Preparation of High Purity V2O5 from a Typical Low-Grade Refractory Stone Coal Using a Pyro-Hydrometallurgical Process
Abstract
:1. Introduction
2. Experimental
2.1. Materials and Apparatus
2.2. Procedure
2.2.1. Roasting of Stone Coal
2.2.2. Leaching of Vanadium
2.2.3. Solvent Extraction and Stripping of Vanadium
2.2.4. Precipitation and Thermal Decomposition
3. Results and Discussion
3.1. Characterization of Raw Stone Coal
3.2. Effects of Roasting Conditions on Vanadium Leaching
3.3. Sulfuric Acid Leaching of Roasted Stone Coal
3.4. Solvent Extraction of Vanadium from Acid Leaching Solution
3.5. Preparation of V2O5
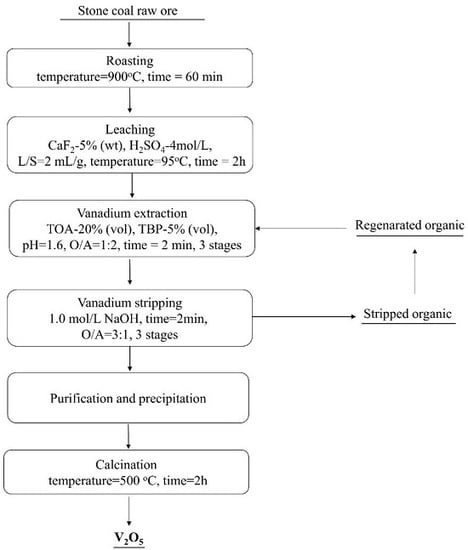
3.6. Establishment of Process Flow Sheet for Vanadium Extraction
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Moskalyk, R.R.; Alfantazi, A.M. Processing of vanadium: A review. Miner. Eng. 2003, 16, 793–805. [Google Scholar] [CrossRef]
- Zhang, Y.M.; Bao, S.X.; Liu, T.; Chen, T.J.; Huang, J. The technology of extracting vanadium from stone coal in China: History, current status and future prospects. Hydrometallurgy 2011, 109, 116–124. [Google Scholar] [CrossRef]
- Zhang, Y.; Bao, S.; Liu, T.; Huang, J.; Chen, T. Research status and prospect of vanadium extraction from stone coal in China. Non-Ferrous Metals Extr. Metall. 2015, 2, 24–29. (In Chinese) [Google Scholar]
- Hu, Y.; Zhang, Y.; Bao, S.; Tao, L. Effects of the mineral phase and valence of vanadium on vanadium extraction from stone coal. Int. J. Miner. Metall. Mater. 2012, 19, 893–897. [Google Scholar] [CrossRef]
- Liu, Y.; Yang, C.; Li, P.; Li, S. A new process of extracting vanadium from stone coal. Int. J. Miner. Metall. Mater. 2010, 17, 381–388. [Google Scholar] [CrossRef]
- Li, X.; Xie, B. Extraction of vanadium from high calcium vanadium slag using direct roasting and soda leaching. Int. J. Miner. Metall. Mater. 2012, 19, 595–601. [Google Scholar] [CrossRef]
- Chen, T.; Zhang, Y.; Song, S. Improved extraction of vanadium from a Chinese vanadium-bearing stone coal using a modified roast-leach process. Asia-Pac. J. Chem. Eng. 2010, 5, 778–784. [Google Scholar] [CrossRef]
- He, D.; Feng, Q.; Zhang, G.; Ou, L.; Lu, Y. An environmentally-friendly technology of vanadium extraction from stone coal. Miner. Eng. 2007, 2011, 84–1186. [Google Scholar] [CrossRef]
- Wang, T.; Xu, L.; Liu, C.; Zhang, Z. Calcified roasting-acid leaching process of vanadium from low-grade vanadium-containing stone coal. Chin. J. Geochem. 2014, 33, 163–167. [Google Scholar] [CrossRef]
- Wang, M.; Wang, X.; Shen, J.; Wu, R. Extraction of vanadium from stone coal by modified salt-roasting process. J. Cent. South Univ. Technol. 2011, 18, 1940–1944. [Google Scholar] [CrossRef]
- Wang, M.; Xiao, L.; Li, Q.; Wang, X.; Xiang, X. Leaching of vanadium from stone coal with sulfuric acid. Rare Metals 2009, 28, 1–4. [Google Scholar] [CrossRef]
- Deng, Z.; Wei, C.; Fan, G.; Li, M.; Li, C.; Li, X. Extracting vanadium from stone-coal by oxygen pressure acid leaching and solvent extraction. Trans. Non-Ferrous Metall. Soc. China 2010, 20, s118–s122. [Google Scholar] [CrossRef]
- Wang, M.; Xiang, X.; Zhang, L.; Xiao, L. Effect of vanadium occurrence state on the choice of extracting vanadium technology from stone coal. Rare Metals 2008, 27, 112–115. [Google Scholar] [CrossRef]
- Bei, S.; Wang, Z.; Li, Q.; Zhang, Y. Review of vanadium extraction from stone coal by roasting technique with sodium chloride and calcium oxide. J. Cent. South Univ. Technol. 2010, 34, 291–297. (In Chinese) [Google Scholar]
