Methods to Assess Fat Mass in Infants and Young Children: A Comparative Study Using Skinfold Thickness and Air-Displacement Plethysmography
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Procedures
2.2.1. Anthropometrics
2.2.2. Air-Displacement Plethysmography (ADP)
2.3. Statistical Analyses
3. Results
3.1. Subject Characteristics
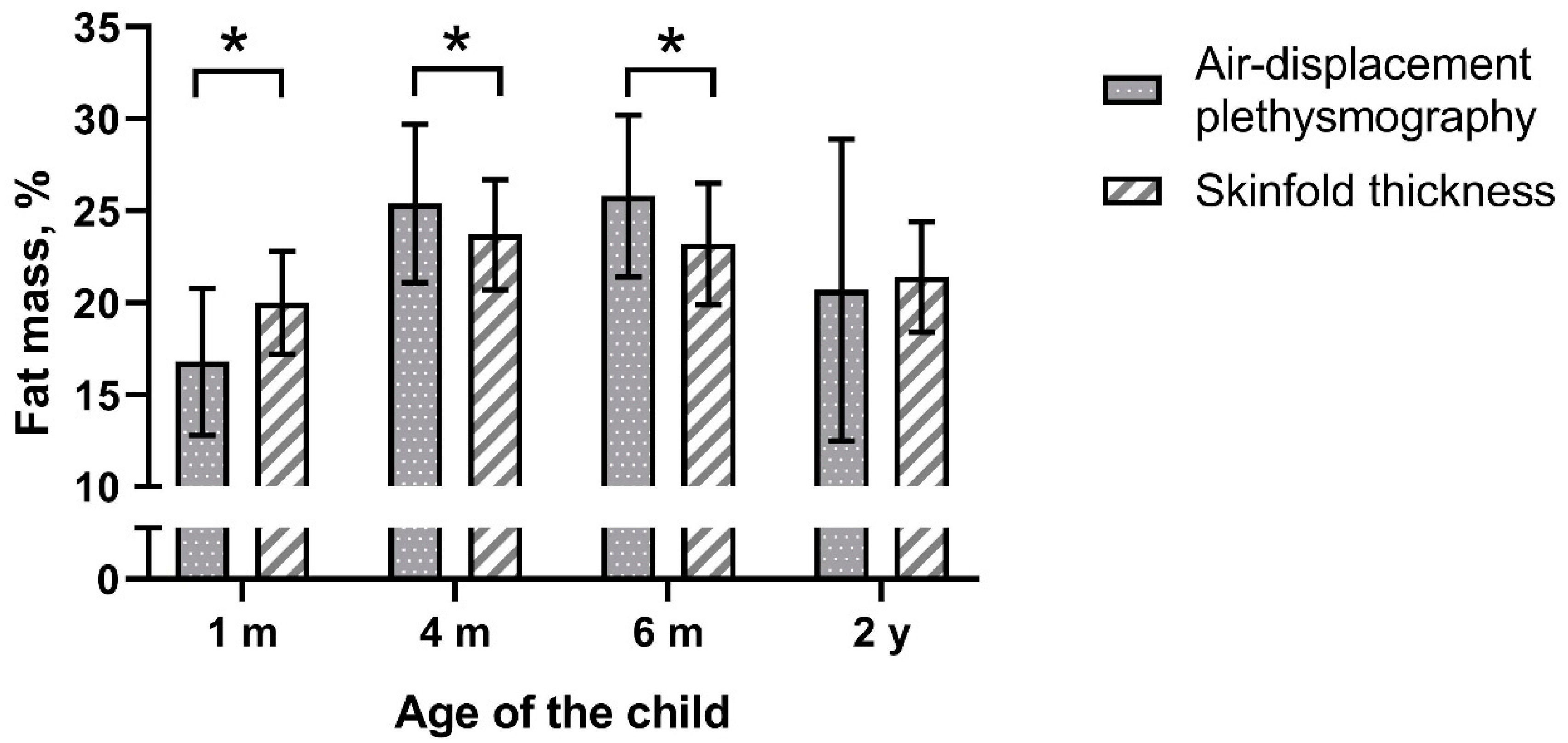
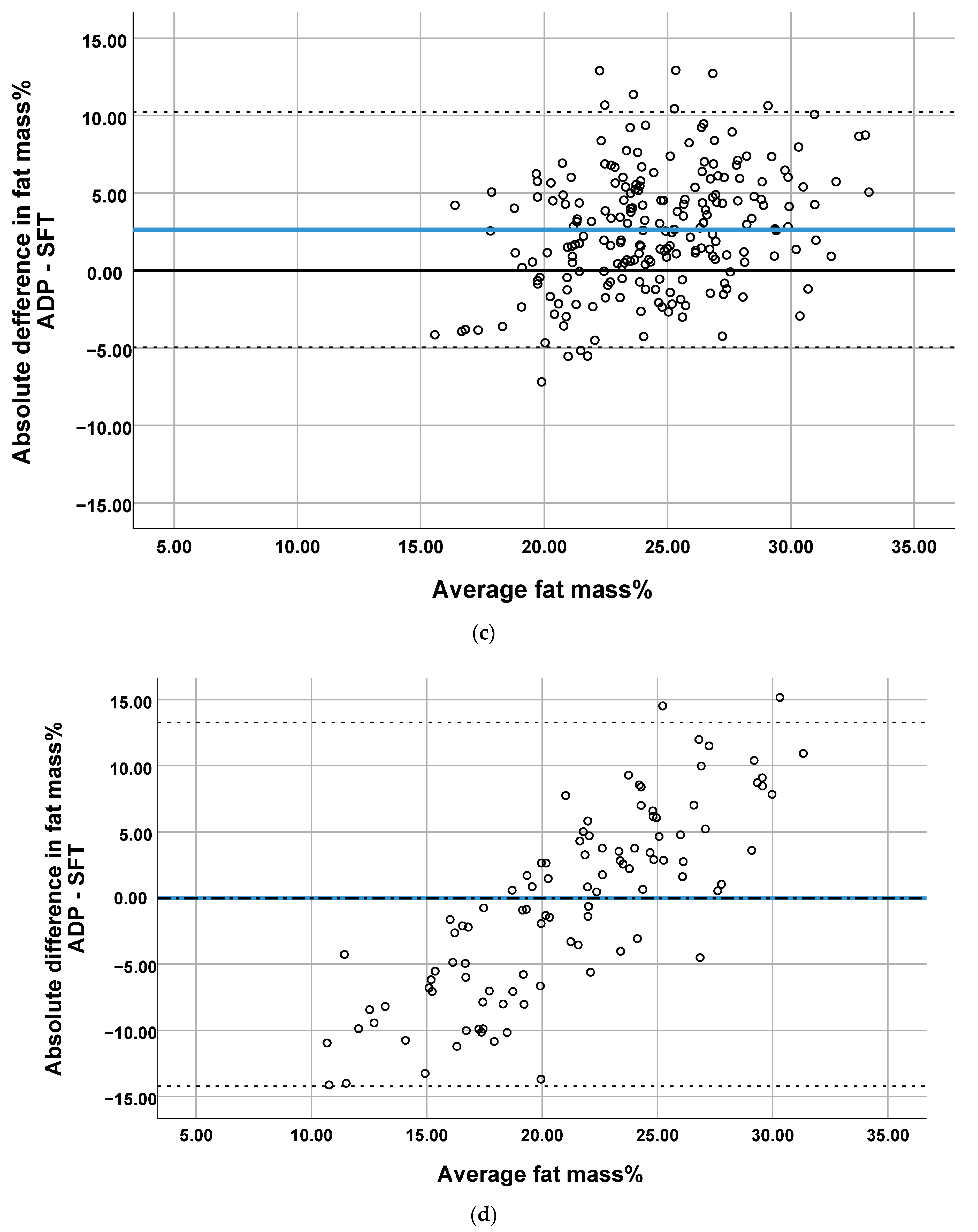
3.2. Comparison of the Methods
3.3. Inter-Rater Reliability Tests–Skinfold Thickness Measurements
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Franks, P.W.; Hanson, R.L.; Knowler, W.C.; Sievers, M.L.; Bennett, P.H.; Looker, H.C. Childhood obesity, other cardiovascular risk factors, and premature death. N. Engl. J. Med. 2010, 362, 485–493. [Google Scholar] [CrossRef] [PubMed]
- Hannon, T.S.; Rao, G.; Arslanian, S.A. Childhood obesity and type 2 diabetes mellitus. Pediatrics 2005, 116, 473–480. [Google Scholar] [CrossRef] [PubMed]
- Hayman, L.L.; Meininger, J.C.; Daniels, S.R.; McCrindle, B.W.; Helden, L.; Ross, J.; Dennison, B.A.; Steinberger, J.; Williams, C.L.; American Heart Association Committee on Atherosclerosis; et al. Primary prevention of cardiovascular disease in nursing practice: Focus on children and youth: A scientific statement from the American Heart Association Committee on Atherosclerosis, Hypertension, and Obesity in Youth of the Council on Cardiovascular Disease in the Young, Council on Cardiovascular Nursing, Council on Epidemiology and Prevention, and Council on Nutrition, Physical Activity, and Metabolism. Circulation 2007, 116, 344–357. [Google Scholar] [PubMed]
- Lobstein, T.; Baur, L.; Uauy, R.; TaskForce, I.I.O. Obesity in children and young people: A crisis in public health. Obes. Rev. 2004, 5 (Suppl. 1), 4–104. [Google Scholar] [CrossRef]
- Magarey, A.M.; Daniels, L.A.; Boulton, T.J.; Cockington, R.A. Predicting obesity in early adulthood from childhood and parental obesity. Int. J. Obes. Relat. Metab. Disord. 2003, 27, 505–513. [Google Scholar] [CrossRef]
