Controlling Factors of Meteoric Diagenesis in Karst Reservoirs: An Example from the Majiagou Formation, Ordos Basin, China
Abstract
1. Introduction
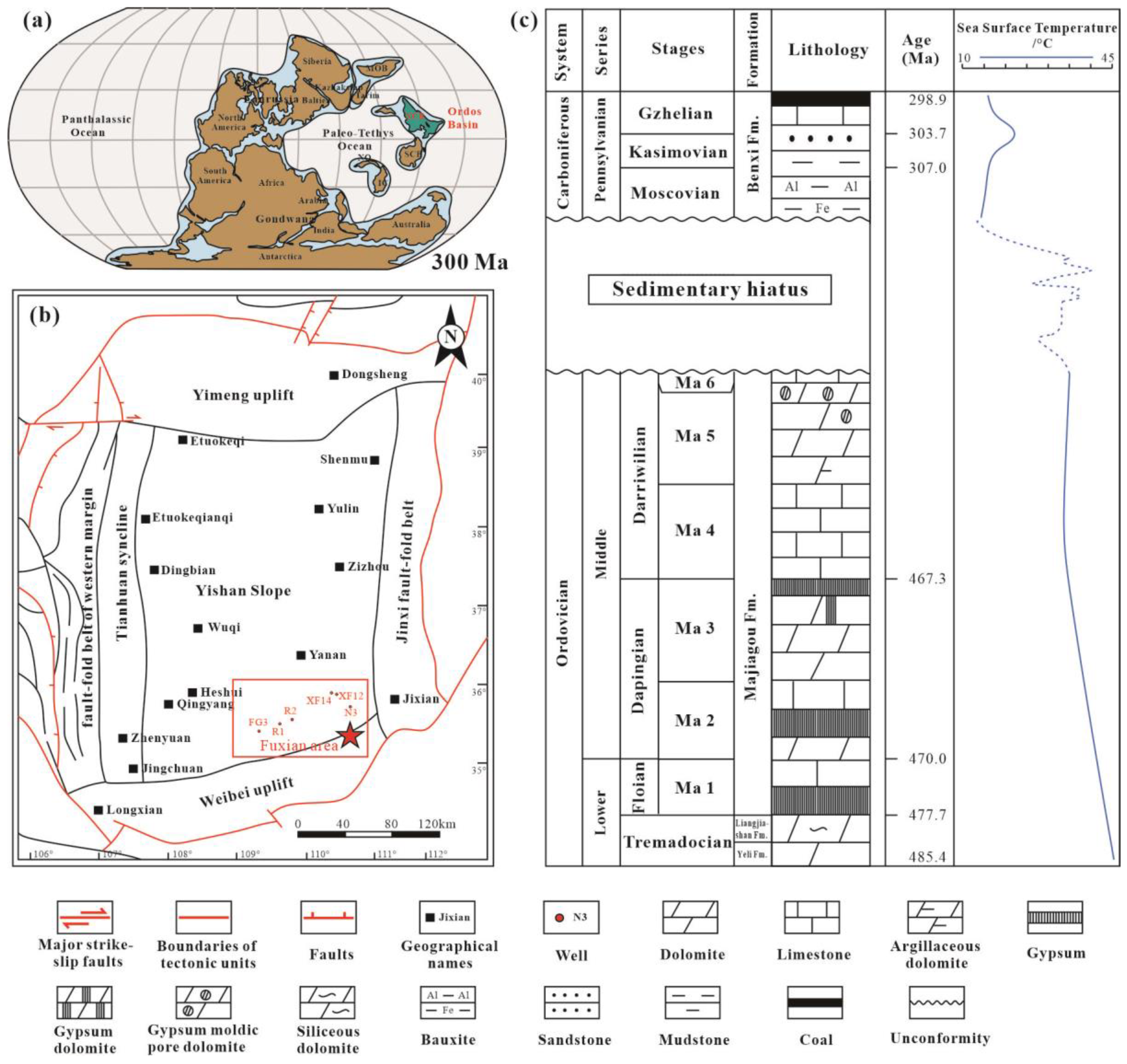
2. Geological Setting
3. Samples and Methods
3.1. Samples Selected
3.2. In Situ Elemental Analysis
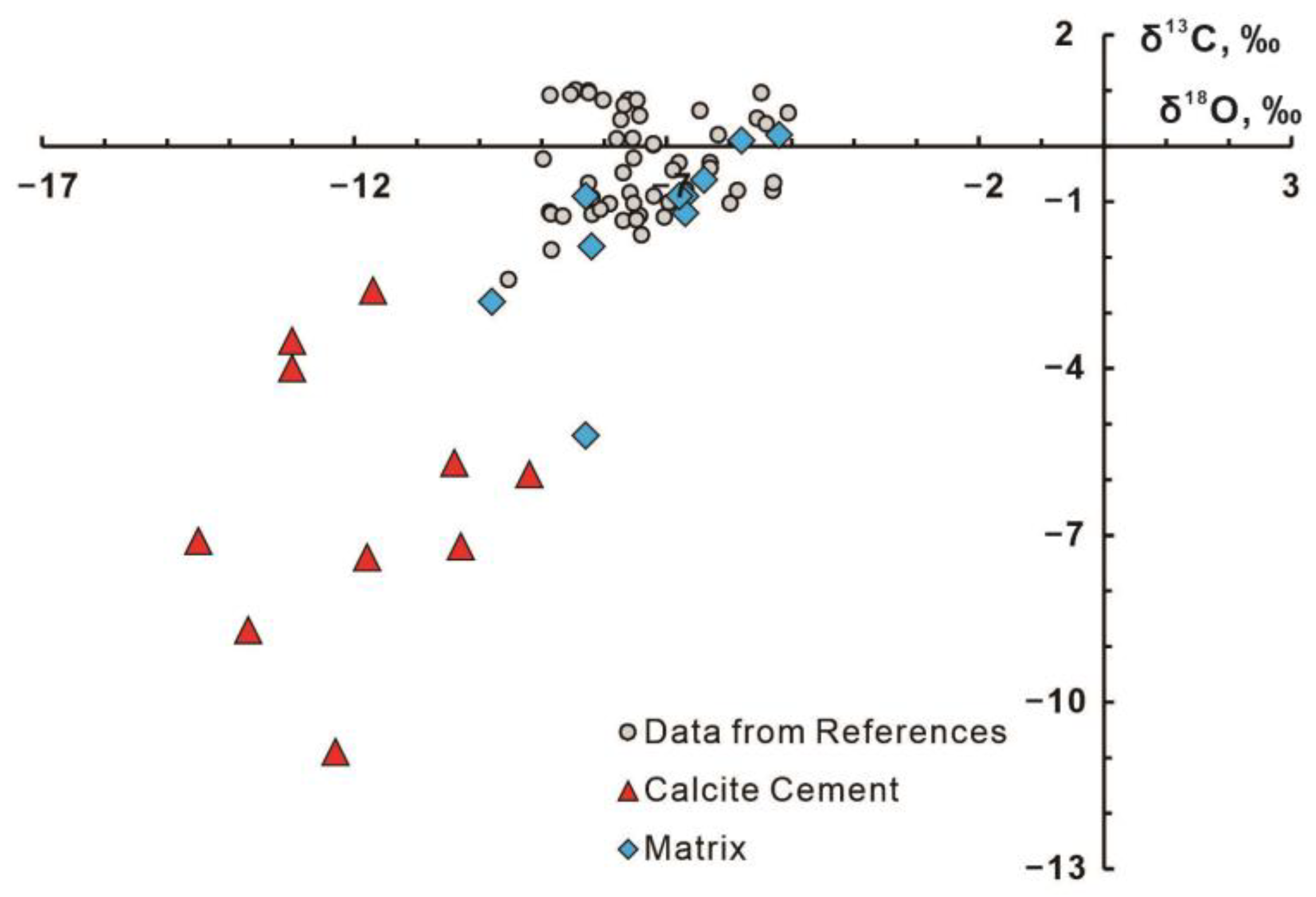
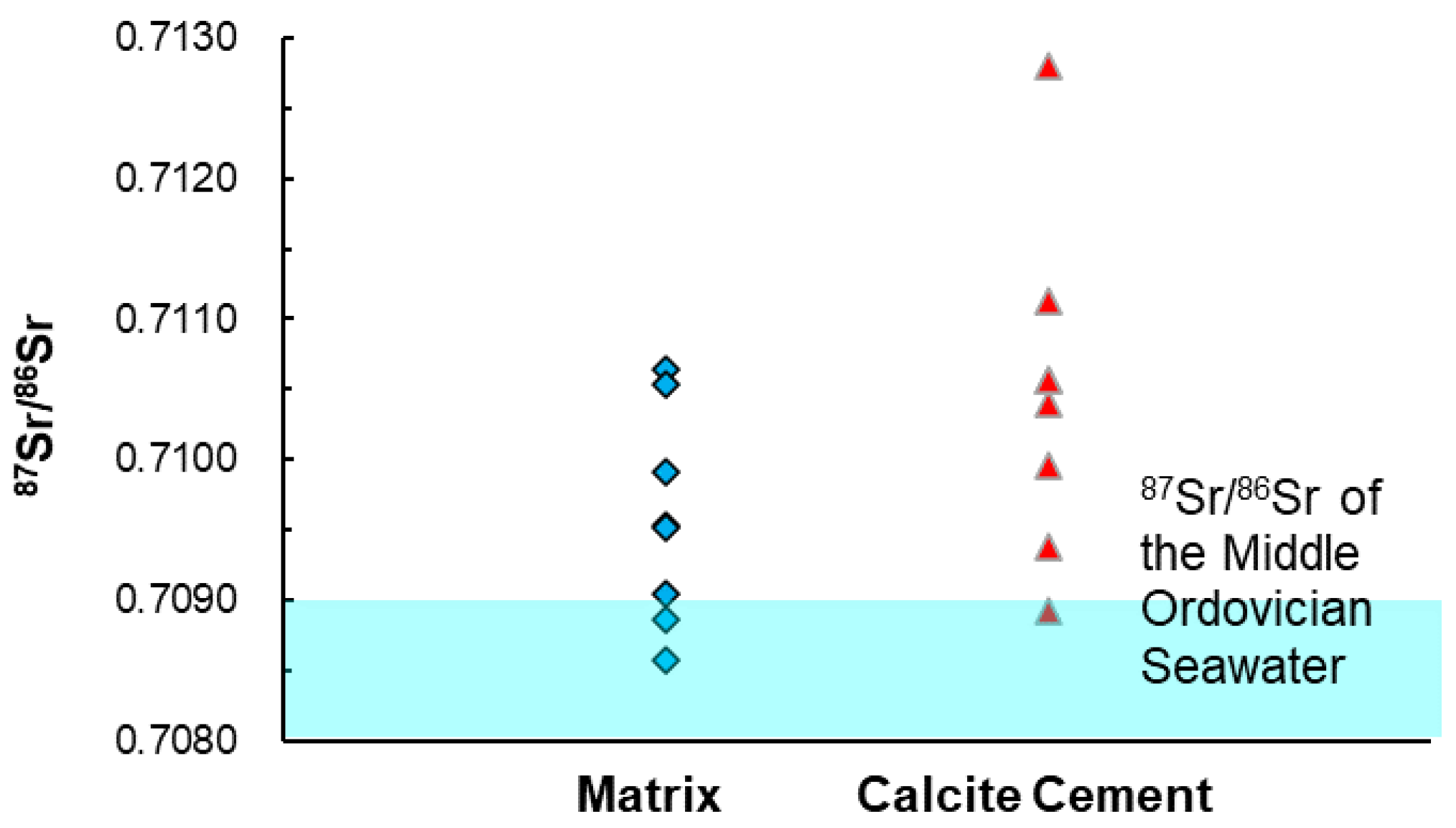
3.3. Bulk C, O, and Sr Isotopes
4. Results
4.1. Petrological Characteristics
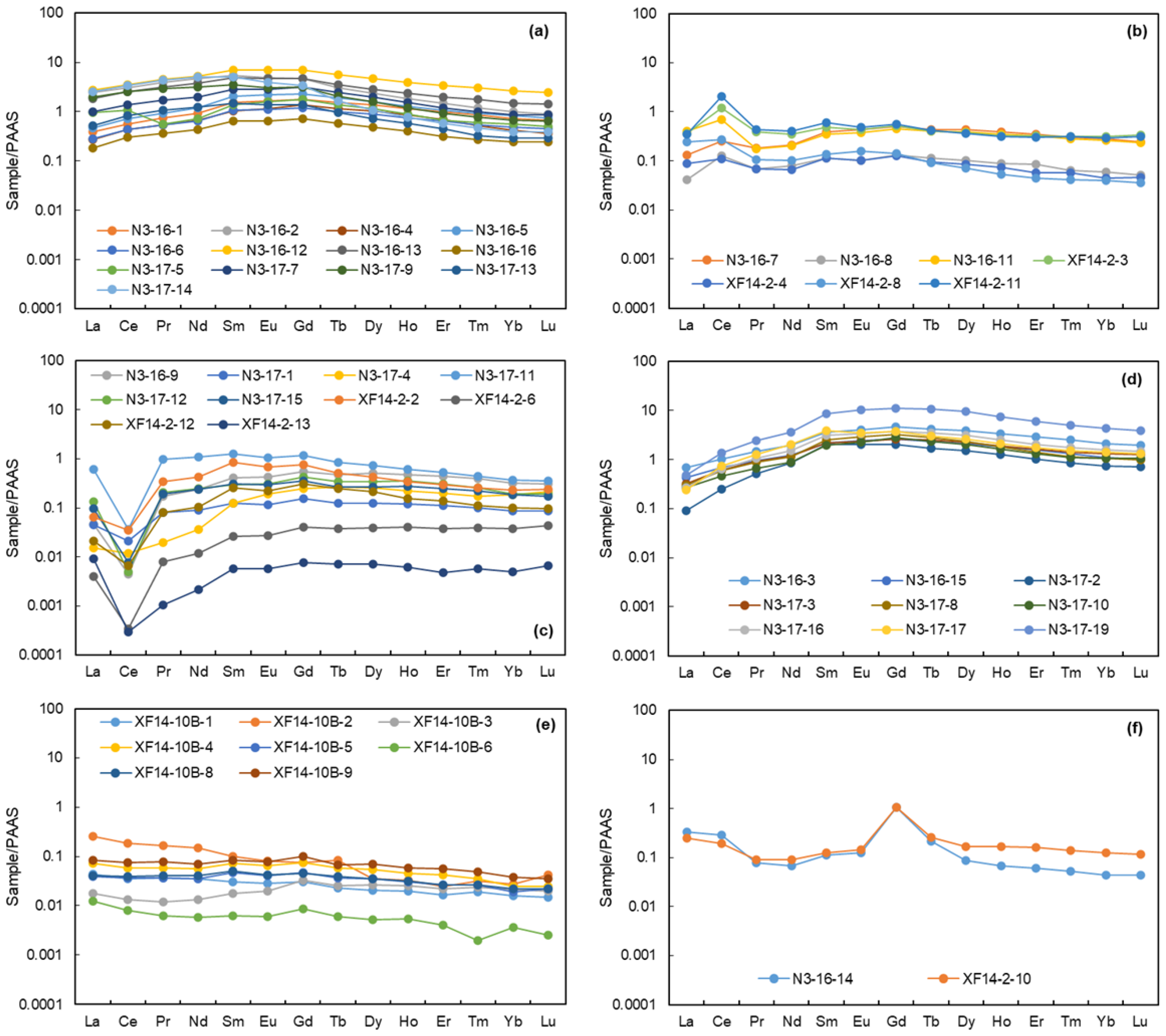
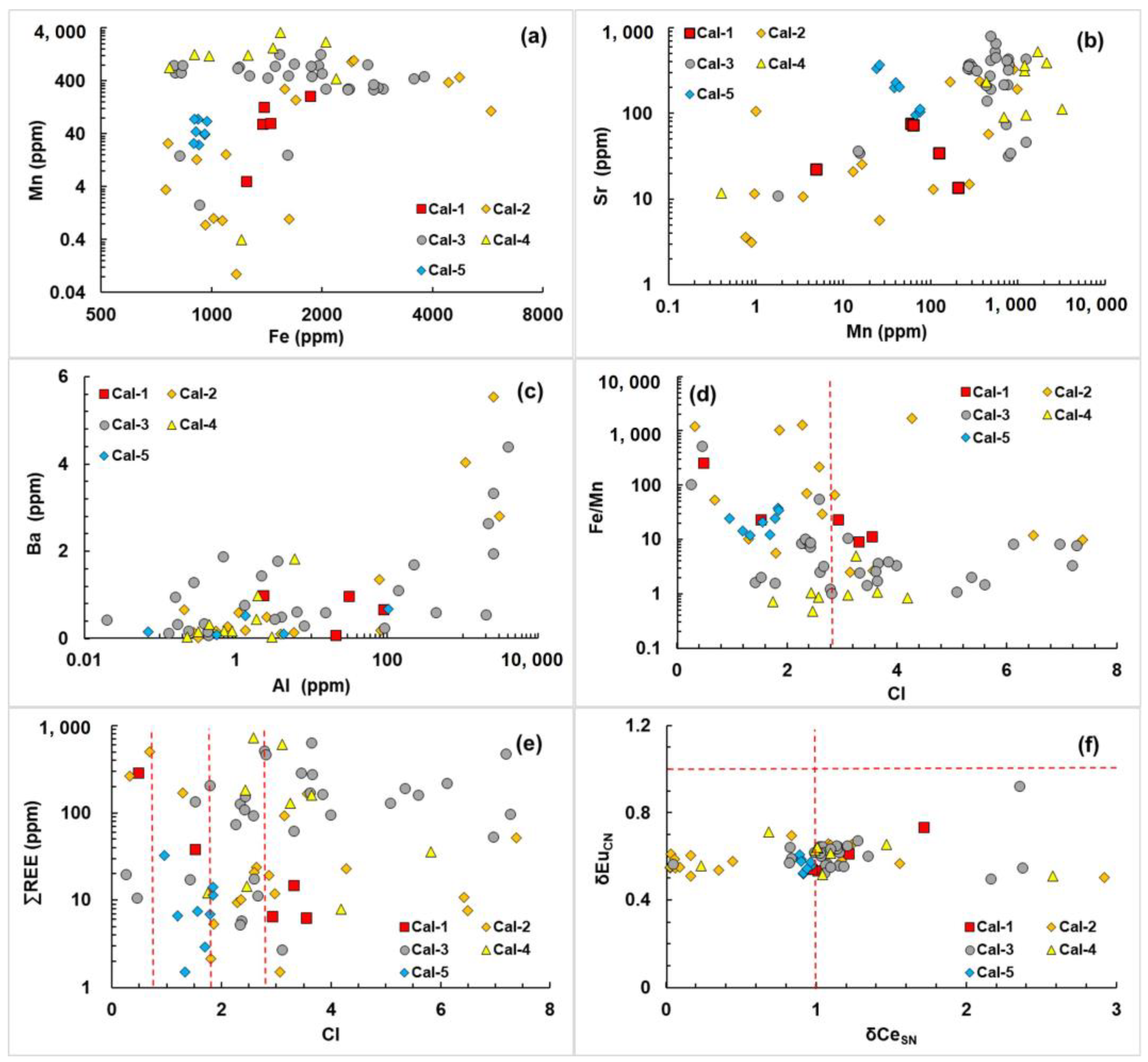
4.2. In Situ Elemental Characteristics
4.3. Isotopic Characteristics
5. Discussion
5.1. Characteristics and Origin of Cements in Karst Reservoirs
5.1.1. Evidence of Meteoric Diagenesis Transformation
5.1.2. Characteristics of Multi-Period Meteoric Calcite Cements
