Neutrophil Recruitment and Participation in Severe Diseases Caused by Flavivirus Infection
Abstract
1. Flaviviruses
2. Neutrophils and Their Effector Functions
3. Participation of Neutrophils in Immune Responses against Viruses
4. Participation of Neutrophils and Neutrophil-Associated Molecules in Flaviviral Diseases
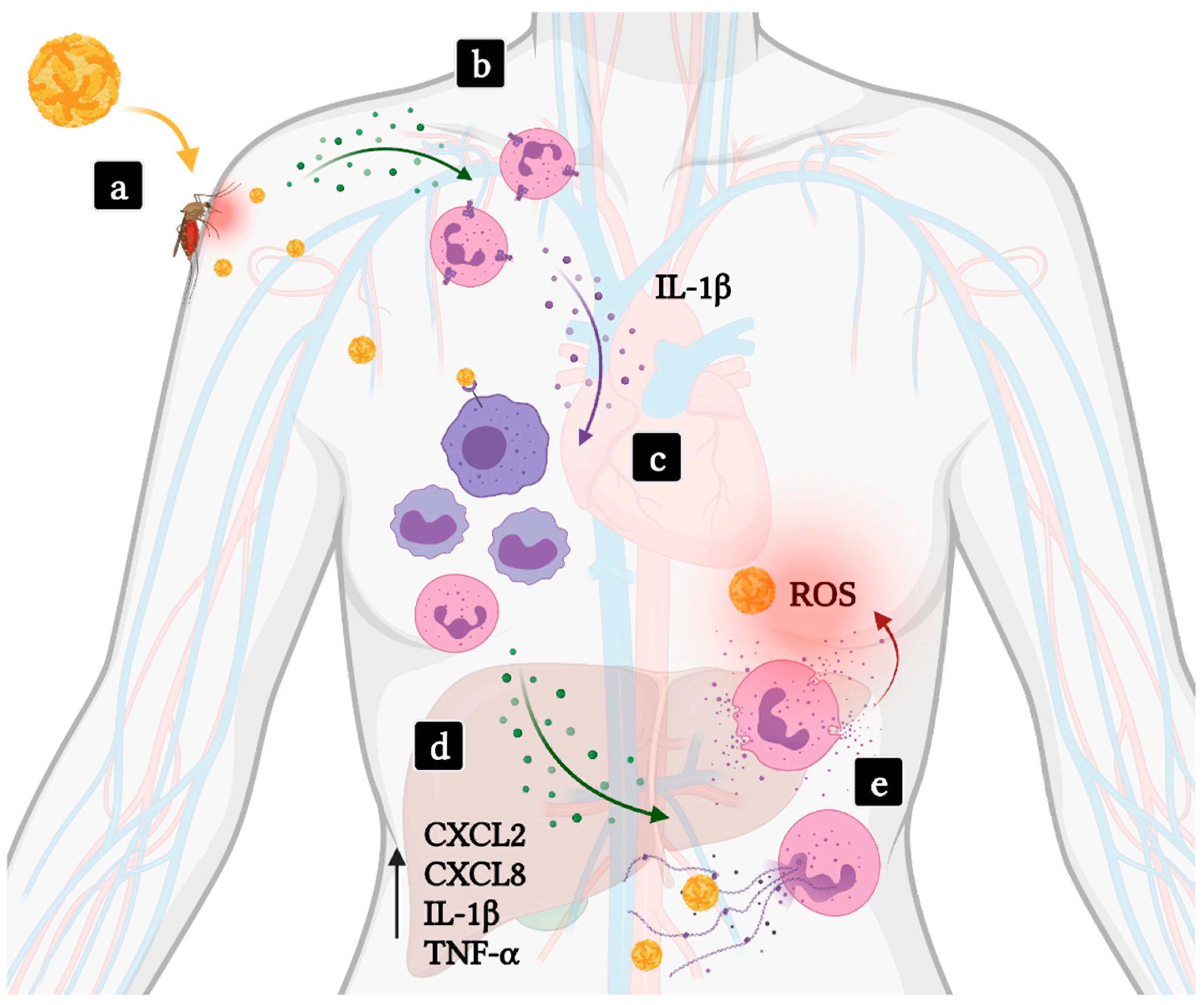
4.1. Hemorrhagic Fevers
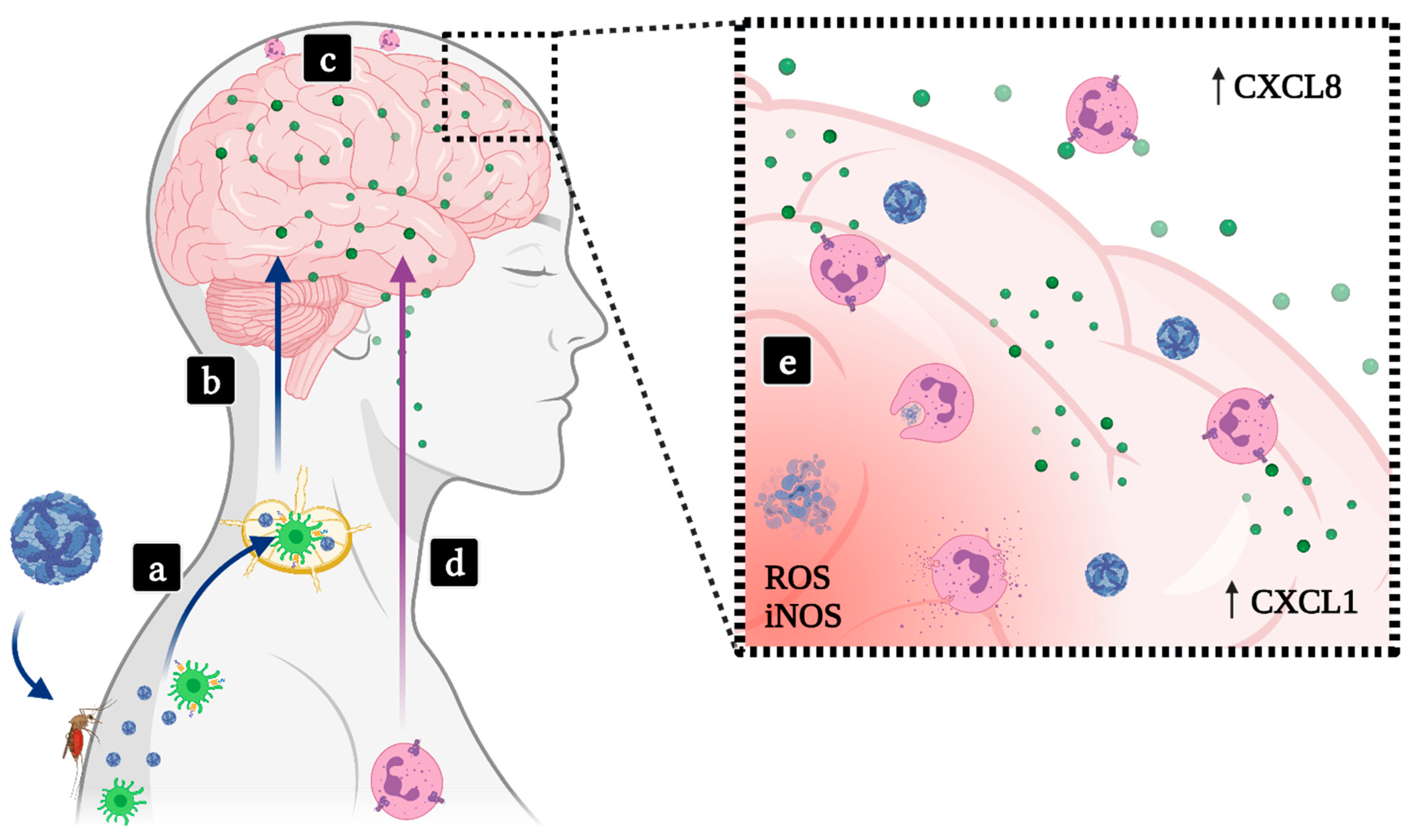
4.2. Encephalitis
4.2.1. SLEV
4.2.2. JEV
4.2.3. WNV
4.2.4. MVEV
4.2.5. TBEV
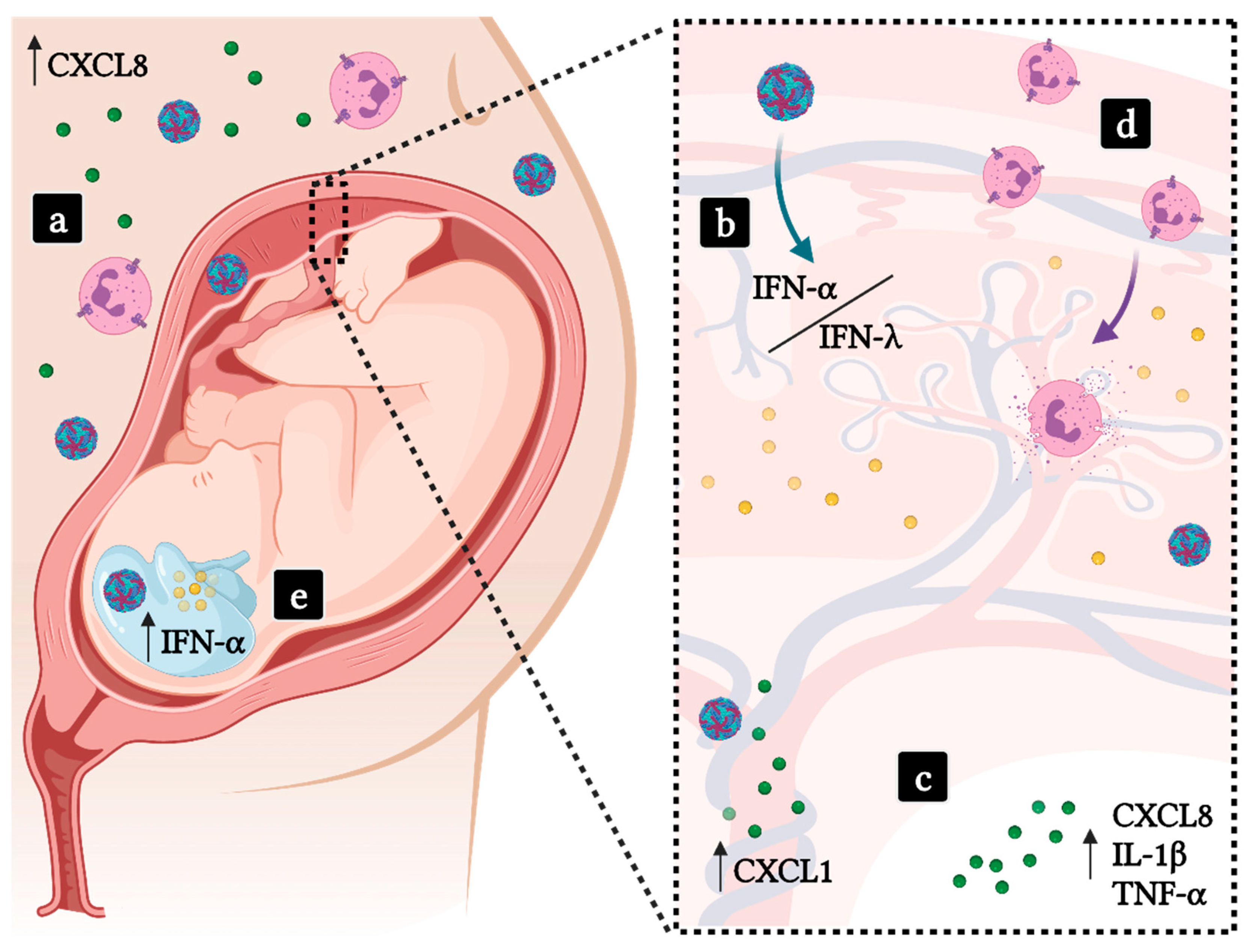
4.3. Implications in Pregnancy
5. Final Considerations
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Marques, R.E.; Guabiraba, R.; Cisalpino, D.; Teixeira, M.M.; Souza, D.G. Dengue. Colloquium Series on Integrated Systems Physiology: From Molecule to Function; Morgan & Claypool Life Sciences: San Rafael, CA, USA, 2014; Volume 6, pp. 1–104. [Google Scholar] [CrossRef]
- Rothman, A.L. Immunity to Dengue Virus: A Tale of Original Antigenic Sin and Tropical Cytokine Storms. Nat. Rev. Immunol. 2011, 11, 532–543. [Google Scholar] [CrossRef] [PubMed]
- Rosa, R.; Costa, E.A.; Marques, R.E.; Oliveira, T.S.; Furtini, R.; Bomfim, M.R.Q.; Teixeira, M.M.; Paixão, T.A.; Santos, R.L. Isolation of Saint Louis Encephalitis Virus from a Horse with Neurological Disease in Brazil. PLoS Negl. Trop. Dis. 2013, 7, e2537. [Google Scholar] [CrossRef]
- Ribeiro, L.S.; Marques, R.E.; de Jesus, A.M.R.; de Almeida, R.P.; Teixeira, M.M. Zika Crisis in Brazil: Challenges in Research and Development. Curr. Opin. Virol. 2016, 18, 76–81. [Google Scholar] [CrossRef]
- Zuchi, N.; da Silva Heinen, L.B.; dos Santos, M.A.M.; Pereira, F.C.; Slhessarenko, R.D. Molecular Detection of Mayaro Virus during a Dengue Outbreak in the State of Mato Grosso, Central-West Brazil. Mem. Inst. Oswaldo Cruz 2014, 109, 820–823. [Google Scholar] [CrossRef] [PubMed]
- Tamba, M.; Bonilauri, P.; Bellini, R.; Calzolari, M.; Albieri, A.; Sambri, V.; Dottori, M.; Angelini, P. Detection of Usutu Virus within a West Nile Virus Surveillance Program in Northern Italy. Vector Borne Zoonotic Dis. 2011, 11, 551–557. [Google Scholar] [CrossRef] [PubMed]
- Pierson, T.C.; Diamond, M.S. The Continued Threat of Emerging Flaviviruses. Nat. Microbiol. 2020, 5, 796–812. [Google Scholar] [CrossRef]
- Rocha, R.F.; Del Sarto, J.L.; Marques, R.E.; Costa, V.V.; Teixeira, M.M. Host Target-Based Approaches against Arboviral Diseases. Biol. Chem. 2018, 399, 203–217. [Google Scholar] [CrossRef]
- Kantari, C.; Pederzoli-Ribeil, M.; Witko-Sarsat, V. The Role of Neutrophils and Monocytes in Innate Immunity. Contrib. Microbiol. 2008, 15, 118–146. [Google Scholar] [CrossRef]
- Németh, T.; Sperandio, M.; Mócsai, A. Neutrophils as Emerging Therapeutic Targets. Nat. Rev. Drug Discov. 2020, 19, 253–275. [Google Scholar] [CrossRef]
- Németh, T.; Mócsai, A. Feedback Amplification of Neutrophil Function. Trends Immunol. 2016, 37, 412–424. [Google Scholar] [CrossRef]
- Lämmermann, T.; Afonso, P.V.; Angermann, B.R.; Wang, J.M.; Kastenmüller, W.; Parent, C.A.; Germain, R.N. Neutrophil Swarms Require LTB4 and Integrins at Sites of Cell Death In Vivo. Nature 2013, 498, 371–375. [Google Scholar] [CrossRef]
- McDonald, B.; Kubes, P. Cellular and Molecular Choreography of Neutrophil Recruitment to Sites of Sterile Inflammation. J. Mol. Med. 2011, 89, 1079–1088. [Google Scholar] [CrossRef]
- Futosi, K.; Fodor, S.; Mócsai, A. Reprint of Neutrophil Cell Surface Receptors and Their Intracellular Signal Transduction Pathways. Int. Immunopharmacol. 2013, 17, 1185–1197. [Google Scholar] [CrossRef] [PubMed]
- Petri, B.; Sanz, M.-J. Neutrophil Chemotaxis. Cell Tissue Res. 2018, 371, 425–436. [Google Scholar] [CrossRef] [PubMed]
- Zlotnik, A.; Yoshie, O. The Chemokine Superfamily Revisited. Immunity 2012, 36, 705–716. [Google Scholar] [CrossRef]
- Plagge, M.; Laskay, T. Early Production of the Neutrophil-Derived Lipid Mediators LTB4 and LXA4 Is Modulated by Intracellular Infection with Leishmania major. BioMed Res. Int. 2017, 2017, 2014583. [Google Scholar] [CrossRef] [PubMed]
- Tecchio, C.; Micheletti, A.; Cassatella, M.A. Neutrophil-Derived Cytokines: Facts beyond Expression. Front. Immunol. 2014, 5, 508. [Google Scholar] [CrossRef]
- Michalec, L.; Choudhury, B.K.; Postlethwait, E.; Wild, J.S.; Alam, R.; Lett-Brown, M.; Sur, S. CCL7 and CXCL10 Orchestrate Oxidative Stress-Induced Neutrophilic Lung Inflammation. J. Immunol. 2002, 168, 846–852. [Google Scholar] [CrossRef]
- Dupré-Crochet, S.; Erard, M.; Nüβe, O. ROS Production in Phagocytes: Why, When, and Where? J. Leukoc. Biol. 2013, 94, 657–670. [Google Scholar] [CrossRef]
- Cowland, J.B.; Borregaard, N. Granulopoiesis and Granules of Human Neutrophils. Immunol. Rev. 2016, 273, 11–28. [Google Scholar] [CrossRef]
