Molecular Correlation between Larval, Deutonymph and Adult Stages of the Water Mite Arrenurus (Micruracarus) Novus
Abstract
:1. Introduction
2. Materials and Methods
2.1. Taxon Sampling
2.2. Morphological Identification of Culicids
2.3. Morphological Identification of Water Mites
2.3.1. Rearing of Adult Female Water Mites
2.3.2. Morphological Identification of Water Mite Adults, Deutonymphs and Larvae
2.3.3. Laser Scanning Confocal Microscopy Imaging
2.4. Molecular Analyses
3. Results
3.1. Morphological Identification of Culicids
3.2. Morphological and Molecular Identification of Water Mites
3.2.1. Rearing of Adult Female Water Mites
3.2.2. Larval Description
3.2.3. Morphological Identification of Adult and Deutonymph Water Mites
3.3. Molecular Analyses
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Thorp, J.H.; Rogers, D.C.; Dimmick, W.W. Introduction to invertebrates of inland waters. In Freshwater Invertebrates, 4th ed.; Elsevier BV: Amsterdam, The Netherlands, 2015; pp. 1–19. [Google Scholar]
- Gerecke, R.; Gledhill, T.; Pešić, V.; Smit, H. Süßwasserfauna von Mitteleuropa, Bd. 7/2-3 Chelicerata; Springer Spektrum: Heidelberg, Germany, 2016. [Google Scholar]
- Smith, I.M.; Cook, D.R.; Smith, B.P. Water mites (Hydrachnidiae) and other arachnids. In Ecology and Classification of North American Freshwater Invertebrates; Elsevier BV: Amsterdam, The Netherlands, 2010; pp. 485–586. [Google Scholar]
- Cook, W.J.; Smith, B.P.; Brooks, R.J. Allocation of reproductive effort in female Arrenurus spp. water mites (Acari: Hydrachnidia; Arrenuridae). Oecologia 1989, 79, 184–188. [Google Scholar] [CrossRef]
- Tuzovsky, P. A new water mite species of the genus Arrenurus Dugès, 1834 (Acariformes: Hydrachnidia: Arrenuridae) from Eastern Palaearctic. Acarina 2012, 20, 173–179. [Google Scholar]
- Zawal, A. Phoresy and parasitism: Water mite larvae of the genus Arrenurus (Acari: Hydrachnidia) on Odonata from lake Binowskie (NW Poland). Biol. Lett. 2006, 43, 257–276. [Google Scholar]
- Zawal, A. Morphological characteristics of water mite larvae of the genus Arrenurus Duges, 1834, with notes on the phylogeny of the genus and an identification key. Zootaxa 2008, 1765, 1–75. [Google Scholar] [CrossRef]
- Zawal, A. Morphology of the larval stages of Arrenurus affinis Koenike, 1887, A. neumani Piersig, 1895, and A. vietsi Koenike, 1911 (Acari: Hydrachnidia). Genus 2008, 19, 161–169. [Google Scholar]
- Bohonak, A.J.; Smith, B.P.; Thornton, M. Distributional, morphological and genetic consequences of dispersal for temporary pond water mites. Freshw. Boil. 2004, 49, 170–180. [Google Scholar] [CrossRef]
- Lanciani, C.A. Sexual bias in host selection by parasitic mites of the mosquito anopheles crucians (Diptera: Culicidae). J. Parasitol. 1988, 74, 768. [Google Scholar] [CrossRef]
- Markmann, M.; Tautz, D. Reverse taxonomy: An approach towards determining the diversity of meiobenthic organisms based on ribosomal RNA signature sequences. Philos. Trans. R. Soc. B: Boil. Sci. 2005, 360, 1917–1924. [Google Scholar] [CrossRef] [Green Version]
- Alarcón-Elbal, P.M.; Murillo, J.M.S.; Estrella, S.D.; Ruiz-Arrondo, I.; Prieto, R.P.; Curdi, J.L. Asociación de vector del VNO e hidrófito invasor: Culex pipiens Linnaeus, 1758 y Ludwigia grandiflora (Michaux) Greuter and Burdet en el marjal de Xeraco-Xeresa, Valencia. Anales Biol. 2013, 35, 17–27. [Google Scholar] [CrossRef]
- Service, M.W. A critical review of procedures for sampling populations of adult mosquitoes. Bull. Entomol. Res. 1977, 67, 343–382. [Google Scholar] [CrossRef]
- Valdecasas, A.G.; Abad, A. Morphological confocal microscopy in arthropods and the enhancement of autofluorescence after proteinase K extraction. Microsc. Microanal. 2010, 17, 109–113. [Google Scholar] [CrossRef] [PubMed]
- Pešić, V.; Valdecasas, A.G.; García-Jimenez, R. Simultaneous evidence for a new species of Torrenticola Piersig, 1896 (Acari, Hydrachnidia) from Montenegro. Zootaxa 2012, 3515, 38–50. [Google Scholar] [CrossRef]