- Li, C.; Zhou, X.; Wang, H.; Zhang, T.; Li, J.; Ou, X.; Jiang, X. Effect of oxidation on vanadium extraction from stone coal with calcified roasting. J. Cent. South Univ. Technol. 2011, 42, 7–10. (In Chinese) [Google Scholar]
- Chen, X.; Lan, X.; Zhang, Q.; Ma, H.; Zhou, J. Leaching vanadium by high concentration sulfuric acid from stone coal. Trans. Non-Ferrous Metall. Soc. China 2010, 20, s123–s126. [Google Scholar] [CrossRef]
- Vitolo, S.; Seggiani, M.; Filippi, S.; Brocchini, C. Recovery of vanadium from heavy oil and Orimulsion fly ashes. Hydrometallurgy 2000, 57, 141–149. [Google Scholar] [CrossRef]
- Nguyen, T.H.; Lee, M.S. Recovery of molybdenum and vanadium with high purity from sulfuric acid leach solution of spent hydrodesulfurization catalysts by ion exchange. Hydrometallurgy 2014, 147–148, 142–147. [Google Scholar] [CrossRef]
- Nguyen, T.H.; Lee, M.S. Separation of molybdenum and vanadium from acid solutions by ion exchange. Hydrometallurgy 2013, 136, 65–70. [Google Scholar] [CrossRef]
- Barik, S.P.; Park, K.H.; Nam, C.W. Process development for recovery of vanadium and nickel from an industrial solid waste by a leaching-solvent extraction technique. J. Environ. Manag. 2014, 146, 22–28. [Google Scholar] [CrossRef] [PubMed]
- Noori, M.; Rashchi, F.; Babakhani, A.; Vahidi, E. Selective recovery and separation of nickel and vanadium in sulfate media using mixtures of D2EHPA and Cyanex 272. Sep. Purif. Technol. 2014, 136, 265–273. [Google Scholar] [CrossRef]
- Nguyen, T.; Lee, M. Solvent extraction of vanadium (V) from sulfate solutions using LIX 63 and PC 88A. J. Ind. Eng. Chem. 2015, 31, 118–123. [Google Scholar] [CrossRef]
- Hu, G.; Chen, D.; Wang, L.; Liu, J.; Zhao, H.; Liu, Y.; Qi, T.; Zhang, C.; Yu, P. Extraction of vanadium from chloride solution with high concentration of iron by solvent extraction using D2EHPA. Sep. Purif. Technol. 2014, 125, 59–65. [Google Scholar] [CrossRef]
- Tavakoli, M.R.; Dreisinger, D.B. Separation of vanadium from iron by solvent extraction using acidic and neutral organophosporus extractants. Hydrometallurgy 2014, 141, 17–23. [Google Scholar] [CrossRef]
- Cote, G. Hydrometallurgy of strategic metals. Solvent Extr. Ion Exch. 2000, 18, 703–727. [Google Scholar] [CrossRef]
- Kim, H.I.; Lee, K.W.; Mishra, D.; Yi, K.M.; Hong, J.H.; Jun, M.K.; Park, H.K. Separation and recovery of vanadium from leached solution of spent residue hydrodesulfurization (RHDS) catalyst using solvent extraction. J. Ind. Eng. Chem. 2014, 20, 4457–4462. [Google Scholar] [CrossRef]
- Lee, K.W.; Mishra, D.; Yi, K.M.; Hong, J.H.; Jun, M.K.; Park, H.K. Separation of molybdenum and vanadium from oxalate leached solution of spent residue hydrodesulfurization (RHDS) catalyst by liquid-liquid extraction using amine extractant. J. Ind. Eng. Chem. 2015, 21, 1265–1269. [Google Scholar]
- Kumar, J.; Shin, S.; Yoon, H.; Nam, C.; Chung, K.; Lee, J.; Park, J. Separation and recovery of vanadium from synthetic leach liquor solutions containing iron, calcium, sodium, aluminum and manganese by the solvent extraction technique. Sep. Sci. Technol. 2014, 49, 819–828. [Google Scholar] [CrossRef]
- Ning, P.; Lin, X.; Wang, X.; Cao, H. High-efficient extraction of vanadium and its application in the utilization of the chromium-bearing vanadium slag. Chem. Eng. J. 2016, 301, 132–138. [Google Scholar] [CrossRef]
