Abstract
Ab initio thermodynamic properties, equation of state and phase stability of periclase (MgO, B1-type structure) have been investigated in a broad P–T range (0–160 GPa; 0–3000 K) in order to set a model reference system for phase equilibria simulations under deep Earth conditions. Phonon dispersion calculations performed on large supercells using the finite displacement method and in the framework of quasi-harmonic approximation highlight the performance of the Becke three-parameter Lee-Yang-Parr (B3LYP) hybrid density functional in predicting accurate thermodynamic functions (heat capacity, entropy, thermal expansivity, isothermal bulk modulus) and phase reaction boundaries at high pressure and temperature. A first principles Mie–Grüneisen equation of state based on lattice vibrations directly provides a physically-consistent description of thermal pressure and P–V–T relations without any need to rely on empirical parameters or other phenomenological formalisms that could give spurious anomalies or uncontrolled extrapolations at HP–HT. The post-spinel phase transformation, Mg2SiO4 (ringwoodite) = MgO (periclase) + MgSiO3 (bridgmanite), is taken as a computational example to illustrate how first principles theory combined with the use of hybrid functionals is able to provide sound results on the Clapeyron slope, density change and P–T location of equilibrium mineral reactions relevant to mantle dynamics.
1. Introduction
Building a reliable mineralogical model for the deep Earth and planetary interiors requires accurate knowledge of thermodynamic properties of the constituent phases at extreme conditions of pressure and temperature in order to obtain meaningful stability relations. Most of the attempts made so far in the literature [1,2,3,4,5,6,7,8] rely on phase equilibrium models, which are based on the assessment of experimental datasets, while very few of them use first principles data to calculate phase diagrams [9,10,11,12]. However, a full ab initio thermodynamic database for phase equilibria calculation at high pressure and temperature (HP–HT) has not been created yet, even for simple compositional systems. The first step to achieving this goal is predicting the thermodynamic properties of the main mineralogical constituents, and using them to define P–T stability fields and model phase transition and reaction boundaries by Gibbs free energy minimization. This, in turn, provides the energetic ground to understand the role of phase stability on the origin of seismic discontinuities and large-scale geodynamic processes in the deep Earth [13,14].
Periclase (MgO with fcc or B1-type cubic structure), along with with silicate perovskites (bridgmanite and Ca-perovskite), is one of the main constituents of the Earth’s lower mantle [15] and, much more than silicate perovskites, controls its rheological properties [16]. Due to its simple structure and large stability field, it has always been considered to be a milestone for computational investigation, also because of the possibility of comparing calculations with a great variety of experimental data in a broad range of P–T conditions. Nevertheless, despite this apparent simplicity, MgO could display complex features under HP–HT conditions (like a strong single-crystal elastic anisotropy [17,18]), and some aspects related to its thermodynamic and thermophysical properties under those conditions are still poorly defined [19].
In this work a first principles, comprehensive thermodynamic investigation of MgO is carried out up to HP–HT conditions (i.e., 0–160 GPa; 0–3000 K) using hybrid density functional theory (DFT). Hybrid functionals, such as B3LYP [20], have been shown to provide excellent results, and a real improvement with respect to pure local density approximation (LDA) and generalized-gradient approximation (GGA) methods with regard to the simulation of elastic, vibrational and thermodynamic properties of insulating solids (such as Mg–Al silicates and oxides) at HP–HT conditions [21,22,23,24,25]. A comparison between ab initio thermodynamic properties of minerals calculated by hybrid functionals and state-of-the-art experimental results obtained by calorimetry, vibrational spectroscopy and X-ray diffraction techniques has been made by several studies in the literature, which have pointed out that the level of accuracy is within 1–2%, on average, for the majority of thermodynamic properties [26,27,28,29,30,31,32,33]. On the other hand, the main sources of error (such as volume overestimate in GGA-based DFT calculations), are usually well-known and under control [34,35,36].
Some key aspects on the thermodynamics of MgO are emphasized in this work. First, a detailed survey on the accuracy of quasi-harmonic lattice dynamics calculations performed on large supercells with the B3LYP hybrid density functional is carried out by comparing the results obtained on thermodynamic and themophysical properties (i.e., heat capacities, entropy, bulk moduli, thermal expansivity, Grüneisen parameter) with selected experimental data and other theoretical calculations. Second, a first principles Mie–Grüneisen equation of state (FPMG-EOS) based on lattice dynamics is developed to provide physically-consistent P–V–T relations for solids at HP–HT conditions and the results are compared with those obtained by the use of a finite strain equation of state (like the 3rd order Birch–Murnaghan EOS). Finally, the computed thermodynamic dataset is used to predict the phase equilibrium boundary of the post-spinel transformation, Mg2SiO4 (ringwoodite) = MgO (periclase) + MgSiO3 (bridgmanite), which is one of the most relevant to the physics of the Earth’s interior.
2. Materials and Methods
Ab initio calculations in this work have been performed with the CRYSTAL14 code [37] by using an all-electron Gaussian-type basis set and the hybrid B3LYP functional [19,38]. Hybrid functionals, which contain a fraction of exact Hartree–Fock exchange (i.e., 20% in the case of B3LYP) mixed to the GGA exchange-correlation, are widely and successfully used in solid-state chemistry [34,35].
Magnesium and oxygen are described by (8s)-(511sp)-(1d) and (8s)-(411sp)-(1d) contractions, respectively. The exponents (in bohr−2) of the most diffuse functions are αsp = 0.2245 and αd = 0.50 for Mg, αsp = 0.181 and αd = 0.60 for O. These basis functions have been optimized and successfully used in previous works on magnesium silicates [23,28,31,33].
The level of accuracy in evaluating the Coulomb and Hartree-Fock exchange series is controlled by five parameters [37], for which the 8 8 8 9 18 values are used here. The threshold on the self-consistent field (SCF) energy is set to 10−8 Hartree for geometry optimization and equation of state calculation, and to 10−10 Hartree for computation of vibrational frequencies. The reciprocal space is sampled according to a regular sublattice of k-points according to the Monkhorst–Pack scheme [39]. The sampling of electronic states in the reciprocal space is defined by a shrinking factor set to 8 (along the three lattice vectors) [37], corresponding to 29 independent k vectors in the irreducible part of the Brillouin zone (BZ). The DFT exchange-correlation contribution is evaluated by numerical integration over the unit cell volume. In CRYSTAL, radial and angular points of the grid are generated through Gauss–Legendre radial quadrature and Lebedev two-dimensional angular point distributions. A (75,974)p grid is used, corresponding to a pruned grid with 75 radial and 974 angular points (see [37] for details). Accuracy in the integration can be estimated by the error Δe on the integrated electronic charge density in the unit cell (i.e., Δe = 4 × 10−5 on a total of 20 electrons per unit cell).
Geometry optimization is performed by using analytical energy gradients with respect to atomic coordinates and unit cell parameters within a quasi-Newton scheme combined with the Broyden-Fletcher-Godfarb-Shanno (BFGS) algorithm for Hessian updating [40,41,42]. Convergence is tested on the root-mean-square (RMS) and the absolute value of the largest component of both the gradients and nuclear displacements. The thresholds for RMS forces and nuclear displacements are set to 0.00015 a.u. and 0.0001 a.u., which are 2 and 10 times more severe than the default values, respectively [37].
The calculation of the vibrational frequencies at the Γ point of the BZ (i.e., k = 0) is performed within the harmonic approximation by constructing the mass-weighted Hessian matrix via numerical integration of the analytical gradients with respect to the atomic Cartesian coordinates (see [43] for further details). The optimized equilibrium geometry is taken as reference for the calculation of vibrational frequencies. Phonon dispersion calculations at k-points other than Γ are performed on large supercells with a direct-space approach [44,45], as implemented in the CRYSTAL14 program [37]. The k-point mesh used to sample the reciprocal space in phonon dispersion calculation is different from that used for the electronic states, as it depends from the shape and the size of the adopted supercell. In general, the bigger the supercell, the denser the sampling of the phonon dispersion in reciprocal space. To check the numerical convergence of thermodynamic properties, isotropic supercells of increasing size have been considered (see below): a 5 × 5 × 5 supercell with 250 atoms and 750 normal modes sampled at 125 k-points in the first BZ turns out to be enough to ensure convergence on harmonic thermodynamic functions (e.g., isochoric heat capacity and vibrational entropy) in the whole P–T range.
Mode-gamma Grüneisen parameters (γi), which define the volume dependence of the vibrational frequencies in the framework of the quasi-harmonic approximation (QHA), are computed by least-square fitting of the vibrational frequencies determined at eight different volume conditions (i.e., V/V0 = 0.65, 0.70, 0.75, 0.80. 0.85, 0.90, 0.95 and 1.00). Third-order polynomial fitting turns out to accurately describe the volume dependence of all the vibrational frequencies at different k-points in the first BZ. The same volume conditions are then considered to calculate zero-point correction and thermal pressure contributions and define the full set of P–V–T relations of MgO.
3. Results and Discussion
Selected results obtained for thermophysical and thermodynamic properties and P–V–T equation of state of periclase in a broad range of P–T conditions (i.e., 0–160 GPa; 0–3000 K) are presented and discussed in the following sections. The computed dataset is then used to simulate the post-spinel reaction boundary, Mg2SiO4 (ringwoodite) = MgO (periclase) + MgSiO3 (bridgmanite), and hence to test the capability of ab initio thermodynamic data to predict reliable phase equilibria under deep Earth conditions.
3.1. Thermal Expansion and Thermophysical Properties
Thermophysical properties of solids can be derived by the statistical mechanics analysis of vibrational modes of the crystal lattice [46]. The Helmholtz free energy, F(V,T), for instance, can be expressed as:
where U0(V) is the static energy calculated at different volumes (the so-called “potential well”); h and k are the Planck and Boltzmann constants, respectively; n represents the number of atoms in the (primitive) unit cell of the crystal; νi is the frequency of the ith vibrational mode in the crystal lattice; and
is the zero-point correction energy related to the atomic vibrations at T = 0 K. By applying the thermodynamic identity:
we obtain the following expression of the product αKT (i.e., the coefficient of volume thermal expansion or thermal expansivity, α, times the isothermal bulk modulus, KT):
where R = kN0 is the universal gas constant (N0 is the Avogadro number); Z is the number of unit formula in the (primitive) unit cell; V is the molar volume (in J/bar); Xi is the adimensionalized frequency, defined as:
and γi is the the mode-gamma Grüneisen parameter of the ith vibrational mode of the crystal lattice, defining its volume dependence in the framework of quasi-harmonic approximation (QHA) [47], i.e.,
If the summation in Equation (4) is taken over all the vibrational modes sampled at discrete k-points in the first BZ, Equation (4) can be rewritten as:
Both the effect of phonon dispersion and the contribution of the acoustic modes are thus straightforwardly included in Equation (7).
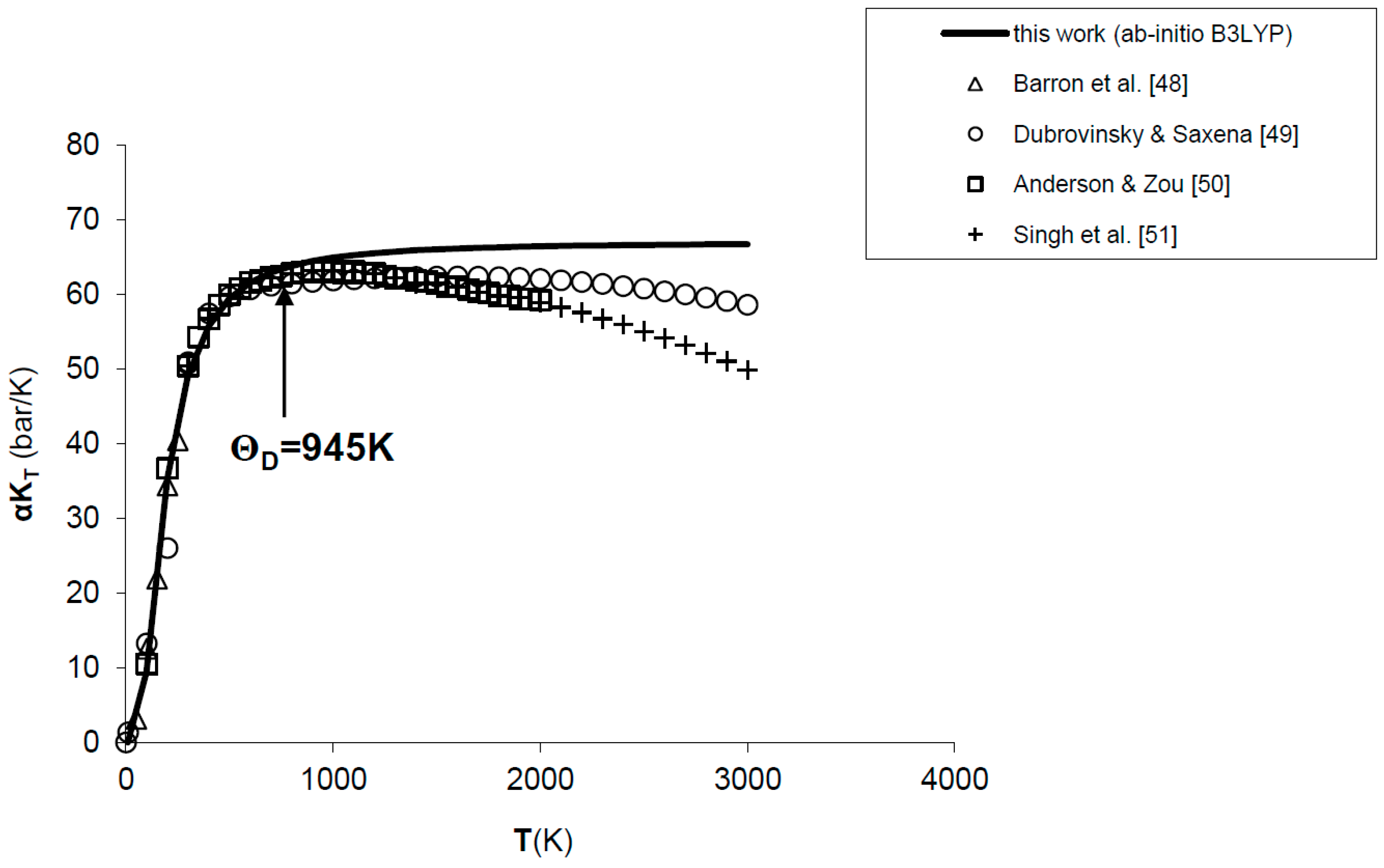
The values calculated for the product αKT of MgO are shown in Figure 1. The ab initio quasi-harmonic calculation reproduces well the experimental data [48,49] and thermodynamic assessments [50,51] up to the Debye temperature of the phase (i.e., ΘD = 945 K); then, a deviation occurs due to a common drawback of QHA at low-pressure and high-temperature conditions. In particular, while experimental observations point out a decrease of αKT values beyond T ≈ 1000 K, ab initio results attain an asymptotic value due to the functional form of Equation (4) or (7).
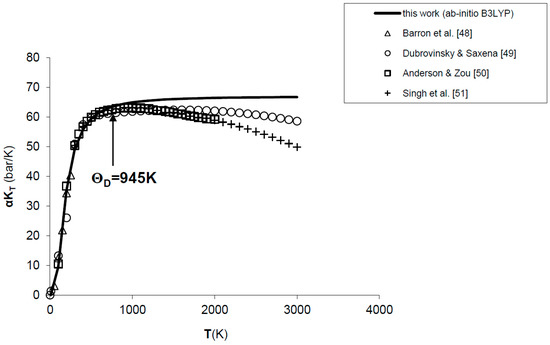
Figure 1.
Ab initio B3LYP results for the product αKT of MgO, as compared to experimental data [48,49] and thermodynamic assessments [50,51]. ΘD = 945 K is the Debye temperature.
Once the values of the product αKT are known, thermal expansivity (α) can be calculated from the values of KT (i.e., the isothermal bulk modulus at dicrete T conditions). The latter can be obtained by fitting F–V–T (or P–V–T) values according to:
So, once free energies at different volumes are available (cf. Equation (1)), one can proceed with the finite difference calculation of the second derivatives of the free energy to derive KT. In principle, this should be the best choice, but a practical problem is that the amount of F–V–T data to fit is usually not so high, due to the high computational cost of full phonon dispersion calculations. Another problem is that the definition of K′T (i.e., the T-dependence of the bulk modulus pressure derivative or, in other words, the mixed P–T derivative of the bulk modulus) is often poor, and is affected by large uncertainties [52], so that K′ is often assumed to be independent of temperature (which is not always the case). There are two different ways to overcome these problems. The first is to use the polynomial functions which define the mode-gamma Grüneisen parameters in Equation (6) to fit the vibrational frequencies under any volume conditions, and then to detail the grid of F–V–T (or P–V–T) data. The second is to use a phenomelogical EOS formalism (like the 3rd- or 4th-order Birch-Murnaghan equation of state, BM3-EOS or BM4-EOS) to fit the data and derive the thermoelastic parameters [53,54]; this is a common practice in both experimental and computational works [55,56] and, in principle, allows for the determination of K′T values, too. The basic difference between the two approaches is that the former is mostly numerical, while the latter is fully analytical.
The ab initio results of KT and K′T obtained for MgO in this work, as compared to experimental measurements and other theoretical calculations, are shown in Table 1 and Table 2 and Figure 2.

Table 1.
Ab initio B3LYP isothermal bulk modulus (KT) of MgO. Values obtained in this work are compared to experiments [57] and other ab initio simulations at LDA [58,59] and GGA [61] level of theory.

Table 2.
Ab initio B3LYP temperature dependence of the isothermal bulk modulus pressure derivative (K′T) of MgO. Values obtained in this work are compared to other ab initio simulations at LDA [59] and GGA [61] level of theory and thermodynamic assessment [63].
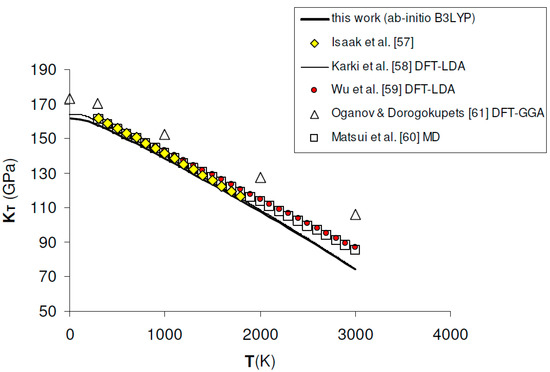
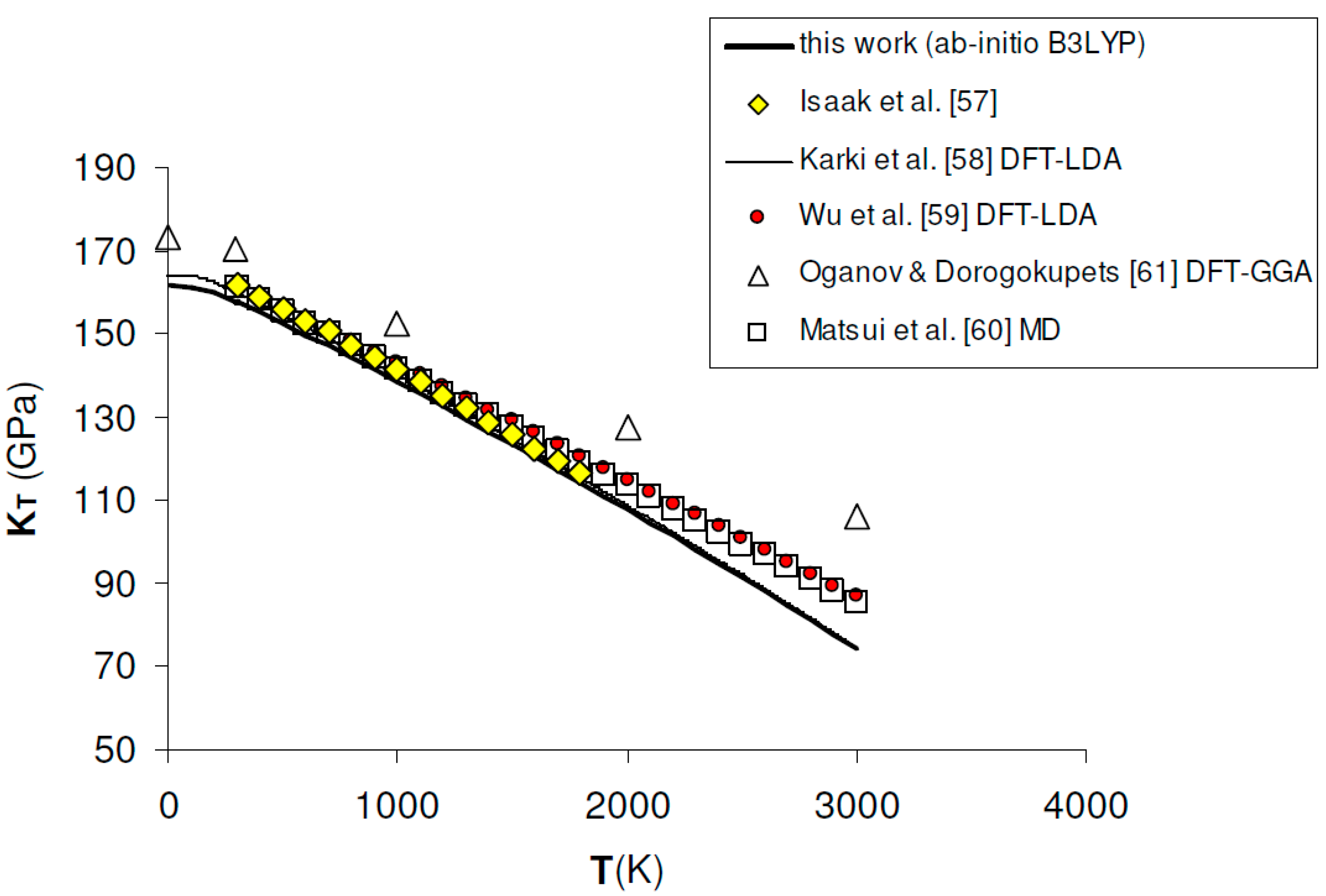
Figure 2.
Ab initio B3LYP isothermal bulk modulus (KT) of MgO. Calculated results in this work are compared to experimental data [57], other ab initio LDA [58,59] and GGA [61] calculations and semi-empirical MD simulation [60].
