Equations of State of Ca-Silicates and Phase Diagram of the CaSiO3 System under Upper Mantle Conditions
Abstract
:1. Introduction
2. Thermodynamic Model
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gasparik, T.; Wolf, K.; Smith, C.M. Experimental determination of phase relations in the CaSiO3 system from 8 to 15 GPa. Am. Mineral. 1994, 79, 1219–1222. [Google Scholar]
- Swamy, V.; Dubrovinsky, L.S. Thermodynamic data for the phases in the CaSiO3 system. Geochim. Cosmochim. Acta 1997, 61, 1181–1191. [Google Scholar] [CrossRef]
- Joswig, W.; Stachel, T.; Harris, J.W.; Baur, W.H.; Brey, G.P. New Ca-silicate inclusions in diamonds—Tracers from the lower mantle. Earth Planet. Sci. Lett. 1999, 173, 1–6. [Google Scholar] [CrossRef]
- Stachel, T.; Harris, J.W.; Brey, G.P.; Joswig, W. Kankan diamonds (Guinea) II: Lower mantle inclusion parageneses. Contrib. Mineral. Petrol. 2000, 140, 16–27. [Google Scholar] [CrossRef]
- Anzolini, C.; Angel, R.J.; Merlini, M.; Derzsi, M.; Tokar, K.; Milani, S.; Krebs, M.Y.; Brenker, F.E.; Nestola, F.; Harris, J.W. Depth of formation of CaSiO3-walstromite included in super-deep diamonds. Lithos 2016, 265, 138–147. [Google Scholar] [CrossRef]
- Nestola, F.; Korolev, N.; Kopylova, M.; Rotiroti, N.; Pearson, D.G.; Pamato, M.G.; Alvaro, M.; Peruzzo, L.; Gurney, J.J.; Moore, A.E.; et al. CaSiO3 perovskite in diamond indicates the recycling of oceanic crust into the lower mantle. Nature 2018, 555, 237–241. [Google Scholar] [CrossRef] [Green Version]
- Woodland, A.B.; Girnis, A.V.; Bulatov, V.K.; Brey, G.P.; Hofer, H.E. Breyite inclusions in diamond: Experimental evidence for possible dual origin. Eur. J. Mineral. 2020, 32, 171–185. [Google Scholar] [CrossRef] [Green Version]
- Yang, H.; Prewitt, C.T. On the crystal structure of pseudowollastonite (CaSiO3). Am. Mineral. 1999, 84, 929–932. [Google Scholar] [CrossRef]
- Ringwood, A.E.; Major, A. Some high-pressure transformations of geophysical significance. Earth Planet. Sci. Lett. 1967, 2, 106–110. [Google Scholar] [CrossRef]
- Brenker, F.; Nestola, F.; Brenker, L.; Peruzzo, L.; Secco, L.; Harris, J.W. Breyite, IMA 2018-062, CNMNC Newsletter. Mineral. Mag. 2018, 82, 1225–1232. [Google Scholar]
- Essene, E. High-Pressure transformations in CaSiO3. Contrib. Mineral. Petrol. 1974, 45, 247–250. [Google Scholar] [CrossRef]
- Akaogi, M.; Yano, M.; Tejima, Y.; Iijima, M.; Kojitani, H. High-pressure transitions of diopside and wollastonite: Phase equilibria and thermochemistry of CaMgSi2O6, CaSiO3 and CaSi2O5–CaTiSiO5 system. Phys. Earth Planet. Inter. 2004, 143–144, 145–156. [Google Scholar] [CrossRef]
