Ore–Bearing Fluids of the Blagodatnoye Gold Deposit (Yenisei Ridge, Russia): Results of Fluid Inclusion and Isotopic Analyses
Abstract
:1. Introduction
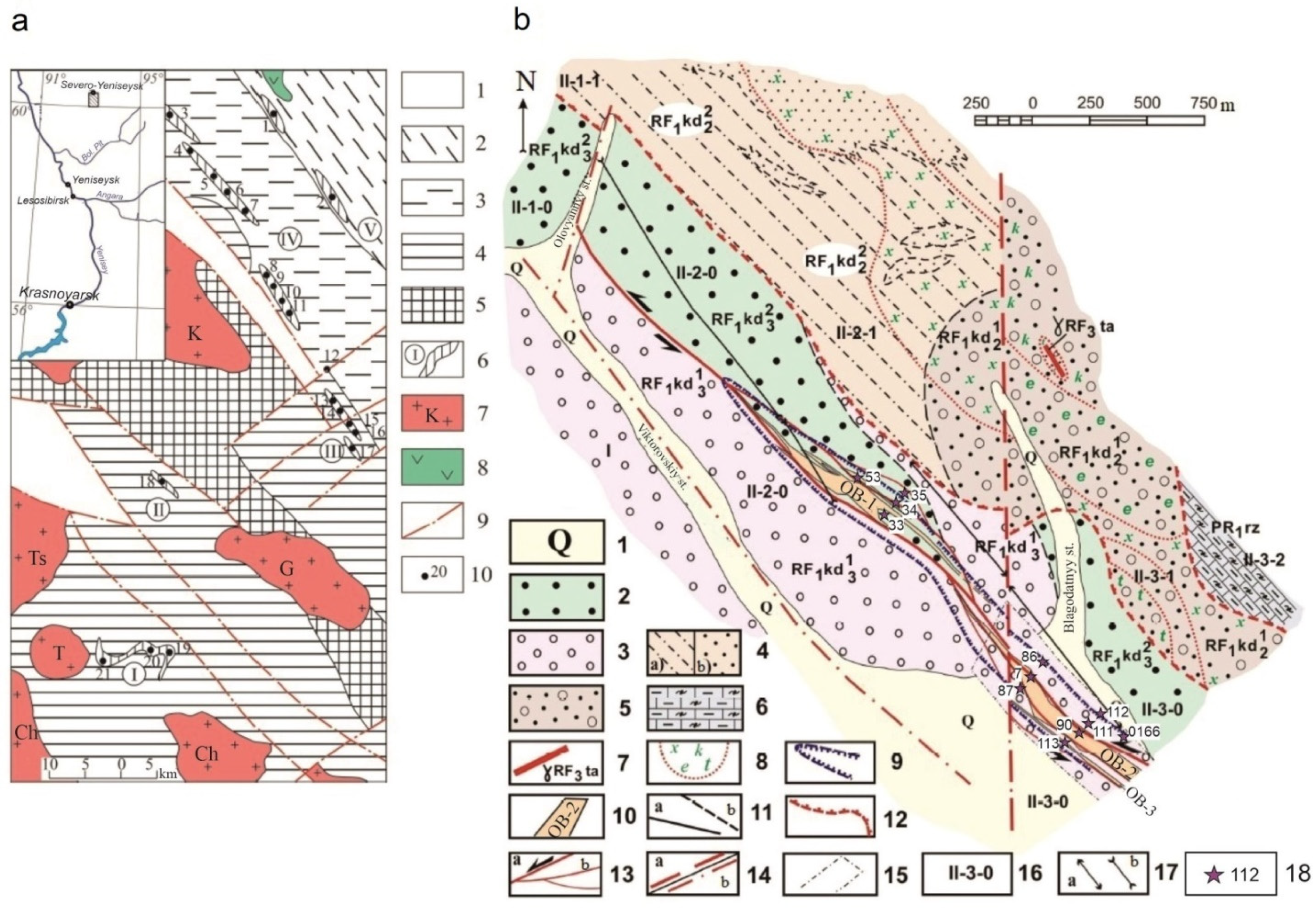
2. Geological Setting
3. Samples and Methods
4. Results
4.1. Fluid Inclusion Types
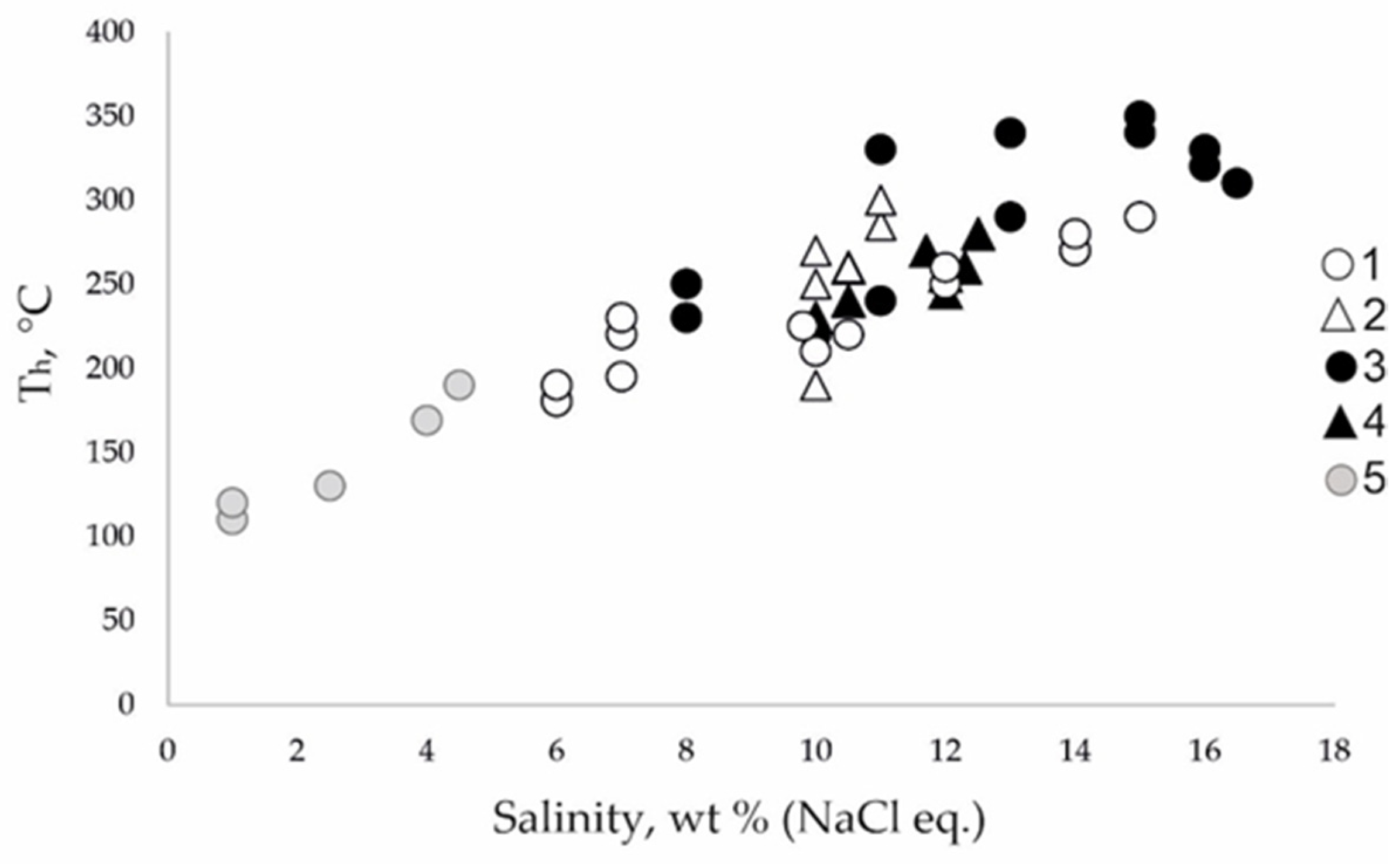
4.2. Homogenization Temperatures, Composition, Salinity and Pressure of Fluid
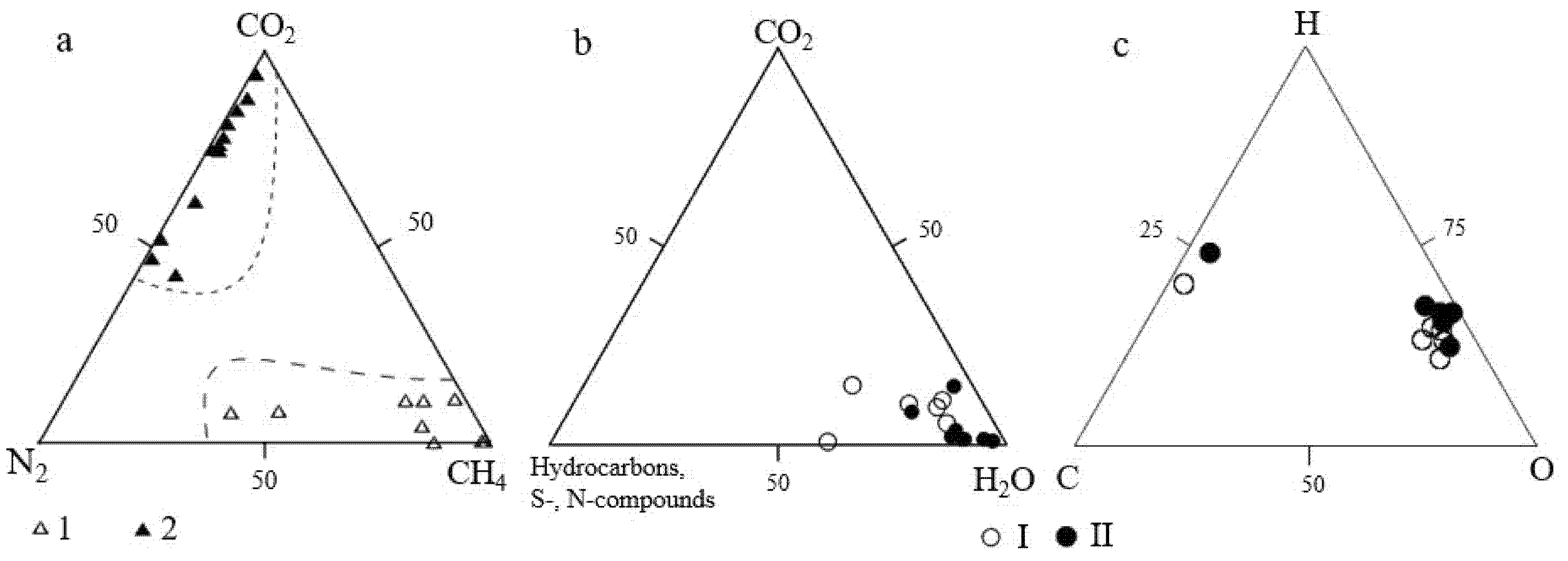
4.3. Composition of the Gaseous Phase of Fluid Inclusions
4.4. Isotopic Characteristics
5. Discussion
6. Conclusions
- The geological, mineralogical and thermobarochemistry data indicate that the formation of the Blagodatnoye deposit took place with the participation of three types of fluid: water–carbon dioxide (oxidized), carbon dioxide–hydrocarbon (reduced) and, to a lesser degree, high salinity fluid. The deposit was formed in the temperature range of 120–350 °C, pressures 0.2–2.6 kbar and salinity from 0.5 to 30 wt.% (NaCl–equiv.).
- Mineral–forming fluids contained H2O, CO2, hydrocarbons and oxygenated organic compounds, S–, N–, halogenated compounds, which are potentially capable to transport ore elements, including gold.