- Schaffner, E.; Angel, G.; Geoffroy, B.; Hervy, J.P.; Rhaiem, A.; Brunhes, J. Les Moustiques d’Europe: Logiciel d’Identification et d’Enseignement = The Mosquitoes of Europe: An Identification and Training Programme; IRD Editions & EID Méditerranée: Montpellier, France, 2001. [Google Scholar]
- George, C.F. The British fresh-water mites. Hardwicke’s Sci. 1884, 20, 80–81. [Google Scholar]
- Viets, K. Zur kenntnis dert Hydracarinen-Fauna von Spanien. Arch. Hydrobiol. 1930. [Google Scholar]
- Folmer, O.; Black, M.; Hoeh, W.; Lutz, R.; Vrijenhoek, R. DNA primers for amplification of mitochondrial cytochrome c oxidase subunit I from diverse metazoan invertebrates. Mol. Mar. Biol. Biotechnol. 1994, 3, 294–299. [Google Scholar] [CrossRef] [PubMed]
- Ernsting, B.R.; Edwards, D.D.; Aldred, K.J.; Fites, J.S.; Neff, C.R. Mitochondrial genome sequence of Unionicola foili (Acari: Unionicolidae): A unique gene order with implications for phylogenetic inference. Exp. Appl. Acarol. 2009, 49, 305–316. [Google Scholar] [CrossRef]
- Kimura, M. A simple method for estimating evolutionary rates of base substitutions through comparative studies of nucleotide sequences. J. Mol. Evol. 1980, 16, 111–120. [Google Scholar] [CrossRef]
- Kumar, S.; Stecher, G.; Tamura, K. MEGA7: Molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol. Boil. Evol. 2016, 33, 1870–1874. [Google Scholar] [CrossRef] [Green Version]
- Azari-Hamidian, S.; Harbach, R.E. Keys to the adult females and fourth-instar larvae of the mosquitoes of Iran (Diptera: Culicidae). Zootaxa 2009, 2078, 1–33. [Google Scholar] [CrossRef]
- Wieçel, M. Effects of the Evolution of Intromission on Courtship Complexity and Male and Female Morphology: Water Mites of the Genus Arrenurus (Acari; Hydrachnida) from Europe and North America. Ph.D. Thesis, Adam Mickiewicz University, Poznan, Poland, 2016. [Google Scholar]
- Valdecasas, A.G.; García-Jiménez, R.; Marín, M. Two rare water mite species (Acari, Parasitengona, Hydrachnidia) new for the Iberian Peninsula. Rev. Ibérica Aracnol. 2019, 35, 33–37. [Google Scholar]
- Pešić, V.; Smit, H. Neumania kyrgyzica sp. nov. a new water mite from Kyrgyzstan based on morphological and molecular data (Acari, Hydrachnidia: Unionicolidae). Syst. Appl. Acarol. 2017, 22, 885. [Google Scholar] [CrossRef]
- Mlynarek, J.J.; Knee, W.; Forbes, M.R. Explaining susceptibility and resistance to a multi-host parasite. Evol. Boil. 2013, 41, 115–122. [Google Scholar] [CrossRef]
- Martin, P. Wassermilben (Hydrachnidia, Acari) und Insekten: Ein Überblick über eine selten betrachtete Beziehung. Entomol. Heute 2008, 20, 45–75. [Google Scholar]
- Scorrano, S.; Aglieri, G.; Boero, F.; Dawson, M.N.; Piraino, S. Unmasking Aurelia species in the Mediterranean Sea: An integrative morphometric and molecular approach. Zool. J. Linn. Soc. 2016, 180, 243–267. [Google Scholar] [CrossRef]
- Horreo, J.L.; Ardura, A.; Pola, I.G.; Martinez, J.L.; Garcia-Vazquez, E. Universal primers for species authentication of animal foodstuff in a single polymerase chain reaction. J. Sci. Food Agric. 2012, 93, 354–361. [Google Scholar] [CrossRef] [PubMed]
- Mullen, G.R. Water mites of the subgenus Truncaturus (Arrenuridae, Arrenurus) in North America. Ithaca Agric. Entomol. 1976, 6, 1–35. [Google Scholar]
- Kirkhoff, C.J.; Simmons, T.W.; Hutchinson, M.; Simmons, T.W. Adult mosquitoes parasitized by larval water mites in Pennsylvania. J. Parasitol. 2013, 99, 31–39. [Google Scholar] [CrossRef] [PubMed]
- Dos Santos, E.B.; Favretto, M.A.; Dos Santos Costa, S.G.; Navarro-Silva, M.A. Mites (Acari: Trombidiformes) parasitizing mosquitoes (Diptera: Culicidae) in an Atlantic Forest area in southern Brazil with a new mite genus country record. Exp. Appl. Acarol. 2016, 69, 323–333. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Atwa, A.A.; Bilgrami, A.L.; Al-Saggaf, A.I. Host–parasite interaction and impact of mite infection on mosquito population. Rev. Bras. Entomol. 2017, 61, 101–106. [Google Scholar] [CrossRef]
- Gerson, U.; Smiley, R.L.; Ochoa, R. Mites (Acari) for Pest Control; Blackewell: Malden, MA, USA, 2003. [Google Scholar]






 Round setae;
Round setae;  solenidum;
solenidum;  other setae.
