Timely and Blood-Based Multiplex Molecular Profiling of Acute Stroke
Abstract
:1. Introduction
2. Circulating Protein Biomarkers for Ischemic Stroke Differential Diagnosis
3. Circulating Protein Biomarkers to Differentiate Acute IS from HS
4. Conventional and Point-of-Care Technologies
5. Future Perspectives
- The recognition of stroke mimics would become more efficient. The frequency of these can vary between 15% and 42% and entail an inappropriate use of the available stroke facilities leading to additional costs and a delayed diagnosis of the actual disease [19,23,24]. Even more, the administration of thrombolytic medication in wrongly diagnosed patients may lead to undesirable side effects such as intracranial hemorrhage [111];
- Stroke chameleons’ recognition at patient admission to the hospital would be more sensitive and specific. The frequency of these can vary between 2% and 26% [18]. The problem of chameleons resides in the lack of proper treatment of stroke patients during the hyper-acute settings due to the fact of missing diagnosis, lowering the chance to administer thrombolytic medication or to undergo mechanical thrombectomy as well as to receive suitable secondary prevention. Consequently, stroke chameleon patients have the worst outcomes at 12 months [10];
- The reperfusion treatments would be hastened. Thrombolytic iv treatment would start right after the first encounter of the paramedic team with the patient, saving over 15 min, depending on the time and distance from the scene to hospital [70], at a significantly lower cost than specialized stroke ambulances with portable imaging devices [112];
- Biomarkers able to anticipate successful recanalization (e.g., reduced levels of inflammation-associated α2-antiplasmin and thrombin-activatable fibrinolysis inhibitor (TAFI) or C-Reactive Protein) [113,114,115], could guide adjuvant therapies (e.g., growth factors administration) [116] to improve the efficacy of thrombolytic iv treatment in centers where mechanical thrombectomy is not readily available or when thrombectomy is not recommended (distal clots with low NIHSS at presentation and high pretreatment modified Rankin scale) [117]. In addition, biomarkers that predict the risk of hemorrhagic transformation after iv thrombolysis or mechanical recanalization (e.g., cellular Fibronectin (c-Fn)) could be measured with POC diagnostic platforms preventing damaging interventions [79];
- The identification of the stroke subtype in the pre-hospital setting would be more sensitive. For instance, the earlier recognition of patients with large vessel occlusions would be possible, and the transport for a comprehensive stroke center would be ensured (Figure 3), reducing the need for secondary transfers (saving up to 100 min) and reducing the time from symptoms onset to mechanical thrombectomy in a timely fashion [32]. The inverse is also applicable to the identification of cases in which mechanical thrombectomy would not be a valuable strategy and would save time and avoid the inappropriate use of comprehensive stroke facilities [118].
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lindsay, M.P.; Norrving, B.; Sacco, R.L.; Brainin, M.; Hacke, W.; Martins, S.; Pandian, J.; Feigin, V. World Stroke Organization (WSO): Global Stroke Fact Sheet. Int. J. Stroke 2019, 14, 806–817. [Google Scholar] [CrossRef]
- Mirzaei, H. Stroke in Women: Risk Factors and Clinical Biomarkers. J. Cell. Biochem. 2017, 118, 4191–4202. [Google Scholar] [CrossRef]
- Arboix, A.; Cartanyà, A.; Lowak, M.; García-Eroles, L.; Parra, O.; Oliveres, M.; Massons, J. Gender differences and woman-specific trends in acute stroke: Results from a hospital-based registry (1986–2009). Clin. Neurol. Neurosurg. 2014, 127, 19–24. [Google Scholar] [CrossRef]