- Parmley, R.T.; Rice, W.G.; Kinkade, J.M., Jr.; Gilbert, C.; Barton, J.C. Peroxidase-Containing Microgranules in Human Neutrophils: Physical, Morphological, Cytochemical, and Secretory Properties. Blood 1987, 70, 1630–1638. [Google Scholar] [CrossRef]
- Rice, W.G.; Kinkade, J.M., Jr.; Parmley, R.T. High Resolution of Heterogeneity among Human Neutrophil Granules: Physical, Biochemical, and Ultrastructural Properties of Isolated Fractions. Blood 1986, 68, 541–555. [Google Scholar] [CrossRef]
- Wong, S.H.; Francis, N.; Chahal, H.; Raza, K.; Salmon, M.; Scheel-Toellner, D.; Lord, J.M. Lactoferrin Is a Survival Factor for Neutrophils in Rheumatoid Synovial Fluid. Rheumatology 2009, 48, 39–44. [Google Scholar] [CrossRef]
- Oseas, R.; Yang, H.H.; Baehner, R.L.; Boxer, L.A. Lactoferrin: A Promoter of Polymorphonuclear Leukocyte Adhesiveness. Blood 1981, 57, 939–945. [Google Scholar] [CrossRef]
- Delclaux, C.; Delacourt, C.; D’Ortho, M.P.; Boyer, V.; Lafuma, C.; Harf, A. Role of Gelatinase B and Elastase in Human Polymorphonuclear Neutrophil Migration across Basement Membrane. Am. J. Respir. Cell Mol. Biol. 1996, 14, 288–295. [Google Scholar] [CrossRef] [PubMed]
- Miller, L.J.; Bainton, D.F.; Borregaard, N.; Springer, T.A. Stimulated Mobilization of Monocyte Mac-1 and p150,95 Adhesion Proteins from an Intracellular Vesicular Compartment to the Cell Surface. J. Clin. Investig. 1987, 80, 535–544. [Google Scholar] [CrossRef]
- Brinkmann, V.; Reichard, U.; Goosmann, C.; Fauler, B.; Uhlemann, Y.; Weiss, D.S.; Weinrauch, Y.; Zychlinsky, A. Neutrophil Extracellular Traps Kill Bacteria. Science 2004, 303, 1532–1535. [Google Scholar] [CrossRef] [PubMed]
- Czaikoski, P.G.; Mota, J.M.S.C.; Nascimento, D.C.; Sônego, F.; Varas e Silva Castanheira, F.; Melo, P.H.; Scortegagna, G.T.; Silva, R.L.; Barroso-Sousa, R.; Souto, F.O.; et al. Neutrophil Extracellular Traps Induce Organ Damage during Experimental and Clinical Sepsis. PLoS ONE 2016, 11, e0148142. [Google Scholar] [CrossRef]
- Colón, D.F.; Wanderley, C.W.; Franchin, M.; Silva, C.M.; Hiroki, C.H.; Castanheira, F.V.S.; Donate, P.B.; Lopes, A.H.; Volpon, L.C.; Kavaguti, S.K.; et al. Neutrophil Extracellular Traps (NETs) Exacerbate Severity of Infant Sepsis. Crit. Care 2019, 23, 113. [Google Scholar] [CrossRef]
- Agraz-Cibrian, J.M.; Giraldo, D.M.; Mary, F.-M.; Urcuqui-Inchima, S. Understanding the Molecular Mechanisms of NETs and Their Role in Antiviral Innate Immunity. Virus Res. 2017, 228, 124–133. [Google Scholar] [CrossRef] [PubMed]
- Cortjens, B.; de Boer, O.J.; de Jong, R.; Antonis, A.F.; Sabogal-Piñeros, Y.S.; Lutter, R.; van Woensel, J.B.; Bem, R.A. Neutrophil Extracellular Traps Cause Airway Obstruction during Respiratory Syncytial Virus Disease. J. Pathol. 2016, 238, 401–411. [Google Scholar] [CrossRef] [PubMed]
- Muraro, S.P.; De Souza, G.F.; Gallo, S.W.; Da Silva, B.K.; De Oliveira, S.D.; Vinolo, M.A.R.; Saraiva, E.M.; Porto, B.N. Respiratory Syncytial Virus Induces the Classical ROS-Dependent NETosis through PAD-4 and Necroptosis Pathways Activation. Sci. Rep. 2018, 8, 14166. [Google Scholar] [CrossRef]
- Hiroki, C.H.; Toller-Kawahisa, J.E.; Fumagalli, M.J.; Colon, D.F.; Figueiredo, L.T.M.; Fonseca, B.A.L.D.; Franca, R.F.O.; Cunha, F.Q. Neutrophil Extracellular Traps Effectively Control Acute Chikungunya Virus Infection. Front. Immunol. 2019, 10, 3108. [Google Scholar] [CrossRef] [PubMed]
- Buckley, C.D.; Gilroy, D.W.; Serhan, C.N. Proresolving Lipid Mediators and Mechanisms in the Resolution of Acute Inflammation. Immunity 2014, 40, 315–327. [Google Scholar] [CrossRef]
- Stark, M.A.; Huo, Y.; Burcin, T.L.; Morris, M.A.; Olson, T.S.; Ley, K. Phagocytosis of Apoptotic Neutrophils Regulates Granulopoiesis via IL-23 and IL-17. Immunity 2005, 22, 285–294. [Google Scholar] [CrossRef]
- Pérez-Figueroa, E.; Álvarez-Carrasco, P.; Ortega, E.; Maldonado-Bernal, C. Neutrophils: Many Ways to Die. Front. Immunol. 2021, 12, 631821. [Google Scholar] [CrossRef]
- Mogensen, T.H. Pathogen Recognition and Inflammatory Signaling in Innate Immune Defenses. Clin. Microbiol. Rev. 2009, 22, 240–273. [Google Scholar] [CrossRef]
- Kawai, T.; Akira, S. Innate Immune Recognition of Viral Infection. Nat. Immunol. 2006, 7, 131–137. [Google Scholar] [CrossRef]
- Taylor, K.E.; Mossman, K.L. Recent Advances in Understanding Viral Evasion of Type I Interferon. Immunology 2013, 138, 190–197. [Google Scholar] [CrossRef] [PubMed]
- Muralidharan, A.; Reid, S.P. Complex Roles of Neutrophils during Arboviral Infections. Cells 2021, 10, 1324. [Google Scholar] [CrossRef] [PubMed]