- Early barren quartz vein zones of the deposit were formed by oxidized water–carbon dioxide fluids.
- Later reduced carbon dioxide–hydrocarbon fluids had a key role in the formation of gold-bearing quartz veins when superimposed on the earlier formed quartz. The stage of productive gold–sulfide mineralization at the deposit lasted for about 50–60 Ma.
- The scales of mineralization depend on the long–term activity of gold–bearing fluids, which is reflected in the degree of saturation of quartz with carbon dioxide–hydrocarbon inclusions.
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Polus Gold. Available online: https://polyus.com/en/ (accessed on 15 September 2021).
- Prokofiev, V.Y.; Bortnikov, N.S.; Garofalo, P. Physicochemical parameters and composition of ore-forming fluids of orogenic gold deposits in Russia. In Proceedings of the XIII International Conference on Thermobarogeochemistry and IV APIFIS Symposium. 22–25 September 2008, IGEM RAN: Moscow, Russia; 2008; Volume 2, pp. 188–191. [Google Scholar]
- Groves, D.I.; Goldfarb, R.J.; Santosh, M. The conjunction of factors that lead to formation of giant gold provinces and deposits in non-arc settings. Geosci. Frontiers. 2016, 7, 303–314. [Google Scholar] [CrossRef] [Green Version]
- Sazonov, A.M.; Kirik, S.D.; Silyanov, S.A.; Bayukov, O.A.; Tishin, P.A. Typomorphism of arsenopyrite from the Blagodatnoe and Olimpiada gold deposits (Yenisei ridge). Mineralogy 2016, 3, 53–70. (In Russian) [Google Scholar]
- Bortnikov, N.S.; Prokov’ev, V.Y.; Razdolina, N.V. Origin of the Charmitan gold–quartz deposit (Uzbekistan). Geol. Ore Depos. 1996, 38, 238–256. [Google Scholar]
- Kryazhev, S.G. Isotope–Geochemical Regime of the Rormation of the Gold Ore Deposit Muruntau; TsNIGRI: Moscow, Russia, 2002. (In Russian) [Google Scholar]
- Safonov, Y.G.; Prokofiev, V.Y. Model of cosedimentation hydrothermal formation of gold–bearing reefs of the Witwatersrand basin. Geol. Ore Depos. 2006, 48, 475–511. [Google Scholar] [CrossRef]
- Ronde, C.E.J.; Faure, K.; Bray, C.J.; Whitford, D.J. Round Hill shear zone–hosted gold deposit, Macraes Flat, Otago, New Zealand; evidence of a magmatic ore fluid. Econ. Geol. 2000, 95, 1025–1048. [Google Scholar]
- Jia, Y.; Li, X.; Kerrich, R. A fluid inclusion study of Au–bearing quartz vein systems in the Central and North Deborah deposits of the Bendigo gold field. Central Victoria. Autralia. Econ. Geol. 2000, 95, 467–494. [Google Scholar] [CrossRef]
- Tomilenko, A.A.; Gibsher, N.A.; Dublaynsky, Y.V.; Dallai, I. Geochemical and isotopic properties of fluid from gold–bearing and barren quartz veins of the Sovetskoye deposit (Siberia, Russia). Econ. Geol. 2010, 105, 375–394. [Google Scholar] [CrossRef]
- Dubessy, J.; Poty, B.; Ramboz, C. Advances in C–O–H–N–S fluid geochemistry based on micro–Raman spectrometric analysis of fluid inclusions. Eur. J. Miner. 1989, 1, 517–534. [Google Scholar] [CrossRef] [Green Version]
- Laverov, N.P.; Prokofiev, V.Y.; Distler, V.V.; Yudovskaya, M.A.; Spiridonov, A.M.; Grebenshchikova, V.I.; Matel, N.L. New data on conditions of ore deposition and composition of ore–forming fluids of the Sukhoi Log gold–platinum deposit. Rep. Acad. Sci. 2000, 371, 88–92. [Google Scholar]
- Prokofiev, V.Y.; Naumov, V.B.; Mironova, O.F. Physicochemical parameters and geochemical features of fluids of Precamrbian gold deposits. Geochem. Int. 2017, 55, 1047–1065. [Google Scholar] [CrossRef]
- Kryazhev, S.G. Genetic Models and Criteria for Prediction of Gold Deposits in Carbon–Terrigenous Complexes. DSci. Thesis, TsNIGRI, Moscow, Russia, 2017. (In Russian). [Google Scholar]
- Ryabukha, M.A.; Gibsher, N.A.; Tomilenko, A.A.; Bul’bak, T.A.; Khomenko, M.O.; Sazonov, A.M. PTX–parameters of metamorphogene and hydrothermal fluids; isotopy and age of formation of the Bogunai gold deposit, southern Yenisei ridge (Russia). Russ. Geol. Geophys. 2015, 56, 903–918. [Google Scholar] [CrossRef]
- Khomenko, M.O.; Gibsher, N.A.; Tomilenko, A.A.; Bul’bak, T.A.; Ryabukha, M.A.; Semenova, D.V. Physicochemical parameters and age of the Vasil’kovskoe gold deposit (Northern Kazakhstan). Russ. Geol. Geophys. 2016, 57, 1728–1749. [Google Scholar] [CrossRef]