- Kanazaki, M.; Stebbins, J.F.; Xue, X. Characterization of quenched high pressure phase in CaSiO3 system by XRD and 29Si NMR. Geophys. Res. Lett. 1991, 18, 463–466. [Google Scholar] [CrossRef]
- Sueda, Y.; Irifune, T.; Yamada, A.; Inoue, T.; Liu, X.; Funakoshi, K. The phase boundary between CaSiO3 perovskite and Ca2SiO4+CaSi2O5 determined by in situ X-ray observations. Geophys. Res. Lett. 2006, 33, L10307. [Google Scholar] [CrossRef]
- Chatterjee, N.D.; Johannes, W.; Leistner, H. The system CaO-Al2O3-SiO2-H2O: New phase equilibria data, some calculated phase relations, and their petrological applications. Contrib. Mineral. Petrol. 1984, 88, 1–13. [Google Scholar] [CrossRef]
- Dorogokupets, P.I.; Dymshits, A.M.; Sokolova, T.S.; Danilov, B.S.; Litasov, K.D. The equations of state of forsterite, wadsleyite, ringwoodite, akimotoite, MgSiO3-perovskite, and postperovskite and phase diagram for the Mg2SiO4 system at pressures of up to 130 GPa. Russ. Geol. Geophys. 2015, 56, 172–189. [Google Scholar] [CrossRef]
- Sokolova, T.S.; Dorogokupets, P.I.; Litasov, K.D.; Danilov, B.S.; Dymshits, A.M. Spreadsheets to calculate P-V-T relations, thermodynamic and thermoelastic properties of silicates in the MgSiO3–MgO system. High Press. Res. 2018, 38, 193–211. [Google Scholar] [CrossRef]
- Sokolova, T.S.; Dorogokupets, P.I.; Litasov, K.D. Self-consistent pressure scales based on the equations of state for ruby, diamond, MgO, B2–NaCl as well as Au, Pt, and other metals to 4 Mbar and 3000 K. Russ. Geol. Geophys. 2013, 54, 181–199. [Google Scholar] [CrossRef]
- Sokolova, T.S.; Dorogokupets, P.I.; Dymshits, A.M.; Danilov, B.S.; Litasov, K.D. Microsoft excel spreadsheets to calculate P-V-T relations and thermodynamic properties from equations of state of nine metals, MgO and diamond used as pressure markers in high-pressure and high-temperature experiments. Comput. Geosci. 2016, 94, 162–169. [Google Scholar] [CrossRef]
- Dorogokupets, P.I.; Dymshits, A.M.; Litasov, K.D.; Sokolova, T.S. Thermodynamics and equations of state of iron to 350 GPa and 6000 K. Sci. Rep. 2017, 7, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Litasov, K.D.; Sharygin, I.S.; Shatskii, A.F.; Gavryushkin, P.N.; Dorogokupets, P.I.; Sokolova, T.S.; Ohtani, E.; Dymshits, A.M.; Alifirova, T.A. P–V–T Equations of state for iron carbides Fe3C and Fe7C3 and their relationships under the conditions of the Earth’s mantle and core. Dokl. Earth Sci. 2013, 453, 1269–1273. [Google Scholar] [CrossRef]
- Dorogokupets, P.I.; Sokolova, T.S.; Dymshits, A.M.; Litasov, K.D. Thermodynamic properties of rock-forming oxides, α-Al2O3, Cr2O3, α-Fe2O3, and Fe3O4 at high temperatures and pressures. Geodynam. Tectonophys. 2016, 7, 459–476. [Google Scholar] [CrossRef] [Green Version]
