Validation of Bromide Leaching through Response Surface Methodology and Separation of Gold from Waste Printed Circuit Boards
Abstract
1. Introduction
2. Experimental Section
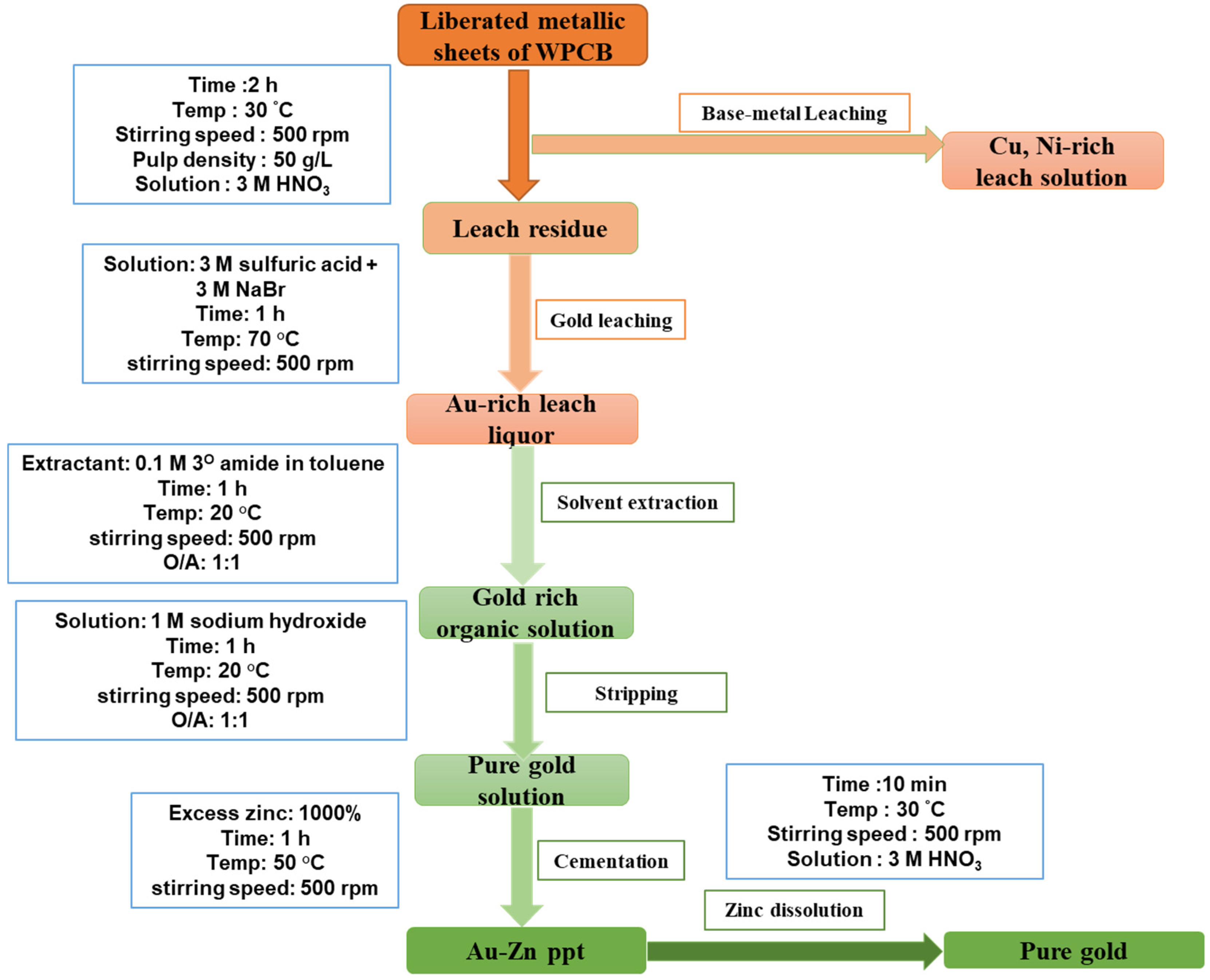
2.1. Pre-Treatment and Removal of Base Metals
2.2. Experimental Design for Gold Leaching
Gold Leaching
2.3. Recovery and Separation of Gold
3. Results and Discussion
3.1. Validation of Bromide Leaching through Response Surface Methodology
3.2. Kinetics of Leaching
3.3. Liquid–Liquid Extraction and Separation of Gold from Leach Liquor
- (a)
- Gold concentration in 5 g of WPCB metal clad: 2.5 mg;
- (b)
- After base metal leaching: 2.5 mg;
- (c)
- After gold leaching (90.73%): 2.27 mg;
- (d)
- After solvent extraction (99% recovery) and stripping (99.9%): 2.25 mg;
- (e)
- After cementation with zinc powder: 2.245 mg;
- (f)
- After zinc dissolution: 2.245 mg.