- Broggi, A.; Tan, Y.; Granucci, F.; Zanoni, I. IFN-λ Suppresses Intestinal Inflammation by Non-Translational Regulation of Neutrophil Function. Nat. Immunol. 2017, 18, 1084–1093. [Google Scholar] [CrossRef]
- Metzemaekers, M.; Cambier, S.; Blanter, M.; Vandooren, J.; de Carvalho, A.C.; Malengier-Devlies, B.; Vanderbeke, L.; Jacobs, C.; Coenen, S.; Martens, E.; et al. Kinetics of Peripheral Blood Neutrophils in Severe Coronavirus Disease 2019. Clin. Transl. Immunol. 2021, 10, e1271. [Google Scholar] [CrossRef] [PubMed]
- Crovatto, M.; Pozzato, G.; Zorat, F.; Pussini, E.; Nascimben, F.; Baracetti, S.; Grando, M.G.; Mazzaro, C.; Reitano, M.; Modolo, M.L.; et al. Peripheral Blood Neutrophils from Hepatitis C Virus-Infected Patients Are Replication Sites of the Virus. Haematologica 2000, 85, 356–361. [Google Scholar]
- Tate, M.D.; Ioannidis, L.J.; Croker, B.; Brown, L.E.; Brooks, A.G.; Reading, P.C. The Role of Neutrophils during Mild and Severe Influenza Virus Infections of Mice. PLoS ONE 2011, 6, e17618. [Google Scholar] [CrossRef] [PubMed]
- Levitt, N.H.; Miller, H.V.; Edelman, R. Interaction of Alphaviruses with Human Peripheral Leukocytes: In Vitro Replication of Venezuelan Equine Encephalomyelitis Virus in Monocyte Cultures. Infect. Immun. 1979, 24, 642–646. [Google Scholar] [CrossRef] [PubMed]
- Abramson, J.S.; Wheeler, J.G.; Parce, J.W.; Rowe, M.J.; Lyles, D.S.; Seeds, M.; Bass, D.A. Suppression of Endocytosis in Neutrophils by Influenza A Virus In Vitro. J. Infect. Dis. 1986, 154, 456–463. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Lu, M.; Lau, L.T.; Lu, J.; Gao, Z.; Liu, J.; Yu, A.C.H.; Cao, Q.; Ye, J.; McNutt, M.A.; et al. Neutrophils May Be a Vehicle for Viral Replication and Dissemination in Human H5N1 Avian Influenza. Clin. Infect. Dis. 2008, 47, 1575–1578. [Google Scholar] [CrossRef]
- Halfhide, C.P.; Flanagan, B.F.; Brearey, S.P.; Hunt, J.A.; Fonceca, A.M.; McNamara, P.S.; Howarth, D.; Edwards, S.; Smyth, R.L. Respiratory Syncytial Virus Binds and Undergoes Transcription in Neutrophils from the Blood and Airways of Infants with Severe Bronchiolitis. J. Infect. Dis. 2011, 204, 451–458. [Google Scholar] [CrossRef]
- Mohamadzadeh, M.; Coberley, S.S.; Olinger, G.G.; Kalina, W.V.; Ruthel, G.; Fuller, C.L.; Swenson, D.L.; Pratt, W.D.; Kuhns, D.B.; Schmaljohn, A.L. Activation of Triggering Receptor Expressed on Myeloid Cells-1 on Human Neutrophils by Marburg and Ebola Viruses. J. Virol. 2006, 80, 7235–7244. [Google Scholar] [CrossRef]
- Larochelle, B.; Flamand, L.; Gourde, P.; Beauchamp, D.; Gosselin, J. Epstein-Barr Virus Infects and Induces Apoptosis in Human Neutrophils. Blood 1998, 92, 291–299. [Google Scholar] [CrossRef]
- Hartshorn, K.L.; Liou, L.S.; White, M.R.; Kazhdan, M.M.; Tauber, J.L.; Tauber, A.I. Neutrophil Deactivation by Influenza A Virus. Role of Hemagglutinin Binding to Specific Sialic Acid-Bearing Cellular Proteins. J. Immunol. 1995, 154, 3952–3960. [Google Scholar] [PubMed]
- Daigneault, D.E.; Hartshorn, K.L.; Liou, L.S.; Abbruzzi, G.M.; White, M.R.; Oh, S.K.; Tauber, A.I. Influenza A Virus Binding to Human Neutrophils and Cross-Linking Requirements for Activation. Blood 1992, 80, 3227–3234. [Google Scholar] [CrossRef]
- Grudzinska, F.S.; Sapey, E. Friend or Foe? The Dual Role of Neutrophils in Lung Injury and Repair. Thorax 2018, 73, 305–307. [Google Scholar] [CrossRef] [PubMed]
- Butterfield, T.A.; Best, T.M.; Merrick, M.A. The Dual Roles of Neutrophils and Macrophages in Inflammation: A Critical Balance between Tissue Damage and Repair. J. Athl. Train. 2006, 41, 457–465. [Google Scholar]
- Johansson, C.; Kirsebom, F.C.M. Neutrophils in Respiratory Viral Infections. Mucosal Immunol. 2021. [Google Scholar] [CrossRef]
- Hayden, F.G.; Fritz, R.; Lobo, M.C.; Alvord, W.; Strober, W.; Straus, S.E. Local and Systemic Cytokine Responses during Experimental Human Influenza A Virus Infection. Relation to Symptom Formation and Host Defense. J. Clin. Investig. 1998, 101, 643–649. [Google Scholar] [CrossRef]
- Lee, N.; Wong, C.K.; Chan, P.K.S.; Chan, M.C.W.; Wong, R.Y.K.; Lun, S.W.M.; Ngai, K.L.K.; Lui, G.C.Y.; Wong, B.C.K.; Lee, S.K.W.; et al. Cytokine Response Patterns in Severe Pandemic 2009 H1N1 and Seasonal Influenza among Hospitalized Adults. PLoS ONE 2011, 6, e26050. [Google Scholar] [CrossRef] [PubMed]
- Fritz, R.S.; Hayden, F.G.; Calfee, D.P.; Cass, L.M.; Peng, A.W.; Alvord, W.G.; Strober, W.; Straus, S.E. Nasal Cytokine and Chemokine Responses in Experimental Influenza A Virus Infection: Results of a Placebo-Controlled Trial of Intravenous Zanamivir Treatment. J. Infect. Dis. 1999, 180, 586–593. [Google Scholar] [CrossRef]
- Bjornson, A.B.; Mellencamp, M.A.; Schiff, G.M. Complement Is Activated in the Upper Respiratory Tract during Influenza Virus Infection. Am. Rev. Respir. Dis. 1991, 143, 1062–1066. [Google Scholar] [CrossRef]