- Bao, S.; Zhang, Y.; Huang, J.; Yang, X.; Hu, Y. Determination of vanadium valency in roasted stone coal by separate dissolve-potentiometric titration method. MRS Proc. 2011, 1380, 98–103. [Google Scholar]
- He, D.; Feng, Q.; Zhang, G.; Luo, W.; Ou, L. Study on leaching vanadium from roasted residue of stone coal. Miner. Metall. Proc. 2008, 25, 181–184. [Google Scholar]
- Wang, Y.; Li, D.; Zhang, H. Main factors affecting precipitation of vanadium with acidic ammonium salt and its countermeasures. Iron Alloy 2012, 4, 12–16. (In Chinese) [Google Scholar]
- Wang, X.; Wang, M.; Shi, L.; Hu, J.; Qiao, P. Recovery of vanadium during ammonium molybdate production using ion exchange. Hydrometallurgy 2010, 104, 317–321. [Google Scholar] [CrossRef]
- Yan, Q.; He, Q.; Li, S.; Ye, Y.; Chen, Z.; Peng, Y.; Li, D. Vanadium pentoxide. Chinese Standard No. YB/T 5304–2011. 2011. (In Chinese) [Google Scholar]









| Composition | V2O5 | Fe2O3 | SiO2 | Al2O3 | MgO | K2O | Na2O | SO3 | CaO | LOI |
|---|---|---|---|---|---|---|---|---|---|---|
| Content | 0.71 | 4.99 | 49.28 | 8.91 | 2.18 | 3.02 | 0.38 | 4.35 | 6.26 | 17.82 |
| NO. | C | O | Mg | Al | Si | S | K | V | Fe | Ba | F | P | Ca |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 15.35 | 17.70 | 1.21 | 7.95 | 46.20 | 1.24 | 6.45 | 1.40 | 2.49 | ||||
| 2 | 27.76 | 0.47 | 8.53 | 51.28 | 0.53 | 8.55 | 0.62 | 0.95 | 1.30 | ||||
| 3 | 26.85 | 11.42 | 9.16 | 0.99 | 3.57 | 26.13 | 21.88 | ||||||
| 4 | 20.35 | 22.76 | 0.53 | 47.80 | 2.15 | 0.37 | 0.51 | 3.31 | 2.22 | ||||
| 5 | 12.00 | 29.68 | 0.60 | 2.47 | 53.90 | 1.34 | |||||||
| 6 | 13.68 | 1.30 | 7.67 | 55.78 | 8.66 | 5.64 | 1.48 | 0.81 | |||||
| 7 | 25.94 | 0.53 | 2.80 | 67.45 | 1.02 | 1.43 | 0.83 | ||||||
| 8 | 9.45 | 8.91 | 1.47 | 2.58 | 18.36 | 23.27 | 1.16 | 34.81 | |||||
| 9 | 24.27 | 7.89 | 55.33 | 0.58 | 8.54 | 3.38 | |||||||
| 10 | 11.43 | 23.22 | 0.91 | 4.18 | 56.16 | 2.89 | 0.49 | 0.71 | |||||
| 11 | 34.02 | 22.01 | 1.13 | 2.63 | 34.20 | 1.52 | 1.54 | 1.12 | 1.83 | ||||
| 12 | 6.60 | 15.67 | 7.89 | 34.13 | 14.39 | 8.23 | 9.02 | 4.07 | |||||
| 13 | 25.68 | 0.88 | 5.51 | 62.61 | 0.99 | 3.27 | 1.06 | ||||||
| 14 | 8.96 | 34.29 | 56.75 |
| Items | V | Fe | Al | Mg | K | Ca | P | Si |
|---|---|---|---|---|---|---|---|---|
| Concentration (g/L) | 1.89 | 3.88 | 12.74 | 3.46 | 3.08 | 0.65 | 1.07 | 0.38 |
| Solution | V | Fe | Al | Mg | P | Si |
|---|---|---|---|---|---|---|
| Stripped solution (g/L) | 25.64 | 0.011 | 0.008 | 0.006 | 0.087 | 0.032 |
| Purified solution (g/L) | 25.32 | 0.002 | 0.001 | 0.001 | 0.011 | 0.008 |
| Component (%) | V2O5 | Si | Fe | P | S | As | K2O + Na2O | |
|---|---|---|---|---|---|---|---|---|
| V2O5 Product in This Study | 99.75 | 0.026 | nill | 0.009 | 0.006 | nill | 0.012 | |
| YB/T5304-2011 | V2O5 99 | ≥99.0 | ≤0.20 | ≤0.20 | ≤0.03 | ≤0.01 | ≤0.01 | ≤1.0 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, X.; Zhang, Y.; Bao, S. Preparation of High Purity V2O5 from a Typical Low-Grade Refractory Stone Coal Using a Pyro-Hydrometallurgical Process. Minerals 2016, 6, 69. https://doi.org/10.3390/min6030069
Yang X, Zhang Y, Bao S. Preparation of High Purity V2O5 from a Typical Low-Grade Refractory Stone Coal Using a Pyro-Hydrometallurgical Process. Minerals. 2016; 6(3):69. https://doi.org/10.3390/min6030069
Chicago/Turabian StyleYang, Xiao, Yimin Zhang, and Shenxu Bao. 2016. "Preparation of High Purity V2O5 from a Typical Low-Grade Refractory Stone Coal Using a Pyro-Hydrometallurgical Process" Minerals 6, no. 3: 69. https://doi.org/10.3390/min6030069