Structural Assessment of Fluorine, Chlorine, Bromine, Iodine, and Hydroxide Substitutions in Lead Arsenate Apatites (Mimetites)–Pb5(AsO4)3X
Abstract
:1. Introduction
2. Materials and Methods
2.1. Synthesis
2.2. Analytical Methods
3. Results
3.1. Preliminary Characteristics of the Synthesized Products
3.2. Rietveld Refinement from High-Resolution Synchrotron Data
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Elliott, J.C.; Wilson, R.M.; Dowker, S.E.P. Apatite structures. Adv. X Ray Anal. 2002, 45, 172–181. [Google Scholar]
- White, T.J.; Dong, Z. Structural derivation and crystal chemistry of apatites. Acta Cryst. B 2003, 59, 1–16. [Google Scholar] [CrossRef]
- White, T.; Ferraris, C.; Kim, J.; Madhavi, S. Apatite–An adaptive framework structure. Rev. Miner. Geochem. 2005, 57, 307–401. [Google Scholar] [CrossRef]
- Pasero, M.; Kampf, A.R.; Ferraris, C.; Pekov, I.V.; Rakovan, J.; White, T.J. Nomenclature of the apatite supergroup minerals. Eur. J. Miner. 2010, 22, 163–179. [Google Scholar] [CrossRef]
- Ptáček, P. Apatites and Their Synthetic Analogues—Synthesis, Structure, Properties and Applications; InTech: Rijeka, Croatia, 2016. [Google Scholar] [CrossRef]
- Elliott, J.C. Structure and Chemistry of the Apatites and Other Calcium Phosphates; Elsevier: Amsterdam, The Netherlands, 1994; ISBN 0-444-81582-1. [Google Scholar]
- Hughes, J.M.; Rakovan, J.F. Structurally robust, chemically diverse: Apatite and apatite supergroup minerals. Elements 2015, 11, 165–170. [Google Scholar] [CrossRef]
- Mackie, P.E.; Elliot, J.C.; Young, R.A. Monoclinic structure of synthetic Ca5(PO4)3Cl, chlorapatite. Acta Cryst. B 1972, 28, 1840–1848. [Google Scholar] [CrossRef]
- Hata, M.; Marumo, F.; Iwai, S.; Aoki, H. Structure of barium chlorapatite. Acta Cryst. B 1979, 35, 2382–2384. [Google Scholar] [CrossRef]
- Hashimoto, H.; Matsumoto, T. Structure refinements of two natural pyromorphites, Pb5(PO4)3Cl, and crystal chemistry of chloroapatite group, M5(PO4)3Cl. Z. Kristallogr. 1998, 213, 585–590. [Google Scholar] [CrossRef]
- Kim, J.Y.; Fenton, R.R.; Hunter, B.A.; Kennedy, B.J. Powder diffraction studies of synthetic calcium and lead apatites. Aust. J. Chem. 2000, 53, 679–686. [Google Scholar] [CrossRef]
- Audubert, F.; Savariault, J.M.; Lacout, J.L. Pentalead tris (vanadate) iodide, a defect vanadinite-type compound. Acta Cryst. C 1999, 55, 271–273. [Google Scholar] [CrossRef]
- Zhang, M.; Maddrell, E.R.; Abraitis, P.K.; Salje, E.K.H. Impact of leach on lead vanado-iodoapatite [Pb5(VO4)3I]: An infrared and Raman spectroscopic study. Mat. Sci. Eng. B Adv. 2007, 137, 149–155. [Google Scholar] [CrossRef]
- Stennett, M.C.; Pinnock, I.J.; Hyatt, N.C. Rapid synthesis of Pb5(VO4)3I, for the immobilisation of iodine radioisotopes, by microwave dielectric heating. J. Nucl. Mater. 2011, 414, 352–359. [Google Scholar] [CrossRef]
- Suetsugu, Y. Synthesis of lead vanadate iodoapatite utilizing dry mechanochemical process. J. Nucl. 2014, 454, 223–229. [Google Scholar] [CrossRef]
- Merker, L.; Wondratschek, H. Bleiverbindungen mit Apatitstruktur, insbesondere Blei–Jod–und Blei–Brom-Apatite. Z. Anorg. Allg. Chem. 1959, 300, 41–50. [Google Scholar] [CrossRef]