- Garcia, C.C.; Weston-Davies, W.; Russo, R.C.; Tavares, L.P.; Rachid, M.A.; Alves-Filho, J.C.; Machado, A.V.; Ryffel, B.; Nunn, M.A.; Teixeira, M.M. Complement C5 Activation during Influenza a Infection in Mice Contributes to Neutrophil Recruitment and Lung Injury. PLoS ONE 2013, 8, e64443. [Google Scholar] [CrossRef]
- Schmitz, N.; Kurrer, M.; Bachmann, M.F.; Kopf, M. Interleukin-1 Is Responsible for Acute Lung Immunopathology but Increases Survival of Respiratory Influenza Virus Infection. J. Virol. 2005, 79, 6441–6448. [Google Scholar] [CrossRef]
- Crowe, C.R.; Chen, K.; Pociask, D.A.; Alcorn, J.F.; Krivich, C.; Enelow, R.I.; Ross, T.M.; Witztum, J.L.; Kolls, J.K. Critical Role of IL-17RA in Immunopathology of Influenza Infection. J. Immunol. 2009, 183, 5301–5310. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, I.G.; de Brito, C.A.; dos Reis, V.M.S.; Sato, M.N.; Pereira, N.Z. SARS-CoV-2 and Other Respiratory Viruses: What Does Oxidative Stress Have to Do with It? Oxid. Med. Cell. Longev. 2020, 2020. [Google Scholar] [CrossRef]
- Hosakote, Y.M.; Liu, T.; Castro, S.M.; Garofalo, R.P.; Casola, A. Respiratory Syncytial Virus Induces Oxidative Stress by Modulating Antioxidant Enzymes. Am. J. Respir. Cell Mol. Biol. 2009, 41, 348–357. [Google Scholar] [CrossRef] [PubMed]
- LeVine, A.M.; Elliott, J.; Whitsett, J.A.; Srikiatkhachorn, A.; Crouch, E.; DeSilva, N.; Korfhagen, T. Surfactant Protein-D Enhances Phagocytosis and Pulmonary Clearance of Respiratory Syncytial Virus. Am. J. Respir. Cell Mol. Biol. 2004, 31, 193–199. [Google Scholar] [CrossRef]
- Hartshorn, K.L.; White, M.R.; Tecle, T.; Holmskov, U.; Crouch, E.C. Innate Defense against Influenza A Virus: Activity of Human Neutrophil Defensins and Interactions of Defensins with Surfactant Protein D. J. Immunol. 2006, 176, 6962–6972. [Google Scholar] [CrossRef] [PubMed]
- Mayadas, T.N.; Cullere, X.; Lowell, C.A. The Multifaceted Functions of Neutrophils. Annu. Rev. Pathol. 2014, 9, 181–218. [Google Scholar] [CrossRef]
- Li, M.; Zhao, L.; Liu, J.; Liu, A.; Jia, C.; Ma, D.; Jiang, Y.; Bai, X. Multi-Mechanisms Are Involved in Reactive Oxygen Species Regulation of mTORC1 Signaling. Cell. Signal. 2010, 22, 1469–1476. [Google Scholar] [CrossRef]
- Tilton, C.; Clippinger, A.J.; Maguire, T.; Alwine, J.C. Human Cytomegalovirus Induces Multiple Means to Combat Reactive Oxygen Species. J. Virol. 2011, 85, 12585–12593. [Google Scholar] [CrossRef]
- Hashimoto, Y.; Moki, T.; Takizawa, T.; Shiratsuchi, A.; Nakanishi, Y. Evidence for Phagocytosis of Influenza Virus-Infected, Apoptotic Cells by Neutrophils and Macrophages in Mice. J. Immunol. 2007, 178, 2448–2457. [Google Scholar] [CrossRef]
- Idilman, R.; Cetinkaya, H.; Savaş, I.; Aslan, N.; Sak, S.D.; Baştemir, M.; Sarioğlu, M.; Soykan, I.; Bozdayi, M.; Colantoni, A.; et al. Bronchoalveolar Lavage Fluid Analysis in Individuals with Chronic Hepatitis C. J. Med. Virol. 2002, 66, 34–39. [Google Scholar] [CrossRef]
- Leber, B.; Balázs, I.; Horvath, A.; Posch, A.; Streit, A.; Spindelböck, W.; Feldbacher, N.; Stiegler, P.; Stauber, R.E.; Rechberger, G.N.; et al. Direct Acting Antiviral Therapy Rescues Neutrophil Dysfunction and Reduces Hemolysis in Hepatitis C Infection. Transl. Res. 2021, 232, 103–114. [Google Scholar] [CrossRef]
- Kozik, J.-H.; Trautmann, T.; Carambia, A.; Preti, M.; Lütgehetmann, M.; Krech, T.; Wiegard, C.; Heeren, J.; Herkel, J. Attenuated Viral Hepatitis in Trem1-/- Mice Is Associated with Reduced Inflammatory Activity of Neutrophils. Sci. Rep. 2016, 6, 28556. [Google Scholar] [CrossRef]
- Veras, F.P.; Pontelli, M.C.; Silva, C.M.; Toller-Kawahisa, J.E.; de Lima, M.; Nascimento, D.C.; Schneider, A.H.; Caetité, D.; Tavares, L.A.; Paiva, I.M.; et al. SARS-CoV-2–triggered Neutrophil Extracellular Traps Mediate COVID-19 Pathology. J. Exp. Med. 2020, 217. [Google Scholar] [CrossRef]
- Saitoh, T.; Komano, J.; Saitoh, Y.; Misawa, T.; Takahama, M.; Kozaki, T.; Uehata, T.; Iwasaki, H.; Omori, H.; Yamaoka, S.; et al. Neutrophil Extracellular Traps Mediate a Host Defense Response to Human Immunodeficiency Virus-1. Cell Host Microbe 2012, 12, 109–116. [Google Scholar] [CrossRef] [PubMed]
- Daigo, K.; Hamakubo, T. Host-Protective Effect of Circulating Pentraxin 3 (PTX3) and Complex Formation with Neutrophil Extracellular Traps. Front. Immunol. 2012, 3, 378. [Google Scholar] [CrossRef] [PubMed]
- Barr, F.D.; Ochsenbauer, C.; Wira, C.R.; Rodriguez-Garcia, M. Neutrophil Extracellular Traps Prevent HIV Infection in the Female Genital Tract. Mucosal Immunol. 2018, 11, 1420–1428. [Google Scholar] [CrossRef]
- Viral Hemorrhagic Fevers (VHFs). Available online: https://www.cdc.gov/vhf/index.html (accessed on 17 June 2021).
