrossRef]
- Pastorello, G.; Trotta, C.; Canfora, E.; Chu, H.; Christianson, D.; Cheah, Y.W.; Poindexter, C.; Chen, J.; Elbashandy, A.; Humphrey, M.; et al. The FLUXNET2015 dataset and the ONEFlux processing pipeline for eddy covariance data. Sci. Data 2020, 7, 225. [Google Scholar] [CrossRef]
- ADC BioScientific. User Manual Automated Soil CO2 Exchange System Reference; ADC BioScientific: Hoddesdon, UK, 2007. [Google Scholar]
- Gomez-Casanovas, N.; Anderson-Teixeira, K.; Zeri, M.; Bernacchi, C.J.; Delucia, E.H. Gap filling strategies and error in estimating annual soil respiration. Glob. Chang. Biol. 2013, 19, 1941–1952. [Google Scholar] [CrossRef]
- Van’t Hoff, J.H. Lectures on theoretical and physical chemistry. In Chemical Dynamics; Edward Arnold: London, UK, 1898; p. 148. [Google Scholar]
- Rodeghiero, M.; Cescatti, A. Main determinants of forest soil respiration along an elevation/temperature gradient in the Italian Alps. Glob. Chang. Biol. 2005, 11, 1024–1041. [Google Scholar] [CrossRef]
- Baty, F.; Ritz, C.; Charles, S.; Brutsche, M.; Flandrois, J.P.; Delignette-Muller, M.L. A Toolbox for Nonlinear Regression in R: The Package nls tools. J. Stat. Softw. 2015, 66, 1–21. [Google Scholar] [CrossRef]
- McKenzie, N.; Coughlan, K.; Cresswell, H. Soil Physical Measurement and Interpretation for Land Evaluation; CSIRO Publishing: Collingwood, VIC, Australia, 2002. [Google Scholar]
- Bieganowski, A.; Ryżak, M. Soil Texture: Measurement Methods. In Encyclopedia of Agrophysics; Encyclopedia of Earth Sciences Series; Gliński, J., Horabik, J., Lipiec, J., Eds.; Springer: Dordrecht, The Netherlands, 2011; pp. 791–794. [Google Scholar]
- Spasić, M.; Vacek, O.; Vejvodová, K.; Tejnecký, V.; Polák, F.; Borůvka, L.; Drábek, O. Determination of physical properties of undisturbed soil samples according to V. Novák. MethodsX 2023, 10, 102133. [Google Scholar] [CrossRef] [PubMed]
- Bremner, J. Determination of nitrogen in soil by the Kjeldahl method. J. Agric. Sci. 1960, 55, 11–33. [Google Scholar] [CrossRef]
- Walkley, A.; Black, I.A. An examination of the Degtjareff method for determining soil organic matter, and a proposed modification of thechromic acid titration method. Soil Sci. 1934, 37, 29–38. [Google Scholar] [CrossRef]
- Shapiro, S.S.; Wilk, M.B. Analysis of variance test for normality. Biometrika 1965, 52, 591–611. [Google Scholar] [CrossRef]
- Levene, H. Contributions to Probability and Statistics: Essays in Honor of Harold Hotelling; Stanford University Press: Palo Alto, CA, USA, 1960; pp. 278–292. [Google Scholar]
- R Development Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2021; Available online: https://www.R-project.org/ (accessed on 18 May 2021).
- Ward, R.B.; Gentile, R.M.; Laubach, J.; Hunt, J.E.; McMillan, A.M.S. Assessment of the carbon and water balances of Sauvignon blanc grapes using eddy covariance. In Nutrient Management in Farmed Landscapes; Occasional Report No. 33; Christensen, C.L., Horne, D.J., Singh, R., Eds.; Farmed Landscapes Research Centre, Massey University: Palmerston North, New Zealand, 2020; p. 11. Available online: http://flrc.massey.ac.nz/publications.html (accessed on 29 August 2023).