- Magomedov, M.N. Variations in thermal properties of diamond under isothermal compression. Tech. Phys. 2017, 62, 661–668. [Google Scholar]
- Angel, R.J.; Miozzi, F.; Alvaro, M. Limits to the validity of thermal-pressure equations of state. Minerals 2019, 9, 562. [Google Scholar] [CrossRef] [Green Version]
- Katsura, T.; Tange, Y. A simple derivation of the Birch–Murnaghan equations of state (EOSs) and comparison with EOSs derived from other definitions of finite strain. Minerals 2019, 9, 745. [Google Scholar] [CrossRef] [Green Version]
- Khishchenko, K.V. Equation of state for rhenium at high pressures. J. Phys. Conf. Ser. 2020, 1556, 012041. [Google Scholar] [CrossRef]
- Zharkov, V.N.; Kalinin, V.A. Equations of State for Solids at High Pressures and Temperatures; Consuitants Bureau: New York, NY, USA, 1971. [Google Scholar]
- Kunc, K.; Loa, I.; Syassen, K. Equation of state and phonon frequency calculations of diamond at high pressures. Phys. Rev. B 2003, 68, 094107. [Google Scholar] [CrossRef] [Green Version]
- Vinet, P.; Ferrante, J.; Rose, J.H.; Smith, J.R. Compressibility of solids. J. Geophys. Res. 1987, 92, 9319–9325. [Google Scholar] [CrossRef]
- Holzapfel, W.B. Physics of solids under strong compression. Rep. Prog. Phys. 1996, 59, 29–90. [Google Scholar] [CrossRef]
- Birch, F. Finite strain isotherm and velocities for single-crystal and polycrystalline NaCl at high pressures and 300 K. J. Geophys. Res. Solid Earth 1978, 83, 1257–1268. [Google Scholar] [CrossRef]
- Dorogokupets, P.I.; Oganov, A.R. Equations of state of Al, Au, Cu, Pt, Ta, and W and revised ruby pressure scale. Dokl. Earth Sci. 2006, 410, 1091–1095. [Google Scholar] [CrossRef]
- Dorogokupets, P.I.; Oganov, A.R. Ruby, metals, and MgO as alternative pressure scales: A semiempirical description of shock-wave, ultrasonic, X-ray, and thermochemical data at high temperatures and pressures. Phys. Rev. B 2007, 75, 024115. [Google Scholar] [CrossRef]
- Al’tshuler, L.V.; Brusnikin, S.E.; Kuz’menkov, E.A. Isotherms and Gruneisen functions of 25 metals. J. Appl. Mech. Tech. Phys. 1987, 28, 129–141. [Google Scholar] [CrossRef]
- Schmetterer, C.; Masset, P.J. Heat capacity of compounds in the CaO-SiO2 System—A Review. J. Phase Equilib. Diffus. 2012, 33, 261–275. [Google Scholar] [CrossRef]
- Swamy, V.; Dubrovinsky, L.S.; Tutti, F. High-temperature Raman spectra and thermal expansion of wollastonite. J. Am. Ceram. Soc. 1997, 80, 2237–2247. [Google Scholar] [CrossRef]
- Vaiday, S.N.; Bailey, S.; Pasternack, T.; Kennedy, G.C. Compressibility of fifteen minerals to 45 kilobars. J. Geophys. Res. 1973, 78, 6893–6898. [Google Scholar] [CrossRef]