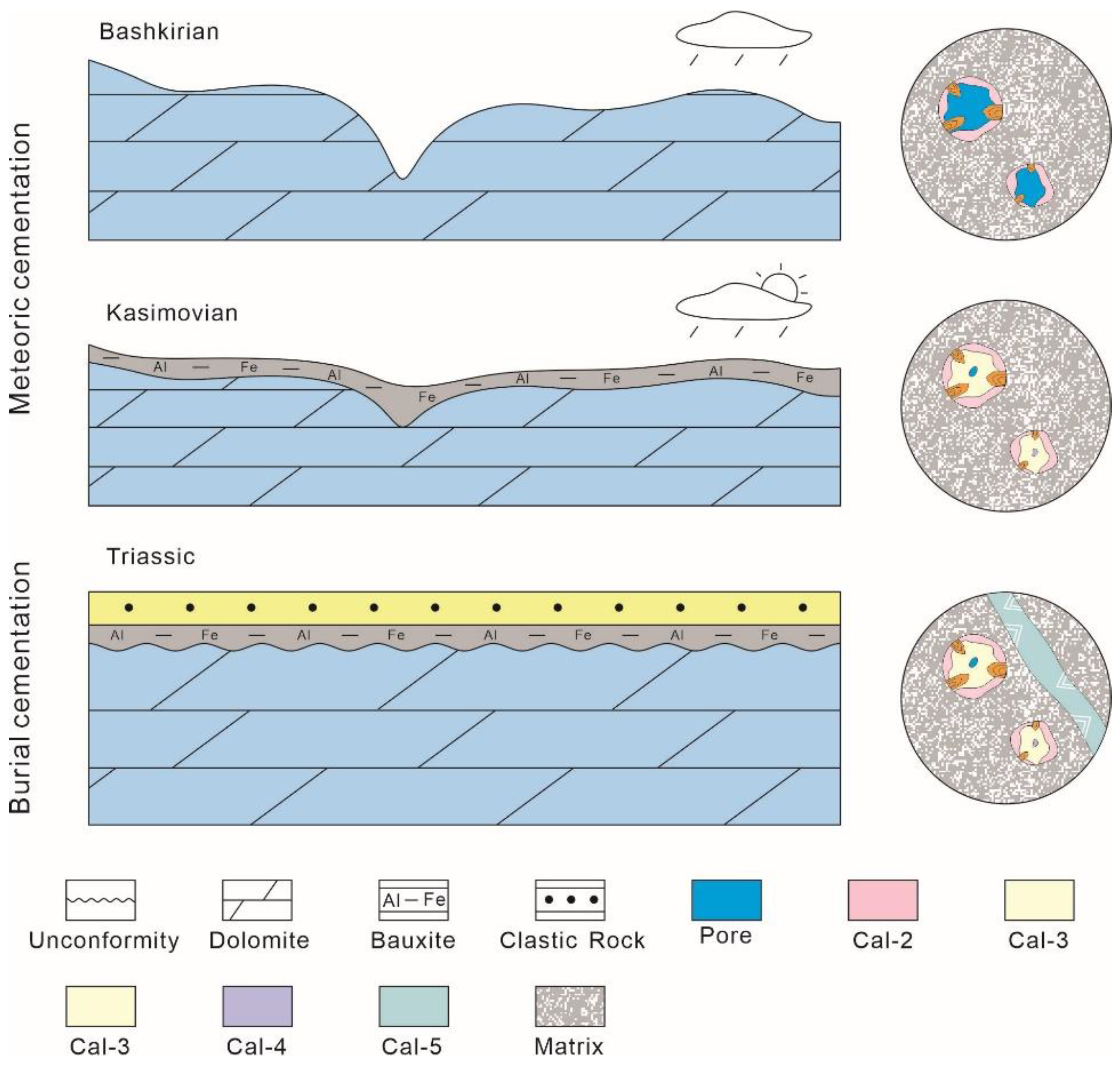
5.2. Evolution Process of the Karst Reservoir Affected by Meteoric Diagenesis
6. Conclusions
- (1)
- The dolomite reservoirs in the Majiagou Formation of the Ordos Basin were formed through a long-term karst process, resulting in high-quality reservoirs. However, the presence of simultaneously precipitated meteoric calcite cement (Cal-1~Cal-4) and burial cementation (Cal-5) has caused reservoir damage.
- (2)
- Cal-1 had a rhombohedral habit, and the REE pattern was flat. Cal-2 had dull luminescence, and had the lowest Fe, Mn, Al, and ∑REE contents; the REE pattern had a negative Ce anomaly. The REE pattern of Cal-3 showed MREE enrichment. Cal-4 had bright red luminescence and the highest contents of Fe, Mn, Al, and ∑REE. In addition, the REE pattern of Cal-5 decreased monotonically.
- (3)
- Cal-1 and Cal-2 formed during the Bashkirian period (c. 319 Ma), while Cal-3 and Cal-4 precipitated during the Kasimovian period (c. 304 Ma) after bauxite precipitation. These meteoric cementation events were influenced by specific geological conditions.
- (4)
- The cementation by meteoric calcite (Cal-1~Cal-4) occurred prior to the oil and gas charging period, causing significant damage to the karst reservoirs compared to Cal-5. Among them, Cal-3 had the most detrimental impact. Therefore, for oil and gas exploration and development in the study region, areas with higher paleogeographical locations during the critical period (Late Carboniferous) should be prioritized.