other setae.
 Round setae;
Round setae;  solenidum;
solenidum;  other setae.
other setae.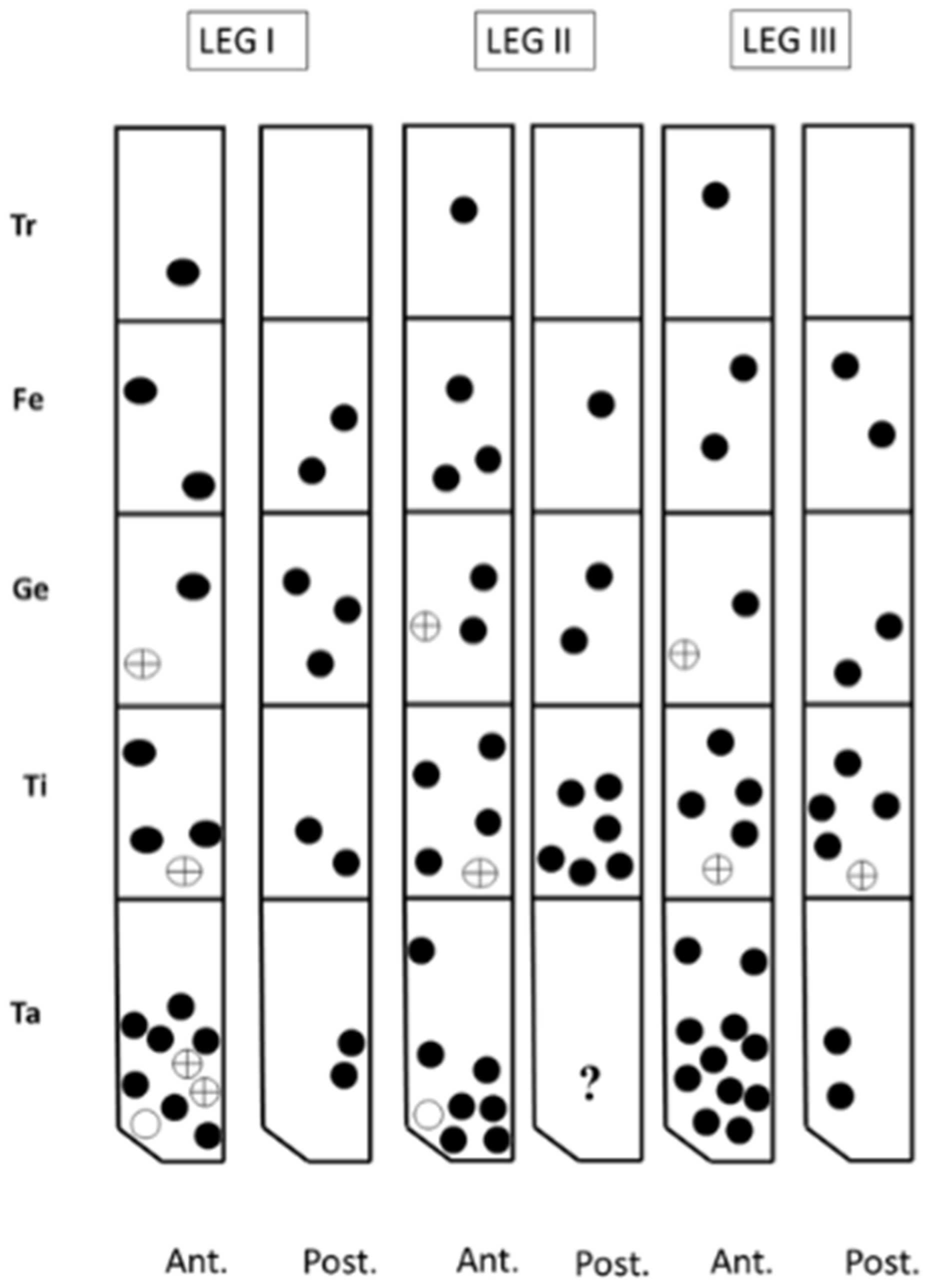





| L1 | L2 | L3 | L4 | L5 | |
|---|---|---|---|---|---|
| Body length | 197 | 182 | 190 | 204 | 200 |
| Body width | 175 | 168 | 168 | 164 | 156 |
| Dorsal plate length | 190 | 186 | 190 | 185 | 185 |
| Dorsal plate width | 171 | 152 | 160 | 148 | 147 |
| CpI medial margin length | 57 | 55 | 61 | 57 | 62 |
| CpII medial margin length | 27 | 30 | 30 | 27 | 26 |
| CpIII medial margin length | 38 | 42 | 44 | 40 | 42 |
| Distance between C1 and CpI median margin | 21 | 19 | 23 | 18 | 20 |
| Distance between C4 and CpIII median margin | 28 | 27 | 24 | 27 | 29 |
| Distance between C1 and C2 | 40 | 36 | 40 | 38 | 39 |
| Excretory pore plate length | 25 | 27 | 26 | 22 | 29 |
| Excretory pore plate width | 32 | 30 | 32 | 30 | 31 |
| Distance between Exp and Expp posterior margin | 11 | 15 | 15 | 12 | 13 |
| PIII length | 27 | 21 | 26 | 31 | 31 |
| Length of PIV claw | 27 | 23 | 14 | 16 | 17 |
| Larvae | Trochanter | Femur | Genu | Tibia | Tarsus | |
|---|---|---|---|---|---|---|
| L1 | leg1 | 25 | 21 | 29 | 42 | 55 |
| leg2 | 30 | 25 | 30 | 46 | 57 | |
| leg3 | 30 | 25 | 30 | 46 | 57 | |
| L2 | leg1 | 23 | 21 | 29 | 40 | 49 |
| leg2 | 23 | 25 | 30 | 44 | 59 | |
| leg3 | 27 | 23 | 25 | 48 | 57 | |
| L3 | leg1 | 30 | 23 | 30 | 38 | 49 |