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mowafy, E.A.; Mohamed, D. Extraction and Separation of Gold(III) from Hydrochloric Acid Solutions Using Long Chain Structurally Tailored Monoamides. Sep. Purif. Technol. 2016, 167, 146–153. [Google Scholar] [CrossRef]
- Hagelüken, C.; Corti, C.W. Recycling of Gold from Electronics: Cost-Effective Use through “Design for Recycling”. Gold Bull. 2010, 43, 209–220. [Google Scholar] [CrossRef]
- Rao, M.D.; Singh, K.K.; Morrison, C.A.; Love, J.B. Challenges and Opportunities in the Recovery of Gold from Electronic Waste. RSC Adv. 2020, 10, 4300–4309. [Google Scholar] [CrossRef]
- Cui, J.; Forssberg, E. Mechanical Recycling of Waste Electric and Electronic Equipment: A Review. J. Hazard. Mater. 2003, 99, 243–263. [Google Scholar] [CrossRef]
- Chaurasia, A.; Singh, K.K.; Mankhand, T.R. Extraction of Tin and Copper by Acid Leaching of PCBs. Int. J. Metall. Eng. 2013, 2, 243–248. [Google Scholar] [CrossRef]
- Bas, A.D.; Deveci, H.; Yazici, E.Y. Treatment of Manufacturing Scrap TV Boards by Nitric Acid Leaching. Sep. Purif. Technol. 2014, 130, 151–159. [Google Scholar] [CrossRef]
- Abhilash; Tabassum, S.; Ghosh, A.; Meshram, P.; van Hullebusch, E.D. Microbial Processing of Waste Shredded PCBs for Copper Extraction Cum Separation—Comparing the Efficacy of Bacterial and Fungal Leaching Kinetics and Yields. Metals 2021, 11, 317. [Google Scholar] [CrossRef]
- Groenewald, T. Potential Application of Thiourea in the Processing of Gold. J. S. Afr. Inst. Min. Metall. 1977, 77, 217–223. [Google Scholar]
- Syed, S. Recovery of Gold from Secondary Sources-A Review. Hydrometallurgy 2012, 115–116, 30–51. [Google Scholar] [CrossRef]
- Barbosa-Filho, O.; Monhemius, A.J. Leaching of Gold in Thiocyanate Solutions-Part 3: Rates and Mechanism of Gold Dissolution. Trans. Inst. Min. Metall. Sect. C Miner. Process. Extr. Metall. 1994, 103, c117–c125. [Google Scholar]
- Oraby, E.A.; Li, H.; Eksteen, J.J. An Alkaline Glycine-Based Leach Process of Base and Precious Metals from Powdered Waste Printed Circuit Boards. Waste Biomass Valorization 2020, 11, 3897–3909. [Google Scholar] [CrossRef]
- Rao, M.D.; Singh, K.K.; Morrison, C.A.; Love, J.B. Recycling Copper and Gold from E-Waste by a Two-Stage Leaching and Solvent Extraction Process. Sep. Purif. Technol. 2021, 263, 118400. [Google Scholar] [CrossRef]
- Harjanto, S.; Pratama, F.W.; Lazuardiyani, A.; Taris, M.; Salam, M.Y. Additional of NaCl on Chloride Leaching of Gold Ore from Indonesian Artisanal Mining. IOP Conf. Ser. Mater. Sci. Eng. 2019, 515, 012032. [Google Scholar] [CrossRef]
- Li, C.; Li, H.; Yang, X.; Wang, S.; Zhang, L. Gold Leaching from a Refractory Gold Concentrate by the Method of Liquid Chlorination. Rare Met. Technol. 2015, 71–77. [Google Scholar] [CrossRef]
- Cui, H.; Anderson, C. Hydrometallurgical Treatment of Waste Printed Circuit Boards: Bromine Leaching. Metals 2020, 10, 462. [Google Scholar] [CrossRef]
- Melashvili, M.; Fleming, C.; Dymov, I.; Manimaran, M.; O’Day, J. Study of Gold Leaching with Bromine and Bromide and the Influence of Sulphide Minerals on This Reaction. In Proceedings of the Conference of Metallurgists (COM 2014), Vancouver, BC, Canada, 28 September–1 October 2014. [Google Scholar]