Ab initio B3LYP values of KT, as obtained by a full phonon dispersion calculation of P–V–T relations fitted with a BM3-EOS (see Section 3.3), are in excellent agreement with the experimental results determined by rectangular parallelepiped resonance technique in the range T = 300–1800 K at P = 1 bar [57] (see Figure 2). Although theoretical data taken from the literature are quite scattered, B3LYP results compare favorably both with LDA results [58,59] and semi-empirical molecular dynamics [60], while GGA values seem to be overestimated over the whole T range [61] (see Table 1 and Figure 2). As expected, KT values slightly deviate from MD results beyond T ≈ 2000 K, which roughly corresponds to 2/3 of the melting temperature of MgO [49]. Zero-point vibrational effects on the isothermal bulk modulus turn out to be remarkable, since KT is lowered by ~5.5 GPa (i.e., ~3.3%) with respect to the B3LYP static value (i.e., K0 = 167.01 GPa). The zero-point correction effect on KT is equal to ~6%, ~4.6% and ~2.3% according to LDA [58], GGA [61] and hybrid WC1LYP [62] calculations, respectively (Table 1). K′T increases with temperature and the temperature dependence predicted in this work is in good agreement both with LDA calculations [59] and thermodynamic assessments [63]. The effect of zero-point motions is again non-negligible, increasing the static value from 3.95 to 4.01 (Table 2). GGA calculations [61] are in good agreement with B3LYP at low temperatures, while they give lower values of K′T in the medium- to high-temperature range.
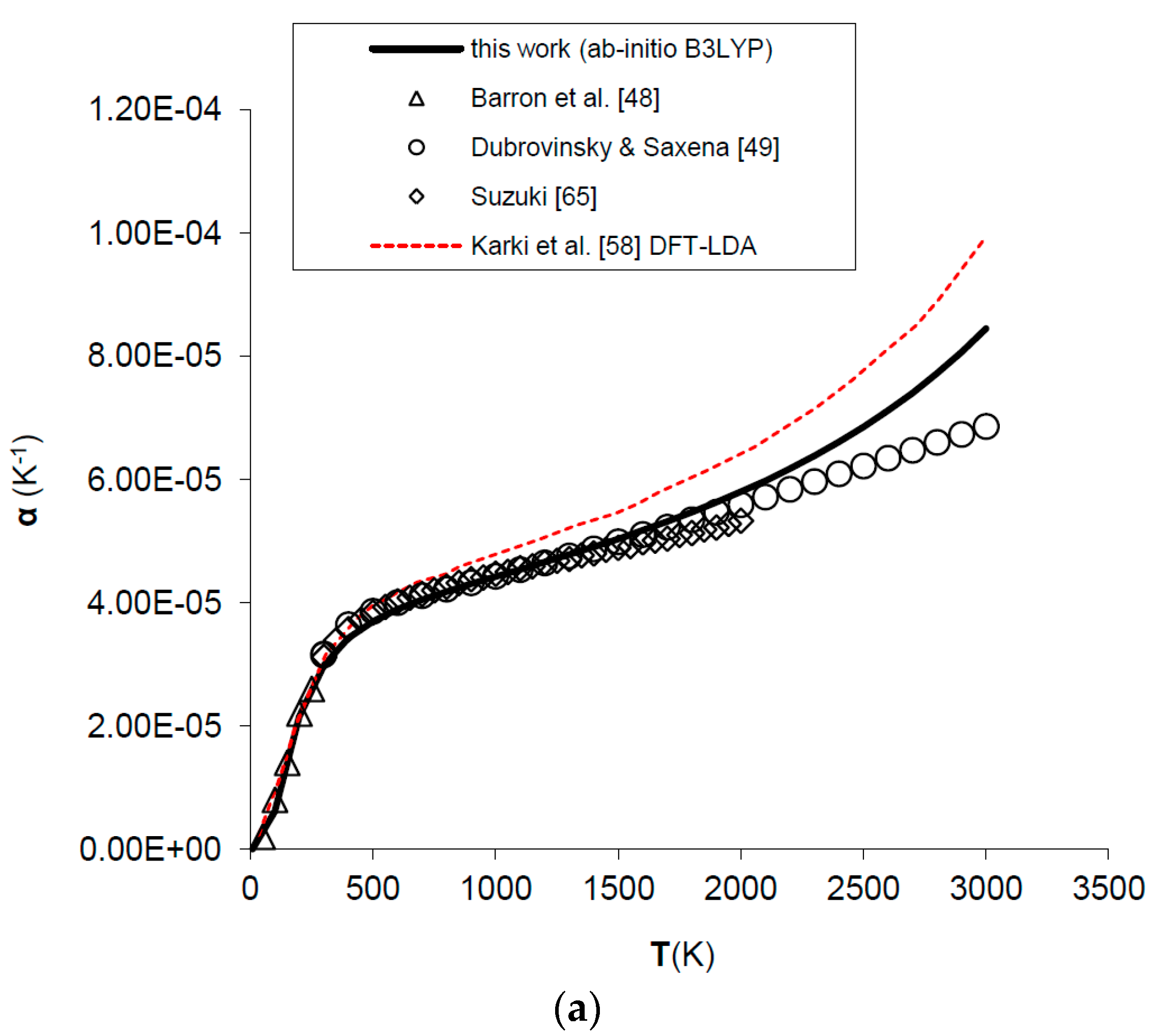
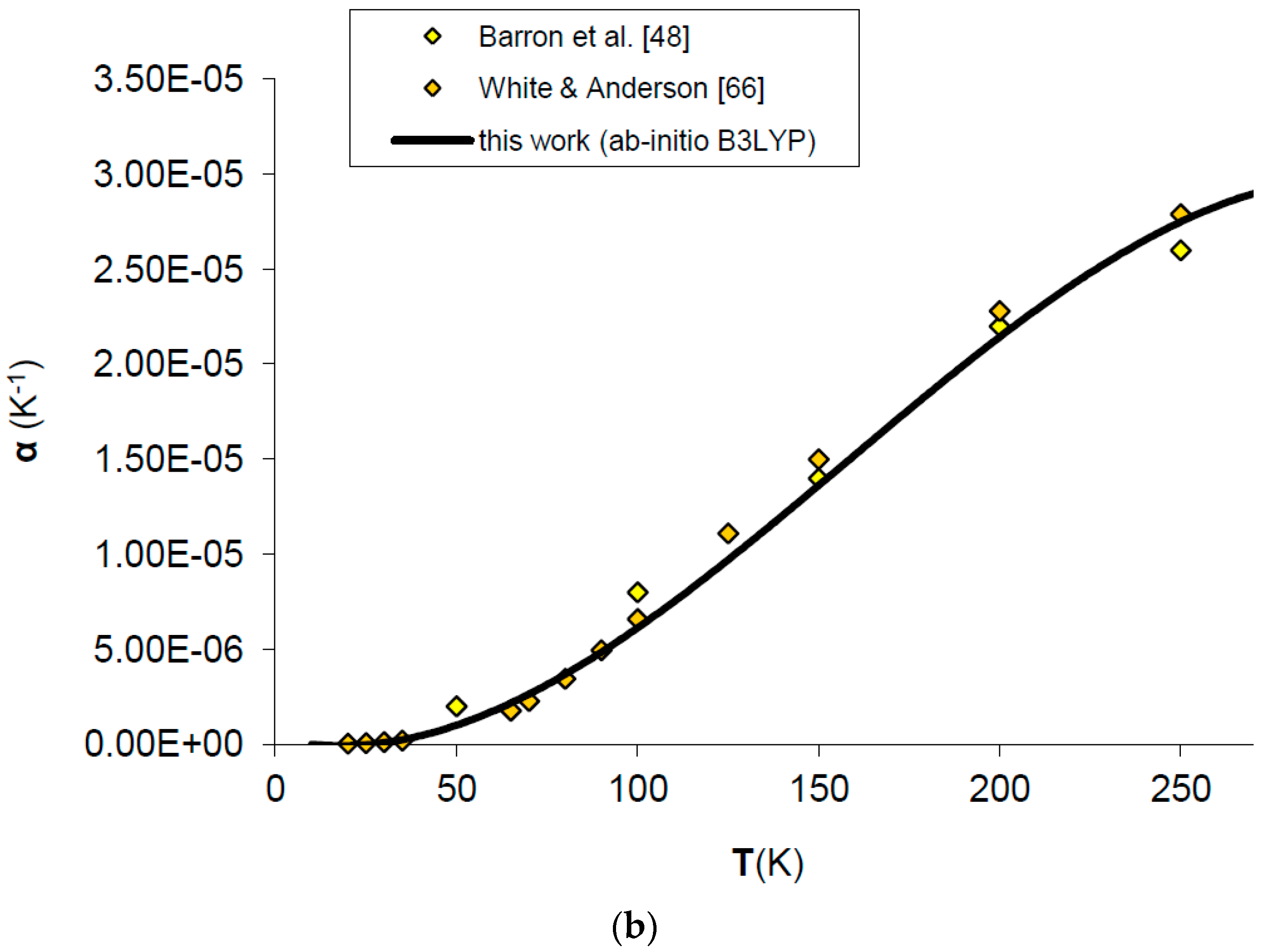
Thermal expansivity (α) can be simply obtained dividing out the values of the product αKT by the values of KT. In spite of the fact that B3LYP systematically overestimates molar volumes by ~2%, the relative volume thermal expansion (i.e., V/V0) and thermal expansivity (α) at discrete temperatures and ambient pressure are reproduced reasonably well up to 2/3 of the melting temperature (i.e., T ≈ 2000 K), then both start to deviate from the observed trend [49,64,65] (see Figure 3 and Figure 4a). The agreement of ab initio B3LYP thermal expansivity with experimental data in the low-T range (i.e., T < 298.15 K) [48,66] is excellent (see Figure 4b). The volume thermal expansion coefficient at ambient conditions as calculated in this work (i.e., α0 = 2.97 × 10−5 K−1) fits well the experimental values, the latter being around α0 = (3.12 ± 0.16) × 10−5 K−1 [49,64,65,66,67]. The limit of QHA in reproducing thermal expansion at very high-temperature conditions is particularly evident in the values of α (Figure 4a); this is the reason that inflection points in the thermal epansivity function α = f(T) at various pressures have been considered by some authors to be a clue to empirically defining the P–T range of validity of QHA [68]. Nevertheless, it’s interesting to note that B3LYP results show a less marked deviation in thermal expansivity at LP–HT with respect to those given by other density functionals (e.g., LDA) (see Figure 4a).
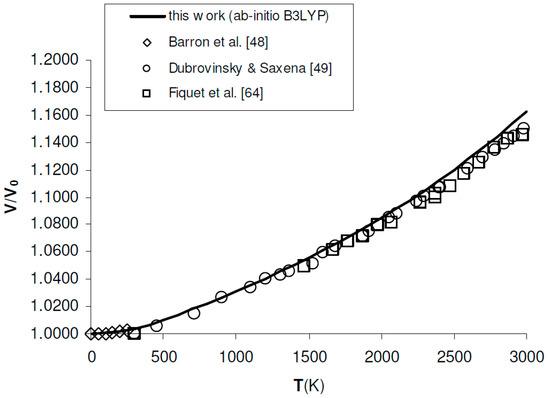
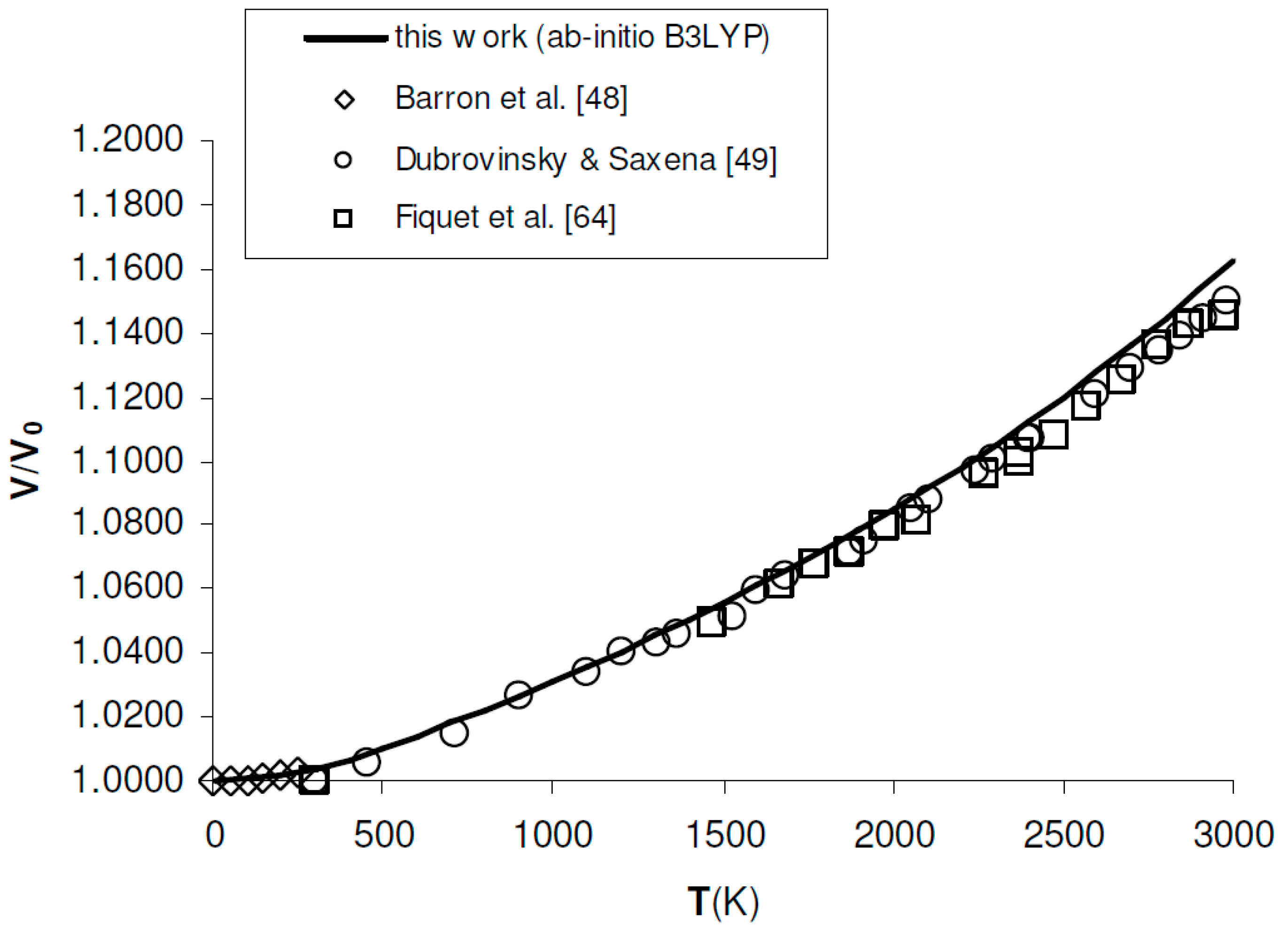
Figure 3.
Ab initio B3LYP relative volume thermal expansion (V/V0) of MgO, as compared to low-T to high-T experimental data [48,49,64]. Reference volume is V0 = 75.762 Å3 = 11.406 cm3/mol (T = 0 K, P = 0 GPa, including ZPC).
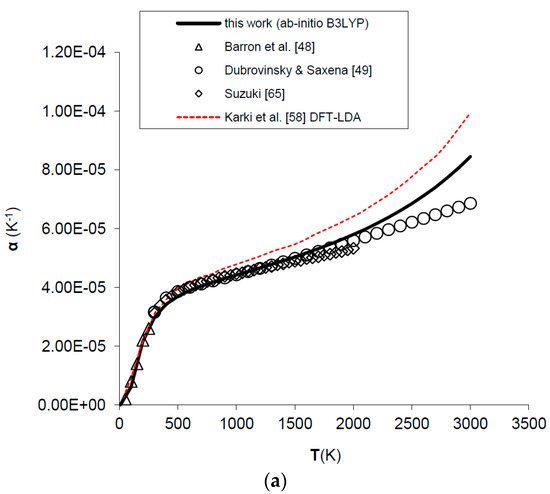
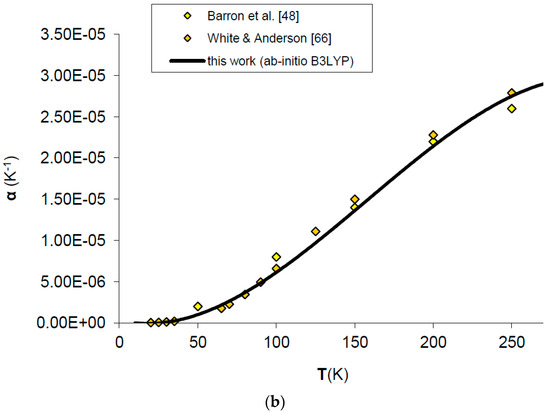
Figure 4.
Ab initio B3LYP thermal expansivity (α) of MgO: calculated results in this work are compared to experimental data and other ab initio LDA calculations (a) in the whole T-range [48,49,58,65]; (b) in the low-T range [48,66].
3.2. Thermodynamic Properties
The calculation of vibrational frequencies (and phonon dispersion) in the harmonic approximation and the mode-gamma Grüneisen parameters in the QHA are the main ingredients also for the computation of thermodynamic properties. If insulating, ordered and stoichiometric crystalline solids (such as MgO) are taken into account, the vibrational contribution is only present in the general partition function [46]. The phonon contribution to the relevant thermodynamic properties, such as Hemholtz free energy (Fvib), internal energy (Uvib), isochoric heat capacity (CV) and entropy (Svib), can thus be determined according to Equation (1) (with νi calculated at different k-points and different volumes) and to the following equations valid for the other thermodynamic functions:
Isobaric heat capacity (CP) and total entropy (S) come out when the anharmonicity contribution is included in Equations (10) and (11), i.e.,
All the other thermodynamic functions (e.g., enthalpy, H; Gibbs free energy, G; etc.) can be derived from the fundamental thermodynamic relations [69].
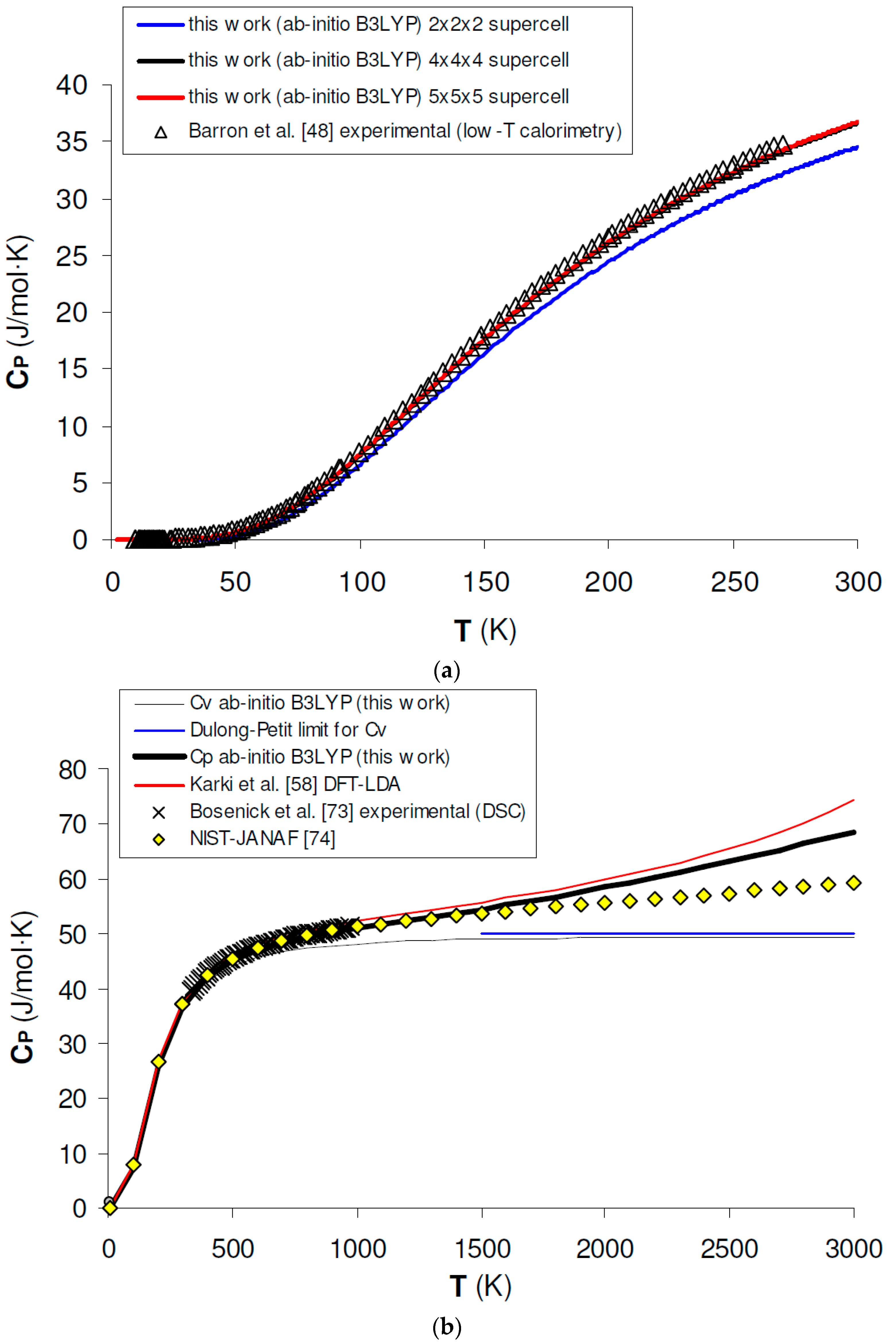
Isobaric heat capacity (CP) is a key thermodynamic property for testing the performance of hybrid DFT in predicting the thermodynamic properties of minerals, because ab initio results can be directly compared with calorimetric measurements in the low-T range and up to intermediate temperature conditions. Figure 5a shows the calculated results for CP of MgO at low T (i.e., T < 300 K) as obtained by a phonon dispersion calculation with supercells of increasing size (i.e., 2 × 2 × 2, 4 × 4 × 4 and 5 × 5 × 5). A few facts are noteworthy, when comparing ab initio B3LYP calculations with low-T calorimetric results [48]. First, a 2 × 2 × 2 supercell calculation gives poor agreement with experimental data; this means that the phonon density of states (PDOS) is not adequately described in this case due to insufficient or overly coarse sampling of the reciprocal space. Second, numerical convergence for CP is fulfilled after a 5 × 5 × 5 supercell calculation (which corresponds to a 125 k-points sampling of the first BZ), since the difference between the calculated heat capacity values becomes negligible beyond this critical size. Finally, 5 × 5 × 5 supercell calculations give an accuracy of ~1% or less in the simulation of the heat capacity and thermodynamic properties in the low-T range. This accuracy is well within the experimental uncertainty of current low-T relaxation calorimetry experiments [70,71,72].