- Henderson, C.M.B.; Bell, A.M.T.; Charnock, J.M.; Knight, K.S.; Wendlandt, R.F.; Plant, D.A.; Harrison, W.J. Synchrotron X-ray absorption spectroscopy and X-ray powder diffraction studies of the structure of johnbaumite [Ca10(AsO4)6(OH,F)2] and synthetic Pb-, Sr- and Ba-arsenate apatites and some comments on the crystal chemistry of the apatite structure type in general. Miner. Mag. 2009, 73, 433–455. [Google Scholar] [CrossRef]
- Epp, T.; Marks, M.A.; Ludwig, T.; Kendrick, M.A.; Eby, N.; Neidhardt, H.; Oelmann, Y.; Markl, G. Crystallographic and fluid compositional effects on the halogen (Cl, F, Br, I) incorporation in pyromorphite-group minerals. Am. Miner. 2019, 104, 1673–1688. [Google Scholar] [CrossRef]
- Wu, P.; Zeng, Y.Z.; Wang, C.M. Prediction of apatite lattice constants from their constituent elemental radii and artificial intelligence methods. Biomaterials 2004, 25, 1123–1130. [Google Scholar] [CrossRef]
- Markl, G.; Marks, M.A.; Holzäpfel, J.; Wenzel, T. Major, minor, and trace element composition of pyromorphite-group minerals as recorder of supergene weathering processes from the Schwarzwald mining district, SW Germany. Am. Miner. 2014, 99, 1133–1146. [Google Scholar] [CrossRef]
- Manecki, M.; Maurice, P.A.; Traina, S.J. Uptake of aqueous Pb by Cl−, F−, and OH− apatites: Mineralogic evidence for nucleation mechanisms. Am. Miner. 2000, 85, 932–942. [Google Scholar] [CrossRef]
- Bajda, T. Solubility of mimetite Pb5(AsO4)3Cl at 5–55 °C. Env. Chem. 2010, 7, 268–278. [Google Scholar] [CrossRef]
- Borkiewicz, O.; Rakovan, J.; Cahill, C.L. Time-resolved in situ studies of apatite formation in aqueous solutions. Am. Miner. 2010, 95, 1224–1236. [Google Scholar] [CrossRef]
- Flis, J.; Borkiewicz, O.; Bajda, T.; Manecki, M.; Klasa, J. Synchrotron-based X-ray diffraction of the lead apatite series Pb10(PO4)6Cl2–Pb10(AsO4)6Cl2. J. Synchrotron Radiat. 2010, 17, 207–214. [Google Scholar] [CrossRef] [PubMed]
- Bajda, T.; Mozgawa, W.; Manecki, M.; Flis, J. Vibrational spectroscopic study of mimetite–pyromorphite solid solutions. Polyhedron 2011, 30, 2479–2485. [Google Scholar] [CrossRef]
- Janicka, U.; Bajda, T.; Manecki, M. Synthesis and solubility of brompyromorphite Pb5(PO4)3Br. Miner. Pol. 2012, 43, 129–135. [Google Scholar] [CrossRef]
- Kwaśniak-Kominek, M.; Matusik, J.; Bajda, T.; Manecki, M.; Rakovan, J.; Marchlewski, T.; Szala, B. Fourier transform infrared spectroscopic study of hydroxylpyromorphite Pb10(PO4)6OH2 –hydroxylmimetite Pb10(AsO4)6(OH)2 solid solution series. Polyhedron 2015, 99, 103–111. [Google Scholar] [CrossRef] [Green Version]
- Puzio, B.; Manecki, M.; Kwaśniak-Kominek, M. Transition from Endothermic to Exothermic Dissolution of Hydroxyapatite Ca5(PO4)3OH–Johnbaumite Ca5(AsO4)3OH Solid Solution Series at Temperatures Ranging from 5 to 65 °C. Minerals 2018, 8, 281. [Google Scholar] [CrossRef] [Green Version]
- Ma, Q.Y.; Traina, S.J.; Logan, T.J.; Ryan, J.A. In situ lead immobilization by apatite. Env. Sci. Technol. 1993, 27, 1803–1810. [Google Scholar] [CrossRef]
- Xu, Y.; Schwartz, F.W. Lead immobilization by hydroxyapatite in aqueous solutions. J. Contam. Hydrol. 1994, 15, 187–206. [Google Scholar] [CrossRef]