- Bray, M. Pathogenesis of Viral Hemorrhagic Fever. Curr. Opin. Immunol. 2005, 17, 399–403. [Google Scholar] [CrossRef] [PubMed]
- LeDuc, J.W. Epidemiology of Hemorrhagic Fever Viruses. Rev. Infect. Dis. 1989, 11 (Suppl. 4), S730–S735. [Google Scholar] [CrossRef]
- Guerrero, D.; Cantaert, T.; Missé, D. Aedes Mosquito Salivary Components and Their Effect on the Immune Response to Arboviruses. Front. Cell. Infect. Microbiol. 2020, 10, 407. [Google Scholar] [CrossRef] [PubMed]
- Pingen, M.; Bryden, S.R.; Pondeville, E.; Schnettler, E.; Kohl, A.; Merits, A.; Fazakerley, J.K.; Graham, G.J.; McKimmie, C.S. Host Inflammatory Response to Mosquito Bites Enhances the Severity of Arbovirus Infection. Immunity 2016, 44, 1455–1469. [Google Scholar] [CrossRef]
- Stegelmeier, A.A.; van Vloten, J.P.; Mould, R.C.; Klafuric, E.M.; Minott, J.A.; Wootton, S.K.; Bridle, B.W.; Karimi, K. Myeloid Cells during Viral Infections and Inflammation. Viruses 2019, 11, 168. [Google Scholar] [CrossRef]
- Findlay, J.S.; Ulaeto, D.; D’Elia, R.V. Cytokines and Viral Hemorrhagic Fever: Potential for Therapeutic Intervention. Future Virol. 2015, 10, 547–557. [Google Scholar] [CrossRef]
- Messaoudi, I.; Basler, C.F. Immunological Features Underlying Viral Hemorrhagic Fevers. Curr. Opin. Immunol. 2015, 36, 38–46. [Google Scholar] [CrossRef] [PubMed]
- Schulz, C.; Gabriel, G.; von Köckritz-Blickwede, M. Detrimental Role of Neutrophil Extracellular Traps during Dengue Virus Infection. Trends Immunol. 2020, 41, 3–6. [Google Scholar] [CrossRef] [PubMed]
- Opasawatchai, A.; Amornsupawat, P.; Jiravejchakul, N.; Chan-In, W.; Spoerk, N.J.; Manopwisedjaroen, K.; Singhasivanon, P.; Yingtaweesak, T.; Suraamornkul, S.; Mongkolsapaya, J.; et al. Neutrophil Activation and Early Features of NET Formation Are Associated with Dengue Virus Infection in Human. Front. Immunol. 2018, 9, 3007. [Google Scholar] [CrossRef]
- Hoang, L.T.; Lynn, D.J.; Henn, M.; Birren, B.W.; Lennon, N.J.; Le, P.T.; Duong, K.T.H.; Nguyen, T.T.H.; Mai, L.N.; Farrar, J.J.; et al. The Early Whole-Blood Transcriptional Signature of Dengue Virus and Features Associated with Progression to Dengue Shock Syndrome in Vietnamese Children and Young Adults. J. Virol. 2010, 84, 12982–12994. [Google Scholar] [CrossRef]
- Thein, T.-L.; Lye, D.C.; Leo, Y.-S.; Wong, J.G.X.; Hao, Y.; Wilder-Smith, A. Severe Neutropenia in Dengue Patients: Prevalence and Significance. Am. J. Trop. Med. Hyg. 2014, 90, 984–987. [Google Scholar] [CrossRef]
- Turtle, L.; Griffiths, M.J.; Solomon, T. Encephalitis Caused by Flaviviruses. QJM Int. J. Med. 2012, 105, 219–223. [Google Scholar] [CrossRef] [PubMed]
- Meyding-Lamadé, U.; Craemer, E.; Schnitzler, P. Emerging and Re-Emerging Viruses Affecting the Nervous System. Neurol. Res. Pract. 2019, 1, 20. [Google Scholar] [CrossRef]
- Salimi, H.; Cain, M.D.; Klein, R.S. Encephalitic Arboviruses: Emergence, Clinical Presentation, and Neuropathogenesis. Neurotherapeutics 2016, 13, 514–534. [Google Scholar] [CrossRef] [PubMed]
- Moore, S.M. The Current Burden of Japanese Encephalitis and the Estimated Impacts of Vaccination: Combining Estimates of the Spatial Distribution and Transmission Intensity of a Zoonotic Pathogen. medRxiv 2021. [Google Scholar] [CrossRef]
- Marques, R.E.; Del Sarto, J.L.; Rocha, R.P.F.; Gomes, G.F.; Cramer, A.; Rachid, M.A.; Souza, D.G.; Nogueira, M.L.; Teixeira, M.M. Development of a Model of Saint Louis Encephalitis Infection and Disease in Mice. J. Neuroinflamm. 2017, 14, 61. [Google Scholar] [CrossRef]
- Richner, J.M.; Gmyrek, G.B.; Govero, J.; Tu, Y.; van der Windt, G.J.W.; Metcalf, T.U.; Haddad, E.K.; Textor, J.; Miller, M.J.; Diamond, M.S. Age-Dependent Cell Trafficking Defects in Draining Lymph Nodes Impair Adaptive Immunity and Control of West Nile Virus Infection. PLoS Pathog. 2015, 11, e1005027. [Google Scholar] [CrossRef]
- Ryan, E.T.; Hill, D.R.; Solomon, T.; Aronson, N.E.; Endy, T.P. (Eds.) 38—Viral CNS Infections. In Hunter’s Tropical Medicine and Emerging Infectious Diseases, 10th ed.; Elsevier: London, UK, 2020; pp. 382–420. ISBN 9780323555128. [Google Scholar]
- Rocha, R.F.; Del Sarto, J.L.; Gomes, G.F.; Gonçalves, M.P.; Rachid, M.A.; Smetana, J.H.C.; Souza, D.G.; Teixeira, M.M.; Marques, R.E. Type I Interferons Are Essential While Type II Interferon Is Dispensable for Protection against St. Louis Encephalitis Virus Infection in the Mouse Brain. Virulence 2021, 12, 244–259. [Google Scholar] [CrossRef] [PubMed]
- Redant, V.; Favoreel, H.W.; Dallmeier, K.; Van Campe, W.; De Regge, N. Efficient Control of Japanese Encephalitis Virus in the Central Nervous System of Infected Pigs Occurs in the Absence of a Pronounced Inflammatory Immune Response. J. Neuroinflamm. 2020, 17, 315. [Google Scholar] [CrossRef] [PubMed]
- Bardina, S.V.; Lim, J.K. The Role of Chemokines in the Pathogenesis of Neurotropic Flaviviruses. Immunol. Res. 2012, 54, 121–132. [Google Scholar] [CrossRef]
- Lannes, N.; Summerfield, A.; Filgueira, L. Regulation of Inflammation in Japanese Encephalitis. J. Neuroinflamm. 2017, 14, 158. [Google Scholar] [CrossRef] [PubMed]
- Su, Q.; Xie, Z.-X.; He, F.; Liu, Z.-C.; Song, X.-J.; Zhao, F.-C.; Li, D.; Che, F.-Y. Adults with Severe Japanese Encephalitis: A Retrospective Analysis of 9 Cases in Linyi, China. Neurol. Sci. 2020. [Google Scholar] [CrossRef]
- Singh, S.; Singh, G.; Tiwari, S.; Kumar, A. CCR2 Inhibition Reduces Neurotoxic Microglia Activation Phenotype After Japanese Encephalitis Viral Infection. Front. Cell. Neurosci. 2020, 14, 230. [Google Scholar] [CrossRef]
- Singh, A.; Kulshreshtha, R.; Mathur, A. Secretion of the Chemokine Interleukin-8 during Japanese Encephalitis Virus Infection. J. Med. Microbiol. 2000, 49, 607–612. [Google Scholar] [CrossRef]
- Srivastava, S.; Khanna, N.; Saxena, S.K.; Singh, A.; Mathur, A.; Dhole, T.N. Degradation of Japanese Encephalitis Virus by Neutrophils. Int. J. Exp. Pathol. 1999, 80, 17–24. [Google Scholar] [CrossRef]
- Debiasi, R.L.; Tyler, K.L. West Nile Virus Meningoencephalitis. Nat. Clin. Pract. Neurol. 2006, 2, 264–275. [Google Scholar] [CrossRef]