- Encinas-Romero, M.A.; Tiburcio-Munive, G.; Yánez-Montaño, M. A Kinetic Study of Gold Leaching in CuBr2-NaBr System. J. Multidiscip. Eng. Sci. Technol. 2015, 2, 2118–2121. [Google Scholar]
- Nguyen, H.H.; Tran, T.; Wong, P. A Kinetic Study of the Cementation of Gold from Cyanide Solutions onto Copper. Hydrometallurgy 1997, 46, 55–69. [Google Scholar] [CrossRef]
- Gomes, C.P.; Almeida, M.F.; Loureiro, M. Gold Recovery with Ion Exchange Used Resins. Sep. Purif. Technol. 2001, 24, 35–57. [Google Scholar] [CrossRef]
- Nakbanpote, W.; Thiravetyan, P.; Kalambahetf, C. Preconcentration of Gold by Rice Husk Ash. Miner. Eng. 2000, 13, 391–400. [Google Scholar] [CrossRef]
- Doidge, E.D.; Carson, I.; Tasker, P.A.; Ellis, R.J.; Morrison, C.A.; Love, J.B. A Simple Primary Amide for the Selective Recovery of Gold from Secondary Resources. Angew. Chem.-Int. Ed. 2016, 55, 12436–12439. [Google Scholar] [CrossRef]
- Doidge, E.D.; Kinsman, L.M.M.; Ji, Y.; Carson, I.; Duffy, A.J.; Kordas, I.A.; Shao, E.; Tasker, P.A.; Ngwenya, B.T.; Morrison, C.A.; et al. Evaluation of Simple Amides in the Selective Recovery of Gold from Secondary Sources by Solvent Extraction. ACS Sustain. Chem. Eng. 2019, 7, 15019–15029. [Google Scholar] [CrossRef]
- Mpinga, C.N.; Bradshaw, S.M.; Akdogan, G.; Snyders, C.A.; Eksteen, J.J. Evaluation of the Merrill-Crowe Process for the Simultaneous Removal of Platinum, Palladium and Gold from Cyanide Leach Solutions. Hydrometallurgy 2014, 142, 36–46. [Google Scholar] [CrossRef]
- Muhtadi, O.A. Metal Extraction (Recovery Systems). In Introduction to Evaluation, Design and Operation of Precious Metal Heap Leaching Projects; Society of Mining Engineers, Inc.: Englewood, CO, USA, 1988; pp. 124–136. [Google Scholar]
- Rao, M.D.; Shahin, C.; Jha, R. Optimization of Leaching of Copper to Enhance the Recovery of Gold from Liberated Metallic Layers of WPCBs. Mater. Today Proc. 2021, 46, 1515–1518. [Google Scholar] [CrossRef]
- Nayebzadeh, H.; Saghatoleslami, N.; Tabasizadeh, M. Optimization of the Activity of KOH/Calcium Aluminate Nanocatalyst for Biodiesel Production Using Response Surface Methodology. J. Taiwan Inst. Chem. Eng. 2016, 68, 379–386. [Google Scholar] [CrossRef]
- Somasundaram, M.; Saravanathamizhan, R.; Ahmed Basha, C.; Nandakumar, V.; Nathira Begum, S.; Kannadasan, T. Recovery of Copper from Scrap Printed Circuit Board: Modelling and Optimization Using Response Surface Methodology. Powder Technol. 2014, 266, 1–6. [Google Scholar] [CrossRef]
- Faraji, F.; Alizadeh, A.; Rashchi, F.; Mostoufi, N. Kinetics of Leaching: A Review. Rev. Chem. Eng. 2020, 38, 113–148. [Google Scholar] [CrossRef]
- Kim, E.Y.; Kim, M.S.; Lee, J.C.; Jeong, J.; Pandey, B.D. Leaching Kinetics of Copper from Waste Printed Circuit Boards by Electro-Generated Chlorine in HCl Solution. Hydrometallurgy 2011, 107, 124–132. [Google Scholar] [CrossRef]












| Element | Cu | Ni | Au | Ag | Sn | Zn | Cd | Pb | Ceramic and Plastic Residue |
|---|---|---|---|---|---|---|---|---|---|
| Delaminated metal clad (wt%) | 84 | 2.3 | 0.05 | 0.03 | 0.4 | 0.4 | 0.2 | 0.1 | 12.4 |
| Parameter | Units | Low (−1) | High (+1) | −α | +α |