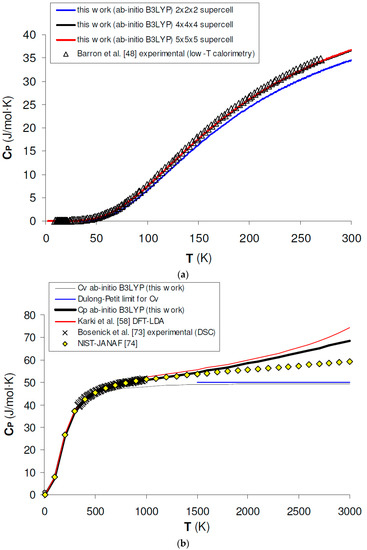
Figure 5.
Ab initio B3LYP heat capacity (CV, CP) of MgO: (a) calculated results by phonon dispersion calculation with 2 × 2 × 2, 4 × 4 × 4 and 5 × 5 × 5 supercells as compared to low-T calorimetry experimental data [48]; (b) calculated results in the whole T range as compared to experimental data by DSC [73], tabulated values by NIST-JANAF [74] and other ab initio LDA calculations [58].
Figure 5b shows as ab initio B3LYP phonon dispersion calculations are able to predict experimental CP values determined by differential scanning calorimetry (DSC) [73] or assessed by thermodynamic tabulations (e.g., the NIST-JANAF, [74]); there is a good agreement up to roughly T ≈ 2000 K, then deviations may occur, essentially due to the QHA overestimation of thermal expansivity (see Section 3.1). This holds also for B3LYP calculations, although the heat capacity overestimation seems to be lesser than that given by other density functionals [58]. The results obtained in the intermediate- to high-temperature range are summarized in Table 3. In this case, the difference between CP values is relevant up to a 4 × 4 × 4 supercell calculation, then becomes negligible because CV tends to reach the Dulong-Petit limit (i.e., 3nR = 49.89 J/mol × K, where n = 2 is the number of atoms in the unit formula of MgO) and the anharmonic term (i.e., TVα2KT) is less sensitive to k-point sampling.

Table 3.
Ab initio B3LYP isobaric heat capacity (CP) of MgO, as obtained in this work by phonon dispersion calculation with 2 × 2 × 2, 4 × 4 × 4 and 5 × 5 × 5 supercells. Tabulated values by NIST-JANAF [74] are also shown for comparison.
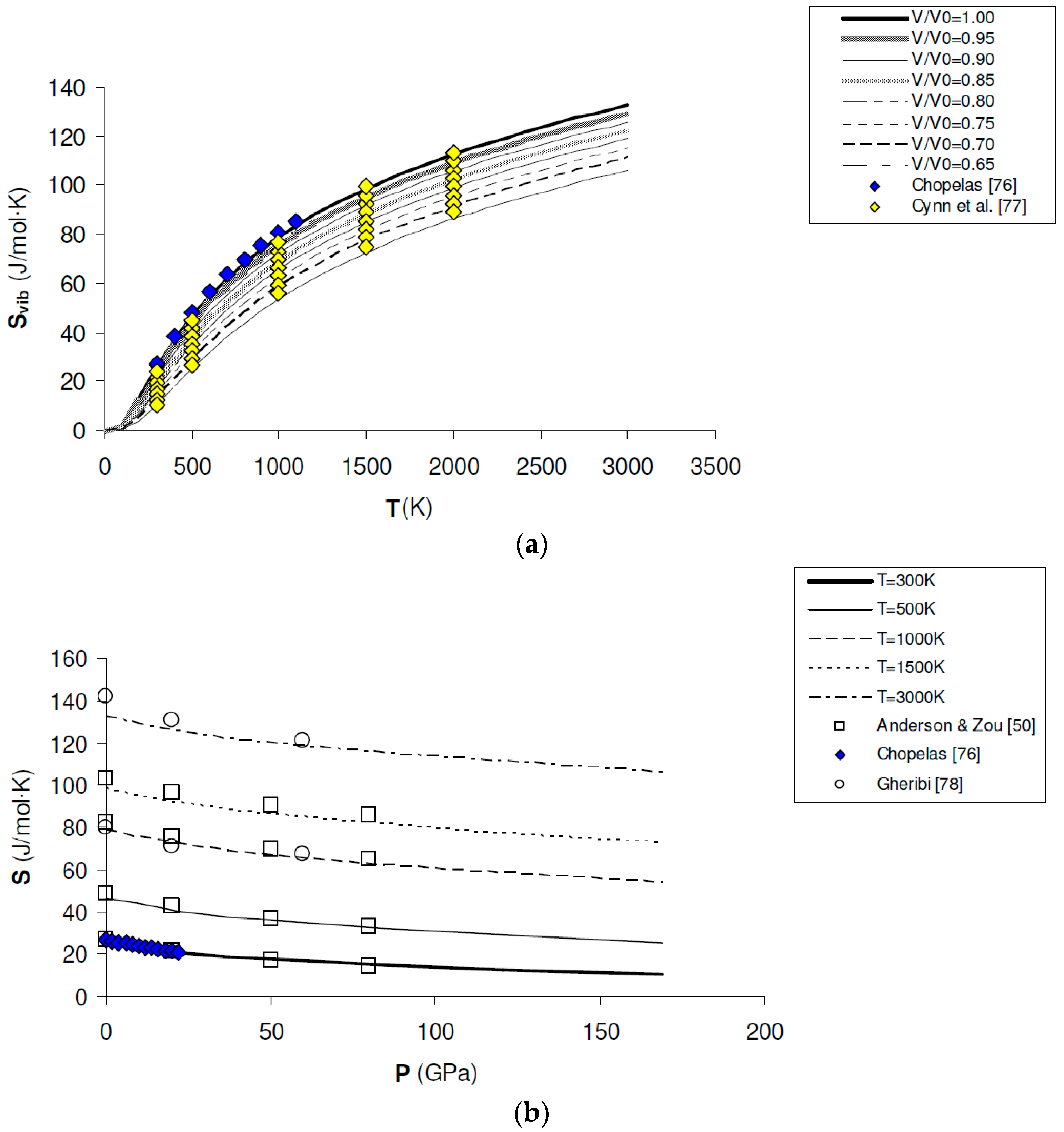
Entropy values (S) can be computed by Equations (11) and (13) and directly compared to those obtained by calorimetric measurements at ambient pressure. The standard-state entropy calculated in this work (i.e., = 26.25 J/mol·K) compares fairly well with the tabulated data (i.e., = 26.924 ± 0.08 J/mol·K and = 26.9 ± 0.2 J/mol·K according to [74,75], respectively). Most interestingly, Svib(V,T) or S(P,T) can be computed in the whole P–T–V range of interest (i.e., up to lower mantle conditions) and compared to entropy values modelled from vibrational spectroscopy experiments [76] and thermodynamic assessments [50,77,78] (see Figure 6a,b). A decrease of entropy with pressure is obtained from first principles, i.e., (∂S/∂P)T < 0 in the whole P–T range investigated in this work (0–160 GPa; 0–3000 K). This result has important implications for the P–V–T equation of state of periclase, as discussed below.
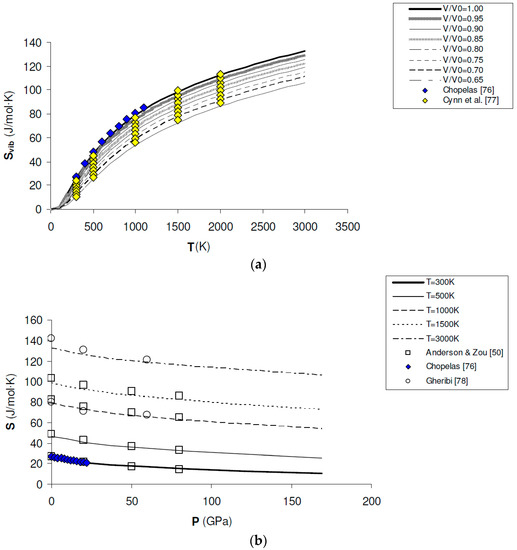
Figure 6.
Ab initio B3LYP vibrational entropy (Svib) and total entropy (S) of MgO: (a) calculated results along isochores (i.e., V/V0 = 0.65, 0.70, 0.75, 0.80, 0.95, 0.90, 0.95 and 1.00), as compared to values modelled from vibrational spectroscopy experiments [76] and thermodynamic assessments [77]; (b) calculated results along isotherms (i.e., T = 300 K, 500 K, 1000 K, 1500 K and 3000 K), as compared to values modelled from vibrational spectroscopy experiments [76] and thermodynamic assessments [50,78].
3.3. P–V–T Equation of State (EOS)
An equation of state (EOS) relates all the physical variables that control the free energy of a thermodynamic system, such as temperature (T), pressure (P) and volume (V). Since the nature of the equation of state is phenomenological—namely, intrinsically phase-dependent—there are plenty of formulations proposed in the literature (e.g., Murnaghan, Birch-Murnaghan, Vinet, natural strain, etc.), which rely on different theoretical assumptions [79,80,81,82]. MgO has always been a key phase for testing the performance of different EOS formalisms due to its simple structure and broad stability field [83,84,85,86]. Isothermal equations of state based on finite strain theory, like the third- or fourth-order Birch-Murnaghan EOS (BM3-EOS and BM4-EOS, respectively) [53], are among the most popular in the Earth Sciences, and have also been applied to high-temperature conditions (HT-BM-EOS) [2].
The Mie-Grüneisen equation of state (MG-EOS) [87] is perhaps the most straightforward way of describing the P–V–T relations of solids from first principles, as it is firmly grounded on the lattice dynamics theory of crystal lattices [88]. The MG-EOS formalism splits pressure into its different contributions (namely static and vibrational, the latter being in turn split into zero-point correction and thermal contributions):
In the statistical mechanics notation [46], Equation (14) can be restated as:
If vibrational frequencies at different volumes and mode-gamma Grüneisen parameters are available from ab initio phonon dispersion calculations), then a first principles Mie–Grüneisen equation of state (FPMG-EOS) will be obtained by plugging Xi(k,V) and γi(k,V) values into Equation (15).
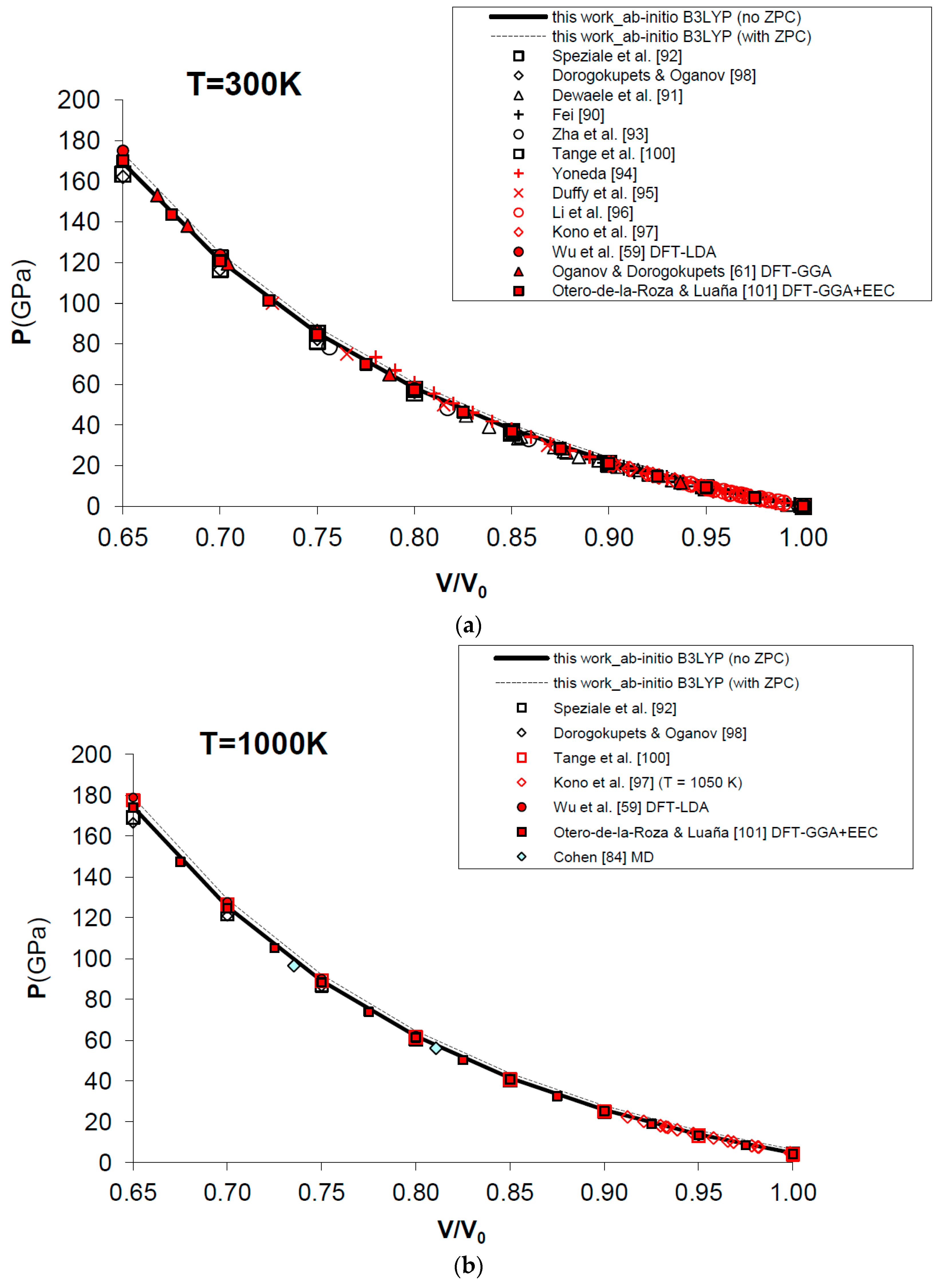
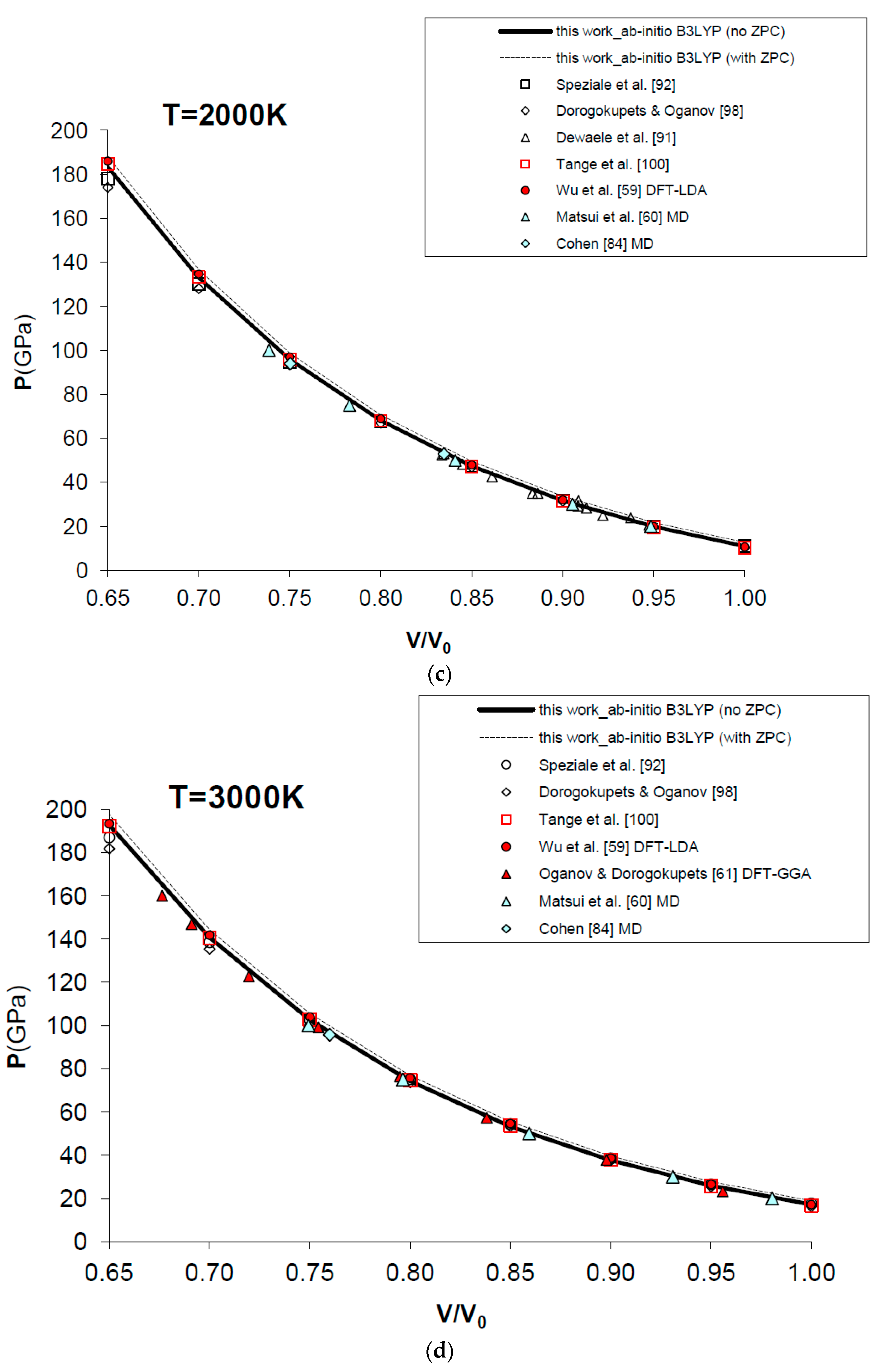
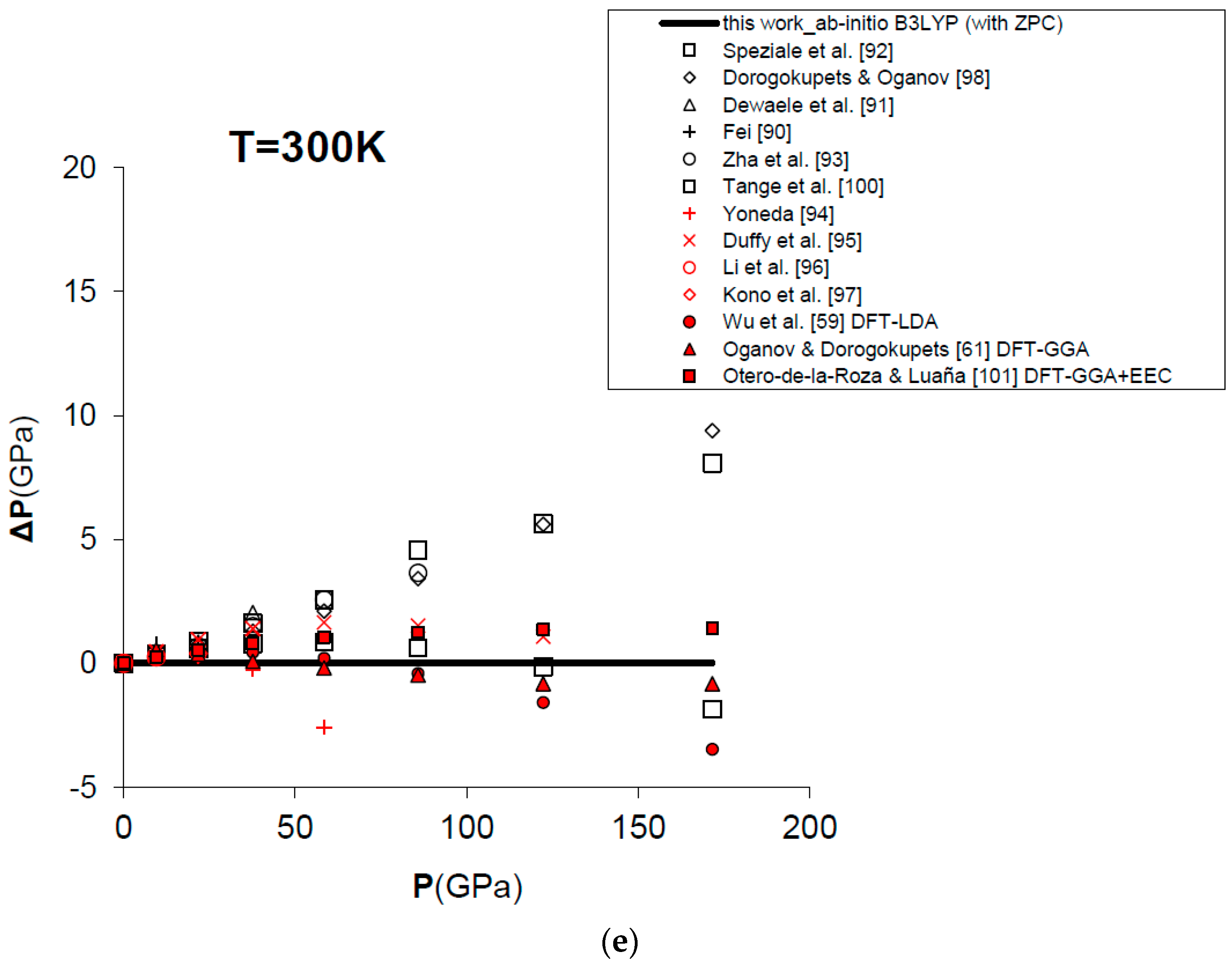
Figure 7 and Table 4 show the results obtained for the P–V–T equation of state of periclase, as compared to different kinds of experimental data (i.e., static compression in the diamond-anvil cell, ultrasonic measurements, etc.) [89,90,91,92,93,94,95,96,97], semi-empirical modelling of experimental data [98,99,100] and other DFT [59,61,101] and MD [60,84] calculations. The EOS of MgO has been calculated according to Equation (15) in a range of conditions compatible with those realized in the Earth’s lower mantle (i.e., P = 0–160 GPa, V/V0 = 0.65–1.00, T = 0–3000 K). Figure 7a displays the calculated results for the EOS at room temperature, and how the P–V–T relations of MgO are perfectly reproduced by means of a first principles Mie-Grüneisen formalism, whether including the zero-point correction term or not (dashed and bold curves in Figure 7a, respectively). It must be noted that ab initio calculations usually make a different choice of the reference state with respect to experimental measurements (i.e., P = 0 GPa, T = 0 K for the former; P = 1 bar, T = 298.15 K for the latter). The effect of the zero-point motions is to slightly raise the P–V–T curve. Ab initio P–V–T data are presented here without any further pressure correction. In other theoretical works, a pressure shift is applied to the calculated values to account for systematic errors of the density functional on volumes. Oganov and Dorogokupets [61], for instance, apply a pressure shift of about −7.7 GPa to the calculated static pressures to account for the systematic overestimation of lattice parameters by GGA (~1–2%) and to match the experimental unit cell volume in ambient conditions. Wu et al. [59] apply a more limited pressure correction to account for the systematic underestimation of volume by LDA (~0.6%) and to reproduce better the experimental results at LP conditions. Those corrections would be reasonable if the fundamental goal was the use of the first principles thermal EOS as a pressure scale (as in the case of MgO). However, even though B3LYP shares with other GGA functionals the same problem of volume overestimation (~2–3%, as already mentioned in Section 1), the aim of the present work is quite different, being the achievement of a first principles physically-consistent P–V–T dataset for the calculation of ΔG values of mineral reactions and the prediction of univariant phase equilibria at HP–HT conditions (see below). This is why theoretical results as directly provided by Equation (15) are considered throughout this work.