- Bai, F.; Kong, K.-F.; Dai, J.; Qian, F.; Zhang, L.; Brown, C.R.; Fikrig, E.; Montgomery, R.R. A Paradoxical Role for Neutrophils in the Pathogenesis of West Nile Virus. J. Infect. Dis. 2010, 202, 1804–1812. [Google Scholar] [CrossRef] [PubMed]
- Paul, A.M.; Acharya, D.; Duty, L.; Thompson, E.A.; Le, L.; Stokic, D.S.; Leis, A.A.; Bai, F. Osteopontin Facilitates West Nile Virus Neuroinvasion via Neutrophil “Trojan Horse” Transport. Sci. Rep. 2017, 7, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Macdonald, F. Murray Valley Encephalitis Infection in the Laboratory Mouse. I. Influence of Age on Susceptibility to Infection. Aust. J. Exp. Biol. Med. Sci. 1952, 30, 319–324. [Google Scholar] [CrossRef]
- Andrews, D.M.; Matthews, V.B.; Sammels, L.M.; Carrello, A.C.; McMinn, P.C. The Severity of Murray Valley Encephalitis in Mice Is Linked to Neutrophil Infiltration and Inducible Nitric Oxide Synthase Activity in the Central Nervous System. J. Virol. 1999, 73, 8781–8790. [Google Scholar] [CrossRef]
- McMinn, P.C.; Dalgarno, L.; Weir, R.C. A Comparison of the Spread of Murray Valley Encephalitis Viruses of High or Low Neuroinvasiveness in the Tissues of Swiss Mice after Peripheral Inoculation. Virology 1996, 220, 414–423. [Google Scholar] [CrossRef] [PubMed]
- Matthews, V.; Robertson, T.; Kendrick, T.; Abdo, M.; Papadimitriou, J.; McMinn, P. Morphological Features of Murray Valley Encephalitis Virus Infection in the Central Nervous System of Swiss Mice. Int. J. Exp. Pathol. 2000, 81, 31–40. [Google Scholar] [CrossRef]
- Hombach, J.; Barrett, A.D.T.; Kollaritsch, H. 59—Tickborne Encephalitis Vaccines. In Plotkin’s Vaccines, 7th ed.; Plotkin, S.A., Orenstein, W.A., Offit, P.A., Edwards, K.M., Eds.; Elsevier: Amsterdam, The Netherlands, 2018; pp. 1080–1094.e5. ISBN 9780323357616. [Google Scholar]
- Calvert, A.; Quenby, M.; Heath, P.T. Chapter 9—Vaccination in pregnancy in specific circumstances. In Maternal Immunization; Leuridan, E.E., Nunes, M.C., Jones, C.E., Eds.; Academic Press: Cambridge, MA, USA, 2020; pp. 191–210. ISBN 9780128145821. [Google Scholar]
- Plekhova, N.G.; Somova, L.M.; Lyapun, I.N.; Krylova, N.V.; Leonova, G.N. Neutrophil Apoptosis Induction by Tick-Borne Encephalitis Virus. Bull. Exp. Biol. Med. 2012, 153, 105–108. [Google Scholar] [CrossRef] [PubMed]
- Thangamani, S.; Hermance, M.E.; Santos, R.I.; Slovak, M.; Heinze, D.; Widen, S.G.; Kazimirova, M. Transcriptional Immunoprofiling at the Tick-Virus-Host Interface during Early Stages of Tick-Borne Encephalitis Virus Transmission. Front. Cell. Infect. Microbiol. 2017, 7, 494. [Google Scholar] [CrossRef] [PubMed]
- Wilson, J.G. Experimental Studies on Congenital Malformations. J. Chronic Dis. 1959, 10, 111–130. [Google Scholar] [CrossRef]
- Barlow, S. Handbook of Teratology, Volume 1: General Principles and Etiology. Biochem. Soc. Trans. 1978, 6, 473–474. [Google Scholar] [CrossRef]
- Alwan, S.; Chambers, C.D. Identifying Human Teratogens: An Update. J. Pediatr. Genet. 2015, 4, 39–41. [Google Scholar] [CrossRef]
- Nausheen, F.; Bashir, S.S.; Atapattu, D.N. Pregnancy Associated Arbovirus Infections. Clin. Microbiol. Infect. 2018, 3, 1–9. [Google Scholar] [CrossRef]
- Coyne, C.B.; Lazear, H.M. Zika Virus—Reigniting the TORCH. Nat. Rev. Microbiol. 2016, 14, 707–715. [Google Scholar] [CrossRef] [PubMed]
- Ander, S.E.; Diamond, M.S.; Coyne, C.B. Immune Responses at the Maternal-Fetal Interface. Sci. Immunol. 2019, 4. [Google Scholar] [CrossRef]
- Mor, G.; Aldo, P.; Alvero, A.B. The Unique Immunological and Microbial Aspects of Pregnancy. Nat. Rev. Immunol. 2017, 17, 469–482. [Google Scholar] [CrossRef]
- Abu-Raya, B.; Michalski, C.; Sadarangani, M.; Lavoie, P.M. Maternal Immunological Adaptation during Normal Pregnancy. Front. Immunol. 2020, 11, 575197. [Google Scholar] [CrossRef]
- Kourtis, A.P.; Read, J.S.; Jamieson, D.J. Pregnancy and Infection. N. Engl. J. Med. 2014, 370, 2211–2218. [Google Scholar] [CrossRef]
- Morelli, S.S.; Mandal, M.; Goldsmith, L.T.; Kashani, B.N.; Ponzio, N.M. The Maternal Immune System during Pregnancy and Its Influence on Fetal Development. Res. Rep. Biol. 2015, 6, 171–189. [Google Scholar] [CrossRef]
- Thomson, A.J.; Telfer, J.F.; Young, A.; Campbell, S.; Stewart, C.J.; Cameron, I.T.; Greer, I.A.; Norman, J.E. Leukocytes Infiltrate the Myometrium during Human Parturition: Further Evidence That Labour Is an Inflammatory Process. Hum. Reprod. 1999, 14, 229–236. [Google Scholar] [CrossRef]
- Gomez-Lopez, N.; Guilbert, L.J.; Olson, D.M. Invasion of the Leukocytes into the Fetal-Maternal Interface during Pregnancy. J. Leukoc. Biol. 2010, 88, 625–633. [Google Scholar] [CrossRef]
- Kruse, A.; Martens, N.; Fernekorn, U.; Hallmann, R.; Butcher, E.C. Alterations in the Expression of Homing-Associated Molecules at the Maternal/fetal Interface during the Course of Pregnancy. Biol. Reprod. 2002, 66, 333–345. [Google Scholar] [CrossRef][Green Version]
- Sacks, G.P.; Studena, K.; Sargent, K.; Redman, C.W. Normal Pregnancy and Preeclampsia Both Produce Inflammatory Changes in Peripheral Blood Leukocytes Akin to Those of Sepsis. Am. J. Obstet. Gynecol. 1998, 179, 80–86. [Google Scholar] [CrossRef]
- Giaglis, S.; Stoikou, M.; Sur Chowdhury, C.; Schaefer, G.; Grimolizzi, F.; Rossi, S.W.; Hoesli, I.M.; Lapaire, O.; Hasler, P.; Hahn, S. Multimodal Regulation of NET Formation in Pregnancy: Progesterone Antagonizes the Pro-NETotic Effect of Estrogen and G-CSF. Front. Immunol. 2016, 7, 565. [Google Scholar] [CrossRef]
- Wira, C.R.; Fahey, J.V.; Rodriguez-Garcia, M.; Shen, Z.; Patel, M.V. Regulation of Mucosal Immunity in the Female Reproductive Tract: The Role of Sex Hormones in Immune Protection against Sexually Transmitted Pathogens. Am. J. Reprod. Immunol. 2014, 72, 236–258. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Herrera, G.G.; Tam, K.K.; Lizarraga, J.S.; Beedle, M.-T.; Winuthayanon, W. Estrogen Action in the Epithelial Cells of the Mouse Vagina Regulates Neutrophil Infiltration and Vaginal Tissue Integrity. Sci. Rep. 2018, 8, 11247. [Google Scholar] [CrossRef]
- Aagaard-Tillery, K.M.; Silver, R.; Dalton, J. Immunology of Normal Pregnancy. Semin. Fetal Neonatal Med. 2006, 11, 279–295. [Google Scholar] [CrossRef] [PubMed]