- Ma, Q.Y.; Logan, T.J.; Traina, S.J. Lead immobilization from aqueous solutions and contaminated soils using phosphate rocks. Env. Sci. Technol. 1995, 29, 1118–1126. [Google Scholar] [CrossRef]
- Ma, L.Q. Factors influencing the effectiveness and stability of aqueous lead immobilization by hydroxyapatite. J. Env. Qual. 1996, 25, 1420–1429. [Google Scholar] [CrossRef]
- Laperche, V.; Traina, S.J.; Gaddam, P.; Logan, T.J. Chemical and mineralogical characterizations of Pb in a contaminated soil: Reactions with synthetic apatite. Env. Sci. Technol. 1996, 30, 3321–3326. [Google Scholar] [CrossRef]
- Laperche, V.; Logan, T.J.; Gaddam, P.; Traina, S.J. Effect of apatite amendments on plant uptake of lead from contaminated soil. Env. Sci. Technol. 1997, 31, 2745–2753. [Google Scholar] [CrossRef]
- Kumpiene, J.; Lagerkvist, A.; Maurice, C. Stabilization of As, Cr, Cu, Pb and Zn in soil using amendments–a review. Waste Manag. 2008, 28, 215–225. [Google Scholar] [CrossRef] [PubMed]
- Liang, Y.; Cao, X.; Zhao, L.; Arellano, E. Biochar- and phosphate-induced immobilization of heavy metals in contaminated soil and water: Implication on simultaneous remediation of contaminated soil and groundwater. Env. Sci. Pollut. 2014, 21, 4665–4674. [Google Scholar] [CrossRef] [PubMed]
- Zeng, G.; Wan, J.; Huang, D.; Hu, L.; Huang, C.; Cheng, M.; Xue, W.; Gong, X.; Wang, R.; Jiang, D. Precipitation, adsorption and rhizosphere effect: The mechanisms for phosphate-induced Pb immobilization in soils—a review. J. Hazard. Mater. 2017, 339, 354–367. [Google Scholar] [CrossRef]
- Manecki, M. Lead in Water and Soil: Speciation, Toxicity, and Treatment Technologies. In Encyclopedia of Water: Science, Technology, and Society; Maurice, P.A., Ed.; Willey: Hoboken, NJ, USA, 2020; pp. 1713–1727. ISBN 978-1-119-30075-5. [Google Scholar]
- Harrison, W.J.; Wendlandt, R.F.; Wendlandt, A.E. Low temperature aqueous solubility and stability of apatite-structure arsenates of lead, barium, and strontium and uptake of arsenic by hydroxylapatite. In Proceedings of the 18th General Meeting of the International Mineralogical Association, Edinburgh, Scotland, 1–6 September 2002; p. 185, Programme with Abstracts, Abstract A18-10. [Google Scholar]
- Wendlandt, A.E.; Harrison, W.J.; Wendlandt, R.F. Investigation of hydroxylapatite as a means of removing dissolved arsenic from potable water. In Proceedings of the Geological Society of America Annual Meeting, Denver, CO, USA, 27–31 October 2002. [Google Scholar]
- Harrison, W.J.; Wendlandt, R.F.; Charnock, J.M.; Henderson, C.M.B. Spectroscopic investigations of the adsorption of As onto bovine bone. In Geochimica et Cosmochimica Acta, 69, Proceedings of the 15th Annual Goldschmidt Conference, Moscow, ID, USA, 21–25 May 2005; Elsvier: Amsterdam, The Netherlands, 2005; p. A65. [Google Scholar]
- Bundschuh, J.; Holländer, H.M.; Ma, Q. In-Situ Remediation of Arsenic-Contaminated Sites; CRC Press: Boca Raton, FL, USA, 2017; p. 208. ISBN 9781138747753. [Google Scholar]
- Rakovan, J.F.; Hughes, J.M. Strontium in the apatite structure: Strontian fluorapatite and belovite-(Ce). Can. Miner. 2000, 38, 839–845. [Google Scholar] [CrossRef]