- Steinberger, J.; Daniels, S.R.; American Heart Association Atherosclerosis; Hypertension, and Obesity in the Young Committee (Council on Cardiovascular Disease in the Young); American Heart Association Diabetes Committee (Council on Nutrition, Physical Activity, and Metabolism). Obesity, insulin resistance, diabetes, and cardiovascular risk in children: An American Heart Association scientific statement from the Atherosclerosis, Hypertension, and Obesity in the Young Committee (Council on Cardiovascular Disease in the Young) and the Diabetes Committee (Council on Nutrition, Physical Activity, and Metabolism). Circulation 2003, 107, 1448–1453. [Google Scholar]
- Chiarelli, F.; Mohn, A. Early diagnosis of metabolic syndrome in children. Lancet Child Adolesc. Health 2017, 1, 86–88. [Google Scholar] [CrossRef]
- Leunissen, R.W.; Kerkhof, G.F.; Stijnen, T.; Hokken-Koelega, A. Timing and tempo of first-year rapid growth in relation to cardiovascular and metabolic risk profile in early adulthood. JAMA 2009, 301, 2234–2242. [Google Scholar] [CrossRef]
- De Fluiter, K.S.; van Beijsterveldt, I.; Breij, L.M.; Acton, D.; Hokken-Koelega, A.C.S. Association Between Fat Mass in Early Life and Later Fat Mass Trajectories. JAMA Pediatr. 2020, 174, 1141–1148. [Google Scholar] [CrossRef]
- Rundle, A.G.; Factor-Litvak, P.; Suglia, S.F.; Susser, E.S.; Kezios, K.L.; Lovasi, G.S.; Cirillo, P.M.; Cohn, B.A.; Link, B.G. Tracking of Obesity in Childhood into Adulthood: Effects on Body Mass Index and Fat Mass Index at Age 50. Child Obes. 2020, 16, 226–233. [Google Scholar] [CrossRef]
- Ronque, E.R.V.; Werneck, A.O.; Bueno, M.R.O.; Cyrino, E.S.; Stanganelli, L.C.R.; Arruda, M. Tracking of body adiposity indicators from childhood to adolescence: Mediation by BMI. PLoS ONE 2018, 13, e0191908. [Google Scholar] [CrossRef] [PubMed]
- Sainz, R.D.; Urlando, A. Evaluation of a new pediatric air-displacement plethysmograph for body-composition assessment by means of chemical analysis of bovine tissue phantoms. Am. J. Clin. Nutr. 2003, 77, 364–370. [Google Scholar] [CrossRef] [PubMed]
- Yao, M.; Nommsen-Rivers, L.; Dewey, K.; Urlando, A. Preliminary evaluation of a new pediatric air displacement plethysmograph for body composition assessment in infants. Acta Diabetol. 2003, 40 (Suppl. 1), S55–S58. [Google Scholar] [CrossRef]