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Li, Z.Q.; Goldstein, R.H.; Franseen, E.K. Climate, duration, and mineralogy controls on meteoric diagenesis, La Molata, southeast Spain. Interpretation 2014, 2, SF111–SF123. [Google Scholar] [CrossRef]
- Csoma, A.E.; Goldstein, R.H.; Mindszenty, A.; Simone, L. Diagenetic salinity cycles and sea level along a major unconformity, Monte Composauro, Italy. J. Sediment. Res. 2004, 74, 889–903. [Google Scholar] [CrossRef]
- Li, Z.Q.; Goldstein, R.H.; Franseen, E.K. Meteoric calcite cementation: Diagenetic response to relative fall in sea-level and effect on porosity and permeability, Las Negras area, southeastern Spain. Sediment. Geol. 2017, 348, 1–18. [Google Scholar] [CrossRef]
- Fritz, R.D.; Wilson, J.L.; Yurewicz, D.A. Preface. In Paleokarst Related Hydrocarbon Reservoirs; Fritz, R.D., Wilson, J.L., Yurewicz, D.A., Eds.; SEPM: Tulsa, OK, USA, 1993. [Google Scholar]
- Cazarin, C.L.; Bezerra, F.H.R.; Borghi, L.; Santos, R.V.; Favoreto, J.; Brod, J.A.; Auler, A.S.; Srivastava, N.K. The conduit-seal system of hypogene karst in Neoproterozoic carbonates in northeastern Brazil. Mar. Petrol. Geol. 2019, 101, 90–107. [Google Scholar] [CrossRef]
- Ennes-Silva, R.A.; Bezerra, F.H.R.; Nogueira, F.C.C.; Balsamo, F.; Klimchouk, A.; Cazarin, C.L.; Auler, A.S. Superposed folding and associated fracturing influence hypogene karst development in Neoproterozoic carbonates, São Francisco Craton, Brazil. Tectonophysics 2016, 666, 244–259. [Google Scholar] [CrossRef]
- Klimchouk, A.; Auler, A.S.; Bezerra, F.H.R.; Cazarin, C.L.; Balsamo, F.; Dublyansky, Y. Hypogenic origin, geologic controls and functional organization of a giant cave system in Precambrian carbonates, Brazil. Geomorphology 2016, 253, 385–405. [Google Scholar] [CrossRef]
- Miranda, T.S.; Santos, R.F.; Barbosa, J.A.; Gomes, I.F.; Alencar, M.L.; Correia, O.J.; Falcão, T.C.; Gale, J.F.W.; Neumann, V.H. Quantifying aperture, spacing and fracture intensity in a carbonate reservoir analogue: Crato Formation, NE Brazil. Mar. Petrol. Geol. 2018, 97, 556–567. [Google Scholar] [CrossRef]
- Bagherpour, B.; Mehrabi, H.; Faghih, A.; Vaziri-Moghaddam, H.; Omidvar, M. Tectono-eustatic controls on depositional setting and spatial facies distribution of Coniacian–Santonian sequences of the Zagros Basin in Fars area, S. Iran. Mar. Petrol. Geol. 2021, 129, 105072. [Google Scholar] [CrossRef]
- He, Z.L.; Wei, X.C.; Qian, Y.X.; Bao, Z.Y.; Fan, M.; Jiao, C.L.; Peng, S.T.; Chen, D. Forming mechanism and distribution prediction of quality marine carbonate reservoirs (in Chinese with English abstract). Oil Gas Geol. 2011, 32, 489–498. [Google Scholar]
- Jiang, Q.C.; Hu, S.Y.; Wang, Z.C.; Chi, Y.L.; Yang, Y.; Lu, W.H.; Wang, H.Z.; Li, Q.F. Paleokarst landform of the weathering crust of Middle Permian Maokou Formation in Sichuan Basin and selection of exploration regions. Acta Petrol. Sin. 2012, 33, 949–960. [Google Scholar] [CrossRef]
- Zhao, W.Z.; Shen, A.J.; Pan, W.Q.; Zhang, B.M.; Qiao, Z.F.; Zheng, J.F. A research on carbonate karst reservoirs classification and its implication on hydrocarbon exploration: Case studies from Tarim Basin (in Chinese with English abstract). Acta Petrol. Sin. 2013, 29, 3213–3222. [Google Scholar]
- Zhang, J.; He, Z.; Yu, X.; Sun, Y.; Jin, X.; Chen, X. Genesis of iron-rich dolostones in the 5th member of the Majiagou Formation of the Ordovician in Ordos Basin (in Chinese with English abstract). Oil Gas Geol. 2017, 38, 776–783. [Google Scholar] [CrossRef]
- He, Z.L.; Ma, Y.S.; Zhu, D.Y.; Duan, T.Z.; Geng, J.H.; Zhang, J.T.; Ding, Q.; Qian, Y.X.; Wo, Y.J.; Gao, Z.Q. Theoretical and technological process and research direction of deep and ultra-deep carbonate reservoirs (in Chinese with English abstract). Oil Gas Geol. 2021, 42, 533–546. [Google Scholar] [CrossRef]
- Xiao, D.; Cao, J.; Tan, X.; Xiong, Y.; Zhang, D.; Dong, G.; Lu, Z. Marine carbonate reservoirs formed in evaporite sequences in sedimentary basins: A review and new model of epeiric basin-scale moldic reservoirs. Earth–Sci. Rev. 2021, 223, 103860. [Google Scholar] [CrossRef]
- Fang, S.; He, J.; Hou, F.; Yang, X.; Qiao, L.; Fu, S.; Yao, J.; Wu, Z.; Yan, R.; Xu, L. Reservoirs pore space types and evolution in M55 to M51 submembers of Majiagou Formation of Middle Ordovician in central gasfield area of Ordos basin. Acta Petrol. Sin. 2009, 25, 2425–2441. [Google Scholar]
- He, J.; Zhao, Z.; Qiao, L.; Wei, W.; Wang, Y.; Feng, C.; Chen, B.; Chen, C. Occurrence, genesis and evolution of evaporite minerals in dolomite reservoir: A case study of the Majiagou Formation in gas fields of the central Ordos Basin (in Chinese with English abstract). Oil Gas Geol. 2013, 34, 659–666. [Google Scholar] [CrossRef]
- Liu, X.; Zhang, D.; Dong, G.; Wang, B.; Xiao, D. Petrography and facies distribution of Middle Ordovician Ma 51+2 tight dolomite reservoirs in the Ordos Basin, Central China. Energ. Explor. Exploit. 2019, 37, 473–492. [Google Scholar] [CrossRef]
- Fu, S.Y.; Zhang, C.G.; Chen, H.D.; Chen, A.Q.; Zhang, J.X.; Su, Z.T.; Yang, S.; Wang, G.; Mi, W.T. Characteristics, formation and evolution of pre-salt dolomite reservoirs in the fifth member of the Ordovician Majiagou Formation, mid-east Ordos Basin, NW China. Petrol. Explor. Dev. 2019, 46, 1087–1098. [Google Scholar] [CrossRef]
- Liu, L.; Wang, C.; Du, Z.; Gong, D. Minerals Filling in Anhydrite Dissolution Pores and Their Origins in the Ordovician Majiagou Formation of the Southeastern Ordos Basin, China. Geofluids 2021, 2021, 5527299. [Google Scholar] [CrossRef]
- Luo, Q.; Liu, B.; Shi, K.; Luo, N.; Yang, Y.; Shen, Y.; Lu, F.; Wei, L.; Wu, C.; Zhao, Q. The moldic pore evolution of the Middle Ordovician sabkha dolostone in Ordos Basin, China: A study based on the petrographic and geochemical characteristics of pore fillings. Geol. J. 2022, 57, 2812–2827. [Google Scholar] [CrossRef]
- Zhou, J.; Yu, Z.; Wu, D.; Ren, J.; Zhang, D.; Wang, S.; Yin, C.; Liu, Y. Restoration of formation processes of dolomite reservoirs based on laser U-Pb dating: A case study of Ordovician Majiagou Formation, Ordos Basin, NW China. Petrol. Explor. Dev. 2022, 49, 327–338. [Google Scholar] [CrossRef]
- Swart, P.K. The geochemistry of carbonate diagenesis: The past, present and future. Sedimentology 2015, 62, 1233–1304. [Google Scholar] [CrossRef]