|---|---|---|---|---|---|
| Time (A) | min | 10 | 60 | 10 | 60 |
| Temperature (B) | °C | 30 | 90 | 30 | 90 |
| Stirring speed (C) | rpm | 100 | 500 | 100 | 500 |
| Parameter 1 | Parameter 2 | Parameter 3 | Response 1 | Error (%) | |
|---|---|---|---|---|---|
| Run | A:Time | B:Temp | C:Stirring Speed | Gold Leaching | |
| min | °C | rpm | % | ||
| 1 | 35 | 60 | 500 | 87 | 1 |
| 2 | 35 | 60 | 300 | 85 | - |
| 3 | 10 | 90 | 100 | 73 | 1 |
| 4 | 35 | 60 | 300 | 84 | - |
| 5 | 60 | 90 | 100 | 86 | 1 |
| 6 | 60 | 90 | 500 | 88 | 0 |
| 7 | 35 | 60 | 300 | 84 | - |
| 8 | 10 | 90 | 500 | 75 | 0 |
| 9 | 35 | 60 | 300 | 85 | - |
| 10 | 35 | 60 | 100 | 83 | 0 |
| 11 | 35 | 90 | 300 | 84 | 0 |
| 12 | 35 | 60 | 300 | 86 | - |
| 13 | 10 | 30 | 500 | 68 | 1 |
| 14 | 35 | 30 | 300 | 75 | 0 |
| 15 | 10 | 60 | 300 | 73 | 1 |
| 16 | 60 | 30 | 500 | 82 | 2 |
| 17 | 60 | 30 | 100 | 80 | 1 |
| 18 | 10 | 30 | 100 | 66 | 1 |
| 19 | 60 | 60 | 300 | 89 | 2 |
| 20 | 35 | 60 | 300 | 85 | - |
| Source | Sum of Squares | df | Mean Square | F-Value | p-Value | |
|---|---|---|---|---|---|---|
| Model | 863.49 | 9 | 95.94 | 93.07 | <0.0001 | significant |
| A-Time | 490.00 | 1 | 490.00 | 475.31 | <0.0001 | |
| B-Temp | 122.50 | 1 | 122.50 | 118.83 | <0.0001 | |
| C-Stirring speed | 14.40 | 1 | 14.40 | 13.97 | 0.0039 | |
| AB | 0.5000 | 1 | 0.5000 | 0.4850 | 0.5020 | |
| AC | 0.0000 | 1 | 0.0000 | 0.0000 | 1.0000 | |
| BC | 0.0000 | 1 | 0.0000 | 0.0000 | 1.0000 | |
| A2 | 30.28 | 1 | 30.28 | 29.37 | 0.0003 | |
| B2 | 63.84 | 1 | 63.84 | 61.93 | <0.0001 | |
| C2 | 1.28 | 1 | 1.28 | 1.24 | 0.2915 | |
| Residual | 10.31 | 10 | 1.03 | |||
| Lack of Fit | 7.48 | 5 | 1.50 | 2.64 | 0.1553 | not significant |
| Pure Error | 2.83 | 5 | 0.5667 | |||
| Cor Total | 873.80 | 19 |
| S. No. | Name of the Factor | % Leaching | |||
|---|---|---|---|---|---|
| 1. | Time (min) | Temperature (°C) | Stirring speed (rpm) | Experimental | Calculated |
| 2. | 60 | 70 | 499 | 93 | 90.73 |
| S. No. | Concentration of Gold (ppm) | |
|---|---|---|
| 1. | Strip solution (Before cementation) | 409 |
| 2. | Solution after cementation | 0.74 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rao, M.D.; Meshram, P.; Abhilash; Singh, K.K. Validation of Bromide Leaching through Response Surface Methodology and Separation of Gold from Waste Printed Circuit Boards. Minerals 2023, 13, 579. https://doi.org/10.3390/min13040579
Rao MD, Meshram P, Abhilash, Singh KK. Validation of Bromide Leaching through Response Surface Methodology and Separation of Gold from Waste Printed Circuit Boards. Minerals. 2023; 13(4):579. https://doi.org/10.3390/min13040579
Chicago/Turabian StyleRao, Mudila Dhanunjaya, Pratima Meshram, Abhilash, and Kamalesh K. Singh. 2023. "Validation of Bromide Leaching through Response Surface Methodology and Separation of Gold from Waste Printed Circuit Boards" Minerals 13, no. 4: 579. https://doi.org/10.3390/min13040579
APA StyleRao, M. D., Meshram, P., Abhilash, & Singh, K. K. (2023). Validation of Bromide Leaching through Response Surface Methodology and Separation of Gold from Waste Printed Circuit Boards. Minerals, 13(4), 579. https://doi.org/10.3390/min13040579