- Urlando, A.; Dempster, P.; Aitkens, S. A new air displacement plethysmograph for the measurement of body composition in infants. Pediatr. Res. 2003, 53, 486–492. [Google Scholar] [CrossRef] [PubMed]
- Ma, G.; Yao, M.; Liu, Y.; Lin, A.; Zou, H.; Urlando, A.; Wong, W.W.; Nommsen-Rivers, L.; Dewey, K.G. Validation of a new pediatric air-displacement plethysmograph for assessing body composition in infants. Am. J. Clin. Nutr. 2004, 79, 653–660. [Google Scholar] [CrossRef]
- Fields, D.A.; Allison, D.B. Air-displacement plethysmography pediatric option in 2-6 years old using the four-compartment model as a criterion method. Obesity (Silver Spring) 2012, 20, 1732–1737. [Google Scholar] [CrossRef] [PubMed]
- Ellis, K.J.; Yao, M.; Shypailo, R.J.; Urlando, A.; Wong, W.W.; Heird, W.C. Body-composition assessment in infancy: Air-displacement plethysmography compared with a reference 4-compartment model. Am. J. Clin. Nutr. 2007, 85, 90–95. [Google Scholar] [CrossRef]
- Cauble, J.S.; Dewi, M.; Hull, H.R. Validity of anthropometric equations to estimate infant fat mass at birth and in early infancy. BMC Pediatr. 2017, 17, 88. [Google Scholar] [CrossRef]
- Josefson, J.L.; Nodzenski, M.; Talbot, O.; Scholtens, D.M.; Catalano, P. Fat mass estimation in neonates: Anthropometric models compared with air displacement plethysmography. Br. J. Nutr. 2019, 121, 285–290. [Google Scholar] [CrossRef]
- Elberg, J.; McDuffie, J.R.; Sebring, N.G.; Salaita, C.; Keil, M.; Robotham, D.; Reynolds, J.C.; Yanovski, J.A. Comparison of methods to assess change in children’s body composition. Am. J. Clin. Nutr. 2004, 80, 64–69. [Google Scholar] [CrossRef]
- Henriksson, H.; Eriksson, B.; Forsum, E.; Flinke, E.; Henriksson, P.; Lof, M. Longitudinal assessment of body composition in healthy Swedish children from 1 week until 4 years of age. Eur. J. Clin. Nutr. 2017. [Google Scholar] [CrossRef] [PubMed]
- Kouwenhoven, S.M.P.; Antl, N.; Finken, M.J.J.; Twisk, J.W.R.; van der Beek, E.M.; Abrahamse-Berkeveld, M.; van de Heijning, B.J.M.; Schierbeek, H.; Holdt, L.M.; van Goudoever, J.B.; et al. A modified low-protein infant formula supports adequate growth in healthy, term infants: A randomized, double-blind, equivalence trial. Am. J. Clin. Nutr. 2019, 111, 962–974. [Google Scholar] [CrossRef] [PubMed]