- Allaire, S.E.; Dufour-L’Arrivée, C.; Lafond, J.A.; Lalancette, R.; Brodeur, J. Carbon dioxide emissions by urban turfgrass areas. Can. J. Soil Sci. 2008, 88, 529–532. [Google Scholar] [CrossRef]
- Verhoeven, E.; Pereira, E.; Decock, C.; Garland, G.; Kennedy, T.; Suddick, E.; Horwath, W.; Six, J. N2O emissions from California farmlands: A review. Calif. Agric. 2017, 71, 148–159. [Google Scholar] [CrossRef]
- Wagle, P.; Kakani, V.G. Confounding Effects of Soil Moisture on the Relationship Between Ecosystem Respiration and Soil Temperature in Switchgrass. Bioenerg. Res. 2014, 7, 789–798. [Google Scholar] [CrossRef]
- Sarma, D.; Burman, P.K.D.; Chakraborty, S.; Gogoi, N.; Bora, A.; Metya, A.; Datye, A.; Murkute, C.; Karipot, A. Quantifying the net ecosystem exchange at a semi-deciduous forest in northeast India from intra-seasonal to the seasonal time scale. Agric. For. Meteorol. 2022, 314, 108786. [Google Scholar] [CrossRef]
- Reichstein, M.; Bahn, M.; Ciais, P.; Frank, D.; Mahecha, M.D.; Seneviratne, S.I.; Zscheischler, J.; Beer, C.; Buchmann, N.; Frank, D.C.; et al. Climate extremes and the carbon cycle. Nature 2013, 500, 287–295. [Google Scholar] [CrossRef]
- Grossiord, C.; Buckley, T.N.; Cernusak, L.A.; Novick, K.A.; Poulter, B.; Siegwolf, R.T.; Sperry, J.S.; McDowell, N.G. Plant responses to rising vapor pressure deficit. New Phytol. 2020, 226, 1550–1566. [Google Scholar] [CrossRef] [PubMed]
- Reichstein, M.; Ciais, P.; Papale, D.; Valentini, R.; Running, S.; Viovy, N. Reduction of ecosystem productivity and respiration during the European summer 2003 climate anomaly: A joint flux tower, remote sensing and modelling analysis. Glob. Chang. Biol. 2007, 13, 634–651. [Google Scholar] [CrossRef]
- Konings, A.G.; Williams, A.P.; Gentine, P. Sensitivity of grassland productivity to aridity controlled by stomatal and xylem regulation. Nat. Geosci. 2017, 10, 284–288. [Google Scholar] [CrossRef]
- Yu, T.; Jiapaer, G.; Bao, A.; Zheng, G.; Zhang, J.; Li, X.; Yuan, Y.; Huang, X.; Umuhoza, J. Disentangling the relative effects of soil moisture and vapor pressure deficit on photosynthesis in dryland Central Asia. Ecol. Indic. 2022, 137, 108698. [Google Scholar] [CrossRef]
- Larsen, K.S.; Ibrom, A.; Beier, C.; Jonasson, S.; Michelsen, A. Ecosystem respiration depends strongly onphotosynthesis in a temperate heath. Biogeochemistry 2007, 85, 201–213. [Google Scholar] [CrossRef]
- Wolf, A.; Anderegg, W.R.L.; Pacala, S.W. Optimal stomatal behavior with competition for water and risk of hydraulic impairment. Proc. Natl. Acad. Sci. USA 2016, 113, E7222–E7230. [Google Scholar] [CrossRef] [PubMed]
- Davidson, E.; Belk, E.; Boone, R.D. Soil water content and temperature as independent or confounded factors controlling soil respiration in a temperate mixed hardwood forest. Glob. Chang. Biol. 1998, 4, 217–227. [Google Scholar] [CrossRef]
- Qi, Y.; Xu, M. Separating the effects of moisture and temperature on soil CO2 efflux in a coniferous forest in the Sierra Nevada mountains. Plant Soil 2001, 237, 15–23. [Google Scholar] [CrossRef]