- Yang, H.; Prewitt, C.T. Crystal structure and compressibility of a two-layer polytype of pseudowollastonite (CaSiO3). Am. Mineral. 1999, 84, 1902–1905. [Google Scholar] [CrossRef]
- Southard, J.C. A modified calorimeter for high temperatures. The heat content of silica, wollastonite and thorium dioxide above 25°. J. Am. Chem. Soc. 1941, 63, 3142–3146. [Google Scholar] [CrossRef]
- Krupka, K.M.; Robie, R.A.; Hemingway, B.S.; Kerrick, D.M. Low-temperature heat capacities an derived thermodynamic properties of anthophyllite, diopside, enstatite, bronzite, and wollastonite. Am. Mineral. 1985, 70, 249–260. [Google Scholar]
- Krupka, K.M.; Robie, R.A.; Hemingway, B.S.; Kerrick, D.M. High-temperature heat capacities and derived thermodynamic properties of anthophyllite, diopside, dolomite, enstatite, bronzite, talc, tremolite, and wollastonite. Am. Mineral. 1985, 70, 261–271. [Google Scholar]
- Richet, P.; Fiquet, G. High-Temperature heat capacity and premelting of minerals in the system MgO-CaO-Al2O3-SiO2. J. Geophys. Res. 1991, 96, 445–456. [Google Scholar] [CrossRef]
- Holland, T.J.B.; Powell, R. An improved and extended internally consistent thermodynamic dataset for phases of petrological interest, involving a new equation of state for solids. J. Metamorph. Geol. 2011, 29, 333–383. [Google Scholar] [CrossRef]
- Richet, P.; Mysen, B.O.; Ingrib, J. High-temperature X-ray diffraction and Raman spectroscopy of diopside and pseudowollastonite. Phys. Chem. Miner. 1998, 25, 401–414. [Google Scholar] [CrossRef]
- White, W.P. Silicate specific heats, Second series. Am. J. Sci. 1919, 47, 1–44. [Google Scholar] [CrossRef]
- Parks, G.S.; Kelley, K.K. The heat capacity of calcium silicate. J. Phys. Chem. 1926, 30, 1175–1178. [Google Scholar] [CrossRef]
- Berman, R.G.; Brown, T.H. Heat capacity of minerals in the system Na2O-K2O-CaO-MgO-FeO-Fe2O3-A12O3-SiO2-TiO2-H2O-CO2: Representation, estimation, and high temperature extrapolation. Contrib. Mineral. Petrol. 1985, 89, 168–183. [Google Scholar] [CrossRef]
- Huang, W.-L.; Wyllie, P.J. Melting and subsolidus phase relationships for CaSiO3 to 35 kilobars pressure. Am. Mineral. 1975, 60, 213–217. [Google Scholar]
- Joswig, W.; Paulus, E.F.; Winkler, B.; Milman, V. The crystal structure of CaSiO3-walstromite, a special isomorph of wollastonite-II. Z. Krist. 2003, 218, 811–818. [Google Scholar] [CrossRef]
- Liu, X.; Wang, S.; He, Q.; Chen, J.; Wang, H.; Li, S.; Peng, F.; Zhang, L.; Fei, Y. Thermal elastic behavior of CaSiO3-walstromite: A powder X-ray diffraction study up to 900 °C. Am. Mineral. 2012, 97, 262–267. [Google Scholar] [CrossRef]
- Remy, C.; Andrault, D.; Madon, M. High-temperature, high-pressure X-ray investigation of dicalcium silicate. J. Am. Ceram. Soc. 1997, 80, 851–860. [Google Scholar] [CrossRef]