- Weststrate, J.A.; Deurenberg, P. Body composition in children: Proposal for a method for calculating body fat percentage from total body density or skinfold-thickness measurements. Am. J. Clin. Nutr. 1989, 50, 1104–1115. [Google Scholar] [CrossRef]
- Schmelzle, H.R.; Fusch, C. Body fat in neonates and young infants: Validation of skinfold thickness versus dual-energy X-ray absorptiometry. Am. J. Clin. Nutr. 2002, 76, 1096–1100. [Google Scholar] [CrossRef]
- Fomon, S.J.; Haschke, F.; Ziegler, E.E.; Nelson, S.E. Body composition of reference children from birth to age 10 years. Am. J. Clin. Nutr. 1982, 35 (Suppl. 5), 1169–1175. [Google Scholar] [CrossRef] [PubMed]
- Fidanza, F.; Keys, A.; Anderson, J.T. Density of body fat in man and other mammals. J. Appl. Physiol. 1953, 6, 252–256. [Google Scholar] [CrossRef]
- Fields, D.A.; Higgins, P.B.; Hunter, G.R. Assessment of body composition by air-displacement plethysmography: Influence of body temperature and moisture. Dyn. Med. 2004, 3, 3. [Google Scholar] [CrossRef]
- Higgins, P.B.; Fields, D.A.; Hunter, G.R.; Gower, B.A. Effect of scalp and facial hair on air displacement plethysmography estimates of percentage of body fat. Obes. Res. 2001, 9, 326–330. [Google Scholar] [CrossRef]
- Bland, J.M.; Altman, D.G. Statistical methods for assessing agreement between two methods of clinical measurement. Lancet 1986, 1, 307–310. [Google Scholar] [CrossRef]
- Butte, N.F.; Hopkinson, J.M.; Wong, W.W.; Smith, E.O.; Ellis, K.J. Body composition during the first 2 years of life: An updated reference. Pediatr. Res. 2000, 47, 578–585. [Google Scholar] [CrossRef]
- Slaughter, M.H.; Lohman, T.G.; Boileau, R.A.; Horswill, C.A.; Stillman, R.J.; Van Loan, M.D.; Bemben, D.A. Skinfold equations for estimation of body fatness in children and youth. Hum. Biol. 1988, 60, 709–723. [Google Scholar] [PubMed]