- Jia, X.; Mu, Y.; Zha, T.; Wang, B.; Qin, S.; Tian, Y. Seasonal and interannual variations in ecosystem respiration in relation to temperature, moisture, and productivity in a temperate semi-arid shrubland. Sci. Total Environ. 2020, 709, 136210. [Google Scholar] [CrossRef] [PubMed]
- Franck, F.; Morales, J.P.; Arancibia-Avendaño, D.; de Cortázar, V.G.; Perez-Quezada, J.F.; Zurita-Silva, A.; Pastenes, C. Seasonal fluctuations in vitis vinifera root respiration in the field. New Phytol. 2011, 192, 939–951. [Google Scholar] [CrossRef]
- Ceccon, C.; Panzacchi, P.; Scandellari, F.; Prandi, L.; Ventura, M.; Russo, B.; Tagliavini, M. Spatial and temporal effects of soil temperature and moisture and the relation to fine root density on root and soil respiration in a mature apple orchard. Plant Soil 2010, 342, 195–206. [Google Scholar] [CrossRef]
- Wu, D.; Liu, S.; Wu, X.; Yang, X.; Xu, T.; Xu, Z.; Shi, H. Diagnosing the temperature sensitivity of ecosystem respiration in northern high-latitude regions. J. Geophys. Res. Biogeosci. 2021, 126, e2020JG005998. [Google Scholar] [CrossRef]
- Gudasz, G.; Karlsson, J.; Bastviken, D. When does temperature matter for ecosystem respiration? Environ. Res. Commun. 2021, 3, 121001. [Google Scholar] [CrossRef]
- Ma, M.; Zang, Z.; Xie, Z.; Chen, Q.; Xu, W.; Zhao, C.; Shen, G. Soil respiration of four forests along elevation gradient in northern subtropical China. Ecol. Evol. 2019, 9, 12846–12857. [Google Scholar] [CrossRef]
- Zhao, P.; Pumpanen, J.; Kang, S. Spatio-temporal variability and controls of soil respiration in a furrow-irrigated vineyard. Soil Tillage Res. 2020, 196, 104424. [Google Scholar] [CrossRef]
- Lardo, E.; Palese, A.M.; Nuzzo, V.; Xiloyannis, C.; Celano, G. Variability of total soil respiration in a Mediterranean vineyard. Arid. Soil Res. Rehabil. 2015, 53, 531–541. [Google Scholar] [CrossRef]
- Escalona, J.M.; Tomás, M.; Martorell, S.; Medrano, H.; Ribas-Carbó, M.; Flexas, J. Carbon balance in grapevines under different soil water supply: Importance of whole plant respiration. Aust. J. Grape Wine Res. 2012, 18, 308–318. [Google Scholar] [CrossRef]
- Lohila, A.; Aurela, M.; Regina, K.; Laurila, T. Soil and total ecosystem respiration in agricultural fields: Effect of soil and crop type. Plant Soil 2003, 251, 303–317. [Google Scholar] [CrossRef]
- Reichstein, M.; Rey, A.; Freibauer, A.; Tenhunen, J.; Valentini, R.; Banza, R. Modeling temporal and largescale spatial variability of soil respiration from soil water availability, temperature and vegetation productivity indices. Glob. Biogeochem. Cycles 2003, 17, 1104. [Google Scholar] [CrossRef]
- Chen, Q.S.; Wang, X.B.; Han, X.G.; Wan, S.Q.; Li, L.H. Temporal and spatial variability and controls of soil respiration in a temperate steppe in northern China. Glob. Biogeochem. Cycles 2010, 24, GB2010. [Google Scholar] [CrossRef]