- Yamnova, N.A.; Zubkova, N.V.; Eremin, N.N.; Zadov, A.E.; Gazeev, V.M. Crystal structure of larnite β-Ca2SiO4 and specific features of polymorphic transitions in dicalcium orthosilicate. Crystallogr. Rep. 2011, 56, 210–220. [Google Scholar] [CrossRef]
- Tilley, C.E. On larnite (calcium orthosilicate, a new mineral) and its associated minerals from the limestone contact-zone of Scawt Hill, Co. Antrim. Mineral. Mag. 1929, 22, 77–86. [Google Scholar] [CrossRef]
- Todd, S.S. Low-temperature heat capacities and entropies at 298.16 °K of crystalline calcium orthosilicate, zinc orthosilicate and tricalcium silicate. J. Am. Chem. Soc. 1951, 73, 3277–3278. [Google Scholar] [CrossRef]
- Coughlin, J.P.; O’Brien, C.J. High temperature heat contents of calcium orthosilicate. J. Phys. Chem. 1957, 61, 767–769. [Google Scholar] [CrossRef]
- Fukuda, K.; Maki, I.; Ito, S. Anisotropic thermal expansion of β-Ca2SiO4 monoclinic crystal. J. Am. Ceram. Soc. 1997, 80, 1595–1598. [Google Scholar] [CrossRef]
- Xiong, Z.; Liu, X.; Shieh, S.R.; Wang, S.; Chang, L.; Tang, J.; Hong, X.; Zhang, Z.; Wang, H. Some thermodynamic properties of larnite (β-Ca2SiO4) constrained by high T/P experiment and/or theoretical simulation. Am. Miner. 2016, 101, 277–288. [Google Scholar] [CrossRef]
- Angel, R.J.; Kunz, M.; Miletich, R.; Woodland, A.B.; Koch, M.; Knoche, R.L. Effect of isovalent Si, Ti substitution on the bulk moduli of Ca(Ti1-xSix)SiO5 titanites. Am. Miner. 1999, 84, 282–287. [Google Scholar] [CrossRef]
- Ringwood, A.E.; Major, A. Synthesis of majorite and other high pressure garnets and perovskites. Earth Planet. Sci. Lett. 1971, 12, 411–418. [Google Scholar] [CrossRef]
- Sun, N.; Mao, Z.; Yan, S.; Wu, X.; Prakapenka, V.B.; Lin, J.-F. Confirming a pyrolitic lower mantle using self-consistent pressure scales and new constraints on CaSiO3 perovskite. Phys. Res. Solid Earth 2016, 121, 4876–4894. [Google Scholar] [CrossRef] [Green Version]
- Chen, H.; Shim, S.-H.; Leinenweber, K.; Prakapenka, V.; Meng, Y.; Prescher, C. Crystal structure of CaSiO3 perovskite at 28–62 GPa and 300 K under quasi-hydrostatic stress conditions. Am. Miner. 2018, 103, 462–468. [Google Scholar] [CrossRef]
- Kojitani, H.; Navrotsky, A.; Akaogi, M. Calorimetric study of perovskite solid solutions in the CaSiO3-CaGeO3 system. Phys. Chem. Miner. 2001, 28, 413–420. [Google Scholar] [CrossRef]
- Noguchi, M.; Komabayashi, T.; Hirose, K.; Ohishi, Y. High-temperature compression experiments of CaSiO3 perovskite to lowermost mantle conditions and its thermal equation of state. Phys. Chem. Miner. 2013, 40, 81–91. [Google Scholar] [CrossRef]