- Shimoya, K.; Matsuzaki, N.; Taniguchi, T.; Kameda, T.; Koyama, M.; Neki, R.; Saji, F.; Tanizawa, O. Human Placenta Constitutively Produces Interleukin-8 during Pregnancy and Enhances Its Production in Intrauterine Infection. Biol. Reprod. 1992, 47, 220–226. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Lopez, N.; Romero, R.; Xu, Y.; Leng, Y.; Garcia-Flores, V.; Miller, D.; Jacques, S.M.; Hassan, S.S.; Faro, J.; Alsamsam, A.; et al. Are Amniotic Fluid Neutrophils in Women with Intraamniotic Infection And/or Inflammation of Fetal or Maternal Origin? Am. J. Obstet. Gynecol. 2017, 217, 693.e1–693.e16. [Google Scholar] [CrossRef] [PubMed]
- Szarka, A.; Rigó, J., Jr.; Lázár, L.; Beko, G.; Molvarec, A. Circulating Cytokines, Chemokines and Adhesion Molecules in Normal Pregnancy and Preeclampsia Determined by Multiplex Suspension Array. BMC Immunol. 2010, 11, 59. [Google Scholar] [CrossRef] [PubMed]
- Gupta, A.K.; Hasler, P.; Holzgreve, W.; Gebhardt, S.; Hahn, S. Induction of Neutrophil Extracellular DNA Lattices by Placental Microparticles and IL-8 and Their Presence in Preeclampsia. Hum. Immunol. 2005, 66, 1146–1154. [Google Scholar] [CrossRef]
- Vokalova, L.; van Breda, S.V.; Ye, X.L.; Huhn, E.A.; Than, N.G.; Hasler, P.; Lapaire, O.; Hoesli, I.; Rossi, S.W.; Hahn, S. Excessive Neutrophil Activity in Gestational Diabetes Mellitus: Could It Contribute to the Development of Preeclampsia? Front. Endocrinol. 2018, 9, 542. [Google Scholar] [CrossRef] [PubMed]
- Staples, J.E.; Monath, T.P. Yellow Fever: 100 Years of Discovery. JAMA 2008, 300, 960–962. [Google Scholar] [CrossRef]
- Machado, C.R.; Machado, E.S.; Rohloff, R.D.; Azevedo, M.; Campos, D.P.; de Oliveira, R.B.; Brasil, P. Is Pregnancy Associated with Severe Dengue? A Review of Data from the Rio de Janeiro Surveillance Information System. PLoS Negl. Trop. Dis. 2013, 7, e2217. [Google Scholar] [CrossRef]
- Tan, P.C.; Soe, M.Z.; Si Lay, K.; Wang, S.M.; Sekaran, S.D.; Omar, S.Z. Dengue Infection and Miscarriage: A Prospective Case Control Study. PLoS Negl. Trop. Dis. 2012, 6, e1637. [Google Scholar] [CrossRef] [PubMed]
- Paixão, E.S.; Teixeira, M.G.; Costa, M.d.C.N.; Rodrigues, L.C. Dengue during Pregnancy and Adverse Fetal Outcomes: A Systematic Review and Meta-Analysis. Lancet Infect. Dis. 2016, 16, 857–865. [Google Scholar] [CrossRef]
- Sirinavin, S.; Nuntnarumit, P.; Supapannachart, S.; Boonkasidecha, S.; Techasaensiri, C.; Yoksarn, S. Vertical Dengue Infection: Case Reports and Review. Pediatr. Infect. Dis. J. 2004, 23, 1042–1047. [Google Scholar] [CrossRef] [PubMed]
- Basurko, C.; Carles, G.; Youssef, M.; Guindi, W.E.L. Maternal and Fetal Consequences of Dengue Fever during Pregnancy. Eur. J. Obstet. Gynecol. Reprod. Biol. 2009, 147, 29–32. [Google Scholar] [CrossRef]
- Sicé, A.; Rodallec, C. Manifestations Hémorragiques de: La Fièvre Jaune (typhus Amaril). Répercussions de L’infection Maternelle Sur L’organisme Foetal. Bull. Soc. Pathol. Exot. 1940, 33, 66–69. [Google Scholar]
- Bentlin, M.R.; de Barros Almeida, R.A.M.; Coelho, K.I.R.; Ribeiro, A.F.; Siciliano, M.M.; Suzuki, A.; Fortaleza, C.M.C.B. Perinatal Transmission of Yellow Fever, Brazil, 2009. Emerg. Infect. Dis. 2011, 17, 1779–1780. [Google Scholar] [CrossRef]
- Burns, K.F. Congenital Japanese B Encephalitis Infection of Swine. Proc. Soc. Exp. Biol. Med. 1950, 75, 621–625. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, T.; Kawakami, Y.; Fukuhara, S.; Matumoto, M. Experimental Stillbirth in Pregnant Swine Infected with Japanese Encephalitis Virus. Jpn. J. Exp. Med. 1954, 24, 363–375. [Google Scholar]
- Chaturvedi, U.C.; Mathur, A.; Chandra, A.; Das, S.K.; Tandon, H.O.; Singh, U.K. Transplacental Infection with Japanese Encephalitis Virus. J. Infect. Dis. 1980, 141, 712–715. [Google Scholar] [CrossRef]
- Andersen, A.A.; Hanson, R.P. Experimental Transplacental Transmission of St. Louis Encephalitis Virus in Mice. Infect. Immun. 1970, 2, 320–325. [Google Scholar] [CrossRef] [PubMed]
- Charlier, C.; Beaudoin, M.-C.; Couderc, T.; Lortholary, O.; Lecuit, M. Arboviruses and Pregnancy: Maternal, Fetal, and Neonatal Effects. Lancet Child Adolesc. Health 2017, 1, 134–146. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention (CDC). Intrauterine West Nile Virus Infection—New York, 2002. MMWR Morb. Mortal. Wkly. Rep. 2002, 51, 1135–1136. [Google Scholar]
- Alpert, S.G.; Fergerson, J.; Noël, L.P. Intrauterine West Nile Virus: Ocular and Systemic Findings. Am. J. Ophthalmol. 2003, 136, 733–735. [Google Scholar] [CrossRef]
- O’Leary, D.R.; Kuhn, S.; Kniss, K.L.; Hinckley, A.F.; Rasmussen, S.A.; Pape, W.J.; Kightlinger, L.K.; Beecham, B.D.; Miller, T.K.; Neitzel, D.F.; et al. Birth Outcomes Following West Nile Virus Infection of Pregnant Women in the United States: 2003–2004. Pediatrics 2006, 117, e537–e545. [Google Scholar] [CrossRef]
- Platt, D.J.; Smith, A.M.; Arora, N.; Diamond, M.S.; Coyne, C.B.; Miner, J.J. Zika Virus-Related Neurotropic Flaviviruses Infect Human Placental Explants and Cause Fetal Demise in Mice. Sci. Transl. Med. 2018, 10. [Google Scholar] [CrossRef]
- Morrison, T.E.; Diamond, M.S. Animal Models of Zika Virus Infection, Pathogenesis, and Immunity. J. Virol. 2017, 91, e00009-17. [Google Scholar] [CrossRef] [PubMed]
- Brasil, P.; Pereira, J.P., Jr.; Moreira, M.E.; Ribeiro Nogueira, R.M.; Damasceno, L.; Wakimoto, M.; Rabello, R.S.; Valderramos, S.G.; Halai, U.-A.; Salles, T.S.; et al. Zika Virus Infection in Pregnant Women in Rio de Janeiro. N. Engl. J. Med. 2016, 375, 2321–2334. [Google Scholar] [CrossRef] [PubMed]
- Schuler-Faccini, L.; Ribeiro, E.M.; Feitosa, I.M.L.; Horovitz, D.D.G.; Cavalcanti, D.P.; Pessoa, A.; Doriqui, M.J.R.; Neri, J.I.; Monteiro de Pina Neto, J.; Wanderley, H.Y.C.; et al. Possible Association Between Zika Virus Infection and Microcephaly—Brazil, 2015. MMWR Morb. Mortal. Wkly. Rep. 2016, 65, 59–62. [Google Scholar] [CrossRef] [PubMed]
- Leal, M.C.; Muniz, L.F.; Ferreira, T.S.A.; Santos, C.M.; Almeida, L.C.; Van Der Linden, V.; Ramos, R.C.F.; Rodrigues, L.C.; Neto, S.S.C. Hearing Loss in Infants with Microcephaly and Evidence of Congenital Zika Virus Infection—Brazil, November 2015–May 2016. MMWR Morb. Mortal. Wkly. Rep. 2016, 65, 917–919. [Google Scholar] [CrossRef] [PubMed]