- Luo, J.; Chen, Y.C.; Wu, Y.H.; Shi, P.L.; She, J.; Zhou, P. Respiration in different primary succession stages on glacier forehead in Gongga mountain, China. PLoS ONE 2012, 7, e42354. [Google Scholar] [CrossRef] [PubMed]
- Oertel, C.; Matschullat, J.; Zurba, K.; Zimmermann, F.; Erasmi, S. Greenhouse gas emissions from soil—A review. Geochemistry 2016, 76, 327–352. [Google Scholar] [CrossRef]
- Irvine, J.; Law, B.E. Contrasting soil respiration in young and old-growth ponderosa pine forests. Glob. Chang. Biol. 2002, 8, 1183–1194. [Google Scholar] [CrossRef]
- Zhao, X.; Li, F.; Zhang, W.; Ai, Z.; Shen, H.; Liu, X.; Cao, J.; Manevski, K. Soil Respiration at Different Stand Ages (5, 10, and 20/30 Years) in Coniferous (Pinus tabulaeformis Carrière) and Deciduous (Populus davidiana Dode) Plantations in a Sandstorm Source Area. Forests 2016, 7, 153. [Google Scholar] [CrossRef]
- Yan, M.; Zhang, X.; Zhou, G.; Gong, J.; You, X. Temporal and spatial variation in soil respiration of poplar plantations at different developmental stages in Xinjiang, China. J. Arid. Environ. 2011, 75, 51–57. [Google Scholar] [CrossRef]
- Gong, J.R.; Ge, Z.W.; An, R.; Duan, Q.W.; You, X.; Huang, Y.M. Soil respiration in poplar plantations in northern China at different forest ages. Plant Soil 2012, 360, 109–122. [Google Scholar] [CrossRef]




| Soil C (kg C m−2) | Soil N (kg N m−2) | Soil pH | BD (g cm−3) | Soil Porosity (%) | Soil Water Retention Capacity (%) | |
|---|---|---|---|---|---|---|
| Mean | 3.04 | 0.23 | 6.31 | 1.32 | 47 | 29 |
| Standard deviation (SD) | 0.41 | 0.06 | 0.20 | 0.06 | 4 | 3 |
| Parameters | Flux | April | May | June | July | August | September |
|---|---|---|---|---|---|---|---|
| Q10 | Rsoil | 3.24 | 2.41 | 2.36 | 2.54 | 1.88 | 2.98 |
| Reco | 3.20 | 1.30 | 1.32 | 1.63 | 1.35 | 3.16 | |
| Rref | Rsoil | 0.70 | 0.75 | 0.67 | 0.60 | 0.98 | 0.64 |
| Reco | 3.96 | 5.80 | 4.80 | 2.40 | 4.65 | 2.01 | |
| RAG | 2.03 ± 0.46 | 4.01 ± 0.11 | 1.51 ± 0.54 | 0.90 ± 0.06 | 1.94 ± 0.47 | 2.62 ± 0.47 | |
| Rsoil fraction to Reco (%) | 27 | 30 | 59 | 70 | 52 | 29 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Badraghi, A.; Novotná, B.; Frouz, J.; Krištof, K.; Trakovický, M.; Juriga, M.; Chvila, B.; Montagnani, L. Temporal Dynamics of CO2 Fluxes over a Non-Irrigated Vineyard. Land 2023, 12, 1925. https://doi.org/10.3390/land12101925
Badraghi A, Novotná B, Frouz J, Krištof K, Trakovický M, Juriga M, Chvila B, Montagnani L. Temporal Dynamics of CO2 Fluxes over a Non-Irrigated Vineyard. Land. 2023; 12(10):1925. https://doi.org/10.3390/land12101925
Chicago/Turabian StyleBadraghi, Aysan, Beáta Novotná, Jan Frouz, Koloman Krištof, Martin Trakovický, Martin Juriga, Branislav Chvila, and Leonardo Montagnani. 2023. "Temporal Dynamics of CO2 Fluxes over a Non-Irrigated Vineyard" Land 12, no. 10: 1925. https://doi.org/10.3390/land12101925
APA StyleBadraghi, A., Novotná, B., Frouz, J., Krištof, K., Trakovický, M., Juriga, M., Chvila, B., & Montagnani, L. (2023). Temporal Dynamics of CO2 Fluxes over a Non-Irrigated Vineyard. Land, 12(10), 1925. https://doi.org/10.3390/land12101925