- Ewing, R.C. The design and evaluation of nuclear-waste forms: Clues from mineralogy. Can. Miner. 2001, 39, 697–715. [Google Scholar] [CrossRef] [Green Version]
- Pan, Y.; Fleet, M.E. Compositions of the apatite-group minerals: Substitution mechanisms and controlling factors. Rev. Miner. Geochem. 2002, 48, 13–49. [Google Scholar] [CrossRef]
- Rakovan, J.; Reeder, R.J.; Elzinga, E.J.; Cherniak, D.J.; Tait, C.D.; Morris, D.E. Structural characterization of U(VI) in apatite by X-ray absorption spectroscopy. Env. Sci. Technol. 2002, 36, 3114–3117. [Google Scholar] [CrossRef]
- Luo, Y.; Hughes, J.M.; Rakovan, J.; Pan, Y. Site preference of U and Th in Cl, F, and Sr apatites. Am. Miner. 2009, 94, 345–351. [Google Scholar] [CrossRef]
- Audubert, F.; Carpena, J.; Lacout, J.L.; Tetard, F. Elaboration of an iodine-bearing apatite iodine diffusion into a Pb3(VO4)2 matrix. Solid State Ion. 1997, 95, 113–119. [Google Scholar] [CrossRef]
- Uno, M.; Kosuga, A.; Masuo, S.; Imamura, M.; Yamanaka, S. Thermal and mechanical properties of AgPb9(VO4)6I and AgBa9(VO4)6I. J. Alloy. Compd. 2004, 384, 300–302. [Google Scholar] [CrossRef]
- Campayo, L.; Grandjean, A.; Coulon, A.; Delorme, R.; Vantelon, D.; Laurencin, D. Incorporation of iodates into hydroxyapatites: A new approach for the confinement of radioactive iodine. J. Mater. Chem. 2011, 21, 17609–17611. [Google Scholar] [CrossRef]
- Yao, T.; Lu, F.; Sun, H.; Wang, J.; Ewing, R.C.; Lian, J. Bulk iodoapatite ceramic densified by spark plasma sintering with exceptional thermal stability. J. Am. Ceram. Soc. 2014, 97, 2409–2412. [Google Scholar] [CrossRef]
- Mercier, P.H.; Le Page, Y.; Whitfield, P.S.; Mitchell, L.D.; Davidson, I.J.; White, T.J. Geometrical parameterization of the crystal chemistry of P63/m apatites: Comparison with experimental data and ab initio results. Acta Cryst. B 2005, 61, 635–655. [Google Scholar] [CrossRef] [Green Version]
- Vegas, A.; Jansen, M. Structural relationships between cations and alloys; an equivalence between oxidation and pressure. Acta Cryst. B 2002, 58, 38–51. [Google Scholar] [CrossRef]
- Mercier, P.H.; Le Page, Y.; Whitfield, P.S.; Mitchell, L.D. Geometrical parameterization of the crystal chemistry of P63/m apatite. II. Precision, accuracy and numerical stability of the crystal-chemical Rietveld refinement. J. Appl. Cryst. 2006, 39, 369–375. [Google Scholar] [CrossRef] [Green Version]
- Baker, W.E. An X-ray diffraction study of synthetic members of the pyromorphite series. Am. Miner. 1966, 51, 1712–1721. [Google Scholar]
- Marciniak, H.; Diduszko, R.; Kozak, M. XRAYAN Program do Rentgenowskiej Analizy Fazowej, Wersja 4.0.1; Koma: Warszawa, Poland, 2006. (In Polish) [Google Scholar]
- Holland, T.J.B.; Redfern, S.A.T. UNITCELL: A nonlinear least-squares program for cell-parameter refinement and implementing regression and deletion diagnostics. J. App. Cryst. 1997, 30, 84. [Google Scholar] [CrossRef] [Green Version]
- Larson, A.C.; Von Dreele, R.B. General Structure Analysis System; Report LAUR 86-748; Los Alamos National Laboratory: Los Almos, NM, USA, 1994.