- Brandano, M.; Cornacchia, I.; Raffi, I.; Tomassetti, L. The Oligocene–Miocene stratigraphic evolution of the Majella carbonate platform (Central Apennines, Italy). Sediment. Geol. 2016, 333, 1–14. [Google Scholar] [CrossRef]
- Ying, Y.; Chen, W.; Simonetti, A.; Jiang, S.; Zhao, K. Significance of hydrothermal reworking for REE mineralization associated with carbonatite: Constraints from in situ trace element and C-Sr isotope study of calcite and apatite from the Miaoya carbonatite complex (China). Geochim. Cosmochim. Acta 2020, 280, 340–359. [Google Scholar] [CrossRef]
- Esteban, M.; Klappa, C.F. Subaerial exposure environments. In Carbonate Depositional Environments; Scholle, P.A., Bebout, D.G., Moore, C.H., Eds.; American Association of Petroleum Geologists: Tulsa, OK, USA, 1983. [Google Scholar] [CrossRef]
- Andrews, J.E.; Pedley, H.M.; Dennis, P.F. Stable isotopic record of palaeoclimatic change in a British Holocene tufa. Holocene 1994, 4, 349–355. [Google Scholar] [CrossRef]
- Muchez, P.; Nielsen, P.; Sintubin, M.; Lagrou, D. Conditions of meteoric calcite formation along a Variscan fault and their possible relation to climatic evolution during the Jurassic–Cretaceous. Sedimentology 1998, 45, 845–854. [Google Scholar] [CrossRef]
- Defliese, W.; Lohmann, K. Evaluation of meteoric calcite cements as a proxy material for mass-47 clumped isotope thermometry. Geochim. Cosmochim. Acta 2016, 173, 126–141. [Google Scholar] [CrossRef]
- Webb, G.; Nothdurft, L.D.; Kamber, B.S.; Kloprogge, J.T.; Zhao, J. Rare earth element geochemistry of scleractinian coral skeleton during meteoric diagenesis: A sequence through neomorphism of aragonite to calcite. Sedimentology 2009, 56, 1433–1463. [Google Scholar] [CrossRef]
- Himmler, T.; Bach, W.; Bohrmann, G.; Peckmann, J. Rare earth elements in authigenic methane-seep carbonates as tracers for fluid composition during early diagenesis. Chem. Geol. 2010, 277, 126–136. [Google Scholar] [CrossRef]
- Barnaby, R.J.; Rimstidt, J.D. Redox conditions of calcite cementation interpreted from Mn and Fe contents of authigenic calcites. Geol. Soc. Am. Bull. 1989, 101, 795–804. [Google Scholar] [CrossRef]
- Wei, D.; Gao, Z.; Fan, T.; Niu, Y.; Guo, R. Volcanic events-related hydrothermal dolomitisation and silicification controlled by intra-cratonic strike-slip fault systems: Insights from the northern slope of the Tazhong Uplift, Tarim Basin, China. Basin Res. 2021, 33, 2411–2434. [Google Scholar] [CrossRef]
- Huang, B.; Yan, Y.; Piper, J.D.A.; Zhang, D.; Yi, Z.; Yu, S.; Zhou, T. Paleomagnetic constraints on the paleogeography of the East Asian blocks during Upper Paleozoic and Lower Mesozoic times. Earth Sci. Rev. 2018, 186, 8–36. [Google Scholar] [CrossRef]
- Trotter, J.; Williams, I.; Barnes, C.; Lecuyer, C.; Nicoll, R. Did Cooling Oceans Trigger Ordovician Biodiversification? Evidence From Conodont Thermometry. Science 2008, 321, 550–554. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Al-Aasm, I. Karst-controlled diagenesis and reservoir development: Example from the Ordovician main-reservoir carbonate rocks on the eastern margin of the Ordos basin, China. AAPG Bull. 2002, 86, 1639–1658. [Google Scholar] [CrossRef]
- Yang, P.; Ren, Z.; Zhou, R.; Cui, J.; Qi, K.; Fu, J.; Li, J.; Liu, X.; Li, W.; Wang, K. Tectonic evolution and controls on natural gas generation and accumulation in the Ordovician system of the Ordos Basin, North China. Energy Rep. 2021, 7, 6887–6898. [Google Scholar] [CrossRef]
- Yang, Y.; Li, W.; Ma, L. Tectonic and stratigraphic controls of hydrocarbon systems in the Ordos basin: A multicycle cratonic basin in central China. AAPG Bull. 2005, 89, 255–269. [Google Scholar] [CrossRef]
- Yu, Z.; Yu, Z.C.; Wu, D.X.; Dong, Y.; Guo, T.; Liu, W. Sedimentary facies evolution model of Ordovician Majiagou Foramtion, Central-Eastern Ordos Basin (in Chinese with English abstract). Marin. Orig. Petrl. Geol. 2017, 22, 12–22. [Google Scholar] [CrossRef]
- Yang, J.Q.; Zhang, J.T.; He, Z.L.; Luo, N.N.; Jin, X.H.; Zhang, T.; Gu, N.; Huang, K.J.; Gao, J. Fractionation characteristics of magnesium isotope in the ancient weathering crust. Petrol. Sci. 2022, in press. [CrossRef]
- Xiong, Y.; Hou, Z.M.; Tan, X.C.; Luo, J.S.; Yue, Y.; Wu, K.Y. Constraining fluid-rock interactions during eogenetic karst and their impacts on carbonate reservoirs: Insights from reactive transport modeling. Appl. Geochem. 2021, 131, 105050. [Google Scholar] [CrossRef]
- Saltzman, M. Late Paleozoic ice age: Oceanic gateway or pCO2? Geology 2003, 31, 151–154. [Google Scholar] [CrossRef]
- Isbell, J.; Henry, L.; Gulbranson, E.; Limarino, C.; Fraiser, M.; Koch, Z.; Ciccioli, P.; Dineen, A. Glacial paradoxes during the late Paleozoic ice age: Evaluating the equilibrium line altitude as a control on glaciation. Gondwana Res. 2012, 22, 1–19. [Google Scholar] [CrossRef]
- Fielding, C.; Frank, T.; Birgenheier, L.; Rygel, M.; Jones, A.; Roberts, J. Stratigraphic imprint of the late Paleozoic ice age in eastern Australia: A record of alternating glacial and nonglacial climate regime. J. Geol. Soc. Lond. 2008, 165, 129–140. [Google Scholar] [CrossRef]
- Davydov, V.; Cózar, P. The formation of the Alleghenian Isthmus triggered the Bashkirian glaciation: Constraints from warm-water benthic foraminifera. Palaeogeogr. Palaeoclimatrol. Palaeoecol. 2019, 531, 108403. [Google Scholar] [CrossRef]
- Cao, G.; Du, X. Formation Mechanism of the Aluminiferous Rock Series in Benxi Formation, North China Block; Science Press: Beijing, China, 2020; pp. 16–27. [Google Scholar]
- Chen, J.; Montanez, I.; Zhang, S.; Isson, T.; Macarewich, S.; Planavsky, N.; Zhang, F.; Rauzi, S.; Daviau, K.; Yao, L.; et al. Marine anoxia linked to abrupt global warming during Earth’s penultimate icehouse. Proc. Natl. Acad. Sci. USA 2022, 119, e2115231119. [Google Scholar] [CrossRef] [PubMed]
- Rieger, P.; Magnall, J.; Gleeson, S.; Oelze, M.; Wilke, F.; Lilly, R. Differentiating between hydrothermal and diagenetic carbonate using rare earth element and yttrium (REE+Y) geochemistry: A case study from the Paleoproterozoic George Fisher massive sulfide Zn deposit, Mount Isa, Australia. Miner. Depos. 2022, 57, 187–206. [Google Scholar] [CrossRef]