- Barbour, L.A.; Hernandez, T.L.; Reynolds, R.M.; Reece, M.S.; Chartier-Logan, C.; Anderson, M.K.; Kelly, T.; Friedman, J.E.; Van Pelt, R.E. Striking differences in estimates of infant adiposity by new and old DXA software, PEAPOD and skin-folds at 2 weeks and 1 year of life. Pediatr. Obes. 2016, 11, 264–271. [Google Scholar] [CrossRef] [PubMed]
- Lingwood, B.E.; Storm van Leeuwen, A.M.; Carberry, A.E.; Fitzgerald, E.C.; Callaway, L.K.; Colditz, P.B.; Ward, L.C. Prediction of fat-free mass and percentage of body fat in neonates using bioelectrical impedance analysis and anthropometric measures: Validation against the PEA POD. Br. J. Nutr. 2012, 107, 1545–1552. [Google Scholar] [CrossRef]
- Catalano, P.M.; Thomas, A.J.; Avallone, D.A.; Amini, S.B. Anthropometric estimation of neonatal body composition. Am. J. Obstet. Gynecol. 1995, 173, 1176–1181. [Google Scholar] [CrossRef]
- Yumani, D.F.J.; de Jongh, D.; Lafeber, H.N.; van Weissenbruch, M.M. A comparative study using dual-energy X-ray absorptiometry, air displacement plethysmography, and skinfolds to assess fat mass in preterms at term equivalent age. Eur. J. Pediatr. 2020. [Google Scholar] [CrossRef]
- De Bruin, N.C.; van Velthoven, K.A.; Stijnen, T.; Juttmann, R.E.; Degenhart, H.J.; Visser, H.K. Body fat and fat-free mass in infants: New and classic anthropometric indexes and prediction equations compared with total-body electrical conductivity. Am. J. Clin. Nutr. 1995, 61, 1195–1205. [Google Scholar] [CrossRef]

| 1 Month | 4 Months | 6 Months | 2 Years | |
|---|---|---|---|---|
| n = 245 | n = 238 | n = 235 | n = 173 | |
| Boys [n (%)] | 113 (46) | 112 (47) | 111 (47) | 82 (47) |
| Caucasian [n (%)] | 213 (87) | 208 (87) | 205 (87) | 151 (87) |
| Age (mo) | 1.0 ± 0.3 | 3.9 ± 0.1 | 6.0 ± 0.1 | 24.1 ± 0.5 |
| Body weight (gram) | 4207 ± 536 | 6598 ± 685 | 7692 ± 834 | 12415 ± 1315 |
| Length/height (cm) | 54.5 ± 2.2 | 63.7 ± 2.0 | 67.9 ± 2.2 | 86.8 ± 2.8 |
| Formula-fed a [n (%)] | 178 (73) | 173 (73) | 170 (72) | NA |
| Skinfold thickness measurements | ||||
| n = 242−235 | n = 237−236 | n = 235 | n = 169−168 | |
| ∑SFT (mm) b | 23.2 ± 3.8 | 29.4 ± 5.2 | 29.0 ± 5.6 | 27.3 ± 5.2 |
| n = 235 | n = 235 | n = 235 | n = 167 | |