- Marrugat, J.; Arboix, A.; García-Eroles, L.; Salas, T.; Vila, J.; Castell, C.; Tresserras, R.; Elosua, R. Estimación de la incidencia poblacional y la mortalidad de la enfermedad cerebrovascular establecida isquémica y hemorrágica en 2002. Rev. Esp. Cardiol. 2007, 60, 573–580. [Google Scholar] [CrossRef]
- Maas, W.J.; Lahr, M.M.H.; Buskens, E.; Van Der Zee, D.J.; Uyttenboogaart, M. Pathway design for acute stroke care in the era of endovascular thrombectomy: A critical overview of optimization efforts. Stroke 2020, 3452–3460. [Google Scholar] [CrossRef]
- Sandercock, P.; Wardlaw, J.M.; Lindley, R.I.; Dennis, M.; Cohen, G.; Murray, G.; Innes, K.; Venables, G.; Czlonkowska, A.; Kobayashi, A.; et al. The benefits and harms of intravenous thrombolysis with recombinant tissue plasminogen activator within 6 h of acute ischaemic stroke (the third international stroke trial [IST-3]): A randomised controlled trial. Lancet 2012, 379, 2352–2363. [Google Scholar] [PubMed] [Green Version]
- Hacke, W.; Kaste, M.; Bluhmki, E.; Brozman, M.; Dávalos, A.; Guidetti, D.; Larrue, V.; Lees, K.R.; Medeghri, Z.; Machnig, T.; et al. Thrombolysis with Alteplase 3 to 4.5 Hours after Acute Ischemic Stroke. N. Engl. J. Med. 2008, 359, 1317–1329. [Google Scholar] [CrossRef] [Green Version]
- Emberson, J.; Lees, K.R.; Lyden, P.; Blackwell, L.; Albers, G.; Bluhmki, E.; Brott, T.; Cohen, G.; Davis, S.; Donnan, G.; et al. Effect of treatment delay, age, and stroke severity on the effects of intravenous thrombolysis with alteplase for acute ischaemic stroke: A meta-analysis of individual patient data from randomised trials. Lancet 2014, 384, 1929–1935. [Google Scholar] [CrossRef] [Green Version]
- Lever, N.M.; Nyström, K.V.; Schindler, J.L.; Halliday, J.; Wira, C.; Funk, M. Missed opportunities for recognition of ischemic stroke in the emergency department. J. Emerg. Nurs. 2013, 39, 434–439. [Google Scholar] [CrossRef]
- Richoz, B.; Hugli, O.; Dami, F.; Carron, P.N.; Faouzi, M.; Michel, P. Acute stroke chameleons in a university hospital. Neurology 2015, 85, 505–511. [Google Scholar] [CrossRef] [PubMed]
- Park, D.; Joo, S.S.; Lee, H.J.; Choi, K.C.; Kim, S.U.; Kim, Y.B. Microtubule-associated protein 2, an early blood marker of ischemic brain injury. J. Neurosci. Res. 2012, 90, 461–467. [Google Scholar] [CrossRef]
- Lanzer, P.; Widimsky, P.; Gorog, D.A.; Mazighi, M.; Liebeskind, D.; Cognard, C. Endovascular thrombectomy 2020: Open issues. Eur. Heart J. Suppl. 2020, 22, M13–M18. [Google Scholar] [CrossRef]
- Misra, S.; Kumar, A.; Kumar, P.; Yadav, A.K.; Mohania, D.; Pandit, A.K.; Prasad, K.; Vibha, D. Blood-based protein biomarkers for stroke differentiation: A systematic review. Proteom. Clin. Appl. 2017, 11, 1–26. [Google Scholar] [CrossRef]
- Aguiar de Sousa, D.; von Martial, R.; Abilleira, S.; Gattringer, T.; Kobayashi, A.; Gallofré, M.; Fazekas, F.; Szikora, I.; Feigin, V.; Caso, V.; et al. Access to and delivery of acute ischaemic stroke treatments: A survey of national scientific societies and stroke experts in 44 European countries. Eur. Stroke J. 2019, 4, 13–28. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Monbailliu, T.; Goossens, J.; Hachimi-Idrissi, S. Blood protein biomarkers as diagnostic tool for ischemic stroke: A systematic review. Biomark. Med. 2017, 11, 503–512. [Google Scholar] [CrossRef] [PubMed]
- Sharma, R.; Gowda, H.; Chavan, S.; Advani, J.; Kelkar, D.; Kumar, G.S.S.; Bhattacharjee, M.; Chaerkady, R.; Prasad, T.S.K.; Pandey, A.; et al. Proteomic signature of endothelial dysfunction identified in the serum of acute ischemic stroke patients by the iTRAQ-based LC-MS approach. J. Proteome Res. 2015, 14, 2466–2479. [Google Scholar] [CrossRef]
- Jickling, G.C.; Sharp, F.R. Biomarker panels in ischemic stroke. Stroke 2015, 46, 915–920. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liberman, A.L.; Prabhakaran, S. Stroke Chameleons and Stroke Mimics in the Emergency Department. Curr. Neurol. Neurosci. Rep. 2017, 17, 15. [Google Scholar] [CrossRef] [PubMed]