- Greaux, S.; Irifune, T.; Higo, Y.; Tange, Y.; Arimoto, T.; Liu, Z.; Yamada, A. Sound velocity of CaSiO3 perovskite suggests the presence of basaltic crust in the Earth’s lower mantle. Nature 2019, 565, 218–221. [Google Scholar] [CrossRef]
- Dorogokupets, P.I.; Dewaele, A. Equations of state of MgO, Au, Pt, NaCl-B1, and NaCl-B2: Internally consistent high-temperature pressure scales. High Press. Res. 2007, 27, 431–446. [Google Scholar] [CrossRef]
- Fei, Y.W.; Ricolleau, A.; Frank, M.; Mibe, K.; Shen, G.Y.; Prakapenka, V. Toward an internally consistent pressure scale. Proc. Natl. Acad. Sci. USA 2007, 104, 9182–9186. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Matsui, M.; Higo, Y.; Okamoto, Y.; Irifune, T.; Funakoshi, K.I. Simultaneous sound velocity and density measurements of NaCl at high temperatures and pressures: Application as a primary pressure standard. Am. Miner. 2012, 97, 1670–1675. [Google Scholar] [CrossRef]
- Mao, H.K.; Chen, L.C.; Hemley, R.J.; Jephcoat, A.P.; Wu, Y. Stability and equation of state of CaSiO3-perovskite to 134 GPa. J. Geophys. Res. 1989, 94, 17889–17894. [Google Scholar] [CrossRef]
- Shim, S.-H.; Duffy, T.S.; Shen, G. The equation of state of CaSiO3 perovskite to 108 GPa at 300 K. Phys. Earth Planet. Inter. 2000, 120, 327–338. [Google Scholar] [CrossRef]
- Wang, Y.; Weidner, D.J.; Guyot, F. Thermal equation of state of CaSiO3 perovskite. J. Geophys. Res. 1996, 101, 661–672. [Google Scholar] [CrossRef]
- Shim, S.-H.; Duffy, T.; Shen, G. The stability and P-V-T equation of state of CaSiO3 perovskite in the Earth’s lower mantle. J. Geophys. Res. 2000, 105, 25955–25968. [Google Scholar] [CrossRef]
- Wang, Y.; Weidner, D.J. Thermoelasticity of CaSiO3 perovskite and implications for the lower mantle. Geophys. Res. Lett. 1994, 21, 895–898. [Google Scholar] [CrossRef]
- Stixrude, L.; Lithgow-Bertelloni, C.; Kiefer, B.; Fumagalli, P. Phase stability and shear softening in CaSiO3 perovskite at high pressure. Phys. Rew. B 2007, 75, 024108. [Google Scholar] [CrossRef] [Green Version]
- Li, L.; Weidner, D.J.; Brodholt, J.; Alfe, D.; Price, G.D.; Caracas, R.; Wentzcovitch, R. Elasticity of CaSiO3 perovskite at high pressure and high temperature. Phys. Earth Planet. Inter. 2006, 155, 249–259. [Google Scholar] [CrossRef]
- Caracas, R.; Wentzcovitch, R.; Price, G.D.; Brodholt, J. CaSiO3 perovskite at lower mantle pressures. Geophys. Res. Lett. 2005, 32, L06306. [Google Scholar] [CrossRef] [Green Version]
- Karki, B.B.; Crain, J. First-principles determination of elastic properties of CaSiO3 perovskite at lower mantle pressures. Geophys. Res. Lett. 1998, 25, 2741–2744. [Google Scholar] [CrossRef]
- Adams, D.J.; Oganov, A. Ab initio molecular dynamics study of CaSiO3 perovskite at P-T conditions of Earth’s lower mantle. Phys. Rev. B 2006, 73, 184106. [Google Scholar] [CrossRef]