- Gibsher, N.A.; Tomilenko, A.A.; Sazonov, A.M.; Ryabukha, M.A.; Timkina, A.L. The Gerfed gold deposit: Fluids and PT–conditions for quartz vein formation (Yenisei Ridge, Russia). Russ. Geol. Geophys. 2011, 52, 1461–1473. [Google Scholar] [CrossRef]
- Gibsher, N.A.; Ruabukha, M.A.; Tomilenko, A.A.; Sazonov, A.M.; Khomenko, M.O.; Bul’bak, T.A.; Nekrasova, N.A. Metal–bearing fluids and the age of the Panimba gold deposits (Yenisei Ridge, Russia). Russ. Geol. Geophys. 2017, 58, 1366–1383. [Google Scholar] [CrossRef]
- Gibsher, N.A.; Tomilenko, A.A.; Bul’bak, T.A.; Ryabukha, M.A.; Khomenko, M.O.; Shaparenko, E.O.; Sazonov, A.V.; Sil’yanov, S.A.; Nekrasova, N.A. The Olimpiadinskoe gold deposit (Yenisei ridge): Temperature, pressure, composition of ore–forming fluids, δ34S in sulfides, 3He/4He of fluids, Ar–Ar age, and duration of formation. Russ. Geol. Geophys. 2019, 60, 1043–1059. [Google Scholar] [CrossRef]
- Gibsher, N.A.; Tomilenko, A.A.; Sazonov, A.M.; Bul’bak, T.A.; Khomenko, M.O.; Ryabukha, M.A.; Shaparenko, E.O.; Sil’yanov, S.A.; Nekrasova, N.A. Ore–bearing fluids of the Eldorado gold deposit (Yenisei Ridge, Russia). Russ. Geol. Geophys. 2018, 59, 983–996. [Google Scholar] [CrossRef]
- Bul’bak, T.A.; Tomilenko, A.A.; Gibsher, N.A.; Sazonov, A.M.; Shaparenko, E.O.; Ryabukha, M.A.; Khomenko, M.O.; Sil’yanov, S.A.; Nekrasova, N.A. Hydrocarbons in fluid inclusions from native gold, pyrite, and quartz of the Sovetskoe deposit (Yenisei ridge, Russia) according to Pyrolysis–Free Gas Chromatography–Mass Spectrometry Data. Russ. Geol. Geophys. 2020, 11, 1260–1282. [Google Scholar] [CrossRef]
- Vernikovsky, V.A.; Vernikovskaya, A.E.; Kotov, A.B.; Sal’nikova, E.B.; Kovach, V.P. Neoproterozoic accretionary and collisional events on the western margin of the Siberian craton: New geological and geochronological evidence from the Yenisey ridge. Tectonophysics 2003, 375, 147–168. [Google Scholar] [CrossRef]
- Sazonov, A.M.; Ananyev, A.A.; Poleva, T.V.; Khokhlov, A.N.; Vlasov, V.S.; Zvyagina, E.A.; Fedorova, A.V.; Tishin, P.A.; Leontiev, S.I. Gold ore metallogeny of the Yenisei ridge: Geological–structural position, structural types of ore fields. Journal of Siberian Federal University. Eng. Technologies. 2010, 4, 371–395. [Google Scholar]
- Poleva, T.V.; Sazonov, A.M. Geology of the Blagodatnoye Gold Deposit in the Yenisei ridge (In Russian); ITKOR: Moscow, Russia, 2012; p. 292. [Google Scholar]
- Sazonov, A.M.; Gertner, I.F.; Zvyagina, E.A.; Tishin, P.A.; Poleva, T.V.; Leontyev, S.I.; Kolmakov, Y.V.; Krasnova, T.S. Ore-forming conditions of the Blagodat gold deposit in the Riphean metamorphic rocks of the Yenisey ridge according to geochemical and isotopic data. Journal of Siberian Federal University. Eng. Technol. 2009, 2, 203–220. [Google Scholar]
- Sazonov, A.M.; Zvyagina, E.A. Genesis of gold–bearing ores of Blagodatnaya. The state and problems of geological study of the subsoil and the development of the mineral resource base of the Krasnoyarsk Territory. Krasnoyarsk 2003, 247–249. (In Russian) [Google Scholar]
- Brown, P.E.; Lamb, W.M. P–V–T properties of fluids in the system H2O ± CO2 ± NaCl: New graphical presentations and implications for fluid inclusion studies. Geochim. Cosmochim. Acta. 1989, 53, 1209–1231. [Google Scholar] [CrossRef]
- Duan, Z.; Moller, N.; Weare, J.H. A general equation of state for supercritical fluid mixtures and molecular dynamics simulation of mixture PVTX properties. Geochim. Cosmochim. Acta. 1996, 60, 1209–1216. [Google Scholar] [CrossRef]
- Bakker, R.J. Fluids: New software package to handle microthermometric data and to calculate isochors. Memoir Geol. Soc. 2001, 7, 23–25. [Google Scholar]
- Borisenko, A.S. Studying the salt composition of solutions of gas–liquid inclusions in minerals by the method of cryometry. Russ. Geol. Geophys. 1977, 8, 16–27. (In Russian) [Google Scholar]
- Kirgintsev, A.N.; Trushnikova, L.I.; Lavrentieva, V.G. Water Solubility of Inorganic Compounds; A Handbook; Khimiya: Leningrad, Russia, 1972; p. 274. (In Russian) [Google Scholar]