- Moulin, S.; Leys, D. Stroke mimics and chameleons. Curr. Opin. Neurol. 2019, 32, 54–59. [Google Scholar] [CrossRef] [PubMed]
- Renjen, P.N.; Chaudhari, D. Stroke chameleons: Uncommon presentations of a common disease. Apollo Med. 2017, 14, 148. [Google Scholar]
- Montaner, J.; Mendioroz, M.; Ribó, M.; Delgado, P.; Quintana, M.; Penalba, A.; Chacón, P.; Molina, C.; Fernández-Cadenas, I.; Rosell, A.; et al. A panel of biomarkers including caspase-3 and d-dimer may differentiate acute stroke from stroke-mimicking conditions in the emergency department. J. Intern. Med. 2011, 270, 166–174. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sharma, R.; Macy, S.; Richardson, K.; Lokhnygina, Y.; Laskowitz, D.T. A blood-based biomarker panel to detect acute stroke. J. Stroke Cerebrovasc. Dis. 2014, 23, 910–918. [Google Scholar] [CrossRef] [PubMed]
- McClelland, G.; Rodgers, H.; Flynn, D.; Price, C.I. The frequency, characteristics and aetiology of stroke mimic presentations: A narrative review. Eur. J. Emerg. Med. 2019, 26, 2–8. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Briard, J.N.; Zewude, R.T.; Kate, M.P.; Rowe, B.H.; Buck, B.; Butcher, K.; Gioia, L.C. Stroke Mimics Transported by Emergency Medical Services to a Comprehensive Stroke Center: The Magnitude of the Problem. J. Stroke Cerebrovasc. Dis. 2018, 27, 2738–2745. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ramanathan, R.S.; Wisco, D.; Vela-Duarte, D.; Zafar, A.; Taqui, A.; Winners, S.; Buletko, A.B.; Hustey, F.; Reimer, A.; Russman, A.; et al. Pre-hospital diagnosis in mobile stroke unit. J. Stroke Cerebrovasc. Dis. 2021, 30, 105801. [Google Scholar] [CrossRef] [PubMed]
- Kalra, L.P.; Khatter, H.; Ramanathan, S.; Sapehia, S.; Devi, K.; Kaliyaperumal, A.; Bal, D.; Sebastian, I.; Kakarla, R.; Singhania, A.; et al. Serum GFAP for stroke diagnosis in regions with limited access to brain imaging (BE FAST India). Eur. Stroke J. 2021, 6, 176–184. [Google Scholar] [CrossRef]
- Bustamante, A.; Penalba, A.; Orset, C.; Azurmendi, L.; Llombart, V.; Simats, A.; Pecharroman, E.; Ventura, O.; Ribó, M.; Vivien, D.; et al. Blood Biomarkers to Differentiate Ischemic and Hemorrhagic Strokes. Neurology 2021, 96, e1928–e1939. [Google Scholar] [CrossRef] [PubMed]
- Atkinson Jr, A.J.; Colburn, W.A.; DeGruttola, V.G.; DeMets, D.L.; Downing, G.J.; Hoth, D.F.; Oates, J.A.; Peck, C.C.; Schooley, R.T.; Spilker, B.A. Biomarkers and surrogate endpoints: Preferred definitions and conceptual framework. Clin. Pharmacol. Ther. 2001, 69, 89–95. [Google Scholar]
- Wang, L.; Song, Q.; Wang, C.; Wu, S.; Deng, L.; Li, Y.; Zheng, L.; Liu, M. Neutrophil to lymphocyte ratio predicts poor outcomes after acute ischemic stroke: A cohort study and systematic review. J. Neurol. Sci. 2019, 406, 116445. [Google Scholar] [CrossRef] [PubMed]
- Kamtchum-Tatuene, J.; Jickling, G.C. Blood Biomarkers for Stroke Diagnosis and MANAGEMENT. Neuromol. Med. 2020, 21, 344–368. [Google Scholar] [CrossRef]
- Sibon, I.; Rouanet, F.; Meissner, W.; Orgogozo, J.M. Use of the Triage Stroke Panel in a neurologic emergency service. Am. J. Emerg. Med. 2009, 27, 558–562. [Google Scholar] [CrossRef] [PubMed]
- Froehler, M.T.; Saver, J.L.; Zaidat, O.O.; Jahan, R.; Aziz-Sultan, M.A.; Klucznik, R.P.; Haussen, D.C.; Hellinger, F.R.; Yavagal, D.R.; Yao, T.L.; et al. Interhospital Transfer Before Thrombectomy Is Associated With Delayed Treatment and Worse Outcome in the STRATIS Registry (Systematic Evaluation of Patients Treated With Neurothrombectomy Devices for Acute Ischemic Stroke). Circulation 2017, 136, 2311–2321. [Google Scholar] [CrossRef] [PubMed]
- Dagonnier, M.; Donnan, G.A.; Davis, S.M.; Dewey, H.M.; Howells, D.W. Acute Stroke Biomarkers: Are We There Yet? Front. Neurol. 2021, 12, 1–16. [Google Scholar] [CrossRef]
- Kim, M.H.; Kang, S.Y.; Kim, M.C.; Lee, W.I. Plasma biomarkers in the diagnosis of acute ischemic stroke. Ann. Clin. Lab. Sci. 2010, 40, 336–341. [Google Scholar] [PubMed]
