Evaluation of the Impact of Different Pain Medication and Proton Pump Inhibitors on the Osteogenic Differentiation Potential of hMSCs Using 99mTc-HDP Labelling
Abstract
:1. Introduction
2. Results
2.1. 99mTc-HDP Labelling
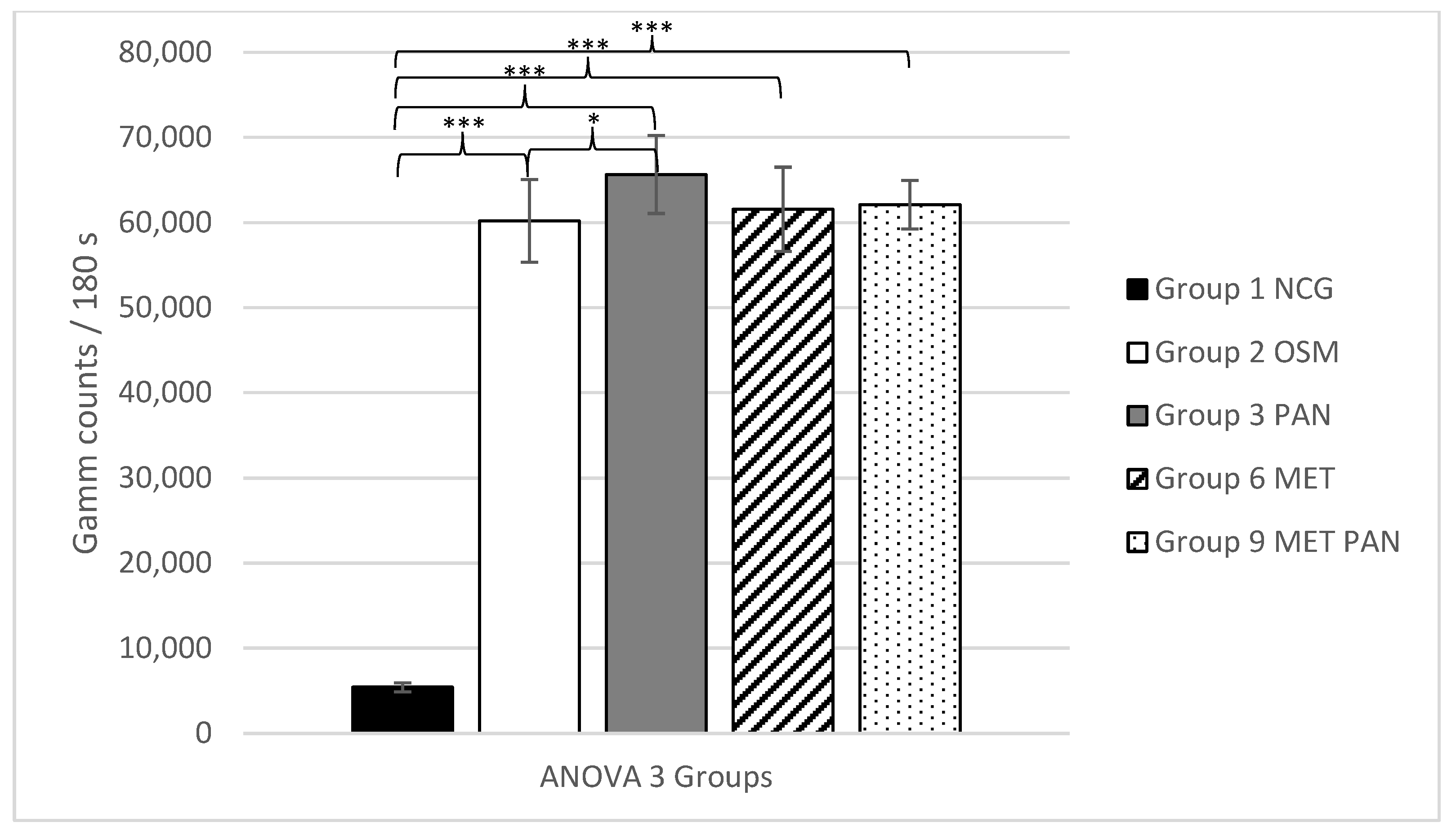
2.2. Statistical Results
3. Discussion
4. Materials and Methods
4.1. Experimental Design at a Glance
- Group 1
- Non-osteogenic negative control group (NCG)
- Group 2
- Osteogenic differentiation media without and drugs (OSM)
- Group 3
- OSM with pantoprazole (PAN)
- Group 4
- OSM with ibuprofen (IBU)
- Group 5
- OSM with diclofenac (DIC)
- Group 6
- OSM with metamizole (MET)
- Group 7
- OSM with ibuprofen and pantoprazole (IBU + PAN)
- Group 8
- OSM with diclofenac and pantoprazole (DIC + PAN)
- Group 9
- OSM with metamizole and pantoprazole (MET + PAN)
- Group 10
- OSM with diclofenac, metamizole and pantoprazole (DIC + MET + PAN)
4.2. hMSC Harvest and Expansion
4.3. Osteogenic Differentiation Assay
4.4. 99mTc-HDP Labelling
4.5. Statistics
4.6. Preparation of Cell Culture Media and Drug Concentrations
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ANOVA | Analysis of variance |
| Bq | Becquerel |
| BM | Bone marrow |
| COX-1 | Cyclooxygenase-1 |
| COX-2 | Cyclooxygenase-2 |
| DIC | diclofenac |
| hMSC | Human mesenchymal stem cells |
| IBU | ibuprofen |
| MET | metamizole |
| MeV | Mega-electron volts |
| MSC | Mesenchymal stem cells |
| NSAID | nonsteroidal anti-inflammatory drugs |
| OSM | Osteogenic treatment group |
| NCG | Non-osteogenic, negative control group |
| OSM | Standard osteogenic media |
| PAN | pantoprazole |
| PEG2 | Prostaglandine 2 |
| PPI | Protone pump inhibitor |
| ROI | Region of interest |
| WHO | World Health Organistaion |
References
- Eden Sebbag, E.; Felten, R.; Sagez, F.; Sibilia1, J.; Devilliers, H.; Arnaud, L. The world-wide burden of musculoskeletal diseases: A systematic analysis of the World Health Organization Burden of Diseases Database. Ann. Rheum. Dis. 2019, 78, 844–848. [Google Scholar] [CrossRef] [PubMed]
- Hsu, J.R.; Mir, H.; Wally, M.K.; Seymour, R.B. Clinical Practice Guidelines for Pain Management in Acute Musculoskeletal Injury. J. Orthop. Trauma. 2019, 33, e158–e182. [Google Scholar] [CrossRef] [Green Version]
- Fikentscher, T.; Grifka, J.; Benditz, A. Perioperative pain therapy in orthopedics. Orthopade 2015, 44, 727–740. [Google Scholar] [CrossRef]
- Chou, R.; Gordon, D.B.; de Leon-Casasola, O.A.; Rosenberg, J.M.; Bickler, S.; Brennan, T.; Carter, T.; Cassidy, C.L.; Chittenden, E.H.; Degenhardt, E.; et al. Management of Postoperative Pain: A Clinical Practice Guideline from the American Pain Society, the American Society of Regional Anesthesia and Pain Medicine, and the American Society of Anesthesiologists’ Committee on Regional Anesthesia, Executive Committee, and Administrative Council. J. Pain 2016, 17, 131–157. [Google Scholar]