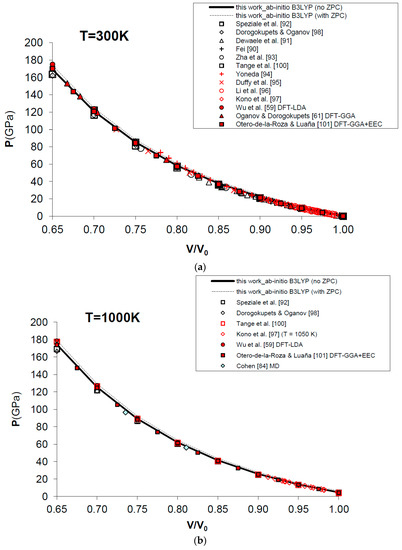
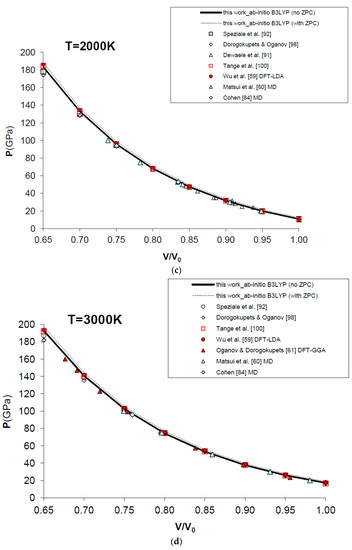
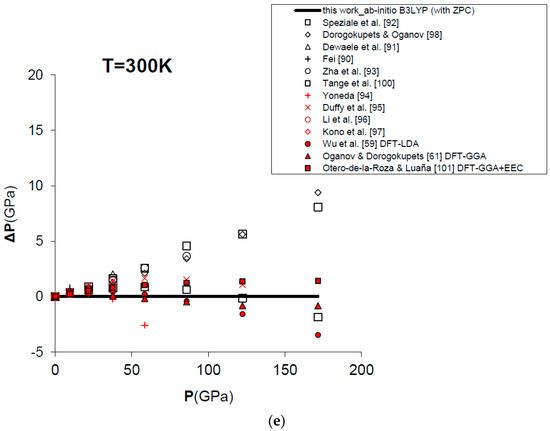
Figure 7.
Ab initio B3LYP P–V–T equation of state (FPMG-EOS) of MgO, calculated with and without zero-point correction (ZPC) (dashed and bold curves, respectively; see Equations (14) and (15): (a) at T = 300 K; (b) at T = 1000 K; (c) at T = 2000 K; (d) at T = 3000 K; (e) deviation plot at T = 300 K. Calculated results of this work are compared to selected experimental data obtained by hydrostatic or quasi-hydrostatic compression in the diamond-anvil cell [90,91,92,93] and ultrasonic measurements [94,95,96,97]. The results of semi-empirical assessments [98,100], other ab initio LDA [59], GGA [61] and empirically-corrected GGA [101] calculations and MD simulations [60,84] are also shown for comparison.

Table 4.
Ab initio B3LYP P–V–T EOS of MgO. Reference volume is V0 = 75.762 Å3 (T = 0 K, P = 0 GPa).
Raising the temperature doesn’t affect the accuracy of the simulation, which is still high and compares favorably with MD results [60,84] at selected high temperatures (i.e., T = 1000 K, 2000 K and 3000 K) and up to the melting point of the substance (see Figure 7b–d).
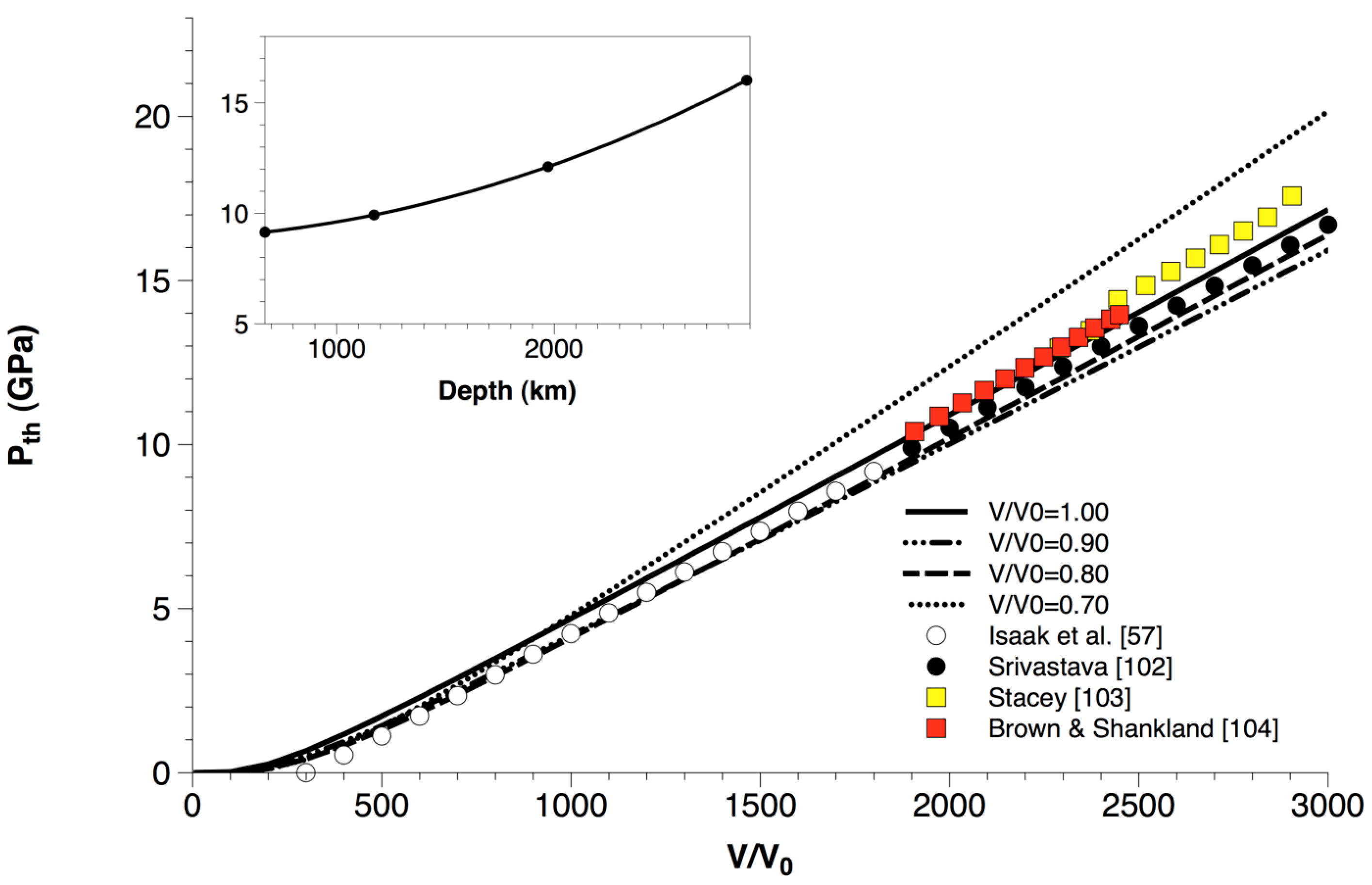
Thermal pressures (Pth in Equation (14)) calculated for MgO up to high volume compression (i.e., V/V0 = 0.70) are compared with ambient-pressure experimental data [57] and a thermodynamic assessment [102] in Figure 8. Furthermore, the thermal pressure values estimated for the lower mantle by different geophysical models [103,104] are superimposed to the ab initio values of MgO. Some relevant considerations can be inferred from the analysis of Figure 8:
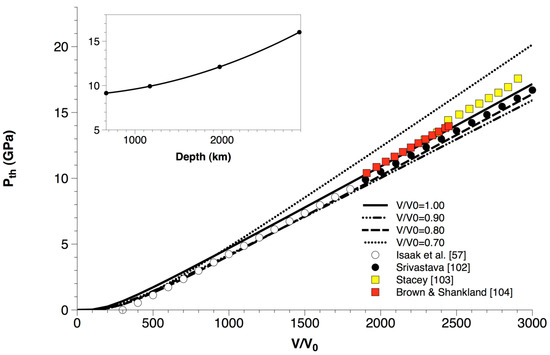
Figure 8.
Ab initio B3LYP thermal pressure (Pth) calculated for MgO along different isochores and up to high volume compression (i.e., V/V0 = 0.70), as compared to ambient-pressure experimental data [57] and a thermodynamic assessment [102]. The thermal pressure values estimated for the lower mantle by different geophysical models [103,104] are superimposed for comparison. The inset shows thermal pressure values calculated for MgO along a typical lower mantle geotherm taken from [104].
- (i)
- ab initio quasi-harmonic thermal pressure, in contrast to thermal expansivity, doesn’t display any deviation at P = 0 (i.e., V/V0 = 1.00) and high-temperature conditions (i.e., T > ΘD ≅ 945 K) (see also Section 3.1). This means that Pth is rather insensitive to QHA deviations at LP-HT conditions, at least for what concerns the accuracy of numerical predictions, as already noted in other theoretical works [61,105,106]. Thermal pressure is quite insensitive to compression, too, especially in the low- to intermediate-T range;
- (ii)
- ab initio quasi-harmonic calculations of thermal pressure reproduce quite well the values assessed from experiments at 1-bar pressure [57,102]. The slight differences with respect to the values of Isaak et al. [57] and Srivastava [102] at low temperature are due to the constraint Pth (300 K, 1 bar) = 0, assumed to be valid in those works; this assumption lacks a theoretical justification, because thermal vibrations obviously occur already at T = 0 K (zero-point motions), and their effects cannot be disregarded;
- (iii)
- Although the lower mantle consists primarily of bridgmanite, the thermal pressure of MgO seems to be representative of that of the lower mantle [103,104] owing to the peculiar thermoelastic and transport properties of periclase. Lower mantle thermodynamics and rheology are thus largely controlled by periclase and its Fe-bearing analogue (ferropericlase) [16,107,108,109,110].
The most relevant thermophysical and thermodynamic functions calculated for MgO at P = 1 bar in the range T = 298.15–3000 K are listed in Appendix A (Table A1).
3.4. Phase Equilibrium Calculation at HP–HT: Modelling High-Pressure Effects on Gibbs Free Energy
Thermodynamic equilibrium of a reversible heterogeneous chemical reaction between fixed-composition (i.e., pure) phases at constant T, P is defined by the principle of Gibbs free energy minimization, i.e.,
where ΔGxt, ΔHrxt, ΔSrxt and ΔVrxt are the Gibbs free energy, enthalpy, entropy and volume changes of the reaction, respectively; Pr = 1 bar is the standard-state reference pressure.
A straightforward way to define the phase equilibrium is to compute the Gibbs free energy of all the phases involved in the reaction at T, P in the so-called “explicit form” and then minimize ΔG of reaction at each T–P conditions according to the following equations:
where Tr = 298.15 K is the reference temperature; (Tr,Pr) is the standard-state enthalpy of the phase, which at Tr,Pr is equal to the enthalpy of formation from the elements at standard state (i.e., = at Tr,Pr); (Tr,Pr) is the standard-state entropy; and xi are the stoichiometric coefficients of the reactants and products in the reaction. In Equations (16)–(18) all thermodynamic entities refer to 1 mole of substance.
An alternative strategy for computing the Gibbs free energy of solid phases involved in the reaction is to define their Helmholtz free energy F(V,T) and then the P-V contribution separately by operating a Legendre transformation (i.e., G = F + PV) [69]. The former can be derived directly from phonon dispersion calculations or, alternatively, by integrating the PDOS at the V,T conditions of interest (see Section 3.1 and Section 3.2); the latter can be computed from the P–V–T curves which define the Mie-Grüneisen EOS of each phase (see Section 3.3). This procedure is formally correct and physically-consistent in the whole P–T–V range (see below), provided that a proper analytical description of the PDOS for all the phases involved in the equilibrium reaction is available from first principles. Phonon dispersion calculations, such as those presented in this work, are very labor intensive, so they might not be affordable for all the relevant mineralogical phases in thermodynamic databases. This is why Gibbs free energy minimization based on Equations (16)–(18) is usually performed at HP–HT [2].
The volume-pressure contribution to the Gibbs free energy is the key entity in phase equilibrium calculations at HP–HT [2,111]. Since pressure is experimentally easier than volume to keep constant in the solid state, the VdP integral in Equation (17) is commonly expressed as a PdV integral and calculated adopting an EOS formalism which relates the P–V–T variables. There are different analytical or numerical procedures to do this (see for instance [112] for details), but the most common way is to use a HT-BM3-EOS [2]. The great advantage in the use of finite strain EOSs to quantify pressure effects on the Gibbs free energy is that they require just ambient-pressure temperature-dependent thermophysical parameters (i.e., volume thermal expansion and isothermal compressibility) to calculate the V(P,T) function. Nevertheless, it has been shown that HT-BM3-EOS is prone to giving spurious effects or anomalous predictions (i.e., negative thermal expansion) at very HP–HT conditions [63,113]. This has led different authors to develop alternative formulations (more or less empirical) to describe the P–V–T relations of minerals up to planetary interior conditions [4,63,78,113,114,115,116,117,118,119]. Even though negative thermal expansion is obviously possible from a physical point of view as different minerals and compounds display a volume decrease with T not just in the low-P range [25,120], this phenomenon is systematically predicted by the HT-BM3-EOS at very HP–HT conditions. In particular, this usually happens beyond a threshold pressure which depends upon the thermophysical properties of the phase (and also on how these properties are defined), but it’s typically around 25–30 GPa [63,113].
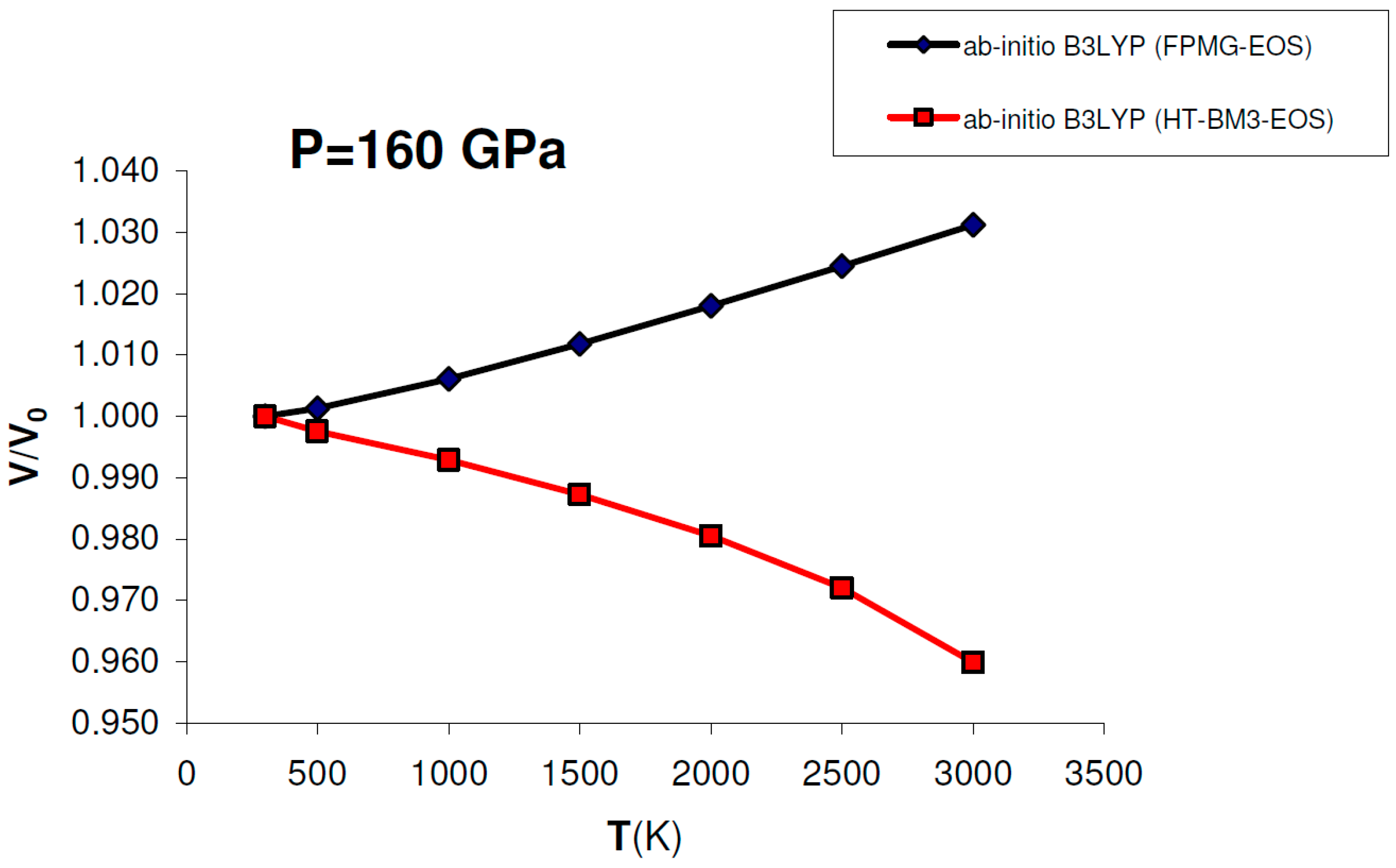
Figure 9 shows that using ab initio B3LYP thermophysical parameters (i.e., V0, K0, K′0, α, dK/dT) into a HT-BM3-EOS gives negative volume thermal expansion for MgO at P = 160 GPa (i.e., at conditions compatible with the base of the lower mantle), while the V(P,T) function obtained by a FPMG-EOS (cf. Equation (15)) always has a positive slope over the whole P–T range, as expected for minerals with a close-packed B1-type structure. If the volume thermal expansion is negative, then entropy will increase with pressure by virtue of the Maxwell relation (∂S/∂P)T = –(∂V/∂T)P. This anomalous behavior, which for MgO occurs around 60 GPa, does not occur if lattice vibrations are properly taken into account in the P–V–T EOS, as done by the FPMG-EOS (see Figure 6b and Figure 9). Therefore a HT-BM3-EOS (and other isothermal finite strain EOSs) should be used with care as P–V–T EOS at very HP–HT conditions, if the thermal or “hot” part of the EOS is not properly integrated with its compression or “cold” part.
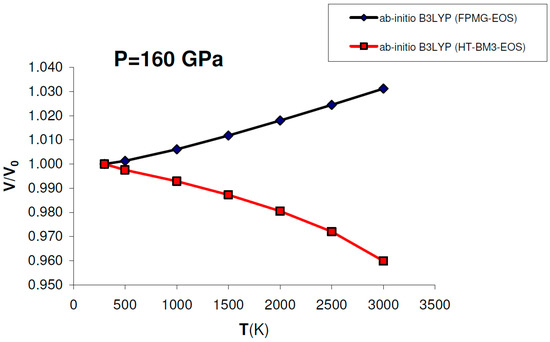
Figure 9.
Ab initio B3LYP relative volume thermal expansion (V/V0) of MgO calculated at P = 160 GPa by: (a) FPMG-EOS (blue) or (b) HT-BM3-EOS (red). While the former gives a V/V0 = f(T) function with a positive slope (i.e., positive thermal expansion) at P = 160 GPa, the latter gives an anomalous negative thermal expansion in the whole T-range at the same pressure (see text for discussion).
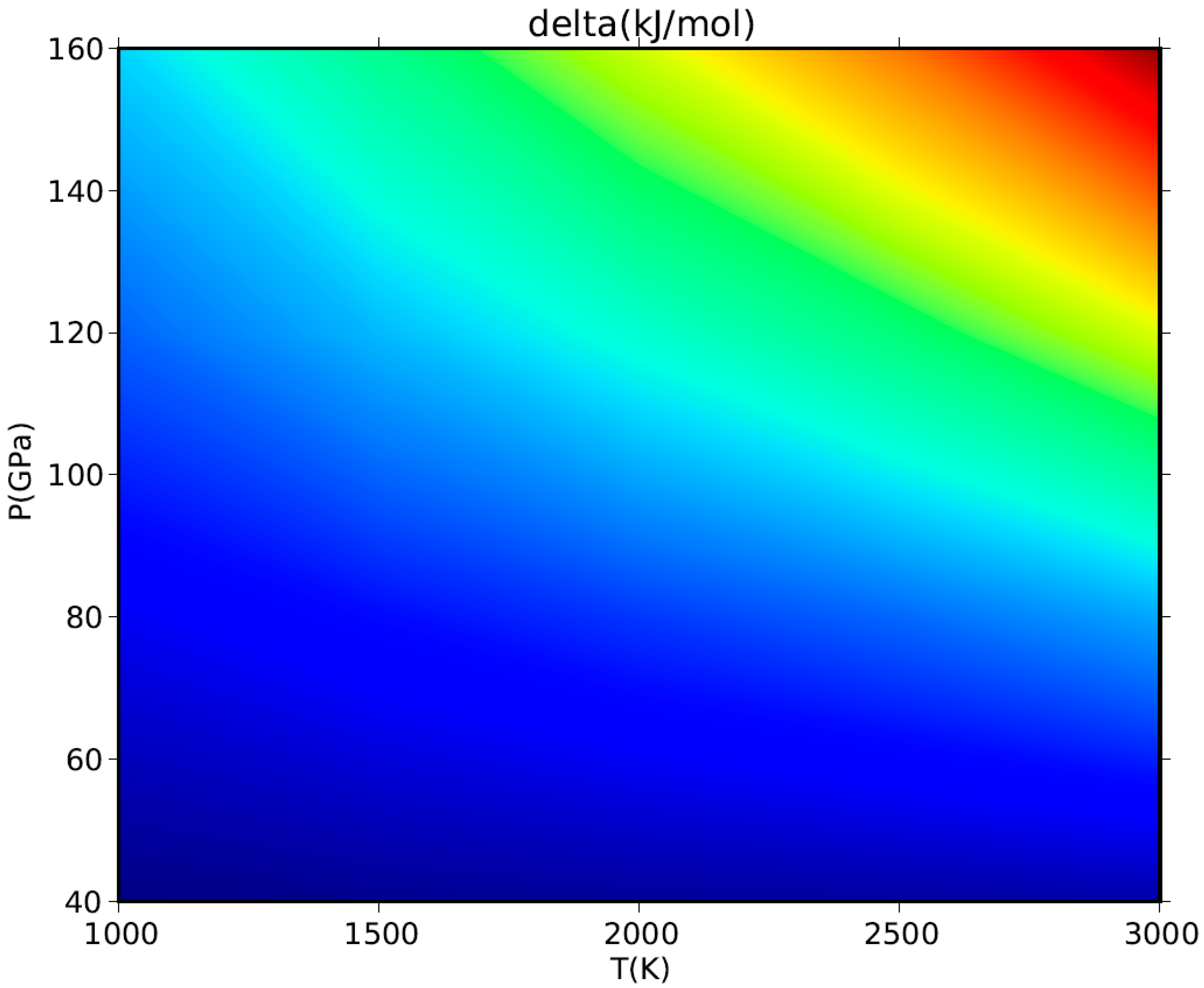
Although a HT-BM3-EOS could give spurious predictions at very HP–HT conditions, it will not eventually affect the phase equilibrium calculation if the thermophysical properties of mineral phases involved in the equilibrium reaction are quite similar and differences among them thus cancel out [113]. A simple numerical exercise shows to what extent the adoption of a HT-BM3-EOS or a FPMG-EOS would affect the calculated values of Gibbs free energy of periclase at HP–HT conditions. The delta values calculated for the volume-pressure integral of MgO in Equation (17) by using the two equations of state are plotted against P and T in Figure 10. The difference turns out to be small (less than 1 kJ/mol) at LP-LT conditions but could sensibly increase (up to few tens of kJ/mol) at HP–HT, namely in the range of conditions where the HT-BM3-EOS could give numerical anomalies in the V(P,T) values. Although there is nothing wrong with fitting P–V–T data with a finite strain EOS like the HT-BM3-EOS to extract thermophysical data such as KT, K′T, etc. (this is a common practice in both experimental and computational works, as already stressed in Section 3.1), it should be noted again that some care is required to avoid physical unsoundness, especially when those data are used to address the problem of phase stability at extreme conditions of P and T.