- Toby, B.H. EXPGUI, a graphical user interface for GSAS. J. Appl. Cryst. 2001, 34, 210–213. [Google Scholar] [CrossRef] [Green Version]
- Momma, K.; Izumi, F. VESTA 3 for three-dimensional visualization of crystal, volumetric and morphology data. J. Appl. Cryst. 2011, 44, 1272–1276. [Google Scholar] [CrossRef]
- Young, R.A. The Rietveld Method; Oxford University Press: New York, NY, USA, 1993; p. 298. [Google Scholar]
- Dai, Y.; Hughes, J.M. Crystal structure refinements of vanadinite and pyromorphite. Can. Miner. 1989, 27, 189–192. [Google Scholar]
- Baikie, T.; Ferraris, C.; Klooster, W.T.; Madhavi, S.; Pramana, S.S.; Pring, A.; Schmidt, G.; White, T.J. Crystal chemistry of mimetite, Pb10(AsO4)6Cl1.48O0. 26, and finnemanite, Pb10(AsO3)6Cl2. Acta Cryst. B 2008, 64, 34–41. [Google Scholar] [CrossRef] [PubMed]
- Dai, Y.; Hughes, J.M.; Moore, P.B. The crystal structure of mimetite and clinomimetite, Pb5(AsO4)3Cl. Can. Miner. 1991, 29, 369–376. [Google Scholar]
- Baikie, T.; Schreyer, M.; Wei, F.; Herrin, J.S.; Ferraris, C.; Brink, F.; Topolska, J.; Piltz, R.O.; Price, J.; White, T.J. The influence of stereochemically active lone-pair electrons on crystal symmetry and twist angles in lead apatite-2H type structures. Miner. Mag. 2014, 78, 325–345. [Google Scholar] [CrossRef]
- Shannon, R.D. Revised effective ionic radii and systematic studies of interatomic distances in halides and chalcogenides. Acta Cryst. A 1976, 32, 751–767. [Google Scholar] [CrossRef]
- Kreidler, E.R.; Hummel, F.A. The crystal chemistry of apatite: Structure fields of fluor- and chlorapatite. Am. Miner. 1970, 55, 170–184. [Google Scholar]
- Lee, Y.J.; Stephens, P.W.; Tang, Y.; Li, W.; Phillips, B.L.; Parise, J.B.; Reeder, R.J. Arsenate substitution in hydroxylapatite: Structural characterization of the Ca5(PxAs1–xO4)3OH solid solution. Am. Miner. 2009, 94, 666–675. [Google Scholar] [CrossRef]
- Sudarsanan, K.; Young, R.A. Structure of strontium hydroxide phosphate, Sr5(PO4)3OH. Acta Cryst. B 1972, 28, 3668–3670. [Google Scholar] [CrossRef]