- He, X.Y.; Shou, J.F.; Shen, A.J.; Wu, X.N.; Wang, Y.S.; Hu, Y.Y.; Zhu, Y.; Wei, D.X. Geochemical characteristics and origin of dolomite: A case study from the middle assemblage of Ordovician Majiagou Formation Member 5 of the west of Jingbian Gas Field, Ordos Basin, North China. Petrol. Explor. Dev. 2014, 41, 417–427. [Google Scholar] [CrossRef]
- Cao, H.X.; Shang, T.; Wu, H.Y.; Wang, N.X.; Feng, Y. Characteristics of carbon and oxygen isotopes of carbonate rocks in Majiagou Formation and their implication, southeastern Ordos Basin (in English abstract). J. Northwest Univ. (Nat. Sci. Ed.) 2018, 48, 578–586. [Google Scholar] [CrossRef]
- He, M.; Huang, W.H.; Jiu, B. Origin and evolution of gypsum dolomite as a favorable reservoir in the Ordos Basin, China (in English abstract). Earth Sci. Front. 2021, 28, 327–336. [Google Scholar] [CrossRef]
- McArthur, J.M.; Howarth, R.J.; Bailey, T.R. Strontium isotope stratigraphy: LOWESS Version 3: Best fit to the marine Sr isotope curve for 0–509 Ma and accompanying look-up table for deriving numerical age. J. Geol. 2001, 109, 155–170. [Google Scholar] [CrossRef]
- Nader, F.H.; Swennen, R.; Keppens, E. Calcitization/dedolomitization of Jurassic dolostones (Lebanon): Results from petrographic and sequential geochemical analyses. Sedimentology 2008, 55, 1467–1485. [Google Scholar] [CrossRef]
- Escorcia, L.C.; Gomez-Rivas, E.; Daniele, L.; Corbella, M. Dedolomitization and reservoir quality: Insights from reactive transport modelling. Geofluids 2013, 13, 221–231. [Google Scholar] [CrossRef]
- Schoenherr, J.; Reuning, L.; Hallenberger, M.; Lüders, V.; Lemmens, L.; Biehl, B.; Lewin, A.; Leupold, M.; Wimmers, K.; Strohmenger, C. Dedolomitization: Review and case study of uncommon mesogenetic formation conditions. Earth Sci. Rev. 2018, 185, 780–805. [Google Scholar] [CrossRef]
- Gao, J.; He, S.; Zhao, J.; He, Z.; Wu, C.; Feng, Y.; Nguyen, A.; Zhou, J.; Yi, Z. Sm-Nd isochron dating and geochemical (rare earth elements, 87Sr/86Sr, δ18O, δ13C) characterization of calcite veins in the Jiaoshiba shale gas field, China: Implications for the mechanisms of vein formation in shale gas systems. Geol. Soc. Am. Bull. 2020, 132, 1722–1740. [Google Scholar] [CrossRef]
- Palmer, M.R.; Edmond, J.M. The strontium isotope budget of the modern ocean. Earth Planet. Sc. Lett. 1989, 92, 11–26. [Google Scholar] [CrossRef]
- Yang, T.; Azmy, K.; He, Z.; Li, S.; Liu, E.; Wu, S.; Wang, J.; Li, T.; Gao, J. Fault-controlled hydrothermal dolomitization of Middle Permian in southeastern Sichuan Basin, SW China, and its temporal relationship with the Emeishan Large Igneous Province: New insights from multi-geochemical proxies and carbonate U–Pb dating. Sediment. Geol. 2022, 439, 106215. [Google Scholar] [CrossRef]
- Ren, Z.; Zhang, S.; Gao, S.; Cui, J.; Xiao, Y.; Xiao, H. Tectonic thermal history and its significance on the formation of oil and gas accumulation and mineral deposit in Ordos Basin. Sci. China Earth Sci. 2007, 50, 27–38. [Google Scholar] [CrossRef]
- Jiu, B.; Huang, W.; Li, Y. The effect of hydrothermal fluids on Ordovician carbonate rocks, southern Ordos Basin, China. Ore Geol. Rev. 2020, 126, 103803. [Google Scholar] [CrossRef]
- Petrash, D.; Bialik, O.; Staudigel, P.; Konhauser, K.; Budd, D. Biogeochemical reappraisal of the freshwater–seawater mixing-zone diagenetic model. Sedimentology 2021, 68, 1797–1830. [Google Scholar] [CrossRef]
- Mishra, P.; Mohanty, S. Geochemistry of carbonate rocks of the Chilpi Group, Bastar Craton, India: Implications on ocean paleoredox conditions at the late Paleoproterozoic Era. Precambrian Res. 2021, 353, 106023. [Google Scholar] [CrossRef]
- Yang, J.Q.; Zhang, J.T.; He, Z.L.; Zhang, T. Paleoenvironment reconstruction of the Middle Ordovician thick carbonate from western Ordos Basin, China. Petrol. Sci. 2023, 20, 48–59. [Google Scholar] [CrossRef]
- Zhang, P.; Wang, Y.; Zhang, X.; Wei, Z.; Wang, G.; Zhang, T.; Ma, H.; Wei, J.; He, W.; Ma, X.; et al. Carbon, oxygen and strontium isotopic and elemental characteristics of the Cambrian Longwangmiao Formation in South China: Paleoenvironmental significance and implications for carbon isotope excursions. Gondwana Res. 2022, 106, 174–190. [Google Scholar] [CrossRef]
- Bau, M.; Möller, P. Rare earth element fractionation in metamorphogenic hydrothermal calcite, magnesite and siderite. Miner. Petrol. 1992, 45, 231–246. [Google Scholar] [CrossRef]
- Bayon, G.; Lambert, T.; Vigier, N.; Deckker, P.; Freslon, N.; Jang, K.; Larkin, C.; Piotrowski, A.; Tachikawa, K.; Thollon, M.; et al. Rare earth element and neodymium isotope tracing of sedimentary rock weathering. Chem. Geol. 2020, 553, 119794. [Google Scholar] [CrossRef]
- Du, J.; Haley, B.A.; Mix, A.C. Neodymium isotopes in authigenic phases, bottom waters and detrital sediments in the Gulf of Alaska and their implications for paleocirculation reconstruction. Geochim. Cosmochim. Acta 2016, 193, 14–35. [Google Scholar] [CrossRef]
- Blattmann, T.M.; Wang, S.L.; Lupker, M.; Märki, L.; Haghipour, N.; Wacker, L.; Chung, L.-H.; Bernasconi, S.M.; Plötze, M.; Eglinton, T.I. Sulphuric acid-mediated weathering on Taiwan buffers geological atmospheric carbon sinks. Sci. Rep. 2019, 9, 2945. [Google Scholar] [CrossRef] [PubMed]
- Lara, R.H.; Monroy, M.G.; Mallet, M.; Dossot, M.; González, M.A.; Cruz, R. An experimental study of iron sulfides weathering under simulated calcareous soil conditions. Environ. Earth Sci. 2015, 73, 1849–1869. [Google Scholar] [CrossRef]
- Franchi, F.; Rovere, M.; Gamberi, F.; Rashed, H.; Vaselli, O.; Tassi, F. Authigenic minerals from the Paola Ridge (southern Tyrrhenian Sea): Evidences of episodic methane seepage. Mar. Petrol. Geol. 2017, 86, 228–247. [Google Scholar] [CrossRef]
- Cornacchia, I.; Brandano, M.; Agostini, S. Miocene paleoceanographic evolution of the Mediterranean area and carbonate production changes: A review. Earth Sci. Rev. 2021, 221, 103785. [Google Scholar] [CrossRef]
- Lopez, O.; Zuddas, P.; Faivre, D. The influence of temperature and seawater composition on calcite crystal growth mechanisms and kinetics: Implications for Mg incorporation in calcite lattice. Geochim. Cosmochim. Acta 2009, 73, 337–347. [Google Scholar] [CrossRef]