- Shen, G.; Lazor, P. Measurement of melting temperatures of some minerals under lower mantle pressures. J. Geophys. Res. 1995, 100, 17699–17713. [Google Scholar] [CrossRef]
- Zerr, A.; Serghiou, G.; Boehler, R. Melting of CaSiO3 perovskite to 430 kbar and first in-situ measurements of lower mantle eutectic temperatures. Geophys. Res. Lett. 1997, 24, 909–912. [Google Scholar] [CrossRef]
- Nomura, R.; Zhou, Y.; Irifune, T. Melting phase relations in the MgSiO3–CaSiO3 system at 24 GPa. Prog. Earth Planet. Sci. 2017, 4, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Braithwaite, J.; Stixrude, L. Melting of CaSiO3 perovskite at high pressure. Geophys. Res. Lett. 2019, 46, 2037–2044. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dziewonski, A.; Anderson, D. Preliminary reference Earth model. Phys. Earth Planet. Inter. 1981, 25, 297–356. [Google Scholar] [CrossRef]
- Stacey, F.D.; Davis, P.M. High pressure equations of state with applications to the lower mantle and core. Phys. Earth Planet. Inter. 2004, 142, 137–184. [Google Scholar] [CrossRef]
- Irifune, T.; Shinmei, T.; McCammon, C.A.; Miyajima, N.; Rubie, D.C.; Frost, D.J. Iron partitioning and density changes of pyrolite in Earth’s lower mantle. Science 2010, 327, 193–195. [Google Scholar] [CrossRef] [PubMed]
- Katsura, T.; Yoneda, A.; Yamazaki, D.; Yoshino, T.; Ito, E. Adiabatic temperature profile in the mantle. Phys. Earth Planet. Inter. 2010, 183, 212–218. [Google Scholar] [CrossRef]
- Sokolova, T.S.; Seredkina, A.I.; Dorogokupets, P.I. Density patterns of the upper mantle under Asia and the Arctic: Comparison of thermodynamic modeling and geophysical data. Pure Appl. Geophys. 2020, 177, 4289–4307. [Google Scholar] [CrossRef]
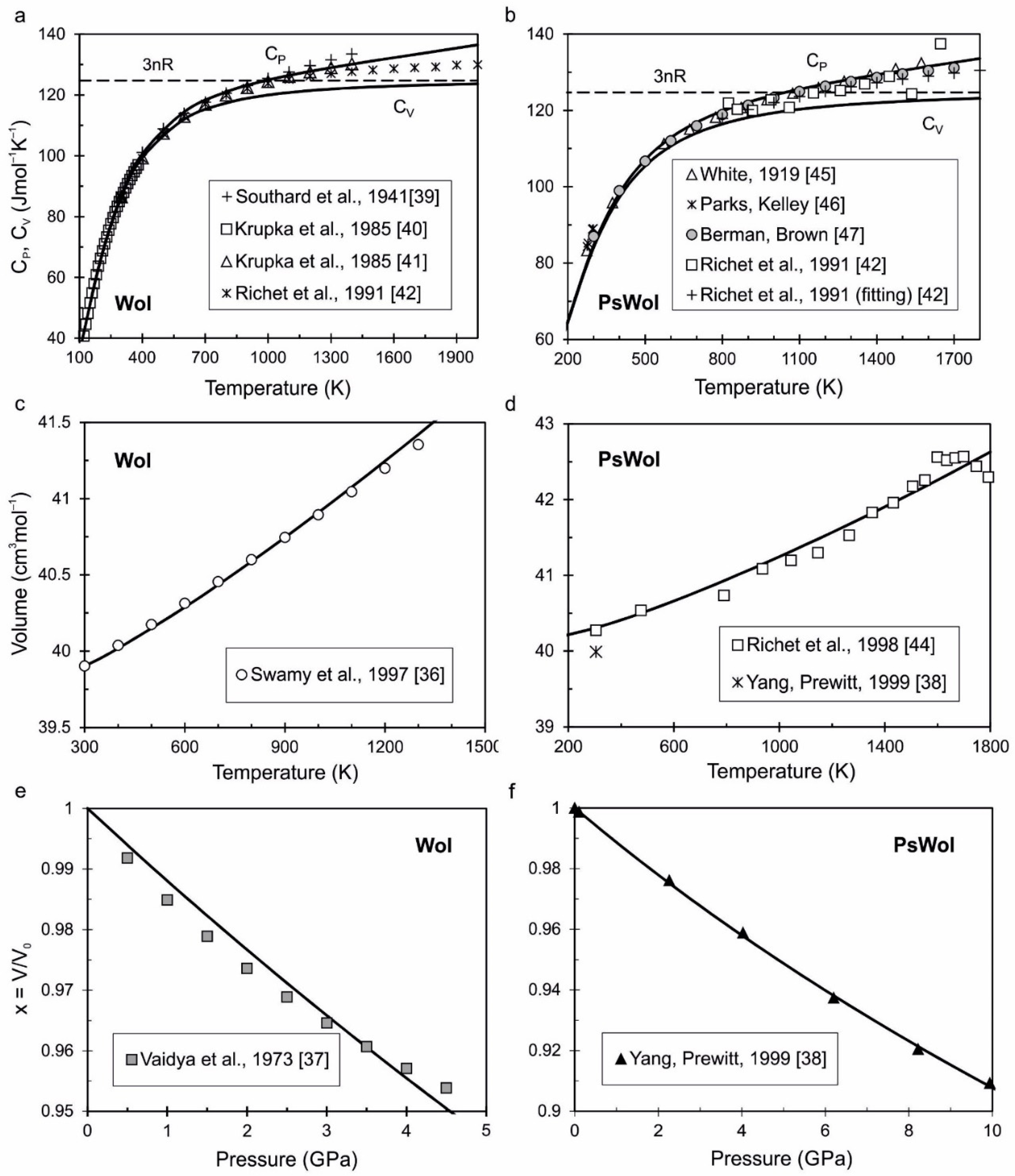






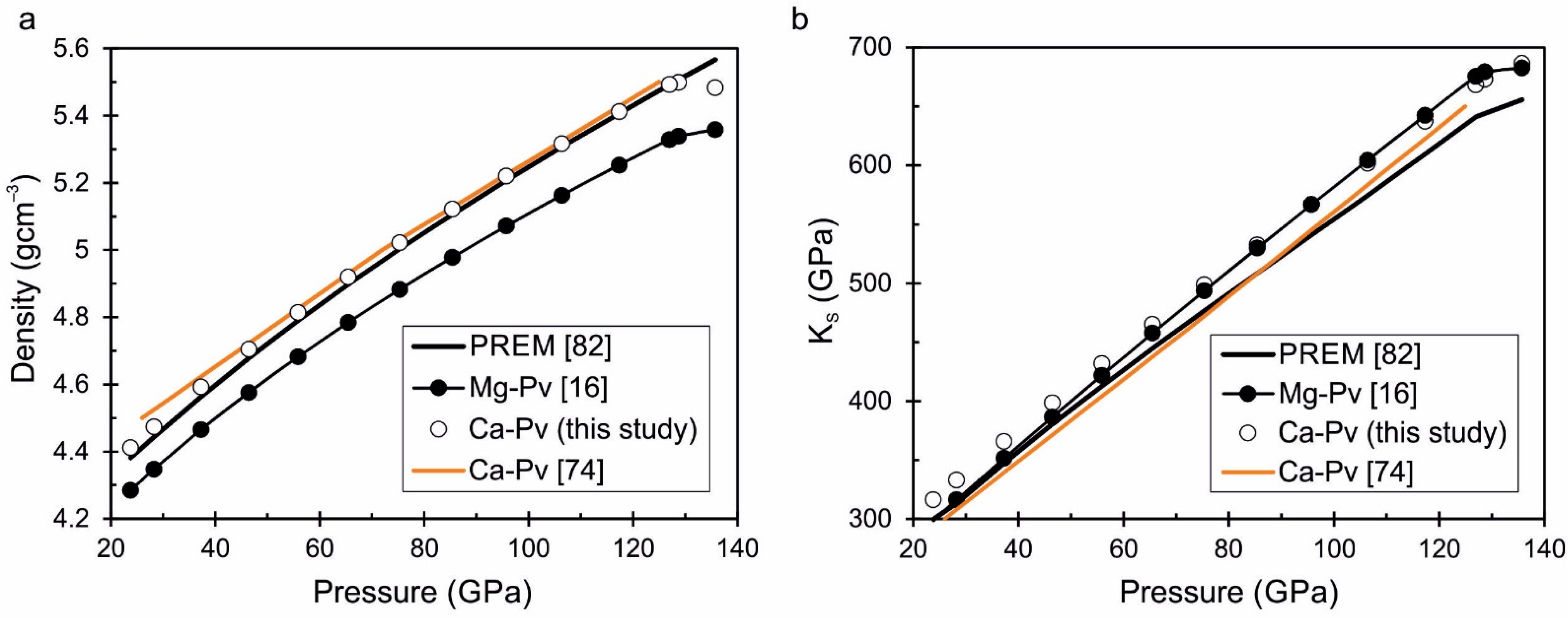
| Parameters | Wol CaSiO3 | PsWol CaSiO3 | Brt CaSiO3 | β-Lrn Ca2SiO4 | Ca-Tit CaSi2O5 | Ca-Pv CaSiO3 |
|---|---|---|---|---|---|---|
| 𝓤0, kJmol−1 | −1658.007 | −1655.531 | −1651.400 | −2344.025 | −2524.000 | −1567.400 |
| V0, cm3mol−1 | 39.9 | 40.3 | 37.78 | 51.88 | 48.19 | 27.403 |
| K0, GPa | 81.5 | 86 | 78.6 | 105 | 178 | 239 |
| K’ | 3.28 | 3.8 | 4 | 6.27 | 4 | 4.13 |
| k | 5 | 5 | 5 | 5 | 5 | 5 |
| Θ01, K | 966 | 1039 | 952 | 838 | 975 | 935 |
| m1 | 7.5 | 7.5 | 7.5 | 10.5 | 12 | 7.5 |
| Θ02, K | 270 | 244 | 280 | 257 | 370 | 388 |
| m2 | 7.5 | 7.5 | 7.5 | 10.5 | 12 | 7.5 |
| γ0 | 1 | 1 | 0.745 | 1.263 | 1.3 | 1.913 |
| γ∞ | 0 | 0 | 0 | 0 | 0 | 0 |
| β | 1 | 1 | 1 | 1.041 | 1 | 0.486 |
| a0, 10−6 K−1 | 20.8 | |||||
| m | 1 |
| P, GPa | T, K | V, cm3 mol−1 | α × 106, K−1 | S, Jmol−1 K−1 | CP, Jmol−1 K−1 | CV, Jmol−1 K−1 | KT, GPa | KS, GPa | ΔG, kJmol−1 |