| leg2 | 30 | 32 | 27 | 42 | 57 | |
| leg3 | 34 | 34 | 27 | 44 | 57 | |
| L4 | leg1 | 29 | 23 | 27 | 44 | 55 |
| leg2 | 21 | 25 | 29 | 46 | 53 | |
| leg3 | 32 | 29 | 29 | 46 | 59 | |
| L5 | leg1 | 24 | 23 | 27 | 40 | 41 |
| leg2 | 29 | 22 | 26 | 45 | 60 | |
| leg3 | 30 | 25 | 30 | 40 | 31 |
| K2P | Adult 1 | Adult 2 | Nymph | Larvae |
|---|---|---|---|---|
| Adult 1 | - | |||
| Adult 2 | 0.006 | - | ||
| Nymph | 0.006 | 0.000 | - | |
| Larvae | 0.000 | 0.006 | 0.006 | - |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alarcón-Elbal, P.M.; García-Jiménez, R.; Peláez, M.L.; Horreo, J.L.; Valdecasas, A.G. Molecular Correlation between Larval, Deutonymph and Adult Stages of the Water Mite Arrenurus (Micruracarus) Novus. Life 2020, 10, 108. https://doi.org/10.3390/life10070108
Alarcón-Elbal PM, García-Jiménez R, Peláez ML, Horreo JL, Valdecasas AG. Molecular Correlation between Larval, Deutonymph and Adult Stages of the Water Mite Arrenurus (Micruracarus) Novus. Life. 2020; 10(7):108. https://doi.org/10.3390/life10070108
Chicago/Turabian StyleAlarcón-Elbal, Pedro María, Ricardo García-Jiménez, María Luisa Peláez, Jose Luis Horreo, and Antonio G. Valdecasas. 2020. "Molecular Correlation between Larval, Deutonymph and Adult Stages of the Water Mite Arrenurus (Micruracarus) Novus" Life 10, no. 7: 108. https://doi.org/10.3390/life10070108