- Tomilenko, A.A.; Chepurov, A.I.; Sonin, V.M.; Bul’bak, T.A.; Zhimulev, E.I.; Chepurov, A.A.; Timina, T.Y.; Pokhilenko, N.P. The synthesis of methane and heavier hydrocarbons in the system graphite–iron–serpentine at 2 and 4 GPa and 1200 °C. High Temp.–High Pressures. 2015, 44, 451–465. [Google Scholar]
- Sokol, A.G.; Palyanov, Y.N.; Tomilenko, A.A.; Bulbak, T.A.; Palyanova, G.A. Carbon and nitrogen speciation in nitrogen–rich C–O–H–N fluids at 5.5–7.8 GPa. Earth Planet. Sci. Lett. 2017, 460, 234–243. [Google Scholar] [CrossRef]
- Pal’yanova, G.A.; Sobolev, E.S.; Reutsky, V.N.; Bortnikov, N.S. Upper Triassic pyritized bivalve mollusks from the Sentachan orogenic gold–antimony deposit, eastern Yakutia: Mineralogy and sulfur isotopic composition. Geol. Ore Depos. 2016, 58, 513–521. [Google Scholar] [CrossRef]
- Tolstikhin, I.N.; Prasolov, E.M. Methodology for the study of isotopes of noble gases from microswitches in rocks and minerals. Investigation of mineral–forming solutions and melts by inclusions in minerals. Work. VNIISIMS. Alexandrov. 1971, 14, 86–98. [Google Scholar]
- Vetrin, V.R.; Kamenskii, I.L.; Ikorskii, S.V.; Gannibal, M.A. Juvenile helium in Archean enderbites and alkali granites of the Kola Peninsula. Geochem. Int. 2003, 7, 699–705. [Google Scholar]
- Ikorsky, S.V.; Gannibal, M.A.; Avedisyan, A.A. High–temperature impregnation of helium into fluid inclusions in minerals: Experiments with quartz and nepheline. Dokl. Earth Sci. 2006, 411, 1299–1302. [Google Scholar] [CrossRef]
- Ikorsky, S.V.; Kamensky, I.L.; Avedisyan, A.A. Helium isotopes in contact zones of alkaline intrusions of different size: A case study of the alkaline–ultrabasic Ozernaya Varaka intrusive and Lovozero massif of nepheline syenites (Kola Peninsula). Dokl. Earth Sci. 2014, 459, 1543–1547. [Google Scholar] [CrossRef]
- Prasolov, E.M.; Sergeev, S.A.; Belitsky, B.V.; Bogomolov, E.S.; Gruzdov, K.A.; Kapitonov, I.N.; Crimean, R.S.; Khalenev, D.O. Isotopic systematics of He, Ar, S, Cu, Ni, Re, Os, Pb, U, Sm, Nd, Rb, Sr, Lu, and Hf in the rocks and ores of the Norilsk deposits. Geochem. Int. 2018, 1, 50–69. [Google Scholar] [CrossRef]
- Travin, A.V. Thermochronology of Early Paleozoic collisional and subduction–collisional structures of Central Asia. Russ. Geol. Geophys. 2016, 57, 553–574. [Google Scholar] [CrossRef]
- Ermakov, N.P.; Dolgov, Y.A. Thermobarogeochemistry; Nedra: Moscow, Russia, 1979; p. 271. (In Russian) [Google Scholar]
- Roedder, E. Fluid Inclusions in Minerals; Mir: Moscow, Russia, 1987; Volume 1, p. 558. (In Russian) [Google Scholar]
- Eirish, L.V. Metallogeny of Gold of Priamurye (Amur Region, Russia); Vladivostok: Dalnauka, Russia, 2002; p. 194. (In Russian) [Google Scholar]
- Robert, F.; Kelly, W.C. Ore–forming fluids in Archean gold–bearing quartz veins at the Sigma Mine, Abitibe green–stone belt, Quebec, Canada. Econ. Geol. 1987, 82, 1464–1482. [Google Scholar] [CrossRef]
- Norman, D.I.; Blamey, N.; Moore, J.N. Interpreting geothermal processes and fluid sources from fluid inclusion organic compounds and CO2/N2 ratios. In Proceedings of the 27th Workshop on Geothermal Reservoir Engineering, Stanford University, Stanford, CA, USA, 28–30 January 2002; pp. 234–243. [Google Scholar]
- Blamey, N.J.F. Composition and evolution of crustal, geothermal and hydrothermal fluids interpreted using quantitative fluid inclusion gas analysis. J. Geochem. Exp. 2012, 116–117, 17–27. [Google Scholar] [CrossRef]
- Marchuk, M.V. Transport of Major Elements and Metals in Reduced Fluids: An Experimental Study. Author’s Abstract. Candidate Thesis (Geology and Mineralogy), Institute of the Earth’s Crust SB RAS, Irkutsk, Russia, 2008. (In Russian). [Google Scholar]
- Bondar, R.A.; Naumko, I.M.; Lyubchak, A.V.; Khokha, Y.V. On the question of the forms of transfer of precious and rare–metal metals to the ore deposition zones of gold deposits. Native Gold, Typomorphism of Mineral Associations, Conditions for the Formation of Deposits, Tasks of Applied Research. IGEM RAS. Moscow. 2010, 1, 76–78. (In Russian) [Google Scholar]