- Burton, E.; Walter, L. Relative precipitation rates of aragonite and Mg calcite from seawater: Temperature or carbonate ion control? Geology 1987, 15, 111–114. [Google Scholar] [CrossRef]
- Gulbranson, E.; Montanez, I.; Schmitz, M.; Limarino, C.; Isbell, J.; Marenssi, S.; Crowley, J. High-precision U-Pb calibration of Carboniferous glaciation and climate history, Paganzo Group, NW Argentina. Geol. Soc. Am. Bull. 2010, 122, 1480–1498. [Google Scholar] [CrossRef]
- Feng, Y.; Xiao, X.M.; Gao, P.; Wang, E.Z.; Hu, D.F.; Liu, R.B.; Li, G.; Lu, C.G. Restoration of sedimentary environment and geochemical features of deep marine Longmaxi shale and its significance for shale gas: A case study of the Dingshan area in the Sichuan Basin, South China. Mar. Pet. Geol. 2023, 151, 106186. [Google Scholar] [CrossRef]

| Lithofacies | Morphology | Th (°C) | Tm (°C) | Salinity (wt% NaCl) | ||
|---|---|---|---|---|---|---|
| Gypsum mold pore | Strip, Quadrilateral, Triangle, Circle | Mean | 115 | −20 | 22.3 | n = 6 |
| Max | 124 | −19 | 23.0 | |||
| Min | 99 | −21 | 21.7 | |||
| Crack | Strip, Quadrilateral, Oval | Mean | 111 | −20 | 22.5 | n = 6 |
| Max | 123 | −19 | 23 | |||
| Min | 96 | −21 | 21.7 |
| Location | Type | Al | Mn | Fe | Sr | Ba | La | Ce | Pr | Nd | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| XF14-2-5 | Cal-1 | 0.3 | 205.0 | 1860 | 13.7 | / | 1.72 | 5.00 | 0.75 | 3.02 | 1.07 | 0.19 | 1.07 | 0.14 | 0.71 | 0.13 | 0.37 | 0.05 | 0.28 | 0.05 |
| XF14-2-7 | 21.3 | 124.1 | 1391 | 34.2 | 0.06 | 0.80 | 1.87 | 0.25 | 1.16 | 0.42 | 0.09 | 0.59 | 0.08 | 0.46 | 0.08 | 0.21 | 0.03 | 0.16 | 0.03 | |
| XF14-2-8 | 91.0 | 59.6 | 1381 | 76.8 | 0.66 | 9.30 | 21.9 | 0.93 | 3.48 | 0.75 | 0.17 | 0.65 | 0.07 | 0.34 | 0.05 | 0.13 | 0.02 | 0.11 | 0.02 | |
| XF14-2-9 | 2.4 | 62.8 | 1450 | 73.1 | 0.99 | 0.88 | 2.72 | 0.30 | 1.28 | 0.36 | 0.07 | 0.33 | 0.05 | 0.23 | 0.04 | 0.10 | 0.02 | 0.07 | 0.01 | |
| N3-16-5 | Cal-2 | 80.0 | 898.0 | 2410 | 326.0 | 1.35 | 18.10 | 57.3 | 8.26 | 40.60 | 11.28 | 2.37 | 10.60 | 1.45 | 7.49 | 1.29 | 3.09 | 0.37 | 2.32 | 0.33 |
| N3-16-6 | 1092.0 | 980.0 | 2440 | 192.0 | 4.04 | 10.80 | 34.00 | 4.74 | 21.90 | 5.85 | 1.22 | 5.52 | 0.77 | 4.20 | 0.74 | 1.81 | 0.22 | 1.35 | 0.20 | |
| N3-16-9 | 0.43 | 1.0 | 1630 | 11.5 | / | 1.77 | 0.35 | 1.53 | 8.05 | 2.31 | 0.46 | 2.59 | 0.37 | 2.43 | 0.48 | 1.28 | 0.16 | 0.89 | 0.13 | |
| N3-16-10 | 0.6 | 28 | 1580 | 14.9 | 0.17 | 0.175 | 0.86 | 0.09 | 0.44 | 0.11 | 0.02 | 0.10 | 0.02 | 0.12 | 0.02 | 0.08 | 0.01 | 0.08 | 0.01 | |
| N3-17-1 | 3.9 | 13.1 | 911 | 20.9 | 0.10 | 1.74 | 1.70 | 0.72 | 3.04 | 0.68 | 0.12 | 0.71 | 0.10 | 0.59 | 0.12 | 0.31 | 0.04 | 0.25 | 0.04 | |
| N3-17-12 | 0.3 | 26.0 | 761 | 5.7 | 0.02 | 5.10 | 0.39 | 1.84 | 8.40 | 1.63 | 0.33 | 2.02 | 0.26 | 1.63 | 0.35 | 0.88 | 0.10 | 0.55 | 0.08 | |
| N3-17-15 | 0.8 | 3.5 | 752 | 10.7 | 0.28 | 3.63 | 0.62 | 1.71 | 8.17 | 1.69 | 0.32 | 1.66 | 0.21 | 1.25 | 0.28 | 0.72 | 0.09 | 0.53 | 0.08 | |
| N3-17-18 | 0.4 | 369.0 | 4420 | 235.0 | 0.16 | 0.63 | 0.86 | 0.09 | 0.87 | 0.96 | 0.28 | 1.55 | 0.19 | 1.02 | 0.18 | 0.44 | 0.06 | 0.38 | 0.05 | |
| XF14-2-6 | 0.2 | 0.1 | 1169 | 5.5 | 0.66 | 0.154 | 0.03 | 0.07 | 0.40 | 0.14 | 0.03 | 0.19 | 0.03 | 0.18 | 0.04 | 0.11 | 0.02 | 0.11 | 0.02 | |
| XF14-2-12 | 1.1 | / | 1152 | 4.8 | 0.59 | 0.80 | 0.53 | 0.70 | 3.51 | 1.44 | 0.24 | 1.46 | 0.20 | 1.00 | 0.16 | 0.39 | 0.05 | 0.28 | 0.04 | |
| XF14-2-13 | / | / | 1079 | 11.9 | 0.54 | 0.35 | 0.02 | 0.01 | 0.07 | 0.03 | 0.01 | 0.04 | 0.01 | 0.03 | 0.01 | 0.01 | 0.00 | 0.01 | 0.00 | |
| N3-16-3 | Cal-3 | 1.3 | 547.0 | 2000 | 520.0 | 0.76 | 26.10 | 82.00 | 13.1 | 65.30 | 19.90 | 4.38 | 21.40 | 3.28 | 18.40 | 3.37 | 8.40 | 1.04 | 5.96 | 0.84 |
| N3-16-4 | 4.0 | 800.0 | 2670 | 367.0 | 0.49 | 10.50 | 34.00 | 4.83 | 22.50 | 5.68 | 1.25 | 6.41 | 0.90 | 4.89 | 0.85 | 1.99 | 0.21 | 1.18 | 0.16 | |
| N3-16-8 | 0.2 | 783.0 | 1960 | 31.5 | 0.32 | 1.58 | 10.00 | 0.60 | 2.68 | 0.63 | 0.11 | 0.60 | 0.09 | 0.47 | 0.09 | 0.24 | 0.03 | 0.17 | 0.02 | |
| N3-16-13 | 2.2 | 1240.0 | 1530 | 427.0 | 1.44 | 71.50 | 201.00 | 28.30 | 127.00 | 27.50 | 5.00 | 21.60 | 2.70 | 13.50 | 2.34 | 5.61 | 0.71 | 4.24 | 0.61 | |
| N3-16-15 | 3.6 | 495.0 | 1270 | 412.0 | 1.78 | 16.70 | 53.80 | 8.50 | 41.20 | 12.20 | 2.59 | 12.20 | 1.83 | 10.00 | 1.80 | 4.49 | 0.54 | 3.13 | 0.44 | |
| N3-16-16 | 15.4 | 760.0 | 1870 | 216.0 | 0.59 | 7.10 | 24.10 | 3.25 | 14.60 | 3.58 | 0.70 | 3.35 | 0.44 | 2.24 | 0.40 | 0.90 | 0.11 | 0.69 | 0.10 | |
| N3-17-2 | / | 488.0 | 3800 | 192.0 | / | 3.50 | 19.90 | 4.60 | 29.20 | 11.10 | 2.19 | 9.60 | 1.30 | 7.10 | 1.24 | 2.93 | 0.35 | 2.10 | 0.31 | |
| N3-17-3 | 0.7 | 485.0 | 1880 | 272.5 | 1.88 | 12.15 | 49.40 | 8.21 | 40.50 | 12.04 | 2.54 | 12.30 | 1.93 | 10.69 | 1.86 | 4.60 | 0.58 | 3.74 | 0.55 | |
| N3-17-7 | 0.1 | 564.0 | 797 | 446.0 | 0.12 | 38.30 | 110.00 | 15.23 | 67.10 | 15.77 | 3.11 | 14.80 | 1.88 | 9.33 | 1.53 | 3.43 | 0.40 | 2.40 | 0.37 | |
| N3-17-8 | 0.4 | 567.0 | 827 | 650.0 | 0.34 | 11.29 | 46.60 | 7.92 | 38.50 | 13.92 | 3.12 | 15.39 | 2.15 | 11.00 | 1.82 | 4.06 | 0.47 | 3.01 | 0.42 | |
| N3-17-9 | 0.2 | 782.0 | 788 | 321.0 | 0.18 | 76.00 | 199.00 | 25.90 | 108.00 | 19.20 | 3.32 | 14.60 | 1.61 | 7.50 | 1.20 | 2.69 | 0.32 | 1.90 | 0.28 | |
| N3-17-10 | 0.4 | 775.0 | 835 | 427.0 | 0.16 | 10.10 | 36.70 | 5.91 | 29.60 | 10.74 | 2.49 | 13.20 | 1.83 | 9.59 | 1.61 | 3.76 | 0.45 | 2.97 | 0.46 | |
| N3-17-11 | 8.0 | 15.0 | 819 | 36.6 | 0.29 | 22.99 | 2.89 | 8.63 | 36.80 | 6.91 | 1.15 | 5.57 | 0.66 | 3.43 | 0.62 | 1.50 | 0.18 | 1.04 | 0.16 | |
| N3-17-14 | 2.4 | 693.0 | 1180 | 215.3 | / | 97.00 | 272.00 | 38.4 | 171.00 | 28.5 | 4.31 | 15.90 | 1.28 | 4.94 | 0.76 | 1.67 | 0.19 | 1.12 | 0.17 | |
| N3-17-16 | 0.3 | 746.0 | 1490 | 413.0 | 1.28 | 10.20 | 51.30 | 9.40 | 50.70 | 17.10 | 3.67 | 17.30 | 2.72 | 14.80 | 2.46 | 5.86 | 0.71 | 4.43 | 0.62 | |
| N3-17-17 | 3.3 | 437.0 | 3570 | 210.0 | 0.44 | 9.10 | 59.00 | 11.20 | 68.00 | 21.40 | 3.83 | 17.60 | 2.32 | 12.40 | 2.07 | 5.10 | 0.62 | 3.93 | 0.58 | |
| N3-17-19 | 6.5 | 490.0 | 1620 | 788.0 | 0.61 | 18.10 | 110.00 | 21.80 | 123.00 | 47.20 | 10.99 | 52.50 | 8.21 | 45.10 | 7.44 | 17.0 | 2.02 | 12.3 | 1.66 | |
| XF14-2-3 | 0.0 | 830.0 | 1681 | 34.2 | 0.43 | 14.00 | 95.00 | 3.43 | 11.80 | 2.68 | 0.48 | 2.42 | 0.32 | 1.77 | 0.32 | 0.94 | 0.13 | 0.90 | 0.15 | |