- Camacho-Zavala, E.; Santacruz-Tinoco, C.; Muñoz, E.; Chacón-Salinas, R.; Salazar-Sanchez, M.I.; Grajales, C.; González-Ibarra, J.; Borja-Aburto, V.H.; Jaenisch, T.; Gonzalez-Bonilla, C.R. Pregnant Women Infected with Zika Virus Show Higher Viral Load and Immunoregulatory Cytokines Profile with CXCL10 Increase. Viruses 2021, 13, 80. [Google Scholar] [CrossRef]
- Ornelas, A.M.M.; Pezzuto, P.; Silveira, P.P.; Melo, F.O.; Ferreira, T.A.; Oliveira-Szejnfeld, P.S.; Leal, J.I.; Amorim, M.M.R.; Hamilton, S.; Rawlinson, W.D.; et al. Immune Activation in Amniotic Fluid from Zika Virus-Associated Microcephaly. Ann. Neurol. 2017, 81, 152–156. [Google Scholar] [CrossRef]
- Lima, M.C.; de Mendonça, L.R.; Rezende, A.M.; Carrera, R.M.; Aníbal-Silva, C.E.; Demers, M.; D’Aiuto, L.; Wood, J.; Chowdari, K.V.; Griffiths, M.; et al. The Transcriptional and Protein Profile from Human Infected Neuroprogenitor Cells Is Strongly Correlated to Zika Virus Microcephaly Cytokines Phenotype Evidencing a Persistent Inflammation in the CNS. Front. Immunol. 2019, 10, 1928. [Google Scholar] [CrossRef]
- Khaiboullina, S.; Uppal, T.; Kletenkov, K.; St. Jeor, S.C.; Garanina, E.; Rizvanov, A.; Verma, S.C. Transcriptome Profiling Reveals Pro-Inflammatory Cytokines and Matrix Metalloproteinase Activation in Zika Virus Infected Human Umbilical Vein Endothelial Cells. Front. Pharmacol. 2019, 10, 642. [Google Scholar] [CrossRef]
- Bayer, A.; Lennemann, N.J.; Ouyang, Y.; Bramley, J.C.; Morosky, S.; Marques, E.T.D.A., Jr.; Cherry, S.; Sadovsky, Y.; Coyne, C.B. Type III Interferons Produced by Human Placental Trophoblasts Confer Protection against Zika Virus Infection. Cell Host Microbe 2016, 19, 705–712. [Google Scholar] [CrossRef]
- Galani, I.E.; Triantafyllia, V.; Eleminiadou, E.-E.; Koltsida, O.; Stavropoulos, A.; Manioudaki, M.; Thanos, D.; Doyle, S.E.; Kotenko, S.V.; Thanopoulou, K.; et al. Interferon-λ Mediates Non-Redundant Front-Line Antiviral Protection against Influenza Virus Infection without Compromising Host Fitness. Immunity 2017, 46, 875–890.e6. [Google Scholar] [CrossRef] [PubMed]
- Blazek, K.; Eames, H.L.; Weiss, M.; Byrne, A.J.; Perocheau, D.; Pease, J.E.; Doyle, S.; McCann, F.; Williams, R.O.; Udalova, I.A. IFN-λ Resolves Inflammation via Suppression of Neutrophil Infiltration and IL-1β Production. J. Exp. Med. 2015, 212, 845–853. [Google Scholar] [CrossRef] [PubMed]
- Azamor, T.; Torrentes-Carvalho, A.; Vasconcelos, Z.; Bom, A.P.D.A.; Melgaço, J.G. Innate Immunity Modulation during Zika Virus Infection on Pregnancy: What We Still Need to Know for Medical Sciences Breakthrough. In Cell Interaction; Singh, B., Ed.; IntechOpen: Rijeka, Croatia, 2021. [Google Scholar]
- Azamor, T.; Cunha, D.P.; da Silva, A.M.V.; de Lima Bezerra, O.C.; Ribeiro-Alves, M.; Calvo, T.L.; de Souza Gomes Kehdy, F.; de Neves Manta, F.S.; Pinto, T.G.; Ferreira, L.P.; et al. Congenital Zika Syndrome Is Associated with Interferon Alfa Receptor 1. bioRxiv 2020, 715862. [Google Scholar] [CrossRef]
- Yockey, L.J.; Jurado, K.A.; Arora, N.; Millet, A.; Rakib, T.; Milano, K.M.; Hastings, A.K.; Fikrig, E.; Kong, Y.; Horvath, T.L.; et al. Type I Interferons Instigate Fetal Demise after Zika Virus Infection. Sci. Immunol. 2018, 3. [Google Scholar] [CrossRef]
- Iwasaki, A.; Pillai, P.S. Innate Immunity to Influenza Virus Infection. Nat. Rev. Immunol. 2014, 14, 315–328. [Google Scholar] [CrossRef]
- Andrade, D.; Kim, M.; Blanco, L.P.; Karumanchi, S.A.; Koo, G.C.; Redecha, P.; Kirou, K.; Alvarez, A.M.; Mulla, M.J.; Crow, M.K.; et al. Interferon-α and Angiogenic Dysregulation in Pregnant Lupus Patients Who Develop Preeclampsia. Arthritis Rheumatol. 2015, 67, 977–987. [Google Scholar] [CrossRef]
| Virus | Chemokines/Cytokines | Involvement |
|---|---|---|
| Flaviviruses transmitted through Aedes mosquito bite | CXCL1, CXCL2, CXCL3, CXCL5, IL-1β, IL-6 | Promote local inflammation and recruitment of target cells to the skin. |
| DENV, YFV | CXCL2, CXCL8, IL-6, TNF-α CCL2, IL-1β | Promote neutrophil activation in dengue. High expression was associated with increased disease severity in yellow fever. |
| SLEV | CXCL1 | Neutrophil recruitment to infected brain tissue. |
| JEV | CCL2, CCL5, TNF-α, IL-1β | High levels were associated to brain damage and neuronal loss. |
| CXCL8 | Accumulation in CSF in severe cases. | |
| WNV | CXCL1, CXCL2 | Involved in PMN recruitment to infected tissues. |
| OPN | Facilitates infiltration of virus and PMNs in the brain. | |
| MVEV | CXCL1, TNF-α | Neutrophil recruitment and activation, also related to increased disease severity. |
| TBEV | CCL2, CCL12, CXCL1, CXCL2, CXCL5, IL-6, IL-10 | Upregulated in cutaneous tissue during early stages of infection. |
| ZIKV | CCL11, CXCL10, CXCL8, CCL2, CCL5, G-CSF, IL-1β, TNF-α | High levels in the amniotic fluid from infected mothers whose children presented microcephaly. |
| CXCL10, CXCL9, IFN-α | Associated with persistent inflammation in the CSF of infants with microcephaly. | |
| CXCL1, CXCL12 | Probably associated with neutrophil recruitment to umbilical cord during ZIKV infection. | |
| Type-III IFN | Protective effect against ZIKV placental infection, also downregulates neutrophils effector functions. | |
| Type-I IFN | Generally protective against virus infections. However, may lead to aberrant neutrophilic response in CZS and placental damage. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fontoura, M.A.; Rocha, R.F.; Marques, R.E. Neutrophil Recruitment and Participation in Severe Diseases Caused by Flavivirus Infection. Life 2021, 11, 717. https://doi.org/10.3390/life11070717
Fontoura MA, Rocha RF, Marques RE. Neutrophil Recruitment and Participation in Severe Diseases Caused by Flavivirus Infection. Life. 2021; 11(7):717. https://doi.org/10.3390/life11070717
Chicago/Turabian StyleFontoura, Marina Alves, Rebeca Fróes Rocha, and Rafael Elias Marques. 2021. "Neutrophil Recruitment and Participation in Severe Diseases Caused by Flavivirus Infection" Life 11, no. 7: 717. https://doi.org/10.3390/life11070717
APA StyleFontoura, M. A., Rocha, R. F., & Marques, R. E. (2021). Neutrophil Recruitment and Participation in Severe Diseases Caused by Flavivirus Infection. Life, 11(7), 717. https://doi.org/10.3390/life11070717