- Lyakhov, Y.V.; Pavlun, N.N. Some Geological and Geochemical Features of Gold Mineralization in Metamorphic and Magmatic Hydrothermal Systems. In Metallogenic Processes: From Genetic Theories to Prediction and Discovery of New Ore Provinces and Deposits; IGEM: Moscow, Russia, 2013; p. 144. (In Russian) [Google Scholar]
- Williams–Jones, A.E.; Bowell, R.J.; Migdisov, A.A. Gold in solution. Element 2009, 5, 281–287. [Google Scholar] [CrossRef]
- Bottrell, S.H.; Miller, M.F. The geochemical behaviour of nitrogen compounds during the formation of black shale hosted quartz–vein gold deposits, North Wales. J. Appl. Geochem. 1990, 5, 289–296. [Google Scholar] [CrossRef]
- Mironova, O.F.; Naumov, V.B.; Salazkin, A.N. Nitrogen in Mineral–Forming Fluids. Gas Chromatography Determination on Fluid Inclusions in Minerals. Geochem. Int. 1992, 7, 979–991. [Google Scholar]
- Yang, Y.; Busigny, V.; Wang, Z.; Xia, Q. The fate of ammonium in phengite at high temperature. Am. Mineral. 2017, 102, 2244–2253. [Google Scholar] [CrossRef]
- Kol’tsov, A.B. Hydrothermal mineralization in the fields of temperature and pressure gradients. Geochem. Int. 2010, 11, 1097–1111. [Google Scholar] [CrossRef]
- Zubkov, V.S. Composition and speciation of fluids in the system C–H–N–O–S at P–T conditions of the upper mantle. Geochem. Int. 2001, 39, 131–145. [Google Scholar]
- Ohmoto, H.; Rye, R.O. Isotopes of Sulfur and Carbon. Geochemistry of Hydrothermal Ore Deposits; Wiley: New York, NY, USA, 1979; pp. 509–567. [Google Scholar]
- Taylor, B.E. Magmatic volatiles: Isotopic variation of C, H, and S. Rev. Mineral. Geochem. 1986, 16, 185–225. [Google Scholar]
- Goldfarb, R.J.; Newberry, R.J.; Pickthorn, W.J.; Gent, C.A. Oxygen, hydrogen, and sulfur isotope studies in the Juneau gold belt, southeastern Alaska: Constraints on the origin of hydrothermal fluids. Econ. Geol. 1991, 86, 66–80. [Google Scholar] [CrossRef]
- Kryazhev, S.G.; Grinenko, V.A. The Isotopic Composition and Sources of Sulfur in Gold–Sulphide Deposits of the Yenisei Ridge. Abstracts of the XVIII Symposium on Geochemistry of Isotopes; GEOKhI RAN: Moscow, Russia, 2007; p. 37. (In Russian) [Google Scholar]
- Ohmoto, H. Stable isotope geochemistry of ore deposits. In Stable Isotopes in High Temperature Geological Processes. Rev. Mineral. Geochem. 1986, 16, 491–560. [Google Scholar]
- Khalenev, V.O. He and Ar isotope compositions as a proxy of metallogenic potential in Plutonic Rocks, Norilsk Region. Author’s Abstract. Candidate Thesis, Geology and Mineralogy, All-Russian Geological Institute, St. Petersburg, Russia, 2010. (In Russian). [Google Scholar]
- Nozhkin, A.D.; Turkina, O.M.; Bobrov, V.A. Radioactive and rare earth elements in metapelites as indicators of composition and evolution of the Precambrian continental crust in the southwestern margin of the Siberian craton. Dokl. Earth Sci. 2003, 391, 718–722. [Google Scholar]
- Nozhkin, A.D. The conditions of placement and geological and geochemical prerequisites for the formation of uranium, gold–uranium, gold and rare metal tools in the Central Metallogenic Belt of the Yenisei Ridge. Sat. Geology and mineral resources of Siberia. Krasnoyarsk 2014, S3, 22–27. [Google Scholar]
- Gertner, I.; Tishin, P.; Vrublevskii, V.; Sazonov, A.; Zvyagina, E.; Kolmakov, Y. Neoproterozoic alkaline igneous rocks, carbonatites and gold deposits of the Yenisei Ridge, Central Siberia: Evidence of mantle plume activity and late collision shear tectonics associated with orogenic gold mineralization. Resour. Geol. 2011, 61, 316–343. [Google Scholar] [CrossRef]
- Vernikovsky, V.A.; Vernikovskaya, A.E. Tectonics and evolution of granitoid magmatism in the Yenisei Ridge. Russ. Geol. Geophys. 2006, 47, 32–50. (In Russian) [Google Scholar]
- Mernagh, T.P.; Bierlein, F.P. Transport and precipitation of gold in Phanerozoic metamorphic terranes from chemical modeling of fluid–rock interaction. Econ. Geol. 2008, 103, 1613–1640. [Google Scholar] [CrossRef]
- Konstantinov, M.M. Ore–forming systems in the earth’s crust. Izv. Vuzov. Geologiya i Razvedka. 2009, 5, 22–28. [Google Scholar]