| XF14-2-11 | / | 1250.0 | 1983 | 45.7 | 0.41 | 13.19 | 164.00 | 3.82 | 13.90 | 3.38 | 0.53 | 2.64 | 0.33 | 1.71 | 0.31 | 0.86 | 0.13 | 0.83 | 0.14 | |
| N3-16-1 | Cal-4 | 6 | 439.0 | 2190 | 234.0 | 1.83 | 15.00 | 43.80 | 6.60 | 31.50 | 8.50 | 1.76 | 8.25 | 1.19 | 6.42 | 1.16 | 2.88 | 0.35 | 2.02 | 0.29 |
| N3-16-2 | 0.3 | 2150.0 | 2050 | 390.0 | 0.15 | 94.00 | 244.90 | 34.50 | 158.60 | 29.70 | 5.29 | 22.10 | 2.35 | 11.03 | 1.85 | 4.28 | 0.48 | 2.85 | 0.39 | |
| N3-16-12 | 3 | 1690.0 | 1470 | 520.0 | 0.04 | 103.00 | 279.00 | 39.40 | 178.00 | 38.30 | 7.41 | 32.10 | 4.29 | 22.10 | 3.88 | 9.70 | 1.23 | 7.46 | 1.08 | |
| N3-17-4 | 0.5 | 1190.0 | 987 | 314.0 | 0.32 | 0.58 | 0.94 | 0.17 | 1.22 | 0.69 | 0.21 | 1.15 | 0.21 | 1.21 | 0.22 | 0.57 | 0.07 | 0.53 | 0.09 | |
| N3-17-5 | 1.9 | 1209.0 | 1260 | 359.0 | 0.45 | 37.00 | 86.00 | 4.93 | 24.70 | 7.78 | 1.72 | 8.14 | 1.07 | 5.32 | 0.88 | 1.94 | 0.24 | 1.56 | 0.22 | |
| N3-17-13 | 0.7 | 702.0 | 767 | 90.1 | 0.17 | 19.80 | 65.10 | 9.48 | 42.00 | 8.21 | 1.47 | 6.41 | 0.74 | 3.42 | 0.58 | 1.27 | 0.13 | 0.81 | 0.13 | |
| XF14-2-1 | 0.9 | 3250.0 | 1541 | 112.0 | 0.17 | 1.30 | 8.20 | 0.41 | 1.67 | 0.59 | 0.10 | 0.64 | 0.09 | 0.54 | 0.11 | 0.32 | 0.04 | 0.24 | 0.04 | |
| XF14-2-2 | 0.2 | 0.4 | 1208 | 11.9 | 0.04 | 2.49 | 2.80 | 2.99 | 14.30 | 4.67 | 0.74 | 3.52 | 0.39 | 1.99 | 0.35 | 0.85 | 0.10 | 0.66 | 0.10 | |
| XF14-10B-1 | Cal-5 | / | 67.5 | 972 | 95.7 | / | 1.66 | 2.83 | 0.32 | 1.20 | 0.17 | 0.03 | 0.14 | 0.02 | 0.10 | 0.02 | 0.05 | 0.01 | 0.04 | 0.01 |
| XF14-10B-2 | / | 38.8 | 958 | 199.0 | 0.04 | 9.79 | 14.60 | 1.46 | 5.02 | 0.55 | 0.09 | 0.35 | 0.07 | 0.17 | 0.03 | 0.07 | 0.01 | 0.08 | 0.02 | |
| XF14-10B-3 | 0.3 | 75.7 | 920 | 103.6 | / | 0.68 | 1.08 | 0.11 | 0.45 | 0.10 | 0.02 | 0.15 | 0.02 | 0.12 | 0.03 | 0.06 | 0.01 | 0.06 | 0.01 | |
| XF14-10B-4 | 103.0 | 24.4 | 922 | 333.0 | 0.68 | 2.79 | 4.71 | 0.52 | 1.92 | 0.40 | 0.07 | 0.35 | 0.05 | 0.25 | 0.05 | 0.12 | 0.01 | 0.07 | 0.01 | |
| XF14-10B-5 | 4.3 | 39.8 | 960 | 226.0 | 0.11 | 1.48 | 2.93 | 0.33 | 1.20 | 0.26 | 0.04 | 0.22 | 0.03 | 0.17 | 0.03 | 0.08 | 0.01 | 0.05 | 0.01 | |
| XF14-10B-6 | 0.6 | 75.9 | 899 | 111.8 | 0.09 | 0.48 | 0.65 | 0.06 | 0.20 | 0.03 | 0.01 | 0.04 | 0.01 | 0.02 | 0.01 | 0.01 | 0.00 | 0.01 | 0.00 | |
| XF14-10B-8 | 0.8 | 44.0 | 908 | 202.0 | 0.15 | 1.58 | 3.17 | 0.36 | 1.385 | 0.28 | 0.05 | 0.21 | 0.03 | 0.16 | 0.03 | 0.07 | 0.01 | 0.06 | 0.01 | |
| XF14-10B-9 | 1.4 | 26.4 | 894 | 367.0 | 0.52 | 3.19 | 6.01 | 0.68 | 2.39 | 0.46 | 0.08 | 0.47 | 0.05 | 0.32 | 0.06 | 0.16 | 0.02 | 0.11 | 0.02 | |
| N3-16-14 | Dol-1 | 1750.0 | 295.1 | 5420 | 91.0 | 3.78 | 12.70 | 22.90 | 0.69 | 2.26 | 0.62 | 0.14 | 4.90 | 0.16 | 0.40 | 0.07 | 0.18 | 0.02 | 0.12 | 0.02 |
| XF14-2-10 | 1310.0 | 502.0 | 3050 | 28.8 | 7.40 | 9.60 | 15.60 | 0.80 | 3.05 | 0.70 | 0.16 | 4.90 | 0.20 | 0.77 | 0.16 | 0.45 | 0.06 | 0.36 | 0.05 | |
| N3-16-7 | Matrix | 442.0 | 880.0 | 2920 | 72.0 | 2.97 | 5.00 | 20.00 | 1.59 | 7.25 | 2.15 | 0.47 | 2.32 | 0.34 | 2.03 | 0.30 | 0.98 | 0.12 | 0.78 | 0.11 |
| N3-16-11 | 518.0 | 967.0 | 1850 | 72.0 | 2.42 | 15.30 | 56.00 | 1.54 | 6.82 | 1.96 | 0.40 | 2.06 | 0.31 | 1.79 | 0.34 | 0.92 | 0.11 | 0.75 | 0.10 | |
| N3-17-6 | 741.0 | 925.0 | 7980 | 75.4 | 2.60 | 4.90 | 9.10 | 0.74 | 3.87 | 1.34 | 0.32 | 1.53 | 0.22 | 1.23 | 0.23 | 0.57 | 0.07 | 0.40 | 0.05 | |
| XF14-10B-7 | 609.0 | 24.5 | 732 | 70.4 | 1.18 | 1.24 | 1.68 | 0.15 | 0.51 | 0.11 | 0.02 | 0.12 | 0.02 | 0.11 | 0.02 | 0.07 | 0.01 | 0.05 | 0.01 | |
| XF14-2-4 | 425.0 | 151.7 | 3360 | 41.1 | 3.39 | 3.40 | 8.60 | 0.60 | 2.23 | 0.63 | 0.11 | 0.60 | 0.07 | 0.40 | 0.07 | 0.17 | 0.02 | 0.13 | 0.02 |
| Sample | Depth, m | Type | C/O Isotopic Compositions, ‰ | Sr isotopic Compositions | |||
|---|---|---|---|---|---|---|---|
| δ13CV–PDB | δ18OV–PDB | δ18OV–SMOW | 87Sr/86Sr | S.D. | |||
| N3-11 | 2967.34 | Matrix | −5.2 | −8.3 | 22.3 | 0.708859 | 0.000015 |
| Calcite cement | −8.7 | −13.7 | 16.8 | 0.708928 | 0.000019 | ||
| R1-21-3 | 2997.45 | Matrix | −2.8 | −9.8 | 20.8 | 0.708567 | 0.000015 |
| Calcite cement | −7.1 | −14.5 | 16 | 0.709384 | 0.000018 | ||
| R1-21-8 | 2987.7 | Matrix | −0.9 | −6.7 | 24 | / | / |
| Calcite cement | −5.9 | −9.2 | 21.4 | / | / | ||
| R1-21-10 | 2987.55 | Matrix | −1.8 | −8.2 | 22.4 | / | / |
| Calcite cement | −5.7 | −10.4 | 20.2 | / | / | ||
| R1-21-13 | 2987.38 | Matrix | −0.9 | −8.3 | 22.4 | / | / |
| Calcite cement | −4 | −13 | 17.5 | / | / | ||
| R2-21-3 | 3092.5 | Matrix | −0.6 | −6.4 | 24.3 | 0.709917 | 0.000013 |
| Calcite cement | −2.6 | −11.7 | 18.8 | 0.712807 | 0.000012 | ||
| R2-21-5 | 3090.56 | Matrix | −1.2 | −6.7 | 24 | 0.709528 | 0.000014 |
| Calcite cement | −3.5 | −13 | 17.5 | 0.711133 | 0.000016 | ||
| R2-21-11 | 3039.13 | Matrix | −0.9 | −6.8 | 23.9 | 0.709042 | 0.000014 |
| Calcite cement | −7.2 | −10.3 | 20.3 | 0.710392 | 0.000016 | ||
| R2-21-21 | 3024 | Matrix | / | / | / | 0.710641 | 0.000014 |
| Calcite cement | / | / | / | 0.710564 | 0.000018 | ||
| FG3-21-3 | 3142.2 | Matrix | 0.1 | −5.8 | 24.9 | 0.710531 | 0.000013 |
| Calcite cement | −7.4 | −11.8 | 18.8 | 0.709961 | 0.000012 | ||
| FG3-21-5 | 3141.58 | Matrix | 0.2 | −5.2 | 25.6 | 0.70952 | 0.000019 |
| Calcite cement | −10.9 | −12.3 | 18.2 | 0.710571 | 0.000016 | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, J.; Zhang, J.; He, Z.; Brandano, M.; Wu, C. Controlling Factors of Meteoric Diagenesis in Karst Reservoirs: An Example from the Majiagou Formation, Ordos Basin, China. Minerals 2023, 13, 812. https://doi.org/10.3390/min13060812
Yang J, Zhang J, He Z, Brandano M, Wu C. Controlling Factors of Meteoric Diagenesis in Karst Reservoirs: An Example from the Majiagou Formation, Ordos Basin, China. Minerals. 2023; 13(6):812. https://doi.org/10.3390/min13060812
Chicago/Turabian StyleYang, Jiaqi, Juntao Zhang, Zhiliang He, Macro Brandano, and Chongyang Wu. 2023. "Controlling Factors of Meteoric Diagenesis in Karst Reservoirs: An Example from the Majiagou Formation, Ordos Basin, China" Minerals 13, no. 6: 812. https://doi.org/10.3390/min13060812
APA StyleYang, J., Zhang, J., He, Z., Brandano, M., & Wu, C. (2023). Controlling Factors of Meteoric Diagenesis in Karst Reservoirs: An Example from the Majiagou Formation, Ordos Basin, China. Minerals, 13(6), 812. https://doi.org/10.3390/min13060812