- Corker, D.L.; Chai, B.H.T.; Nicholls, J.O.H.N.; Loutts, G.B. Neodymium-Doped Sr5(PO4)3F and Sr5(VO4)3F. Acta Cryst. C 1995, 51, 549–551. [Google Scholar] [CrossRef]
- Nötzold, D.; Wulff, H.; Herzog, G. Differenzthermoanalyse der Bildung des Pentastrontiumchloridphosphats und röntgenographische Untersuchung seiner Struktur. J. Alloys Compd. 1994, 215, 281–288. [Google Scholar] [CrossRef]
- Alberius-Henning, P.; Mattsson, C.; Lidin, S. Crystal structure of pentastrontium tris(phosphate) bromide, Sr5(PO4)3Br and of pentabarium tris(phosphate) bromide Ba5(PO4)3Br, two bromoapatites. Z. Krist. Ncs 2000, 215, 345–346. [Google Scholar] [CrossRef]
- Đordević, T.; Šutović, S.; Stojanović, J.; Karanović, L. Sr, Ba and Cd arsenates with the apatite-type structure. Acta Cryst. C 2008, 64, i82–i86. [Google Scholar] [CrossRef]
- Calos, N.J.; Kennard, C.H.; Davis, R.L. Crystal structure of mimetite, Pb5(AsO4)3Cl. Z. Krist. Cryst. Mater. 1990, 191, 125–130. [Google Scholar] [CrossRef]
- Okudera, H. Relationships among channel topology and atomic displacements in the structures of Pb5(BO4)3Cl with B = P (pyromorphite), V (vanadinite), and As (mimetite). Am. Miner. 2013, 98, 1573–1579. [Google Scholar] [CrossRef]






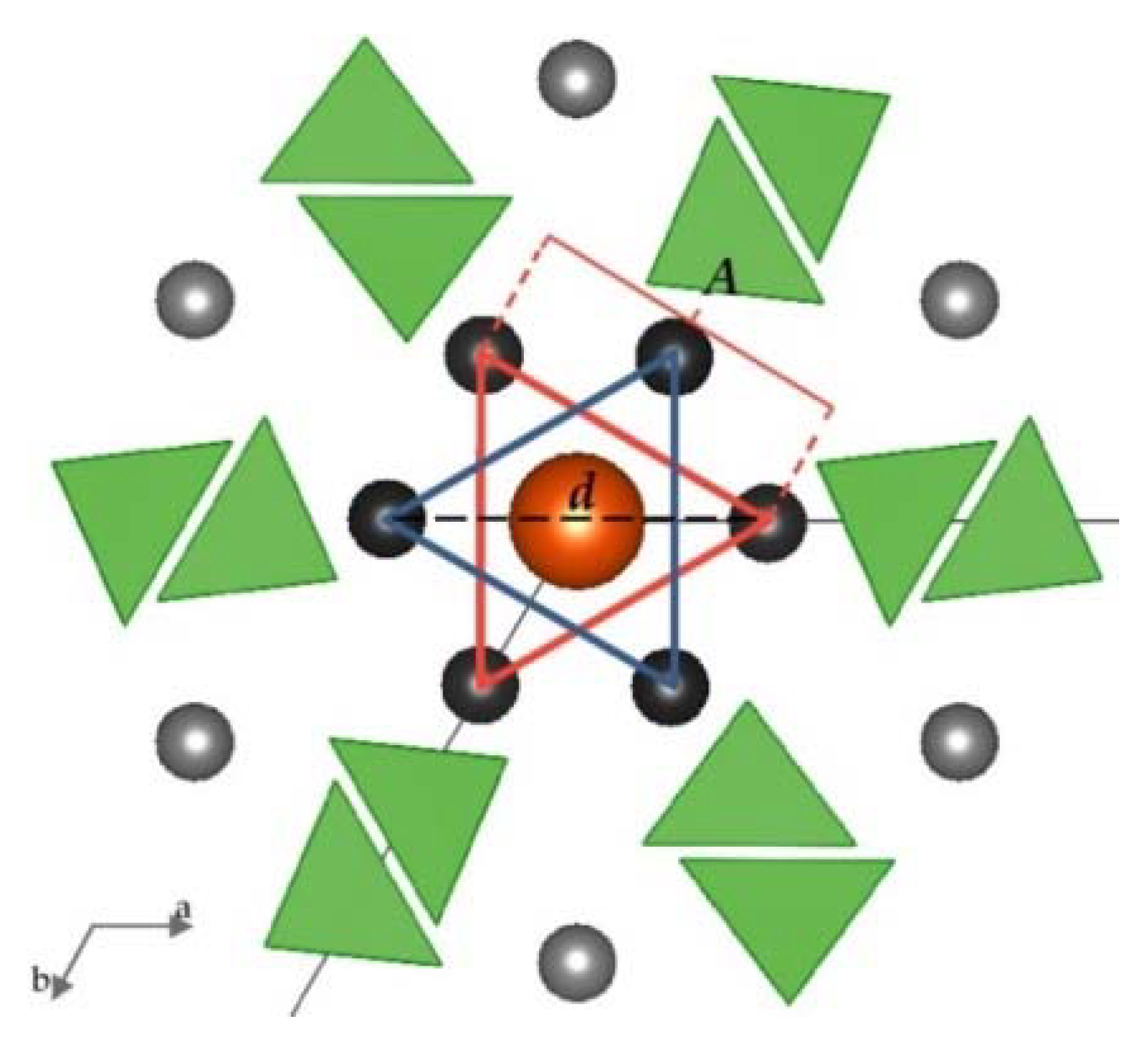


| Intended Chemical Formula | Empirical Chemical Formula | Sample Name |
|---|---|---|
| Pb5(AsO4)3F | Pb5.00(AsO4)2.99F0.98 | mimetite-F |
| Pb5(AsO4)3Cl | Pb5.00(AsO4)2.99Cl1.00 | mimetite-Cl |
| Pb5(AsO4)3Br | Pb5.00(AsO4)2.98Br0.97 | mimetite-Br |
| Pb5(AsO4)3I | Pb5.00(AsO4)3.03I0.60 | mimetite-I |
| Pb5(AsO4)3OH | Pb5.00(AsO4)3.01OH1.00 | mimetite-OH |
| Atom | Parameters | X = F | X = Cl | X = Br | X = I | X = OH | |
|---|---|---|---|---|---|---|---|
| a [Å] | 10.081 | 10.249 | 10.312 | 10.353 | 10.187 | ||
| c [Å] | 7.426 | 7.443 | 7.474 | 7.530 | 7.523 | ||
| V [Å3] | 653.619(8)a | 677.00(4) | 688.320(19) | 699.125(24) | 676.303(13) | ||
| rX [Å]b | 1.33 | 1.81 | 1.96 | 2.20 | 1.37 | ||
| Av CR [Å]c | 1.21 | 1.23 | 1.24 | 1.25 | 1.21 | ||
| Pb(1) | x | 1/3 | 1/3 | 1/3 | 1/3 | 1/3 | |
| y | 2/3 | 2/3 | 2/3 | 2/3 | 2/3 | ||
| z | 0.00463(14) | 0.00506 | 0.00577(34) | 0.00706(27) | 0.00357 | ||