| Fluid Inclusion Type | Generation of Fluid Inclusions * | Th Total, °C | Type of Homogenization ** | Tm, °C | Aqueous Phase | Tm of CO2 ± CH4 ± N2, °C | Partial Th, °C | Type of Homogenization | Pressure, kbar | ||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Teut, °C | Tm, °C | Salinity, wt.%, NaCl-eq. | |||||||||
| Pre-ore stage. Quartz of pre-ore metasomatites. | |||||||||||
| A LH2O + V | P, PS | 180–290 (37) | L | - | −31.3 ÷ −30.5 (19) | −3.0 ÷ −10.0 (15) | 6−15 | - | - | - | 0.2–1.6 |
| S | 140–170 (8) | L | - | −22.0 ÷ −26.3 (3) | −1.0 ÷ −3.0 (3) | 2–6 | - | - | - | ||
| B LH2O + LCO2 ± CH4 ± N2 | P, PS | 250–300 (19) | L | - | −33.4 ÷ −39.0 (11) | −5.9 ÷ −7.0 (11) | 10–11 | −58.1 ÷ −60.3 (9) | −31.8 ÷ +15.1 (9) | L, V | |
| C L(V)CO2 ± CH4 ± N2 | S | - | - | - | - | - | - | −59.3 ÷ −61.3 (7) | −19.0 ÷ −23.8 (7) | L, V | |
| S | - | - | - | - | - | - | −103.5 ÷ 120.3 (5) | −90.5 ÷ −92.5 (5) | V | ||
| D LH2O + V + S | S | 190–210 (9) | L | 170−200 (3) | −49.4 ÷ −56.0 (5) | −21.4 ÷ −20.5 (5) | >30 | - | - | - | |
| Ore stage. Quartz of gold-sulfide veins. | |||||||||||
| A LH2O + V | P, PS | 220–350 (68) | L, V | - | −29.3 ÷ −34.2 (28) | −3.9 ÷ −12.0 (27) | 8–16.5 | - | - | - | 1.8–2.6 |
| S | 130–230 (31) | L | - | −18.0 ÷ −21.5 (15) | −0.5 ÷ −2.5 (15) | 1–8.5 | - | - | - | ||
| B LH2O + LCO2 ± CH4 ± N2 | P, PS | 230–280 (39) | L | - | −36.8 ÷ −45.9 (16) | −6.1 ÷ −8.0 (16) | 10–12.5 | −57.9 ÷ −63.8 (19) | −53.3 ÷ +21.3 (19) | L, V | |
| C L(V)CO2 ± CH4 ± N2 | P, PS | - | - | - | - | - | - | −59.0 ÷ −61.3 (31) | −37.6 ÷ +19.8 (31) | L, V | |
| PS | - | - | - | - | - | - | −90.1 ÷ −105.1 (17) | −78.5 ÷ −96.6 (17) | L, V | ||
| D LH2O + V + S | S | 150–250 (19) | L | 140−190 (15) | −47.2 ÷ −54.3 (11) | −38.1 ÷ −42.1 (11) | >30 | - | - | - | |
| Post-ore stage. Quartz, calcite filamentous veins. | |||||||||||
| A LH2O > >V | P, PS | Quartz | |||||||||
| 110−180 (9) | L | - | −18.5 ÷ −21.0 (5) | −0.5 ÷ −3.0 (6) | 1−6 | - | - | - | - | ||
| B LH2O > >V | P, PS | Calcite | |||||||||
| 120–160 (7) | L | - | - | - | - | - | - | - | - | ||
| No. * | Tm, °C | Th, °C | Type ** of Homogenization | Content, mol.% | CO2/CH4 | ||
|---|---|---|---|---|---|---|---|
| CO2 | CH4 | N2 | |||||
| FI type B (LH2O + L(V)CO2 ± CH4 ± N2) | |||||||
| 111/59.3/1 | −58.1 | +8 | L | 78 | 2.0 | 20.0 | 39.0 |
| 111/59.3/2 | −57.1 | +10 | L | 76 | 2.0 | 22.0 | 38.0 |
| 111/60.8/1 | −56.8 | +13 | L | 94 | 1.0 | 5.0 | 93.0 |
| 111/60.8/2 | −57.3 | +19 | L | 88 | 2.0 | 10.0 | 44.0 |
| 111/90.3/1 | −59.5 | −6.5 | L | 81.5 | 1.0 | 17.5 | 81.5 |
| 111/90.3/2 | −61.2 | −9.5 | L | 74.5 | 2.5 | 23.0 | 29.8 |
| 111/120.6/1 | −60.3 | −8.0 | L | 85 | 1.5 | 13.5 | 56.7 |
| 111/129.5/1 | −64.0 | −9.5 | V | 52 | 1.0 | 47.0 | 52.0 |
| 111/129.5/2 | −63.8 | −32.5 | L | 47 | 1.5 | 51.5 | 31.3 |
| 33/29.3/1 | - | - | - | 74.7 | 0.9 | 24.4 | 83.0 |
| 33/29.3/2 | - | - | - | 42.7 | 9.0 | 48.3 | 4.7 |
| 33/29.3/3 | - | - | - | 61.4 | 4.0 | 34.6 | 15.4 |
| FI type C (L(V)CO2 ± CH4 ± N2) | |||||||
| 111/76.5/1 | - | −90.0 | V | 8.0 | 49.0 | 43.0 | 0.2 |
| 111/90.3/1 | - | −82.0 | L | 10.5 | 80.0 | 9.5 | 0.1 |
| 111/120.6/2 | −94.5 | −79.0 | L | 10.5 | 76.0 | 13.5 | 0.1 |
| 111/120.6/3 | - | −78.5 | V | 0.0 | 87.5 | 12.5 | - |
| 111/120.6/4 | - | −86.5 | V | 0.5 | 98.5 | 1.0 | 0.01 |
| 111/120.6/5 | - | −78.5 | L | 11.0 | 86.5 | 2.5 | 0.1 |
| 111/129.5/3 | - | −83.0 | L | 4.5 | 86.0 | 13.5 | 0.05 |
| 111/129.5/4 | - | −84.0 | V | 0.5 | 98.0 | 1.5 | 0.01 |
| 33/29.3/4 | - | - | - | 7.7 | 38.6 | 53.5 | 0.2 |
| Component | Mineral Association | ||||||
|---|---|---|---|---|---|---|---|
| Gold-Bearing (Au = 1–35.5 g/t) | Barren (Au = 0.2–0.8 g/t) | ||||||
| Quartz | Quartz | Arseno-Pyrite | Arseno-Pyrite | Quartz | Pyrrhotite | Calcite | |
| 111/90.3 | 7/96.0 | 7/96.0 | BKS-10 | 86/107.7 | 34/105.9 | 35/178.1 | |
| Aliphatic hydrocarbons | |||||||
| Paraffins (alkanes) | 1.27 (16) | 7.48 (21) | 2.24 (18) | 3.64 (14) | 1.04 (12) | 0.87 (16) | 0.57 (16) |
| Olefins (alkenes) | 0.07 (17) | 0.12 (19) | 1.08 (20) | 0.11 (19) | 2.77 (31) | 0.96 (27) | 0.06 (19) |
| Cyclic hydrocarbons | |||||||