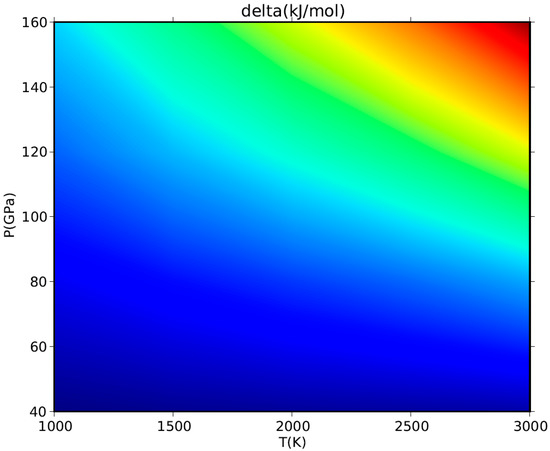
Figure 10.
Effect of P–V–T EOS on the volume-pressure integral of MgO. Delta values of Gibbs free energy (in kJ/mol) calculated by using a HT-BM3-EOS or, alternatively, a FPMG-EOS for the volume-pressure integral (see Equation (17)) are plotted between P = 40–160 GPa and T = 1000–3000 K. Color scale as follows: blue = 6–30 kJ/mol; green = 30–45 kJ/mol, yellow = 45–50 kJ/mol, orange = 50–60 kJ/mol, red > 60 kJ/mol.
3.5. Phase Equilibrium Calculation at HP–HT: The Post-Spinel Phase Transformation
It is widely accepted that the disproportionation reaction of olivine with spinel-type structure (ringwoodite) into an assemblage of bridgmanite and ferropericlase (the so called “post-spinel phase transformation”) is an appropriate candidate to interpret the 660-km depth global seismic discontinuity in the Earth’s mantle [13,121,122]. For the magnesium end-member phases this reaction can be written as:
γ-Mg2SiO4 (ringwoodite) = MgO (periclase) + MgSiO3 (bridgmanite)
The post-spinel phase change turns out to be consistent with the 660-km mantle seismic discontinuity essentially because the stability field and seismic properties of minerals involved in the equilibrium reaction process are compatible with depth, density contrast and seismic velocity jumps associated with the discontinuity, as derived by seismological models (like the Preliminary Reference Earth Model, PREM) [123]. Owing to its geophysical implications, the Mg2SiO4 post-spinel phase transformations have been investigated by several experimental studies in the last decades [124,125,126,127,128,129,130,131,132,133,134,135]. Although experimental results are not consistent with each other concerning the P–T localization of the phase equilibrium boundary, there is general agreement about its most important thermodynamic feature; that is, the negative Clapeyron slope (i.e., dP/dT < 0) or endothermic character (i.e., ∆Hrxt > 0). By virtue of the negative Clapeyron slope, this reaction may cause elevation and depression of the 660-km discontinuity within subducted slabs and the surrounding mantle due to lateral temperature variations, thus working as a potential barrier to the penetration of slabs beneath the mantle transition zone, and favoring stratified convection in the mantle [13].
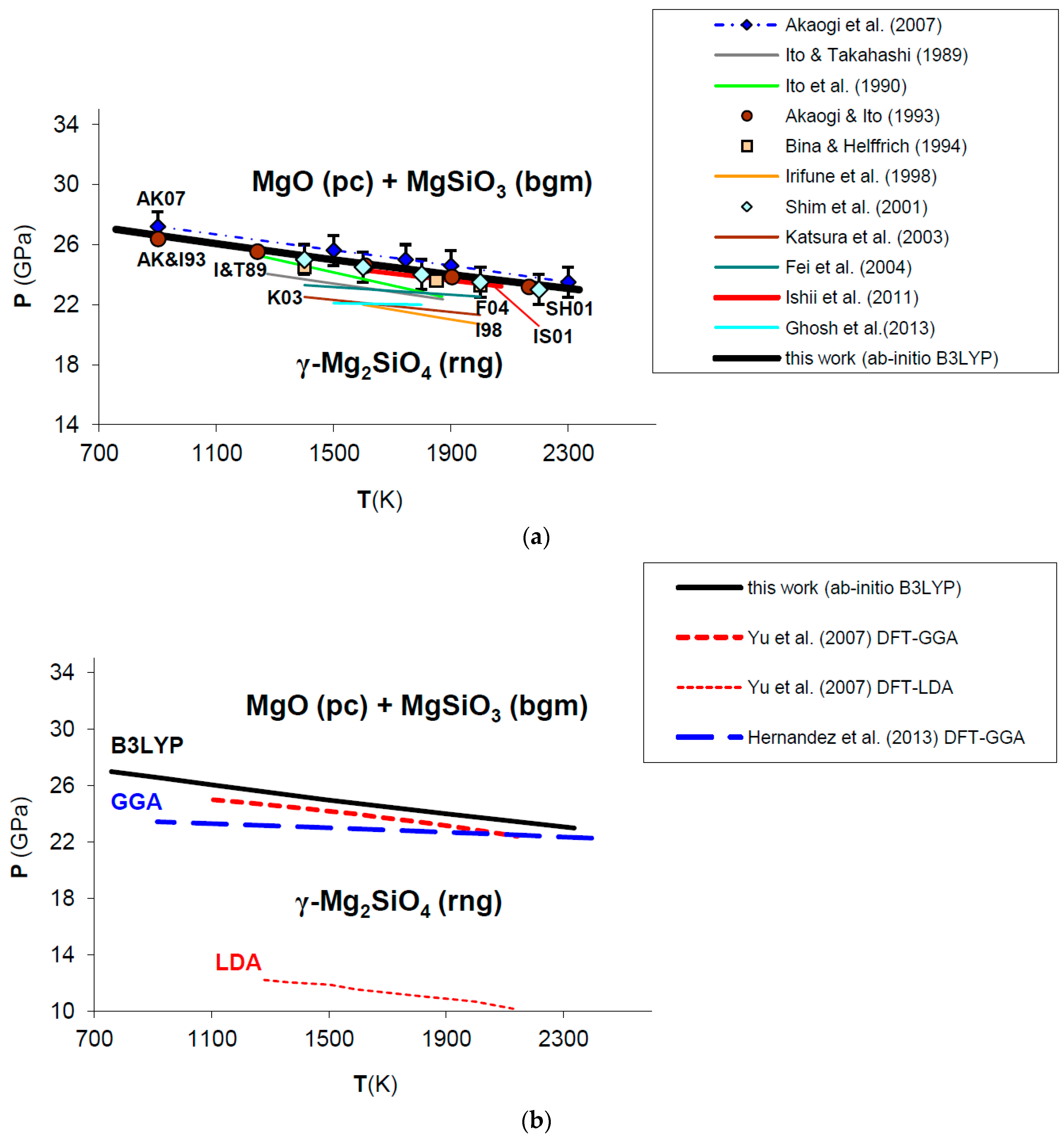
The phase equilibrium boundary of post-spinel phase transformation calculated in this work from first principles is shown in Figure 11. Ab initio B3LYP thermodynamic data for Mg2SiO4 ringwoodite and MgSiO3 bridgmanite have been taken from Ottonello et al. [36] and Belousov [136], respectively. All the details about the calculation, along with a discussion on the possible effects of equation of state on the phase boundary, are given in Appendix B.
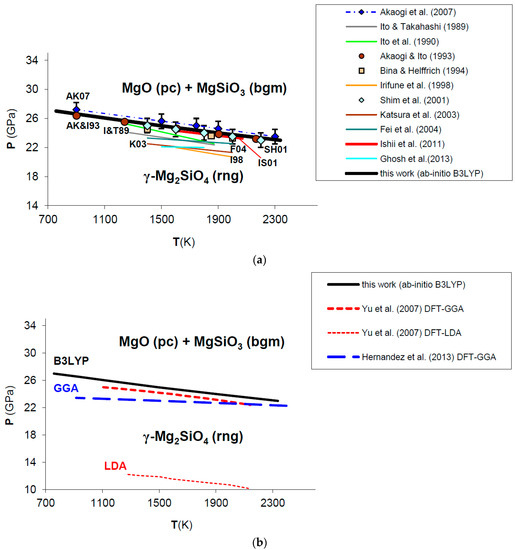
Figure 11.
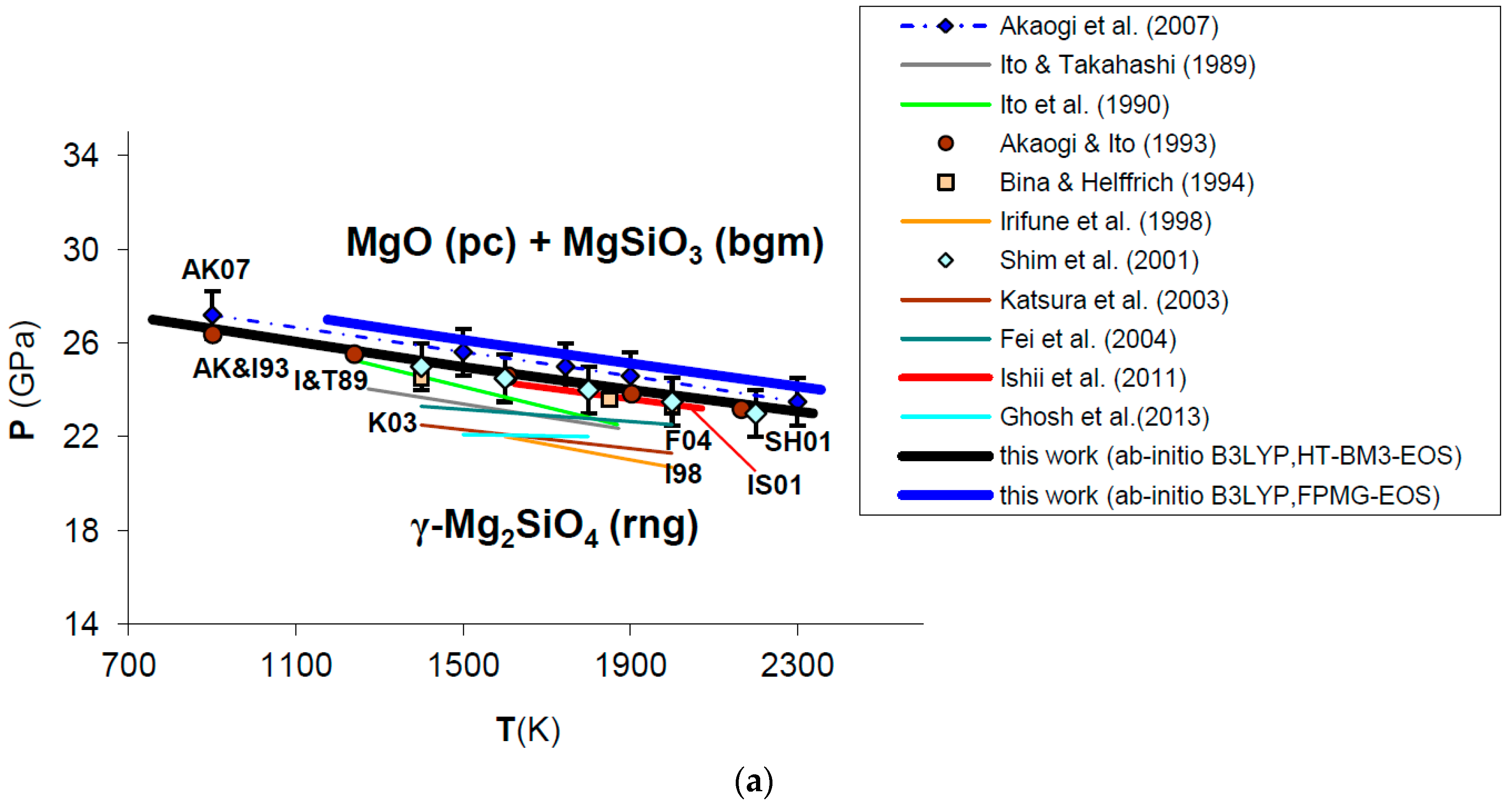
Ab initio B3LYP phase boundary of post-spinel transformation (in bold) as compared to: (a) experimental results (multi-anvil press, laser-heated diamond-anvil cell) [124,127,128,129,130,132,133] and calculations based on calorimetric measurements [121,125,126,131]; (b) other ab initio LDA [137] and GGA [138] calculations. Abbreviations for minerals are: rng = ringwoodite (γ-Mg2SiO4); pc = periclase (MgO); bgm = bridgmanite (MgSiO3).
Ab initio B3LYP results show an excellent agreement with the in situ LH-DAC X-ray diffraction study of Shim et al. [128] and the multi-anvil experiments of Ishii et al. [132], as well as with calculations based on accurate calorimetric measurements by Akaogi and Ito [126] and Akaogi et al. [131] (Figure 11a; see also Figure A2 in Appendix B). This means that calculated values for Δ and Δ of the post-spinel reaction are consistent with those obtained by calorimetry (see Table 5), although a recent study found a lower enthalpy of reaction [134]. The ab initio phase boundary is localized at slightly higher pressures (≈1–2 GPa) than those determined by other HP–HT experiments [124,127,129,130,133] (see Figure 11a). The calculated Clapeyron slope (i.e., dP/dT = −2.53 MPa/K) agrees well with those obtained by the most part of experimental investigations (ranging from −4.0 to −2.0 MPa/K) [121,124,125,126,127,131,132,135], while other works [129,130,133,134] suggest a more gentle slope of the phase boundary (i.e., between −2.0 and −1.0 MPa/K or even less) (see Table 5). B3LYP results are in qualitative agreement with previous GGA calculations [137,138,139], whereas LDA [137] severely underestimates transformation pressures of the post-spinel reaction (see Figure 11b). However, ab initio Clapeyron slopes obtained by this and other DFT studies [137,138,139] seem to be consistent with each other, and less sensitive to the exchange-correlation term. The phase equilibrium boundary calculated in this work is also in good agreement with thermodynamic assessments based on lattice dynamical models [4,10,140]. The effect of lattice vibrations seems to be crucial to correctly predicting the high-pressure phase transformation; ab initio static enthalpy calculations [137,138,139,141] give transition pressures that are overestimated (by ≈1−2 up to 10 GPa) compared with those obtained by including vibrational contributions to thermodynamic properties (i.e., by minimizing Gibbs free energies instead of static enthalpies).

Table 5.
Enthalpy (Δ), entropy (Δ) and volume change (Δ) at T = 298.15 K, P = 1 bar and Clapeyron slope (dP/dT) of the post-spinel phase transformation (Equation (19)). Values obtained in this work are compared to experimental results [121,124,125,126,127,128,129,130,131,132,133,134,135] and other ab initio calculations [137,138,139].
The density change of the post-spinel phase transformation provides useful insights on the interpretation of the 660-km seismic discontinuity in the Earth’s mantle. By assuming this discontinuity is produced primarily by the complete dissociation of Mg2SiO4 and the disproportionation reaction does not affect the stability of other mineralogical phases present in the Earth’s mantle at 660-km depths (such as majorite garnet and Ca-perovskite), the density change (i.e., Δρ/ρ) of the post-spinel transformation (cf. Equation (19)) can be estimated as [137]:
where ρrng and ρpc+bgm are the density of Mg2SiO4 ringwoodite (rng) and the average density of an isotropic aggregate of MgO periclase (pc) plus MgSiO3 bridgmanite (bgm) at T,P of interest, respectively. Table 6 shows the results of the density change calculation at P–T conditions given by the ab initio B3LYP phase boundary of Figure 11 and compatible with those of the Earth’s mantle at 660-km depths [103,104,123], namely P = 23–24 GPa and T = 1900–2337 K. The calculated density jump across the 660-km discontinuity (i.e., 7.1–7.2%) turns out to be sensibly lower and higher, respectively, than those inferred by PREM [123] and the global seismic impedance study of Shearer and Flanagan [142]. This discrepancy can be interpreted in two alternative ways. If the 660-km seismic discontinuity were just due to the post-spinel phase transformation, the observed density jump would be reproduced only by assuming an incomplete dissociation reaction of ringwoodite in a mantle mineral aggregate; in fact, this would lead to an attenuation of the density change [137]. Alternatively, multiple reactions compatible with P–T conditions realized at the base of mantle transition zone (such as majorite garnet to bridgmanite phase transition, besides the post-spinel transformation) could occur simultaneously and could jointly contribute to determine the 660-km seismic discontinuity. This second scenario is nowadays supported by several evidences coming from seismology, geochemistry and mineral physics [122,143,144,145,146,147,148].

Table 6.
Calculated density change (Δρ/ρ; see Equation (20)) of the post-spinel phase transformation across the 660-km mantle seismic discontinuity, as compared to geophysical models [123,142]. The results of another DFT-GGA simulation [137] are also shown for comparison. Abbreviations for minerals are: rng = ringwoodite (γ-Mg2SiO4); pc = periclase (MgO); bgm = bridgmanite (MgSiO3).
4. Conclusions
The main conclusions of this work can be summarized as follows:
- At ambient pressure, ab initio thermophysical properties of MgO (such as α and KT) show a good agreement with experimental data up to relatively high temperatures (i.e., T ≈ 1800–2000 K), then deviations occur due to losing the power of the quasi-harmonic approximation at high-temperature (and low-pressure) conditions. Nevertheless, B3LYP results seem to show a less marked deviation in thermal expansivity with respect to other density functionals (e.g., LDA) under those conditions. Thermodynamic properties at low and up to relatively high temperatures are accurately defined by ab initio B3LYP phonon dispersion calculations. The level of accuracy with regard to current calorimetric results is around the experimental uncertainty (i.e., 1–2%) throughout the whole range of experimental measurement (i.e., T = 298.15–1000 K).
- A first principles Mie-Grüneisen EOS formalism (splitting pressure into static, zero-point and thermal contributions) gives reliable and physically-consistent P–V–T relations for MgO up to lower mantle conditions, provided that an accurate description of lattice vibrations and phonon density of state is available. Quasi-harmonic thermal pressures turn out to be rather insensitive to both volume compression and LP-HT deviations.
- P–V–T equations of state based on finite strain theory (such as HT-BM3-EOS) systematically predict anomalous thermodynamic functions (like negative thermal expansion or entropy increase with pressure) at HP–HT conditions. Nevertheless, spurious effects on the V(P,T) function become relevant for MgO at pressures higher than 60 GPa, but in most cases don’t have a huge impact on phase equilibrium calculations at deep mantle conditions as the ensuing Gibbs free energy differences tend to cancel out.
- Hybrid DFT and statistical mechanics calculations in the quasi-harmonic approximation accurately simulate phase reaction boundaries, such as the post-spinel phase transformation of ringwoodite (Mg2SiO4) to periclase (MgO) and bridgmanite (MgSiO3), at HP–HT conditions. Anharmonic effects, if any, are virtually irrelevant under those conditions. The hybrid B3LYP functional predicts accurate thermodynamic properties and, at the same time, reliable phase stability relations for magnesium oxide (and silicates) at HP–HT.
- The Clapeyron slope of post-spinel phase transformation is correctly predicted by ab initio B3LYP calculations, both in sign and magnitude. The calculated value in this work (i.e., dP/dT = −2.53 MPa/K) is in good agreement with the majority of experimental investigations, especially with those based on calorimetric measurements. Although the Clapeyron slope of the phase boundary is almost insensitive to the exchange-correlation term, its P–T location strongly depends upon the choice of the density functional: B3LYP results are in qualitative agreement with previous GGA calculations, while LDA drastically underestimates transformation pressures.
- The calculated density change across the post-spinel phase boundary at P–T conditions compatible with the 660-km seismic discontinuity in the Earth’s mantle is different from that inferred by geophysical models. This suggests either an incomplete dissociation reaction of ringwoodite to periclase + bridgmanite or the occurrence of multiple mineral phase changes in mantle aggregates at those depths.
Acknowledgments
This research was partially funded by the University of Genova (Fondo di Ricerca di Ateneo, Grant No. 100022-2015-FRA_Belmonte). The constructive remarks and comments of three anonymous reviewers are greatly appreciated. The author is grateful to Giulio Ottonello and Mauro Prencipe for critical reading of the manuscript and insightful discussions.
Conflicts of Interest
The author declares no conflict of interest.
Appendix A
Detailed tabulations of the most relevant thermophysical and thermodynamic properties calculated in this work for MgO (periclase, B1-type structure) are presented in Table A1 and Table A2. The listed values refer to P = 1 bar and T = 298.15–3000 K.

Table A1.
Ab initio B3LYP thermophysical and thermodynamic functions of MgO at P = 1 bar, T = 298.15–3000 K. Symbols: KT = isothermal bulk modulus; α = thermal expansion coefficient (thermal expansivity); VT = molar volume; CV, CP = isochoric and isobaric heat capacity; S = entropy; γth = Grüneisen thermodynamic parameter.