| Body density (kg/L) | 1.0274 ± 0.0050 | 1.0215 ± 0.0055 | 1.0228 ± 0.0060 | 1.0286 ± 0.0057 |
| Fat mass% | 20.0 ± 2.8 | 23.7 ± 3.0 | 23.2 ± 3.3 | 21.4 ± 3.0 |
| Air-displacement plethysmography | ||||
| n = 231 | n = 230 | n = 219 | n = 103 | |
| Body density | 1.0330 ± 0.0073 | 1.0185 ± 0.0077 | 1.0184 ± 0.0079 | 1.0327 ± 0.0154 |
| Fat mass% | 16.8± 4.0 | 25.4 ± 4.3 | 25.8 ± 4.4 | 20.7 ± 8.2 |
| 1 Month | 4 Months | 6 Months | 2 Years | |
|---|---|---|---|---|
| n | 225 | 229 | 219 | 102 |
| Mean difference (bias) | −3.05 | 1.70 | 2.65 | −0.47 |
| Limits of agreement (CI95%) | 3.12 −9.21 | 8.86 −546 | 10.25 −4.96 | 13.29 −14.23 |
| Difference 1 | −3.15 | 1.70 | 2.63 | −0.67 |
| P1 | <0.001 | <0.001 | <0.001 | 0.112 |
| (CI95%) | (−3.75, −2.54) | (1.10, 2.31) | (2.02, 3.24) | (−1.49, 0.16) |
| ICC 2 | 0.42 | 0.47 | 0.42 | 0.33 |
| 1 month | 4 months | 6 months | 2 years |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kouwenhoven, S.M.P.; Antl, N.; Twisk, J.W.R.; Koletzko, B.V.; Finken, M.J.J.; van Goudoever, J.B. Methods to Assess Fat Mass in Infants and Young Children: A Comparative Study Using Skinfold Thickness and Air-Displacement Plethysmography. Life 2021, 11, 75. https://doi.org/10.3390/life11020075
Kouwenhoven SMP, Antl N, Twisk JWR, Koletzko BV, Finken MJJ, van Goudoever JB. Methods to Assess Fat Mass in Infants and Young Children: A Comparative Study Using Skinfold Thickness and Air-Displacement Plethysmography. Life. 2021; 11(2):75. https://doi.org/10.3390/life11020075
Chicago/Turabian StyleKouwenhoven, Stefanie M. P., Nadja Antl, Jos W. R. Twisk, Berthold V. Koletzko, Martijn J. J. Finken, and Johannes B. van Goudoever. 2021. "Methods to Assess Fat Mass in Infants and Young Children: A Comparative Study Using Skinfold Thickness and Air-Displacement Plethysmography" Life 11, no. 2: 75. https://doi.org/10.3390/life11020075
APA StyleKouwenhoven, S. M. P., Antl, N., Twisk, J. W. R., Koletzko, B. V., Finken, M. J. J., & van Goudoever, J. B. (2021). Methods to Assess Fat Mass in Infants and Young Children: A Comparative Study Using Skinfold Thickness and Air-Displacement Plethysmography. Life, 11(2), 75. https://doi.org/10.3390/life11020075