| Uiso [Å2] | 0.01415(10) | 0.01967 | 0.02438 | 0.02871 | 0.02292 | ||
| Pb(2) | x | 0.23638(5) | 0.25085 | 0.25530(12) | 0.25888(12) | 0.24762 | |
| y | 1.00391(9) | 1.00362 | 1.00160(20) | 0.99753(19) | 0.99940 | ||
| z | 1/4 | 1/4 | 1/4 | 1/4 | 1/4 | ||
| Uiso [Å2] | 0.02296(10) | 0.01971 | 0.02286 | 0.03552 | 0.03163 | ||
| As | x | 0.40395(11) | 0.40714 | 0.40896(20) | 0.40491(21) | 0.40060 | |
| y | 0.38267(10) | 0.38331 | 0.37815(17) | 0.380001(17) | 0.37689 | ||
| z | 1/4 | 1/4 | 1/4 | 1/4 | 1/4 | ||
| Uiso [Å2] | 0.00984(23) | 0.00883 | 0.00838 | 0.01515 | 0.01508 | ||
| O(1) | x | 0.3208(6) | 0.32833 | 0.34000h | 0.3567(18) | 0.33388 | |
| y | 0.4936(5) | 0.49474 | 0.49543h | 0.5111(14) | 0.49738 | ||
| z | 1/4 | 1/4 | 1/4 | 1/4 | 1/4 | ||
| Uiso [Å2] | 0.0128(17) | 0.04555 | 0.08219 | 0.09133 | 0.02269 | ||
| O(2) | x | 0.59886(15) | 0.60045 | 0.60049(23) | 0.5960(7) | 0.59424 | |
| y | 0.4962(5) | 0.48128 | 0.4738(12) | 0.4827(12) | 0.48179 | ||
| z | 1/4 | 1/4 | 1/4 | 1/4 | 1/4 | ||
| Uiso [Å2] | 0.0158(18) | 0.02689 | 0.03885 | 0.07495 | 0.06533 | ||
| O(3) | x | 0.3518(4) | 0.35298 | 0.3494(10) | 0.3591(10) | 0.35403 | |
| y | 0.2658(4) | 0.27245 | 0.2733(9) | 0.2650(9) | 0.26182 | ||
| z | 0.0689(5) | 0.06819 | 0.0636(8) | 0.0758(9) | 0.07079 | ||
| Uiso [Å2] | 0.0190(13) | 0.04534 | 0.06731 | 0.11329 | 0.03906 | ||
| X | x | 0 | 0 | 0 | 0 | 0 | |
| y | 0 | 0 | 0 | 0 | 0 | ||
| z | 1/2 | 1/2 | 1/2 | 1/2 | 0.37433 | ||
| Uiso [Å2] | 0.138(7) | 0.00555 | 0.01941 | 0.025f | 0.52451 | ||
| Occ.d | 1.012(19) | 0.80446 | 0.797(5) | 0.4629(29) | 0.86556 | ||
| x | 0e | 0e | 0i | 0e | |||
| y | 0e | 0e | 0i | 0e | |||
| z | 1/2e | 1/2e | 1/2i | 0.37182e | |||
| Uiso [Å2] | 0.025e,f | 0.025e,f | 0.025f,i | 0.03003e,f | |||
| Occ. | 0.0535e | 0.044(5)e | 0.6150(29)i | 0.04227e | |||
| x | 0.74971g | 0.796(5)g | 0.59585g | ||||
| y | 0.78630g | 0.828(5)g | 1.10557g | ||||
| z | 0.12045g | 0.102(5)g | 0.12728g | ||||
| Uiso [Å2] | 0.025f,g | 0.025f,g | 0.03003f,g | ||||
| Occ. | 0.18841g | 0.143(14)g | 0.14682g | ||||
| wRp [%] | 9.64 | 7.64 | 8.38 | 7.46 | 9.53 | ||