Table A1.
Ab initio B3LYP thermophysical and thermodynamic functions of MgO at P = 1 bar, T = 298.15–3000 K. Symbols: KT = isothermal bulk modulus; α = thermal expansion coefficient (thermal expansivity); VT = molar volume; CV, CP = isochoric and isobaric heat capacity; S = entropy; γth = Grüneisen thermodynamic parameter.
| T (K) | KT (GPa) | α × 105 (K−1) | VT 1 (cm3/mol) | CV (J/mol·K) | CP (J/mol·K) | S (J/mol·K) | γth 2 |
|---|---|---|---|---|---|---|---|
| 0 | 161.5 | 0.00 | 11.406 | 0.000 | 0.000 | 0.000 | - |
| 100 | 161.4 | 0.61 | 11.412 | 7.532 | 7.539 | 2.290 | 1.496 |
| 298.15 | 157.7 | 2.97 | 11.444 | 36.142 | 36.616 | 26.251 | 1.483 |
| 300 | 157.7 | 2.98 | 11.444 | 36.272 | 36.753 | 26.478 | 1.483 |
| 400 | 155.2 | 3.42 | 11.481 | 41.339 | 42.174 | 37.870 | 1.475 |
| 500 | 152.5 | 3.70 | 11.522 | 44.069 | 45.269 | 47.640 | 1.474 |
| 600 | 149.8 | 3.89 | 11.566 | 45.674 | 47.246 | 56.080 | 1.475 |
| 700 | 147.0 | 4.04 | 11.613 | 46.687 | 48.641 | 63.474 | 1.478 |
| 800 | 144.2 | 4.18 | 11.660 | 47.364 | 49.711 | 70.042 | 1.483 |
| 900 | 141.3 | 4.30 | 11.710 | 47.837 | 50.592 | 75.947 | 1.488 |
| 1000 | 138.4 | 4.42 | 11.760 | 48.180 | 51.361 | 81.314 | 1.493 |
| 1100 | 135.5 | 4.54 | 11.812 | 48.437 | 52.061 | 86.236 | 1.499 |
| 1200 | 132.5 | 4.66 | 11.866 | 48.633 | 52.724 | 90.787 | 1.505 |
| 1300 | 129.5 | 4.78 | 11.922 | 48.787 | 53.370 | 95.026 | 1.512 |
| 1400 | 126.5 | 4.90 | 11.979 | 48.910 | 54.010 | 98.999 | 1.519 |
| 1500 | 123.4 | 5.03 | 12.039 | 49.009 | 54.657 | 102.744 | 1.526 |
| 1600 | 120.4 | 5.17 | 12.100 | 49.090 | 55.320 | 106.293 | 1.534 |
| 1700 | 117.3 | 5.31 | 12.164 | 49.158 | 56.005 | 109.672 | 1.542 |
| 1800 | 114.1 | 5.47 | 12.231 | 49.215 | 56.719 | 112.902 | 1.550 |
| 1900 | 111.0 | 5.63 | 12.300 | 49.263 | 57.471 | 116.003 | 1.559 |
| 2000 | 107.8 | 5.80 | 12.372 | 49.304 | 58.265 | 118.988 | 1.568 |
| 2100 | 104.6 | 5.98 | 12.446 | 49.340 | 59.109 | 121.872 | 1.577 |
| 2200 | 101.3 | 6.17 | 12.524 | 49.371 | 60.011 | 124.665 | 1.587 |
| 2300 | 98.1 | 6.38 | 12.605 | 49.398 | 60.978 | 127.376 | 1.597 |
| 2400 | 94.8 | 6.61 | 12.688 | 49.421 | 62.020 | 130.013 | 1.607 |
| 2500 | 91.4 | 6.85 | 12.775 | 49.442 | 63.147 | 132.583 | 1.618 |
| 2600 | 88.1 | 7.12 | 12.865 | 49.461 | 64.374 | 135.093 | 1.630 |
| 2700 | 84.7 | 7.40 | 12.959 | 49.477 | 65.712 | 137.546 | 1.642 |
| 2800 | 81.2 | 7.72 | 13.056 | 49.492 | 67.180 | 139.947 | 1.654 |
| 2900 | 77.8 | 8.06 | 13.157 | 49.505 | 68.800 | 142.300 | 1.667 |
| 3000 | 74.3 | 8.45 | 13.262 | 49.517 | 70.594 | 144.608 | 1.680 |
1 Calculated as VT = V0exp∫α(T)dT by fitting ab initio values of α with a polynomial function in T (i.e., α = α0T + α1 + α2T−1 + α3T−2 + α4T−3) (see also Appendix B).2 Grüneisen parameter calculated as γth = αKTVT/CV [47].

Table A2.
Ab initio B3LYP vibrational contribution to thermodynamic functions of MgO at P = 1 bar, T = 298.15-3000 K. Symbols: Uvib = vibrational internal energy; Hvib = vibrational enthalpy; Svib = vibrational entropy; Fvib, Gvib = vibrational Helmholtz and Gibbs free energy, respectively.
Table A2.
Ab initio B3LYP vibrational contribution to thermodynamic functions of MgO at P = 1 bar, T = 298.15-3000 K. Symbols: Uvib = vibrational internal energy; Hvib = vibrational enthalpy; Svib = vibrational entropy; Fvib, Gvib = vibrational Helmholtz and Gibbs free energy, respectively.
| T (K) | Uvib (kJ/mol) | Hvib (kJ/mol) | Svib (J/mol·K) | Fvib (kJ/mol) | Gvib (kJ/mol) |
|---|---|---|---|---|---|
| 0 | 14.555 1 | 14.556 | 0.000 | 14.555 | 14.556 |
| 100 | 14.735 | 14.736 | 2.290 | 14.506 | 14.507 |
| 298.15 | 19.581 | 19.582 | 26.080 | 11.805 | 11.806 |
| 300 | 19.648 | 19.649 | 26.304 | 11.757 | 11.758 |
| 400 | 23.556 | 23.557 | 37.513 | 8.551 | 8.552 |
| 500 | 27.839 | 27.840 | 47.059 | 4.309 | 4.310 |
| 600 | 32.333 | 32.334 | 55.248 | −0.816 | −0.815 |
| 700 | 36.954 | 36.955 | 62.370 | −6.705 | −6.704 |
| 800 | 41.659 | 41.660 | 68.651 | −13.262 | −13.261 |
| 900 | 46.421 | 46.422 | 74.259 | −20.412 | −20.411 |
| 1000 | 51.222 | 51.223 | 79.318 | −28.096 | −28.095 |
| 1100 | 56.054 | 56.055 | 83.922 | −36.261 | −36.259 |
| 1200 | 60.908 | 60.909 | 88.146 | −44.867 | −44.866 |
| 1300 | 65.779 | 65.780 | 92.045 | −53.879 | −53.878 |
| 1400 | 70.664 | 70.665 | 95.665 | −63.267 | −63.266 |
| 1500 | 75.560 | 75.561 | 99.043 | −73.004 | −73.003 |
| 1600 | 80.465 | 80.466 | 102.208 | −83.069 | −83.067 |
| 1700 | 85.378 | 85.379 | 105.187 | −93.439 | −93.438 |
| 1800 | 90.296 | 90.297 | 107.998 | −104.101 | −104.099 |
| 1900 | 95.220 | 95.221 | 110.660 | −115.035 | −115.033 |
| 2000 | 100.149 | 100.150 | 113.188 | −126.228 | −126.226 |
| 2100 | 105.081 | 105.082 | 115.595 | −137.668 | −137.667 |
| 2200 | 110.017 | 110.018 | 117.891 | −149.343 | −149.341 |
| 2300 | 114.955 | 114.956 | 120.086 | −161.243 | −161.241 |
| 2400 | 119.896 | 119.897 | 122.189 | −173.357 | −173.356 |
| 2500 | 124.839 | 124.840 | 124.207 | −185.678 | −185.677 |
| 2600 | 129.784 | 129.785 | 126.146 | −198.196 | −198.195 |
| 2700 | 134.731 | 134.732 | 128.013 | −210.905 | −210.903 |
| 2800 | 139.680 | 139.681 | 129.813 | −223.796 | −223.795 |
| 2900 | 144.630 | 144.631 | 131.550 | −236.865 | −236.863 |
| 3000 | 149.581 | 149.582 | 133.228 | −250.104 | −250.103 |
1 Zero-point correction to internal energy (i.e., UZPC).
Appendix B
The possible effects of the equation of state and thermodynamic parameters used for MgO on the post-spinel phase equilibrium boundary (cf. Equation (19)) are outlined in Figure A1.
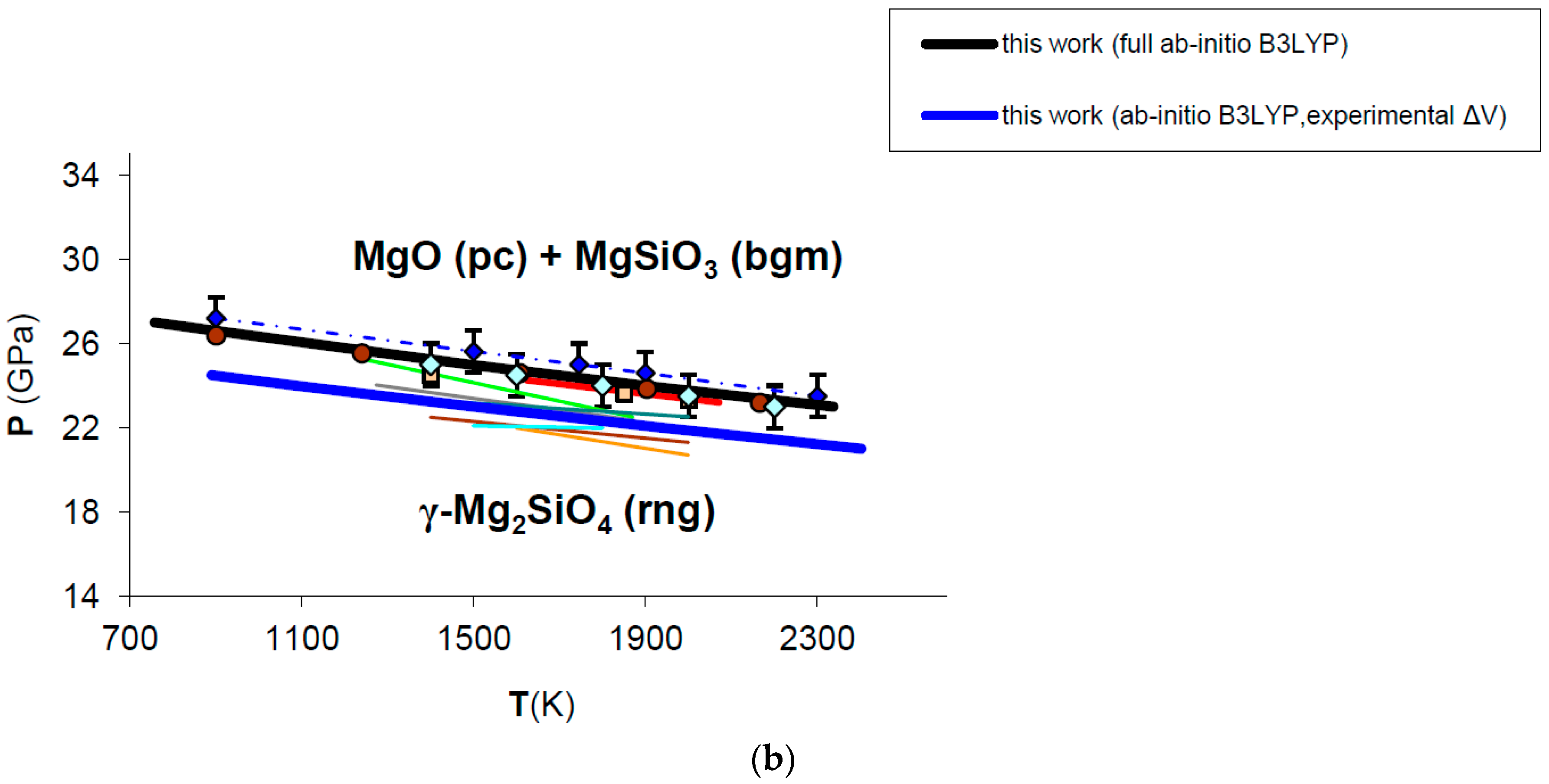
The adoption of HT-BM3-EOS or FPMG-EOS to calculate the volume-pressure contribution to Gibbs free energy of MgO may alter the phase equilibrium boundary by ±1 GPa (black and blue lines in Figure A1a, respectively). Actually, the effect of the EOS on the phase boundary is possibly less marked because P–V–T relations computed by FPMG-EOS (requiring a full ab initio B3LYP phonon dispersion calculation) are currently available for MgO (cf. Table 4 and Figure 7), but not yet for the other phases involved in the reaction (i.e., Mg2SiO4 ringwoodite and MgSiO3 bridgmanite). It is sensible to assume that Gibbs free energy differences tend to cancel out and the computed phase boundaries to converge when the same EOS formalism (i.e., HT-BM3-EOS or FPMG-EOS) is used for all mineralogical phases of the reaction. Therefore the upper phase boundary (in blue in Figure A1a) is computed by using a HT-BM3-EOS for ringwoodite and bridgmanite and a FPMG-EOS for MgO according to the delta values calculated for the VdP integral in Figure 10 (see Section 3.4 for details). The lower phase boundary (in black in Figure A1b) is computed by the self-consistent use of HT-BM3-EOS for all phases in the reaction according to thermophysical properties listed in Table A3 (KT, α and CP polynomial functions are fitted to ab initio values). Enthalpy, entropy and volume changes of the reaction at T = 298.15 K, P = 1 bar are obviously the same for both calculations (see Table 5). This exercise highlights that the effect of the EOS on the post-spinel phase boundary is virtually negligible at those P–T conditions. Furthermore, the phase boundary calculated by adopting a HT-BM3-EOS for all mineral phases is also physically sound in this case, because spurious effects on the V(P,T) function of MgO becomes relevant at much higher pressures (i.e., P > 60 GPa).
Since the post-spinel reaction (cf. Equation (19)) is strongly volume-dependent, the effect of thermodynamic parameters on the phase equilibrium calculation has been tested by changing Δ. Figure A1b shows two different phase boundaries computed by adopting either ab initio volumes (i.e., Δ = −3.52 cm3/mol; cf. Table 5) or experimental volumes (Δ = −3.80 cm3/mol) for periclase [49], bridgmanite [149] and ringwoodite [150] (black and blue lines in Figure A1b, respectively). It is quite evident from the analysis of Figure A1b that even small differences in volume may cause displacements of the phase boundary (~2–3 GPa in this case), which seem to be more relevant than those induced by the equation of state (see Figure A1a for comparison). The internal consistency of thermodynamic parameters (which is always ensured by first principles calculations) is thus crucial to accurately predicting phase equilibrium boundaries in HP–HT conditions.

Table A3.
Ab initio B3LYP thermophysical properties used to calculate the post-spinel phase equilibrium (cf. Equation (19)). K0 = bulk modulus at the athermal limit (i.e., T = 0 K, P = 0 GPa); K′0 = (dK/dP)0 = pressure derivative of the bulk modulus at T = 0 K, P = 0 GPa; (dK/dT)P = temperature derivative of the bulk modulus, with KT = K0 + T·(dK/dT)P; α = thermal expansivity (i.e., α = α0T + α1 + α2T−1 + α3T−2 + α4T−3); CP = isobaric heat capacity (i.e., CP = a + bT + cT−2 + dT2 + eT−0.5). Abbreviations for minerals as in Figure 11.
Table A3.
Ab initio B3LYP thermophysical properties used to calculate the post-spinel phase equilibrium (cf. Equation (19)). K0 = bulk modulus at the athermal limit (i.e., T = 0 K, P = 0 GPa); K′0 = (dK/dP)0 = pressure derivative of the bulk modulus at T = 0 K, P = 0 GPa; (dK/dT)P = temperature derivative of the bulk modulus, with KT = K0 + T·(dK/dT)P; α = thermal expansivity (i.e., α = α0T + α1 + α2T−1 + α3T−2 + α4T−3); CP = isobaric heat capacity (i.e., CP = a + bT + cT−2 + dT2 + eT−0.5). Abbreviations for minerals as in Figure 11.
| Thermophysical Properties | MgO (pc) 1 | MgSiO3 (bgm) 2 | γ-Mg2SiO4 (rng) 3 |
|---|---|---|---|
| K0 (GPa) | 167.01 | 249.2 | 196.4 |
| K′0 | 3.95 | 4.2 | 4.322 |
| (dK/dT)P (bar/K) | −308.0 | −280.0 | −104.108 |
| α0 × 107 | 0.2608 | 0.0818 | 0.0 |
| α 1 × 107 | −104.77954 | 198.2 | 323.0 |
| α 2 × 103 | 42.9578 | 0.0 | −7.3084 |
| α3 | −17.04307 | −0.474 | 1.3745 |
| α 4 | 2122.59 | 0.0 | −150.406 |
| a × 10−2 | 0.44202 | 1.1012 | 1.5346 |
| b × 102 | 0.39852 | 0.95903 | 2.1405 |
| c × 10−6 | −1.2908 | −3.8879 | −4.749 |
| d × 105 | 0.12101 | 0.099651 | −0.308 |
| e × 10−2 | 0.93607 | 1.9259 | 1.2708 |
1 This work; 2 Belousov [136]; 3 Ottonello et al. [36].
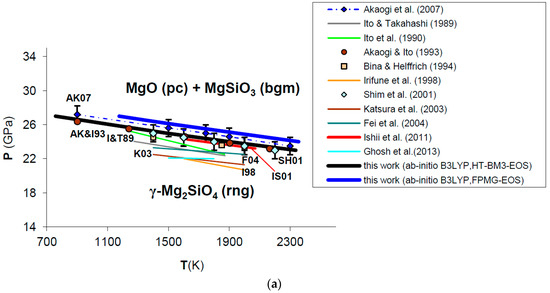
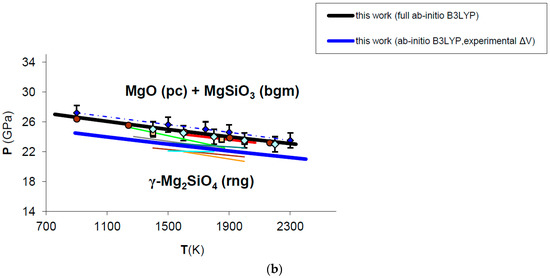
Figure A1.
Effects of EOS and thermodynamic parameters on the post-spinel phase equilibrium: (a) ab initio B3LYP phase boundary calculated by adopting either a FPMG-EOS (blue line, in bold) or a HT-BM3-EOS (black line, in bold) for MgO; (b) ab initio B3LYP phase boundary calculated by adopting either ab initio volumes (black line, in bold) or experimental volumes (blue line, in bold) for all phases in the reaction. Experimental results are shown for comparison (cf. Figure 11). Abbreviations for minerals and labels as in Figure 11.
Figure A1.
Effects of EOS and thermodynamic parameters on the post-spinel phase equilibrium: (a) ab initio B3LYP phase boundary calculated by adopting either a FPMG-EOS (blue line, in bold) or a HT-BM3-EOS (black line, in bold) for MgO; (b) ab initio B3LYP phase boundary calculated by adopting either ab initio volumes (black line, in bold) or experimental volumes (blue line, in bold) for all phases in the reaction. Experimental results are shown for comparison (cf. Figure 11). Abbreviations for minerals and labels as in Figure 11.