|---|---|---|---|---|---|---|---|---|---|
| 0.0001 | 298.15 | 39.900 | 26.458 | 81.358 | 86.715 | 86.037 | 81.50 | 82.14 | −1658.007 |
| 0.0001 | 500 | 40.147 | 33.714 | 132.366 | 108.976 | 107.159 | 79.66 | 81.01 | −1679.826 |
| 0.0001 | 1000 | 40.907 | 40.288 | 214.108 | 124.900 | 119.946 | 74.62 | 77.70 | −1768.341 |
| 0.0001 | 1398 | 41.596 | 43.519 | 256.827 | 130.068 | 122.310 | 70.44 | 74.91 | −1862.474 |
| 0.0001 | 2000 | 42.763 | 48.446 | 304.482 | 136.431 | 123.596 | 63.94 | 70.58 | −2032.169 |
| 3 | 298.15 | 38.536 | 23.175 | 78.447 | 84.890 | 84.327 | 91.20 | 91.81 | −1540.406 |
| 3 | 500 | 38.746 | 29.732 | 128.623 | 107.621 | 106.089 | 89.43 | 90.72 | −1561.542 |
| 3 | 1000 | 39.393 | 35.446 | 209.562 | 123.771 | 119.586 | 84.55 | 87.51 | −1647.956 |
| 3 | 1500 | 40.130 | 38.619 | 260.949 | 129.599 | 122.464 | 79.48 | 84.11 | −1766.352 |
| 3 | 2000 | 40.944 | 41.668 | 298.850 | 134.050 | 123.490 | 74.28 | 80.63 | −1906.702 |
| Pressure Range, GPa | Temperature Range, K | Method | Reference |
|---|---|---|---|
| 0–134 | 300 | XRD | [68] |
| 16.8–108 | 300 | XRD | [69] |
| 28–62 | 300 | XRD | [61] |
| 0.59–13 | 301–1600 | XRD | [70] |
| 18–96 | 1238–2419 | XRD | [71] |
| 51–130 | 700–2300 | XRD | [63] |
| 21.2–156 | 1200–2600 | XRD | [60] |
| 12–23 | 700–1700 | XRD | [64] |
| Depth, km | P, GPa | T, K | Density, gcm−3 | Ks, GPa |
|---|---|---|---|---|
| 2891 | 135.75 | 3739 | 5.483 | 686.56 |
| 2771 | 128.71 | 2838 | 5.499 | 673.01 |
| 2741 | 126.97 | 2740 | 5.492 | 668.35 |
| 2571 | 117.35 | 2668 | 5.412 | 637.46 |
| 2371 | 106.39 | 2596 | 5.317 | 601.82 |
| 2171 | 95.76 | 2525 | 5.220 | 566.86 |
| 1971 | 85.43 | 2452 | 5.122 | 532.51 |
| 1771 | 75.36 | 2379 | 5.022 | 498.60 |
| 1571 | 65.52 | 2302 | 4.920 | 465.04 |
| 1371 | 55.9 | 2227 | 4.814 | 431.71 |
| 1171 | 46.46 | 2144 | 4.705 | 398.53 |
| 971 | 37.29 | 2060 | 4.592 | 365.70 |
| 771 | 28.29 | 1974 | 4.474 | 332.80 |
| 670 | 23.83 | 1931 | 4.412 | 316.19 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sokolova, T.S.; Dorogokupets, P.I. Equations of State of Ca-Silicates and Phase Diagram of the CaSiO3 System under Upper Mantle Conditions. Minerals 2021, 11, 322. https://doi.org/10.3390/min11030322
Sokolova TS, Dorogokupets PI. Equations of State of Ca-Silicates and Phase Diagram of the CaSiO3 System under Upper Mantle Conditions. Minerals. 2021; 11(3):322. https://doi.org/10.3390/min11030322
Chicago/Turabian StyleSokolova, Tatiana S., and Peter I. Dorogokupets. 2021. "Equations of State of Ca-Silicates and Phase Diagram of the CaSiO3 System under Upper Mantle Conditions" Minerals 11, no. 3: 322. https://doi.org/10.3390/min11030322