| Rp [%] | 7.67 | 6.43 | 7.10 | 6.28 | 7.94 | ||
| χ2 | 3.056 | 0.901 | 1.021 | 1.009 | 1.918 |
| Bonds | This Study | Dai et al. [64] | ||||
|---|---|---|---|---|---|---|
| X = F | X = Cl | X = Br | X = I | X = OH | X = Cl | |
| As–O(1) | 1.700 | 1.696 | 1.681 | 1.664(6) | 1.674 | 1.70 |
| As–O(2) | 1.709 | 1.715 | 1.710 | 1.715(6) | 1.710 | 1.72 |
| As–O(3) | 1.689 | 1.673 | 1.679 | 1.673(4) | 1.691 | 1.67 |
| Average As–O | 1.698(9) | 1.689(20) | 1.687(15) | 1.68(1) | 1.691(12) | 1.69 |
| Distortion index | 0.004 | 0.009 | 0.006 | 0.010 | 0.005 | |
| Pb(1)–O(1) | 2.482 | 2.518 | 2.564 | 2.528(5) | 2.534 | 2.52 |
| Pb(1)–O(2) | 2.803(4) | 2.718 | 2.685(9) | 2.793(9) | 2.754 | 2.78 |
| Pb(1)–O(3) | 2.946(4) | 3.002 | 3.054 | 2.963(9) | 2.944 | 2.94 |
| Pb(2)–O(1) | 3.005(5) | 3.063 | 3.137 | 3.264 | 3.058 | 3.07 |
| Pb(2)–O(2) | 2.362(4) | 2.385 | 2.421(11) | 2.322(10) | 2.398 | 2.33 |
| Pb(2)–O(3) | 2.605 | 2.567 | 2.514(6) | 2.643(7) | 2.617 | 2.63 |
| Pb(2)–O(3) | 2.657(4) | 2.762 | 2.831(10) | 2.755(10) | 2.691 | 2.73 |
| Pb(2)–X | 3.005 | 3.158 | 3.222 | 3.286 | 2.693 | 3.16 |
| Twist angle (φ)° | 20.34(20) | 19.04(26) | 16.38(05) | 11.42(16) | 18.60(26) | 18 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sordyl, J.; Puzio, B.; Manecki, M.; Borkiewicz, O.; Topolska, J.; Zelek-Pogudz, S. Structural Assessment of Fluorine, Chlorine, Bromine, Iodine, and Hydroxide Substitutions in Lead Arsenate Apatites (Mimetites)–Pb5(AsO4)3X. Minerals 2020, 10, 494. https://doi.org/10.3390/min10060494
Sordyl J, Puzio B, Manecki M, Borkiewicz O, Topolska J, Zelek-Pogudz S. Structural Assessment of Fluorine, Chlorine, Bromine, Iodine, and Hydroxide Substitutions in Lead Arsenate Apatites (Mimetites)–Pb5(AsO4)3X. Minerals. 2020; 10(6):494. https://doi.org/10.3390/min10060494
Chicago/Turabian StyleSordyl, Julia, Bartosz Puzio, Maciej Manecki, Olaf Borkiewicz, Justyna Topolska, and Sylwia Zelek-Pogudz. 2020. "Structural Assessment of Fluorine, Chlorine, Bromine, Iodine, and Hydroxide Substitutions in Lead Arsenate Apatites (Mimetites)–Pb5(AsO4)3X" Minerals 10, no. 6: 494. https://doi.org/10.3390/min10060494