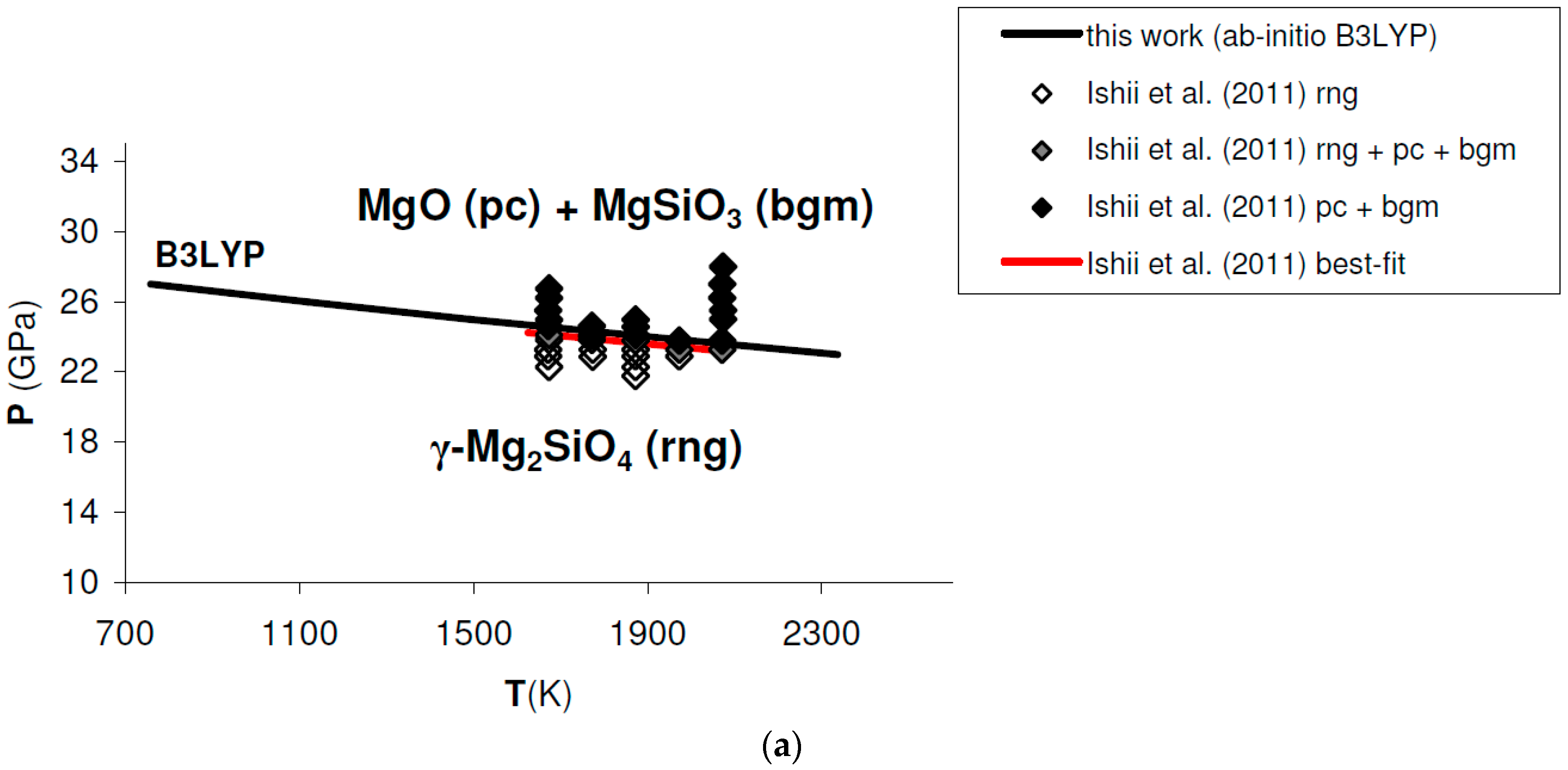
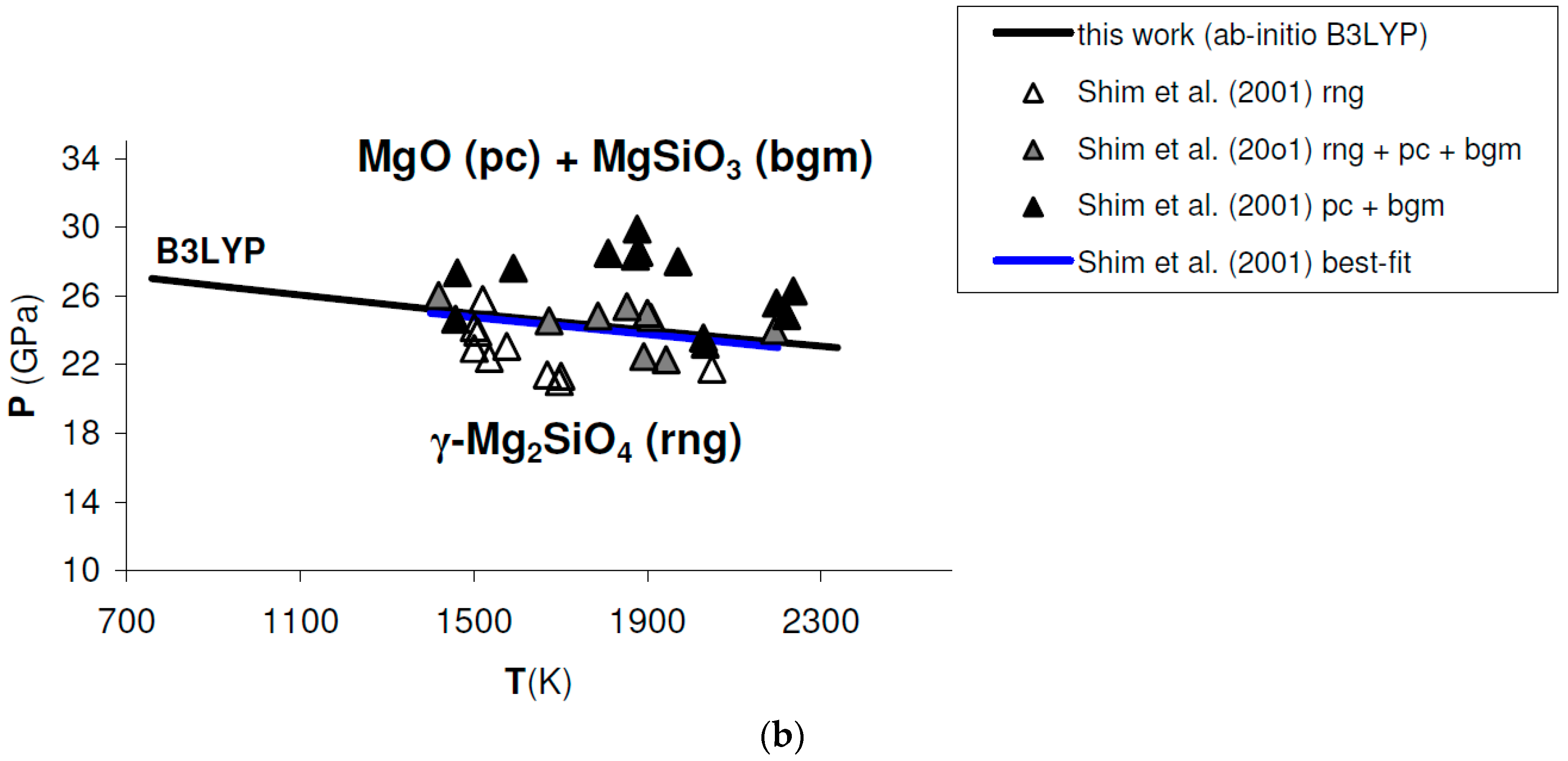
A detailed comparison between the post-spinel phase boundary calculated ab initio in this work and selected experimental results obtained in the laser-heated diamond-anvil cell [128] or multi-anvil apparatus [132] is shown in Figure A2. Linear best-fits of experimental runs are also indicated (red and blue lines in Figure A2a,b, respectively).
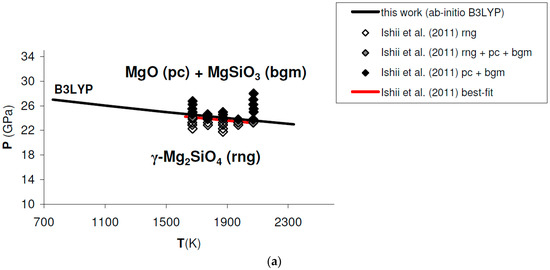
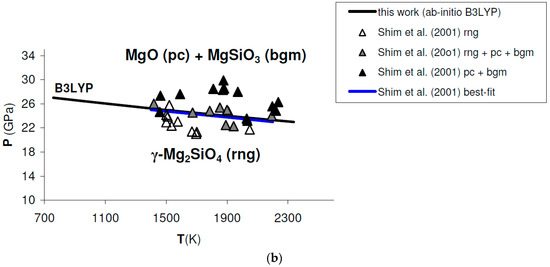
Figure A2.
Ab initio B3LYP phase boundary of post-spinel transformation (in bold) as compared to: (a) results obtained by multi-anvil experiments (diamonds) [132]; (b) results obtained by laser-heated diamond-anvil cell experiments (triangles) [128]. Best-fits of experimental results are also indicated. Abbreviations for minerals as in Figure 11.
Figure A2.
Ab initio B3LYP phase boundary of post-spinel transformation (in bold) as compared to: (a) results obtained by multi-anvil experiments (diamonds) [132]; (b) results obtained by laser-heated diamond-anvil cell experiments (triangles) [128]. Best-fits of experimental results are also indicated. Abbreviations for minerals as in Figure 11.
References
- Kuskov, O.L.; Fabrichnaya, O.B. Constitution of the Moon: 1. Composition and seismic properties of the lower mantle. Phys. Earth Planet. Inter. 1994, 83, 197–216. [Google Scholar] [CrossRef]
- Saxena, S.K. Earth mineralogical model: Gibbs free energy minimization computation in the system MgO-FeO-SiO2. Geochim. Cosmochim. Acta 1996, 60, 2379–2395. [Google Scholar] [CrossRef]
- Bina, C.R. Free energy minimization by simulated annealing with applications to lithospheric slabs and mantle plumes. Pure Appl. Geophys. 1998, 151, 605–618. [Google Scholar] [CrossRef]
- Jacobs, M.H.G.; de Jong, B.H.W.S. Placing constraints on phase equilibria and thermophysical properties in the system MgO-SiO2 by a thermodynamically consistent vibrational method. Geochim. Cosmochim. Acta 2007, 71, 3630–3655. [Google Scholar] [CrossRef]
- Piazzoni, A.S.; Steinle-Neumann, G.; Bunge, H.-P.; Dolejš, D. A mineralogical model for density and elasticity of the Earth’s mantle. Geochem. Geophys. Geosyst. 2007, 8, Q11010. [Google Scholar] [CrossRef]
- Ganguly, J.; Freed, A.M.; Saxena, S.K. Density profiles of oceanic slabs and surrounding mantle: Integrated thermodynamic and thermal modelling, and implications for the fate of slabs at the 660 km discontinuity. Phys. Earth Planet. Inter. 2009, 172, 257–267. [Google Scholar] [CrossRef]
- Khan, A.; Connolly, J.A.D.; Olsen, N. Constraining the composition and thermal state of the mantle beneath Europe from inversion of long-period electromagnetic sounding data. J. Geophys. Res. Solid Earth 2006, 111, B10102. [Google Scholar] [CrossRef]
- Connolly, J.A.D.; Khan, A. Uncertainty of mantle geophysical properties computed from phase equilibrium models. Geophys. Res. Lett. 2016, 43, 5026–5034. [Google Scholar] [CrossRef]
- Stixrude, L.; Lithgow-Bertelloni, C. Thermodynamics of mantle minerals—I. Physical properties. Geophys. J. Int. 2005, 162, 610–632. [Google Scholar] [CrossRef]
- Stixrude, L.; Lithgow-Bertelloni, C. Thermodynamics of mantle minerals—II. Phase equilibria. Geophys. J. Int. 2011, 184, 1180–1213. [Google Scholar] [CrossRef]
- Belmonte, D.; Ottonello, G.; Vetuschi Zuccolini, M.; Attene, M. The system MgO-Al2O3-SiO2 under pressure: A computational study of melting relations and phase diagrams. Chem. Geol. 2017, 461, 54–64. [Google Scholar] [CrossRef]
- Belmonte, D.; Ottonello, G.; Vetuschi Zuccolini, M. Ab initio-assisted assessment of the CaO-SiO2 system under pressure. Calphad 2017, 59, 12–30. [Google Scholar] [CrossRef]
- Christensen, U. Effects of phase transitions on mantle convection. Annu. Rev. Earth Planet. Sci. 1995, 23, 65–87. [Google Scholar] [CrossRef]
- Faccenda, M.; Dal Zilio, L. The role of solid-soild phase transitions in mantle convection. Lithos 2017, 268, 198–224. [Google Scholar] [CrossRef]
- Bina, C.R. Lower mantle mineralogy and the geophysical perspective. In Ultrahigh-Pressure Mineralogy: Physics and Chemistry of the Earth’s Deep Interior. Reviews in Mineralogy; Hemley, R.J., Ed.; Mineralogical Society of America: Chantilly, VA, USA, 1998; Volume 37, pp. 205–240. [Google Scholar]
- Yamazaki, D.; Karato, S.-I. Some mineral physics constraints on the rheology and geothermal structure of Earth’s lower mantle. Am. Mineral. 2001, 86, 385–391. [Google Scholar] [CrossRef]
- Karki, B.B.; Stixrude, L.; Clark, S.J.; Warren, M.C.; Ackland, G.J.; Crain, J. Structure and elasticity of MgO at high pressure. Am. Mineral. 1997, 82, 51–60. [Google Scholar] [CrossRef]
- Tsuchiya, T.; Kawamura, K. Systematics of elasticity: Ab initio study in B1-type alkaline earth oxides. J. Chem. Phys. 2001, 114, 10086–10093. [Google Scholar] [CrossRef]
- Wentzcovitch, R.M.; Wu, Z.; Carrier, P. First principles quasiharmonic thermoelasticity of mantle minerals. In Theoretical and Computational Methods in Mineral Physics: Geophysical Applications. Reviews in Mineralogy and Geochemistry; Wentzcovitch, R.M., Stixrude, L., Eds.; Mineralogical Society of America and Geochemical Society: Chantilly, VA, USA, 2010; Volume 71, pp. 99–128. [Google Scholar]
- Becke, A.D. Density-functional thermochemistry. III. The role of exact exchange. J. Chem. Phys. 1993, 98, 5648–5652. [Google Scholar] [CrossRef]
- Demichelis, R.; Civalleri, B.; Ferrabone, M.; Dovesi, R. On the performance of eleven DFT functionals in the description of the vibrational properties of aluminosilicates. Int. J. Quantum Chem. 2010, 110, 406–415. [Google Scholar] [CrossRef]
- Demichelis, R.; Civalleri, B.; D’Arco, P.; Dovesi, R. Performance of 12 DFT functionals in the study of crystal systems: Al2SiO5 orthosilicates and Al hydroxides as a case study. Int. J. Quantum Chem. 2010, 110, 2260–2273. [Google Scholar] [CrossRef]
- De La Pierre, M.; Orlando, R.; Maschio, L.; Doll, K.; Ugliengo, P.; Dovesi, R. Performance of six functionals (LDA, PBE, PBESOL, B3LYP, PBE0 and WC1LYP) in the simulation of vibrational and dielectric properties of crystalline compounds. The case of forsterite Mg2SiO4. J. Comput. Chem. 2011, 32, 1775–1784. [Google Scholar] [CrossRef] [PubMed]
- De La Pierre, M.; Belmonte, D. Ab initio investigation of majorite and pyrope garnets: Lattice dynamics and vibrational spectra. Am. Mineral. 2016, 101, 162–174. [Google Scholar] [CrossRef]
- Prencipe, M.; Mantovani, L.; Tribaudino, M.; Bersani, D.; Lottici, P.P. The Raman spectrum of diopside: A comparison between ab initio calculated and experimentally measured frequencies. Eur. J. Mineral. 2012, 24, 457–464. [Google Scholar] [CrossRef]
- Prencipe, M.; Scanavino, I.; Nestola, F.; Merlini, M.; Civalleri, B.; Bruno, M.; Dovesi, R. High-pressure thermo-elastic properties of beryl (Al4Be6Si12O36) from ab initio calculations, and observations about the source of thermal expansion. Phys. Chem. Miner. 2011, 38, 223–239. [Google Scholar] [CrossRef]
- Zucchini, A.; Prencipe, M.; Belmonte, D.; Comodi, P. Ab initio study of the dolomite to dolomite-II high-pressure phase transition. Eur. J. Mineral. 2017, 29, 227–238. [Google Scholar] [CrossRef]
- Ottonello, G.; Civalleri, B.; Ganguly, J.; Perger, W.F.; Belmonte, D.; Vetuschi Zuccolini, M. Thermo-chemical and thermo-physical properties of the high-pressure phase anhydrous B (Mg14Si5O24): An ab-initio all-electron investigation. Am. Mineral. 2010, 95, 563–573. [Google Scholar] [CrossRef]
- Ottonello, G.; Vetuschi Zuccolini, M.; Belmonte, D. The vibrational behavior of silica clusters at the glass transition: Ab initio calculations and thermodynamic implications. J. Chem. Phys. 2010, 133, 104508. [Google Scholar] [CrossRef] [PubMed]
- Belmonte, D.; Ottonello, G.; Vetuschi Zuccolini, M. Melting of α-Al2O3 and vitrification of the undercooled alumina liquid: Ab initio vibrational calculations and their thermodynamic implications. J. Chem. Phys. 2013, 138, 064507. [Google Scholar] [CrossRef] [PubMed]
- Belmonte, D.; Ottonello, G.; Vetuschi Zuccolini, M. Ab initio thermodynamic and thermophysical properties of sapphirine end-members in the join Mg4Al8Si2O20-Mg3Al10SiO20. Am. Mineral. 2014, 99, 1449–1461. [Google Scholar] [CrossRef]
- Belmonte, D.; Gatti, C.; Ottonello, G.; Richet, P.; Vetuschi Zuccolini, M. Ab initio thermodynamic and thermophysical properties of sodium metasilicate, Na2SiO3, and their electron-densityand electron-pair-density counterparts. J. Phys. Chem. A 2016, 120, 8881–8895. [Google Scholar] [CrossRef] [PubMed]
- Erba, A.; Mahmoud, A.; Belmonte, D.; Dovesi, R. High pressure elastic properties of minerals from ab initio simulations: The case of pyrope, grossular and andradite silicate garnets. J. Chem. Phys. 2014, 140, 124703. [Google Scholar] [CrossRef] [PubMed]
- Kohanoff, J. Electronic Structure Calculations for Solids and Molecules: Theory and Computational Methods; Cambridge University Press: Cambridge, UK, 2006; p. 372. [Google Scholar]
- Corà, F.; Alfredsson, M.; Mallia, G.; Middlemiss, D.S.; Mackrodt, W.C.; Dovesi, R.; Orlando, R. The Performance of Hybrid Density Functionals in Solid State Chemistry. In Structure and Bonding, Principles and Applications of Density Functional Theory in Inorganic Chemistry II; Kaltsoyannis, N., McGrady, J.E., Eds.; Springer: Berlin/Heidelberg, Germany, 2004; Volume 113, pp. 171–232. [Google Scholar]
- Ottonello, G.; Civalleri, B.; Ganguly, J.; Vetuschi Zuccolini, M.; Nöel, Y. Thermophysical properties of the α-β-γ polymorphs of Mg2SiO4: A computational study. Phys. Chem. Miner. 2009, 36, 87–106. [Google Scholar] [CrossRef]
- Dovesi, R.; Saunders, V.R.; Roetti, C.; Orlando, R.; Zicovich-Wilson, C.M.; Pascale, F.; Civalleri, B.; Doll, K.; Harrison, N.M.; Bush, I.J.; et al. CRYSTAL14 User’s Manual; Università di Torino: Torino, Italy, 2014; 382p. [Google Scholar]
- Lee, C.; Yang, E.; Parr, R.G. Development of the Colle-Salvetti correlation-energy formula into a functional of the electron density. Phys. Rev. B 1988, 37, 785–789. [Google Scholar] [CrossRef]
- Monkhorst, H.J.; Pack, J.D. Special points for Brillouin-zone integrations. Phys. Rev. B 1976, 13, 5188–5192. [Google Scholar] [CrossRef]
- Broyden, C.G. The converge of a class of double-rank minimization algorithms 1. General considerations. IMA J. Appl. Math. 1970, 6, 76–90. [Google Scholar] [CrossRef]
- Civalleri, B.; D’Arco, P.; Orlando, R.; Saunders, V.R.; Dovesi, R. Hartree-Fock geometry optimization of periodic system with the CRYSTAL code. Chem. Phys. Lett. 2001, 348, 131–138. [Google Scholar] [CrossRef]
- Doll, K. Implementation of analytical Hartree-Fock gradients for periodic systems. Comput. Phys. Commun. 2001, 137, 74–88. [Google Scholar] [CrossRef]
- Pascale, F.; Zicovich-Wilson, C.M.; López Gejo, F.; Civalleri, B.; Orlando, R.; Dovesi, R. The calculation of the vibrational frequencies of crystalline compounds and its implementation in the CRYSTAL code. J. Comput. Chem. 2004, 25, 888–895. [Google Scholar] [CrossRef] [PubMed]
- Parlinski, K.; Li, Z.Q.; Kawazoe, Y. First-principles determination of the soft mode in cubic ZrO2. Phys. Rev. Lett. 1997, 78, 4063–4066. [Google Scholar] [CrossRef]
- Alfè, D. PHON: A program to calculate phonons using the small displacement method. Comput. Phys. Commun. 2009, 180, 2622–2633. [Google Scholar] [CrossRef]
- Landau, L.D.; Lifshitz, E.M. Statistical Physics. Part 1, 3rd ed.; Butterworth-Heinemann: Oxford, UK, 1980; pp. 191–224. [Google Scholar]
- Anderson, O.L. Equation of State for Geophysics and Ceramic Science. Oxford Monographs on Geology and Geophysics; Oxford University Press: New York, NY, USA, 1995; Volume 31, pp. 3–18. [Google Scholar]
- Barron, T.H.K.; Berg, W.T.; Morrison, J.A. On the heat capacity of crystalline magnesium oxide. Proc. R. Soc. 1959, 250, 70–83. [Google Scholar] [CrossRef]
- Dubrovinsky, L.S.; Saxena, S.K. Thermal expansion of periclase (MgO) and tungsten (W) to melting temperatures. Phys. Chem. Miner. 1997, 24, 547–550. [Google Scholar] [CrossRef]
- Anderson, O.L.; Zou, K. Thermodynamic functions and properties of MgO at high compression and high temperature. J. Phys. Chem. Ref. Data 1990, 19, 69–81. [Google Scholar] [CrossRef]
- Singh, B.P.; Srivastava, S.K.; Dinesh, K. Relation between thermal expansivity and bulk modulus for ionic solids at high temperatures. Phys. B 2004, 349, 401–407. [Google Scholar] [CrossRef]
- Pavese, A. Pressure-volume-temperature equations of state: A comparative study based on numerical simulations. Phys. Chem. Miner. 2002, 29, 43–51. [Google Scholar] [CrossRef]
- Angel, R.J. Equations of state. In High-Temperature and High-Pressure Crystal Chemistry. Reviews in Mineralogy and Geochemistry; Hazen, R.M., Downs, R.T., Eds.; Mineralogical Society of America and Geochemical Society: Chantilly, VA, USA, 2000; Volume 41, pp. 35–60. [Google Scholar]
- Angel, R.J.; Gonzalez-Platas, J.; Alvaro, M. EosFit7c and a Fortran module (library) for equation of state calculations. Z. Krist. 2014, 229, 405–419. [Google Scholar] [CrossRef]
- Duffy, T.S.; Wang, Y. Pressure-volume-temperature equations of state. In Ultrahigh-Pressure Mineralogy: Physics and Chemistry of the Earth’s Deep Interior. Reviews in Mineralogy; Hemley, R.J., Ed.; Mineralogical Society of America: Chantilly, VA, USA, 1998; Volume 37, pp. 425–457. [Google Scholar]
- Oganov, A.R.; Brodholt, J.P.; Price, G.D. Ab initio theory of phase transitions and thermoelasticity of minerals. In Energy Modelling in Minerals. EMU Notes in Mineralogy; Gramaccioli, C.M., Ed.; Eötvös University Press: Budapet, Hungary, 2002; Volume 4, pp. 83–170. [Google Scholar]
- Isaak, D.G.; Anderson, O.L.; Goto, T. Measured elastic moduli of single-crystal MgO up to 1800 K. Phys. Chem. Miner. 1989, 16, 704–713. [Google Scholar] [CrossRef]
- Karki, B.B.; Wentzcovitch, R.M.; de Gironcoli, S.; Baroni, S. High-pressure lattice dynamics and thermoelasticity of MgO. Phys. Rev. B 2000, 61, 8793–8800. [Google Scholar] [CrossRef]
- Wu, Z.; Wentzcovitch, R.M.; Umemoto, K.; Li, B.; Hirose, K.; Zheng, J.-C. Pressure-volume-temperature relations in MgO: An ultrahigh pressure-temperature scale for planetary sciences applications. J. Geophys. Res. Solid Earth 2008, 113, B06204. [Google Scholar] [CrossRef]
- Matsui, M.; Parker, S.C.; Leslie, M. The MD simulation of the equation of state of MgO: Application as a pressure calibration standard at high temperature and high pressure. Am. Mineral. 2000, 85, 312–316. [Google Scholar] [CrossRef]
- Oganov, A.R.; Dorogokupets, P.I. All-electron and pseudopotential study of MgO: Equation of state, anharmonicity, and stability. Phys. Rev. B 2003, 67, 224110. [Google Scholar] [CrossRef]
- Scanavino, I.; Belousov, R.; Prencipe, M. Ab initio quantum-mechanical study of the effects of the inclusion of iron on thermoelastic and thermodynamic properties of periclase (MgO). Phys. Chem. Miner. 2012, 39, 649–663. [Google Scholar] [CrossRef]
- Jacobs, M.H.G.; Oonk, H.A.J. A realistic equation of state for solids. The high pressure and high temperature thermodynamic properties of MgO. Calphad 2000, 24, 133–147. [Google Scholar] [CrossRef]
- Fiquet, G.; Richet, P.; Montagnac, G. High-temperature thermal expansion of lime, periclase, corunsum and spinel. Phys. Chem. Miner. 1999, 27, 103–111. [Google Scholar] [CrossRef]
- Suzuki, I. Thermal expansion of periclase and olivine, and their anharmonic properties. J. Phys. Earth 1975, 23, 145–159. [Google Scholar] [CrossRef]
- White, G.K.; Anderson, O.L. Gruneisen parameter of magnesium oxide. J. Appl. Phys. 1966, 37, 430–432. [Google Scholar] [CrossRef]
- Touloukian, Y.S.; Kirby, R.K.; Taylor, R.E.; Lee, T.Y.R. Thermal Expansion: Nonmetallic Solids. Thermophysical Properties of Matter, 1st ed.; IFI/Plenum: New York, NY, USA, 1977; Volume 13, p. 1786. [Google Scholar]
- Wentzcovitch, R.M.; Yu, Y.G.; Wu, Z. Thermodynamic properties and phase relations in mantle minerals investigated by first principles quasiharmonic theory. In Theoretical and Computational Methods in Mineral Physics: Geophysical Applications. Reviews in Mineralogy and Geochemistry; Wentzcovitch, R.M., Stixrude, L., Eds.; Mineralogical Society of America and Geochemical Society: Chantilly, VA, USA, 2010; Volume 71, pp. 59–98. [Google Scholar]
- Callen, H.B. Thermodynamics, 1st ed.; John Wiley & Sons, Inc.: New York, NY, USA, 1960; pp. 85–102. [Google Scholar]
- Lashley, J.C.; Hundley, M.F.; Migliori, A.; Sarrao, J.L.; Pagliuso, P.G.; Darling, T.W.; Jaime, M.; Cooley, J.C.; Hults, W.L.; Morales, L.; et al. Critical examination of heat capacity measurements made on a Quantum Design physical property measurement system. Cryogenics 2003, 43, 369–378. [Google Scholar] [CrossRef]
- Dachs, E.; Bertoldi, C. Precision and accuracy of the heat-pulse calorimetric technique: Low-temperature heat capacities of milligram-sized synthetic mineral samples. Eur. J. Mineral. 2005, 17, 251–261. [Google Scholar] [CrossRef]
- Dachs, E.; Benisek, A. A sample-saving method for heat capacity measurements on powders using relaxation calorimetry. Cryogenics 2011, 51, 460–464. [Google Scholar] [CrossRef] [PubMed]
- Bosenick, A.; Geiger, C.; Cemič, L. Heat capacity measurements of synthetic pyrope-grossular garnets between 320 and 1000 K by differential scanning calorimetry. Geochim. Cosmochim. Acta 1996, 60, 3215–3227. [Google Scholar] [CrossRef]
- Chase, M.W., Jr. NIST-JANAF Thermochemical Tables. Journal of Phyical and Chemical Reference Data, Monograph No. 9, Part II. Cr-Zr, 4th ed.; American Chemical Society and American Institute of Physics for the National Institute of Standards and Technology: New York, NY, USA, 1998; pp. 1536–1538. [Google Scholar]
- Robie, R.A.; Hemingway, B.S. Thermodynamic Properties of Minerals and Related Substances at 298.15 K and 1 Bar (105 Pascals) Pressures and at Higher Temperatures, U.S. Geological Survey Bulletin 2131; U.S. Geological Survey: Denver, Colorado, VA, USA, 1995; p. 197.