- Freys, S.M.; Erlenwein, J.; Koppert, W.; Meißner, W.; Pogatzki-Zahn, E.; Schwenk, W.; Simanski, C. Agreement of the Professional Association of German Anesthesiologists and the Professional Association of German Surgeons for the Organization of Postoperative Pain Therapy for Surgical Patients (revised version 2019). Der Unf. 2019, 90, 648–651. [Google Scholar]
- Hartz, C.; Hauser, W.; Schiltenwolf, M. Long-term Application of Opioids for Non-Tumor Associated Pain--Impact of the S3 Guidelines for the Field of Orthopaedic Surgery and Trauma Surgery. Z. Orthop. Unfall 2016, 154, 134–139. [Google Scholar] [PubMed]
- Montané, E.; Vallano, A.; Aguilera, C.; Vidal, X.; Laporte, J. Analgesics for pain after traumatic or orthopaedic surgery: What is the evidence-a systematic review. Eur. J. Clin. Pharmacol. 2006, 62, 971–988. [Google Scholar] [CrossRef]
- Firoozabadi, R.; Alton, T.; Sagi, H.C. Heterotopic Ossification in Acetabular Fracture Surgery. J. Am. Acad. Orthop. Surg. 2017, 25, 117–124. [Google Scholar]
- Joice, M.; Vasileiadis, G.I.; Amanatullah, D.F. Non-steroidal anti-inflammatory drugs for heterotopic ossification prophylaxis after total hip arthroplasty: A systematic review and meta-analysis. Bone Joint J 2018, 100, 915–922. [Google Scholar] [CrossRef]
- Kan, S.L.; Yang, B.; Ning, G.Z.; Chen, L.X.; Li, Y.L.; Gao, S.J.; Chen, X.Y.; Sun, J.C.; Feng, S.Q. Nonsteroidal Anti-inflammatory Drugs as Prophylaxis for Heterotopic Ossification after Total Hip Arthroplasty: A Systematic Review and Meta-Analysis. Medicine 2015, 94, e828. [Google Scholar] [CrossRef] [PubMed]
- Łęgosz, P.; Otworowski, M.; Sibilska, A.; Starszak, K.; Kotrych, D.; Kwapisz, A.; Synder, M. Heterotopic Ossification: A Challenging Complication of Total Hip Arthroplasty: Risk Factors, Diagnosis, Prophylaxis, and Treatment. Biomed Res. Int. 2019, 2019, 3860142. [Google Scholar] [CrossRef] [Green Version]
- Zhu, X.T.; Chen, L.; Lin, J.H. Selective COX-2 inhibitor versus non-selective COX-2 inhibitor for the prevention of heterotopic ossification after total hip arthroplasty: A meta-analysis. Medicine 2018, 97, e11649. [Google Scholar] [CrossRef]
- Rostom, A.; Dube, C.; A Wells, G.; Tugwell, P.; Welch, V.; Jolicoeur, E.; McGowan, J.; Lanas, A. Prevention of NSAID-induced gastroduodenal ulcers. Cochrane Database Syst. Rev. 2002, CD002296. [Google Scholar] [CrossRef]
- Hoff, M.; Skovlund, E.; Skurtveit, S.; Meyer, H.; Langhammer, A.; Søgaard, A.; Syversen, U.; Forsmo, S.; Abrahamsen, B.; Schei, B. Proton pump inhibitors and fracture risk. The HUNT study, Norway. Osteoporos. Int. 2020, 31, 109–118. [Google Scholar] [CrossRef]
- Thong, B.K.S.; Ima-Nirwana, S.; Chin, K.-Y. Proton Pump Inhibitors and Fracture Risk: A Review of Current Evidence and Mechanisms Involved. Int. J. Environ. Res. Public Health 2019, 16, 1571. [Google Scholar] [CrossRef] [Green Version]
