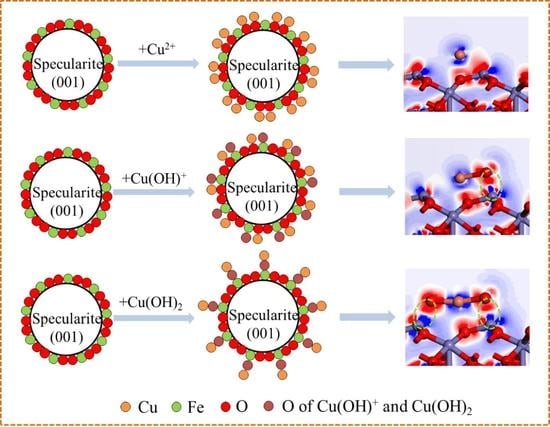
Study of the Effect of Absorbed Cu Species on the Surface of Specularite (0 0 1) by the DFT Calculations
Abstract
Share and Cite
Huangfu, M.; Li, J.; Zhang, X.; Hu, Y.; Deng, J.; Wang, Y.; Wei, P. Study of the Effect of Absorbed Cu Species on the Surface of Specularite (0 0 1) by the DFT Calculations. Minerals 2021, 11, 930. https://doi.org/10.3390/min11090930
Huangfu M, Li J, Zhang X, Hu Y, Deng J, Wang Y, Wei P. Study of the Effect of Absorbed Cu Species on the Surface of Specularite (0 0 1) by the DFT Calculations. Minerals. 2021; 11(9):930. https://doi.org/10.3390/min11090930
Chicago/Turabian StyleHuangfu, Mingzhu, Jiaxin Li, Xi Zhang, Yiming Hu, Jiushuai Deng, Yu Wang, and Pingping Wei. 2021. "Study of the Effect of Absorbed Cu Species on the Surface of Specularite (0 0 1) by the DFT Calculations" Minerals 11, no. 9: 930. https://doi.org/10.3390/min11090930
APA StyleHuangfu, M., Li, J., Zhang, X., Hu, Y., Deng, J., Wang, Y., & Wei, P. (2021). Study of the Effect of Absorbed Cu Species on the Surface of Specularite (0 0 1) by the DFT Calculations. Minerals, 11(9), 930. https://doi.org/10.3390/min11090930