| Cycloalkanes, cycloalkenes, arenes, PAH | 0.03 (17) | 0.09 (14) | 0.44 (24) | 0.14 (22) | 2.27 (16) | 0.68 (28) | 0.04 (20) |
| Oxygenated hydrocarbons | |||||||
| Alcohols, ethers and esters | 0.33 (23) | 0.61 (19) | 0.24 (11) | 2.78 (19) | 28.14 (28) | 0.71 (27) | 1.13 (17) |
| Aldehydes | 0.50 (22) | 0.30 (24) | 0.33 (22) | 1.17 (22) | 0.85 (17) | 1.08 (26) | 0.44 (21) |
| Ketones | 0.29 (21) | 0.21 (15) | 0.08 (11) | 0.13 (18) | 1.47 (12) | 0.91 (21) | 0.08 (19) |
| Carboxylic acids | 0.73 (13) | 0.76 (15) | 0.96 (13) | 0.3 (14) | 0.95 (12) | 5.71 (21) | 0.41 (13) |
| Heterocyclic compounds | |||||||
| Dioxanes, furans | <0.01 (6) | 0.01 (3) | <0.01 (4) | <0.01 (5) | 0.02 (4) | 0.025 (7) | <0.01 (6) |
| Nitrogenated compounds | |||||||
| N2, ammonia, nitriles | 0.85 (19) | 1.44 (7) | 0.38 (3) | 1.21 (19) | 0.78 (4) | 1.71 (22) | 23.28 (18) |
| Sulfonated compounds | |||||||
| H2S, SO2, CS2, COS, thiophenes | 0.06 (8) | 0.08 (8) | 2.59 (10) | 0.24 (10) | 0.46 (5) | 3.85 (11) | 0.22 (9) |
| Inorganic compounds | |||||||
| CO2 | 14.77 | 2.13 | 1.53 | 1.48 | 0.83 | 10.3 | 15.09 |
| H2O | 81.08 | 86.78 | 90.14 | 89.52 | 60.42 | 73.45 | 58.67 |
| Ar | <0.01 | 0.01 | <0.01 | - | <0.01 | <0.01 | <0.01 |
| Number of components | 165 | 149 | 139 | 165 | 144 | 209 | 162 |
| Halogenated compounds: Cl, F, Br | 0.004 (4) | 0.004 (2) | 0.001 (1) | 0.001 (2) | 0.06 (3) | 0.06 (2) | - |
| Alkanes/alkenes | 17.6 | 60.8 | 2.1 | 33.4 | 0.4 | 0.9 | 10.0 |
| CO2/(CO2 + H2O) | 0.15 | 0.02 | 0.02 | 0.02 | 0.01 | 0.12 | 0.2 |
| Σ(C5-C17)/Σ(C1-C4) | 0.1 | 0.5 | 0.31 | 0.04 | 14.8 | 0.14 | 0.63 |
| Sample No. | Mineral | δ34S,‰ CDT |
|---|---|---|
| 34/105.9 | pyrrhotite | 8.3 |
| 53/112.8 | 2.6 | |
| 100/216.5 | 8.9 | |
| 0166/500 | 8.7 | |
| 0166/540 | 8.6 | |
| 111/76.5 | pyrite | 10.1 |
| 111/129.5 | 9.1 | |
| B-10 | 11.0 | |
| B-14-b | 10.1 | |
| 7/42.6 | arsenopyrite | 6.8 |
| 7/48.5 | 9.4 | |
| 7/61.3 | 12.0 | |
| 7/96.0 | 8.9 | |
| 87.69.3 | 6.3 | |
| 100/221 | 9.5 | |
| 100/223 | 8.7 | |
| 111/76.5 | 8.9 | |
| 111/97.1 | 11.0 | |
| 112/191.2 | 12.0 | |
| 0166/340 | 20.1 | |
| 0166/570 | 11.2 | |
| B-2 | 7.7 | |
| B-3 | 9.8 | |
| B-6 | 8.9 | |
| B-11-a | 9.9 | |
| B-16 | 11.6 | |
| B17 | 9.4 |
| Sample No. * | δ13CCO2, ‰ (VPDB) | Au, g/t |
|---|---|---|
| Quartz of gold-bearing associations | ||
| 7/48 | −10.8 | 8.6 |
| 7/61.3 | −2.8 | 1.6 |
| 7/96 | −3.0 | 31.5 |
| 100/216.5 | −9.8 | 10.2 |
| 100/221 | −12.0 | 1.3 |
| 100/223 | −10.8 | 1.6 |
| 111/97.1 | −4.4 | 0.8 |
| 112/191.2 | −3.0 | 4.9 |
| Quartz of barren associations | ||
| 7/55 | −10.0 | 0.6 |
| 86/107.7 | −17.4 | 0.2 |
| 86/131.8 | −3.3 | 0.2 |
| 86/241.4 | −4.6 | 0.3 |
| 69/157 | −20.9 | 0.4 |
| 34/105.9 | −6.4 | 0.5 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shaparenko, E.; Gibsher, N.; Tomilenko, A.; Sazonov, A.; Bul’bak, T.; Ryabukha, M.; Khomenko, M.; Silyanov, S.; Nekrasova, N.; Petrova, M. Ore–Bearing Fluids of the Blagodatnoye Gold Deposit (Yenisei Ridge, Russia): Results of Fluid Inclusion and Isotopic Analyses. Minerals 2021, 11, 1090. https://doi.org/10.3390/min11101090
Shaparenko E, Gibsher N, Tomilenko A, Sazonov A, Bul’bak T, Ryabukha M, Khomenko M, Silyanov S, Nekrasova N, Petrova M. Ore–Bearing Fluids of the Blagodatnoye Gold Deposit (Yenisei Ridge, Russia): Results of Fluid Inclusion and Isotopic Analyses. Minerals. 2021; 11(10):1090. https://doi.org/10.3390/min11101090
Chicago/Turabian StyleShaparenko, Elena, Nadezhda Gibsher, Anatoly Tomilenko, Anatoly Sazonov, Taras Bul’bak, Maria Ryabukha, Margarita Khomenko, Sergey Silyanov, Natalya Nekrasova, and Marina Petrova. 2021. "Ore–Bearing Fluids of the Blagodatnoye Gold Deposit (Yenisei Ridge, Russia): Results of Fluid Inclusion and Isotopic Analyses" Minerals 11, no. 10: 1090. https://doi.org/10.3390/min11101090
APA StyleShaparenko, E., Gibsher, N., Tomilenko, A., Sazonov, A., Bul’bak, T., Ryabukha, M., Khomenko, M., Silyanov, S., Nekrasova, N., & Petrova, M. (2021). Ore–Bearing Fluids of the Blagodatnoye Gold Deposit (Yenisei Ridge, Russia): Results of Fluid Inclusion and Isotopic Analyses. Minerals, 11(10), 1090. https://doi.org/10.3390/min11101090