- Costa-Rodrigues, J.; Reis, S.; Teixeira, S.; Lopes, S.; Fernandes, M.H. Dose-dependent inhibitory effects of proton pump inhibitors on human osteoclastic and osteoblastic cell activity. FEBS J. 2013, 280, 5052–5064. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Prause, M.; Seeliger, C.; Unger, M.; van Griensven, M.; Haug, A.T. Pantoprazole increases cell viability and function of primary human osteoblasts in vitro. Injury 2014, 45, 1156–1164. [Google Scholar] [CrossRef] [PubMed]
- Prause, M.; Seeliger, C.; Unger, M.; Balmayor, E.R.; Van Griensven, M.; Haug, A.T. Pantoprazole decreases cell viability and function of human osteoclasts in vitro. Mediators Inflamm. 2015, 2015, 413097. [Google Scholar] [CrossRef] [PubMed]
- Marquez-Lara, A.; Hutchinson, I.D.; Nuñez, F.; Smith, T.L.; Miller, A.N. Nonsteroidal Anti-Inflammatory Drugs and Bone-Healing: A Systematic Review of Research Quality. JBJS Rev. 2016, 4. [Google Scholar] [CrossRef]
- Pountos, I.; Georgouli, T.; Calori, G.M.; Giannoudis, P.V. Do nonsteroidal anti-inflammatory drugs affect bone healing? A critical analysis. Sci. World J. 2012, 2012, 606404. [Google Scholar] [CrossRef] [Green Version]
- Grossner, T.; Gotterbarm, T.; Gerbaudo, V.H.; Haberkorn, U.; Spector, M. (99m)Tc-Methyl-Diphosphonate Binding to Mineral Deposits in Cultures of Marrow-Derived Mesenchymal Stem Cells in Osteogenic Medium. Tissue Eng. Part C Methods 2019, 25, 49–57. [Google Scholar] [CrossRef]
- Abukawa, H.; Phelps, M.; Jackson, P.; Smith, R.M.; Vacanti, J.P.; Kaban, L.B.; Troulis, M.J. Effect of ibuprofen on osteoblast differentiation of porcine bone marrow-derived progenitor cells. J. Oral Maxillofac. Surg. 2009, 67, 2412–2417. [Google Scholar] [CrossRef]
- Pountos, I.; Giannoudis, P.V.; Jones, E.; English, A.; Churchman, S.; Field, S.; Ponchel, F.; Bird, H.; Emery, P.; McGonagle, D. NSAIDS inhibit in vitro MSC chondrogenesis but not osteogenesis: Implications for mechanism of bone formation inhibition in man. J. Cell. Mol. Med. 2011, 15, 525–534. [Google Scholar] [CrossRef] [Green Version]
- Yoon, D.S.; Yoo, J.H.; Kim, Y.H.; Paik, S.; Han, C.D.; Lee, J.W. The effects of COX-2 inhibitor during osteogenic differentiation of bone marrow-derived human mesenchymal stem cells. Stem. Cells Dev. 2010, 19, 1523–1533. [Google Scholar] [CrossRef]
- Almaawi, A.; Wang, H.T.; Ciobanu, O.; Rowas, S.A.; Rampersad, S.; Antoniou, J.; Mwale, F. Effect of acetaminophen and nonsteroidal anti-inflammatory drugs on gene expression of mesenchymal stem cells. Tissue Eng. Part A 2013, 19, 1039–1046. [Google Scholar] [CrossRef]