- Chopelas, A. Thermal expansion, heat capacity, and entropy of MgO at mantle pressures. Phys. Chem. Miner. 2000, 17, 142–148. [Google Scholar] [CrossRef]
- Cynn, H.; Anderson, O.L.; Isaak, D.G.; Nicol, M. Grüneisen ratio of MgO from the calculation of entropy. J. Phys. Chem. 1995, 99, 7813–7818. [Google Scholar] [CrossRef]
- Gheribi, A.E. Formulation of the thermal volume consistent with Swenson’s concept of thermal pressure. Phys. Earth Planet. Inter. 2009, 177, 59–64. [Google Scholar] [CrossRef]
- Murnaghan, F.D. Finite deformations of an elastic solid. Am. J. Math. 1937, 49, 235–260. [Google Scholar] [CrossRef]
- Birch, F. Finite elastic strain of cubic crystals. Phys. Rev. Lett. 1947, 71, 809–824. [Google Scholar] [CrossRef]
- Vinet, P.; Ferrante, J.; Smith, J.R.; Rose, J.H. A universal equation of state for solids. J. Phys. C Solid State Phys. 1986, 19, L467–L473. [Google Scholar] [CrossRef]
- Poirier, J.-P.; Tarantola, A. A logarithmic equation of state. Phys. Earth Planet. Inter. 1998, 109, 1–8. [Google Scholar] [CrossRef]
- Hama, J.; Suito, K. The search for a universal equation of state correct up to very high pressures. J. Phys. Condens. Matter 1996, 8, 67–81. [Google Scholar] [CrossRef]
- Cohen, R.E. MgO—The simplest oxide. In Physics Meets Mineralogy: Condensed-Matter Physics in Geosciences; Aoki, H., Syono, Y., Hemley, R.J., Eds.; Cambridge University Press: Cambridge, UK, 2000; pp. 95–123. [Google Scholar]
- Ponkratz, U.; Holzapfel, W.B. Equations of state for wide ranges in pressure and temperature. J. Phys. Condens. Matter 2004, 16, S963–S972. [Google Scholar] [CrossRef]
- Bose Roy, P.; Bose Roy, S. Applicability of isothermal three-parameter equations of state of solids—A reappraisal. J. Phys. Condens. Matter 2005, 17, 6193–6216. [Google Scholar]
- Grüneisen, E. Zustand de festen Körpers. In Thermische Eigenschaften der Stoffe; Springer: Berlin/Heidelberg, Germany, 1926; Volume 10, pp. 1–59. [Google Scholar]
- Born, M.; Huang, K. Dynamical Theory of Crystal Lattices, 1st ed.; Oxford University Press: London, UK, 1954; pp. 38–128. [Google Scholar]
- Utsumi, W.; Weidner, D.J.; Liebermann, R.C. Volume measurement of MgO at high pressures and high temperatures. In Properties of Earth and Planetary Materials at High Pressure and Temperature. Geophysical Monograph Series; Manghnani, M.H., Yagi, T., Eds.; American Geophysical Union: Washington, DC, USA, 1998; Volume 101, pp. 327–333. [Google Scholar]
- Fei, Y. Effects of temperature and composition on the bulk modulus of (Mg,Fe)O. Am. Mineral. 1999, 84, 272–276. [Google Scholar] [CrossRef]
- Dewaele, A.; Fiquet, G.; Andrault, D.; Hausermann, D. P–V–T equation of state of periclase from synchrotron radiation measurements. J. Geophys. Res. Solid Earth 2000, 105, 2869–2877. [Google Scholar] [CrossRef]
- Speziale, S.; Zha, C.-S.; Duffy, T.S.; Hemley, R.J.; Mao, H. Quasi-hydrostatic compression of magnesium oxide to 52 GPa: Implications for the pressure-volume-temperature equation of state. J. Geophys. Res. Solid Earth 2001, 106, 515–528. [Google Scholar] [CrossRef]
- Zha, C.-S.; Mibe, K.; Bassett, W.A.; Tschauner, O.; Mao, H.; Hemley, R.J. P–V–T equation of state of platinum to 80 GPa and 1900 K from internal resistive heating/X-ray diffraction measurements. J. Appl. Phys. 2008, 103, 054908. [Google Scholar] [CrossRef]
- Yoneda, A. Pressure derivatives of elastic constants of single crystal MgO and MgAl2O4. J. Phys. Earth 1990, 38, 19–55. [Google Scholar] [CrossRef]
- Duffy, T.S.; Hemley, R.J.; Mao, H. Equation of state and shear strenght at multimegabar pressures: Magnesium oxide to 227 GPa. Phys. Rev. Lett. 1995, 74, 1371–1374. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Woody, K.; Kung, J. Elasticity of MgO to 11 GPa with an independent absolute pressure scale: Implications for pressure calibration. J. Geophys. Res. Solid Earth 2006, 111, B11206. [Google Scholar] [CrossRef]
- Kono, Y.; Irifune, T.; Higo, Y.; Inoue, T.; Barnhoorn, A. P–V–T relations of MgO derived by simultaneous elastic wave velocity and in situ X-ray measurements: A new pressure scale for the mantle transition region. Phys. Earth Planet. Int. 2010, 183, 196–211. [Google Scholar] [CrossRef]
- Dorogokupets, P.I.; Oganov, A.R. Ruby, metals, and MgO as alternative pressure scales: A semiempirical description of shock-wave, ultrasonic, X-ray, and thermochemical data at high temperatures and pressures. Phys. Rev. B 2007, 75, 024115. [Google Scholar] [CrossRef]
- Garai, J.; Chen, J.; Telekes, G. The P–V–T equation of state of periclase. Calphad 2009, 33, 737–743. [Google Scholar] [CrossRef]
- Tange, Y.; Nishihara, Y.; Tsuchiya, T. Unified analyses for P–V–T equation of state of MgO: A solution for pressure-scale problems in high P–T experiments. J. Geophys. Res. Solid Earth 2009, 114, B03208. [Google Scholar] [CrossRef]
- Otero-de-la-Roza, A.; Luaña, V. Treatment of first-principles data for predictive quasiharmonic thermodynamics of solids: The case of MgO. Phys. Rev. B 2011, 84, 024109. [Google Scholar] [CrossRef]
- Srivastava, S.K. Analysis of thermoelastic constants of solids and minerals at high temperatures. J. Phys. Chem. Solids 2006, 67, 2275–2281. [Google Scholar] [CrossRef]
- Stacey, F.D. A thermal model of the Earth. Phys. Earth Planet. Inter. 1977, 15, 341–348. [Google Scholar] [CrossRef]
- Brown, J.M.; Shankland, T.J. Thermodynamic parameters in the Earth as determined from seismic profiles. Geophys. J. Int. 1981, 66, 579–596. [Google Scholar] [CrossRef]
- Karki, B.B. Thermal pressure in MgO and MgSiO3 perovskite at lower mantle conditions. Am. Mineral. 2000, 85, 1447–1451. [Google Scholar] [CrossRef]
- Stacey, F.D.; Isaak, D.G. Anharmonicity in mineral physics: A physical interpretation. J. Geophys. Res. Solid Earth 2003, 108, 2440. [Google Scholar] [CrossRef]
- Scanavino, I.; Prencipe, M. Ab-initio determination of high-pressure and high-temperature thermoelastic and thermodynamic properties of low-spin (Mg1−xFex)O ferropericlase with x in the range [0.06, 0.59]. Am. Mineral. 2013, 98, 1270–1278. [Google Scholar] [CrossRef]
- Merli, M.; Sciascia, L.; Pavese, A.; Diella, V. Modelling of thermo-chemical properties over the sub-solidus MgO-FeO binary, as a function of iron spin configuration, composition and temperature. Phys. Chem. Miner. 2015, 42, 347–362. [Google Scholar] [CrossRef]
- Merli, M.; Bonadiman, C.; Diella, V.; Pavese, A. Lower mantle hydrogen partitioning between periclase and perovskite: A quantum chemical modelling. Geochim. Cosmochim. Acta 2016, 173, 304–318. [Google Scholar] [CrossRef]
- Merli, M.; Bonadiman, C.; Diella, V.; Sciascia, L.; Pavese, A. Fe-periclase reactivity at Earth’s lower mantle conditions: Ab-initio geochemical modelling. Geochim. Cosmochim. Acta 2017, 214, 14–29. [Google Scholar] [CrossRef]
- Pavese, A.; Diella, V. Uncertainties on elastic parameters and occupancy factors: How do they affect the accuracy of the calculated Gibbs free energy of minerals at (P,T) conditions? The case of 3T- versus 2M1-phengite. Phys. Chem. Miner. 2007, 34, 637–645. [Google Scholar] [CrossRef]
- Ganguly, J. Thermodynamics in Earth and Planetary Sciences; Springer: Berlin/Heidelberg, Germany, 2008; pp. 145–147. [Google Scholar]
- Helffrich, G.; Connolly, J.A.D. Physical contradictions and remedies using simple polythermal equations of state. Am. Mineral. 2009, 94, 1616–1619. [Google Scholar] [CrossRef]
- Jacobs, M.H.G.; van den Berg, A.P.; de Jong, B.H.W.S. The derivation of thermo-physical properties and phase equilibria of silicate materials from lattice vibrations: Application to convection in the Earth’s mantle. Calphad 2006, 30, 131–146. [Google Scholar] [CrossRef]
- Brosh, E.; Makov, G.; Shneck, R.Z. Application of CALPHAD to high pressures. Calphad 2007, 31, 173–185. [Google Scholar] [CrossRef]
- Brosh, E.; Shneck, R.Z.; Makov, G. Explicit Gibbs free energy equation of state for solids. J. Phys. Chem. Solids 2008, 69, 1912–1922. [Google Scholar] [CrossRef]
- Karbasi, A.; Saxena, S.K.; Hrubiak, R. The thermodynamics of several elements at high pressure. Calphad 2011, 35, 72–81. [Google Scholar] [CrossRef]
- Holland, T.J.B.; Powell, R. An improved and extended internally consistent thermodynamic dataset for phases of petrological interest, involving a new equation of state for solids. J. Metamorph. Geol. 2011, 29, 333–383. [Google Scholar] [CrossRef]
- Tirone, M. On the use of thermal equations of state and the extrapolation at high temperature and pressure for geophysical and petrological applications. Geophys. J. Int. 2015, 202, 55–66. [Google Scholar] [CrossRef]
- Welche, P.R.L.; Heine, V.; Dove, M.T. Negative thermal expansion in beta-quartz. Phys. Chem. Miner. 1998, 26, 63–77. [Google Scholar] [CrossRef]
- Bina, C.R.; Helffrich, G. Phase transition Clapeyron slopes and transition zone seismic discontinuity topography. J. Geophys. Res. Solid Earth 1994, 99, 15853–15860. [Google Scholar] [CrossRef]
- Deuss, A.; Redfern, S.A.T.; Chambers, K.; Woodhouse, J.H. The nature of the 660-kilometer disconttinuity in Earth’s mantle from global seismic observations of PP precursors. Science 2006, 311, 198–201. [Google Scholar] [CrossRef] [PubMed]
- Dziewonski, A.M.; Anderson, D.L. Preliminary Reference Earth Model. Phys. Earth Planet. Inter. 1981, 25, 297–356. [Google Scholar] [CrossRef]
- Ito, E.; Takahashi, E. Postspinel transformations in the system Mg2SiO4-Fe2SiO4 and some geophysical implications. J. Geophys. Res. Solid Earth 1989, 94, 10637–10646. [Google Scholar] [CrossRef]
- Ito, E.; Akaogi, M.; Topor, L.; Navrotsky, A. Negative pressure-temperature slopes for reactions forming MgSiO3 perovskite from calorimetry. Science 1990, 249, 1275–1278. [Google Scholar] [CrossRef] [PubMed]
- Akaogi, M.; Ito, E. Refinement of enthalpy measurement of MgSiO3 perovskite and negative pressure-temperature slopes for perovskite-forming reactions. Geophys. Res. Lett. 1993, 20, 1839–1842. [Google Scholar] [CrossRef]
- Irifune, T.; Nishiyama, N.; Kuroda, K.; Inoue, T.; Isshiki, M.; Utsumi, W.; Funakoshi, K.; Urakawa, S.; Uchida, T.; Katsura, T.; et al. The postspinel phase boundary in Mg2SiO4 determined by in situ X-ray diffraction. Science 1998, 279, 1698–1700. [Google Scholar] [CrossRef] [PubMed]
- Shim, S.-H.; Duffy, T.S.; Shen, G. The post-spinel transformation in Mg2SiO4 and its relations to the 660-km seismic discontinuity. Nature 2001, 411, 571–574. [Google Scholar] [CrossRef] [PubMed]
- Katsura, T.; Yamada, H.; Shinmei, T.; Kubo, A.; Ono, S.; Kanzaki, M.; Yoneda, A.; Walter, M.J.; Ito, E.; Urakawa, S.; et al. Post-spinel transition in Mg2SiO4 determined by high P–T in situ X-ray diffractometry. Phys. Earth Planet. Inter. 2003, 136, 11–24. [Google Scholar] [CrossRef]
- Fei, Y.; Van Orman, J.; Li, J.; van Westrenen, W.; Sanloup, C.; Minarik, W.; Hirose, K.; Komabayashi, T.; Walter, M.; Funakoshi, K. Experimentally determined postspinel transformation boundary in Mg2SiO4 using MgO as an internal pressure standard and its geophysical implications. J. Geophys. Res. Solid Earth 2004, 109, B02305. [Google Scholar] [CrossRef]
- Akaogi, M.; Takayama, H.; Kojitani, H.; Kawaji, H.; Atake, T. Low-temperature heat capacities, entropies and enthalpies of Mg2SiO4 polymorphs, and α-β-γ and post-spinel phase relations at high pressure. Phys. Chem. Miner. 2007, 34, 169–183. [Google Scholar] [CrossRef]
- Ishii, T.; Kojitani, H.; Akaogi, M. Post-spinel transitions in pyrolite and Mg2SiO4 and akimotoite-perovskite transition in MgSiO3: Precise comparison by high-pressure high-temperature experiments with multi-sample cell technique. Earth Planet. Sci. Lett. 2011, 309, 185–197. [Google Scholar] [CrossRef]
- Ghosh, S.; Ohtani, E.; Litasov, K.D.; Suzuki, A.; Dobson, D.; Funakoshi, K. Effect of water in depleted mantle on post-spinel transition and implication for 660 km seismic discontinuity. Earth Planet. Sci. Lett. 2013, 371, 103–111. [Google Scholar] [CrossRef]
- Kojitani, H.; Inoue, T.; Akaogi, M. Precise measurements of enthalpy of postspinel transition in Mg2SiO4 and application to the phase boundary calculation. J. Geophys. Res. Solid Earth 2016, 121, 729–742. [Google Scholar] [CrossRef]
- Chopelas, A.; Boehler, R.; Ko, T. Thermodynamics and behavior of γ-Mg2SiO4 at high pressure: Implications for Mg2SiO4 phase equilibrium. Phys. Rev. Lett. 1997, 78, 4063–4066. [Google Scholar] [CrossRef]
- Belousov, R. Ab-initio Quantum-Mechanical Simulation of Earth Minerals at Extreme Conditions of Temperature and Pressure. Ph.D. Thesis, University of Torino, Torino, Italy, 2011; p. 91, unpublished. [Google Scholar]
- Yu, Y.; Wentzcovitch, R.M.; Tsuchiya, T.; Umemoto, K.; Weidner, D.J. First principles investigation of the postspinel transition in Mg2SiO4. Geophys. Res. Lett. 2007, 34, L10306. [Google Scholar] [CrossRef]
- Hernández, E.R.; Alfè, D.; Brodholt, J. The incorporation of water into lower-mantle perovskites: A first-principles study. Earth Planet. Sci. Lett. 2013, 364, 37–43. [Google Scholar] [CrossRef]
- Hernández, E.R.; Brodholt, J.; Alfè, D. Structural, vibrational and thermodynamic properties of Mg2SiO4 and MgSiO3 minerals from first-principles simulations. Phys. Earth Planet. Inter. 2015, 240, 1–24. [Google Scholar] [CrossRef]
- Jacobs, M.H.G.; Schmid-Fetzer, R.; van den Berg, A.P. Phase diagrams, thermodynamic properties and sound velocities derived from a multiple Einstein method using vibrational densities of states: An application to MgO-SiO2. Phys. Chem. Miner. 2017, 44, 43–62. [Google Scholar] [CrossRef]
- Parisi, F.; Sciascia, L.; Princivalle, F.; Merli, M. The pressure-induced ringwoodite to Mg-perovskite and periclase post-spinel phase transition: A Bader’s topological analysis of the ab initio electron densities. Phys. Chem. Miner. 2012, 39, 103–113. [Google Scholar] [CrossRef]
- Shearer, P.M.; Flanagan, M.P. Seismic velocities and density jumps across the 410- and 660-kilometer discontinuities. Science 1999, 285, 1545–1548. [Google Scholar] [CrossRef] [PubMed]
- Cammarano, F.; Romanowicz, B. Insights into the nature of the transition zone from physically constrained inversion of long-period seismic data. Proc. Natl. Acad. Sci. USA 2007, 104, 9139–9144. [Google Scholar] [CrossRef] [PubMed]
- Agee, C.B. Petrology of the mantle transition zone. Annu. Rev. Earth Planet. Sci. 1993, 21, 19–41. [Google Scholar] [CrossRef]
- Agee, C.B. Phase transformations and seismic structure in the upper mantle and transition zone. In Ultrahigh-Pressure Mineralogy: Physics and Chemistry of the Earth’s Deep Interior. Reviews in Mineralogy; Hemley, R.J., Ed.; Mineralogical Society of America: Chantilly, VA, USA, 1998; Volume 37, pp. 165–203. [Google Scholar]
- Vacher, P.; Mocquet, A.; Sotin, C. Computation of seismic profiles from mineral physics: The importance of non-olivine components for explaining the 660 km depth discontinuity. Phys. Earth Planet. Inter. 1998, 106, 275–298. [Google Scholar] [CrossRef]
- Weidner, D.J.; Wang, Y. Chemical- and Clapeyron-induced buoyancy at the 660 km discontinuity. J. Geophys. Res. Solid Earth 1998, 103, 7431–7441. [Google Scholar] [CrossRef]
- Hirose, K. Phase transitions in pyrolitic mantle around 670-km depth: Implications for upwelling of plumes from the lower mantle. J. Geophys. Res. Solid Earth 2002, 107, 2078. [Google Scholar] [CrossRef]
- Dobson, D.P.; Jacobsen, S.D. The flux growth of magnesium silicate perovskite single crystals. Am. Mineral. 2004, 89, 807–811. [Google Scholar] [CrossRef]
- Sasaki, S.; Prewitt, C.T.; Sato, Y.; Ito, E. Single-crystal X-ray study of γ-Mg2SiO4. J. Geophys. Res. Solid Earth 1982, 87, 7829–7832. [Google Scholar] [CrossRef]
© 2017 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).