- Oh, N.; Kim, S.; Hosoya, K.; Okumura, M. Compensatory cellular reactions to nonsteroidal anti-inflammatory drugs on osteogenic differentiation in canine bone marrow-derived mesenchymal stem cells. J. Vet. Med. Sci. 2014, 76, 629–636. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pytlik, M.; Cegieła, U.; Nowińska, B.; Folwarczna, J.; Sliwiński, L.; Kaczmarczyk-Sedlak, I.; Bolek, D.; Korzeniowska, H. Bone remodeling after administration of proton pump (H+/K+-ATPase) inhibitors and alendronate in ovariectomized rats. Acta Pol. Pharm. 2012, 69, 113–120. [Google Scholar] [PubMed]
- Joo, M.K.; Park, J.-J.; Lee, B.J.; Kim, J.H.; Yeon, J.E.; Kim, J.S.; Byun, K.S.; Bak, Y.-T. The effect of a proton pump inhibitor on bone metabolism in ovariectomized rats. Mol. Med. Rep. 2013, 7, 1267–1272. [Google Scholar] [CrossRef] [Green Version]
- Grossner, T.L.; Haberkorn, U.; Gotterbarm, T. (99m)Tc-Hydroxydiphosphonate quantification of extracellular matrix mineralization in 3D human mesenchymal stem cell cultures. Bone Joint Res. 2019, 8, 333–341. [Google Scholar] [CrossRef] [PubMed]
- Jaiswal, N.; Haynesworth, S.E.; Caplan, A.I.; Bruder, S.P. Osteogenic differentiation of purified, culture-expanded human mesenchymal stem cells in vitro. J. Cell. Biochem. 1997, 64, 295–312. [Google Scholar] [CrossRef]
- Takeda_GmbH. Fachinformation für Pantozol® 40 mg Magensaftresistente Tabletten. 2019. Available online: https://www.fachinfo.de/suche/fi/006633 (accessed on 5 May 2020).
- Sanofi-Aventis_GmbH. Fachinformation für Novalgin Tropfen. 2019. Available online: https://www.fachinfo.de/suche/fi/014710 (accessed on 5 May 2020).



| Group | N | Mean | Minimum | Maximum | Standard Deviation |
|---|---|---|---|---|---|
| Group_1_NCG | 12 | 328 | 2 | 1170 | 322 |
| Group_2_OSM | 12 | 54,880 | 46,194 | 60,107 | 5061 |
| Group_3_PAN | 12 | 60,333 | 46,627 | 64,402 | 4791 |
| Group_4_IBU | 12 | 59,124 | 49,896 | 63,246 | 4403 |
| Group_5_DIC | 12 | 61,048 | 51,602 | 65,606 | 3939 |
| Group_6_MET | 12 | 56,246 | 45,070 | 60,775 | 5159 |
| Group_7_IBU_PAN | 12 | 58,624 | 50,548 | 62,402 | 3859 |
| Group_8_DIC_PAN | 12 | 56,694 | 43,372 | 64,111 | 5349 |
| Group_9_MET_PAN | 12 | 56,778 | 50,133 | 60,326 | 2972 |
| Group_10_DIC_MET_PAN | 12 | 55,940 | 47,865 | 61,387 | 4562 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Grossner, T.; Haberkorn, U.; Gotterbarm, T. Evaluation of the Impact of Different Pain Medication and Proton Pump Inhibitors on the Osteogenic Differentiation Potential of hMSCs Using 99mTc-HDP Labelling. Life 2021, 11, 339. https://doi.org/10.3390/life11040339
Grossner T, Haberkorn U, Gotterbarm T. Evaluation of the Impact of Different Pain Medication and Proton Pump Inhibitors on the Osteogenic Differentiation Potential of hMSCs Using 99mTc-HDP Labelling. Life. 2021; 11(4):339. https://doi.org/10.3390/life11040339
Chicago/Turabian StyleGrossner, Tobias, Uwe Haberkorn, and Tobias Gotterbarm. 2021. "Evaluation of the Impact of Different Pain Medication and Proton Pump Inhibitors on the Osteogenic Differentiation Potential of hMSCs Using 99mTc-HDP Labelling" Life 11, no. 4: 339. https://doi.org/10.3390/life11040339