- Lynch, J.R.; Blessing, R.; White, W.D.; Grocott, H.P.; Newman, M.F.; Laskowitz, D.T. Novel Diagnostic Test for Acute Stroke. Stroke 2004, 35, 57–63. [Google Scholar] [CrossRef] [Green Version]
- Reynolds, M.A.; Kirchick, H.J.; Dahlen, J.R.; Anderberg, J.M.; McPherson, P.H.; Nakamura, K.K.; Laskowitz, D.T.; Valkirs, G.E.; Buechler, K.F. Early biomarkers of stroke. Clin. Chem. 2003, 49, 1733–1739. [Google Scholar] [CrossRef] [PubMed]
- Jickling, G.C.; Sharp, F.R. Blood Biomarkers of Ischemic Stroke. Neurotherapeutics 2011, 8, 349–360. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maestrini, I.; Ducroquet, A.; Moulin, S.; Leys, D.; Cordonnier, C.; Bordet, R. General review Blood biomarkers in the early stage of cerebral ischemia. Rev. Neurol. 2016, 172, 198–219. [Google Scholar] [CrossRef] [PubMed]
- Katan, M.; Elkind, M.S. The potential role of blood biomarkers in patients with ischemic stroke. Clin. Transl. Neurosci. 2018, 2. [Google Scholar] [CrossRef] [Green Version]
- Makris, K.; Haliassos, A.; Chondrogianni, M.; Tsivgoulis, G. Blood biomarkers in ischemic stroke: Potential role and challenges in clinical practice and research. Crit. Rev. Clin. Lab. Sci. 2018, 55, 294–328. [Google Scholar] [CrossRef] [PubMed]
- Dambinova, S.A.; Bettermann, K.; Glynn, T.; Tews, M.; Olson, D.; Weissman, J.D.; Sowell, R.L. Diagnostic potential of the NMDA receptor peptide assay for acute ischemic stroke. PLoS ONE 2012, 7, e42362. [Google Scholar] [CrossRef] [Green Version]
- Rahmati, M.; Azarpazhooh, M.R.; Ehteram, H.; Ferns, G.A.; Ghayour-Mobarhan, M.; Ghannadan, H.; Mobarra, N. The elevation of S100B and downregulation of circulating miR-602 in the sera of ischemic stroke (IS) patients: The emergence of novel diagnostic and prognostic markers. Neurol. Sci. 2020, 41, 2185–2192. [Google Scholar] [CrossRef] [PubMed]
- Park, K.Y.; Ay, I.; Avery, R.; Caceres, J.A.; Siket, M.S.; Pontes-Neto, O.M.; Zheng, H.; Rost, N.S.; Furie, K.L.; Sorensen, A.G.; et al. New biomarker for acute ischaemic stroke: Plasma glycogen phosphorylase isoenzyme BB. J. Neurol. Neurosurg. Psychiatry 2018, 89, 404–409. [Google Scholar] [CrossRef]
- Dambinova, S.A.; Khounteev, G.A.; Izykenova, G.A.; Zavolokov, I.G.; Ilyukhina, A.Y.; Skoromets, A.A. Blood Test Detecting Autoantibodies to N -Methyl- D -aspartate Neuroreceptors for Evaluation of Patients with Transient Ischemic Attack and Stroke. Clin. Chem. 2003, 49, 1752–1762. [Google Scholar] [CrossRef] [Green Version]
- Allard, L.; Burkhard, P.R.; Lescuyer, P.; Burgess, J.A.; Walter, N.; Hocnstrasser, D.F.; Sanchez, J.C. PARK7 and nucleoside diphosphate kinase A as plasma markers for the early diagnosis of stroke. Clin. Chem. 2005, 51, 2043–2051. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Yu, Y.; Xu, W.; Dong, L.; Wang, Y.; Gao, B.; Li, G.; Zhang, W. Apolipoprotein A1-unique peptide as a diagnostic biomarker for acute ischemic stroke. Int. J. Mol. Sci. 2016, 17, 458. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hasan, N.; McColgan, P.; Bentley, P.; Edwards, R.J.; Sharma, P. Towards the identification of blood biomarkers for acute stroke in humans: A comprehensive systematic review. Br. J. Clin. Pharmacol. 2012, 74, 230–240. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- An, S.A.; Kim, J.; Kim, O.J.; Kim, J.K.; Kim, N.K.; Song, J.; Oh, S.H. Limited clinical value of multiple blood markers in the diagnosis of ischemic stroke. Clin. Biochem. 2013, 46, 710–715. [Google Scholar] [CrossRef]
- Laskowitz, D.T.; Kasner, S.E.; Saver, J.; Remmel, K.S.; Jauch, E.C. Clinical usefulness of a biomarker-based diagnostic test for acute stroke: The Biomarker Rapid Assessment in Ischemic Injury (BRAIN) study. Stroke 2009, 40, 77–85. [Google Scholar] [CrossRef] [Green Version]
- Glickman, S.W.; Phillips, S.; Anstrom, K.J.; Laskowitz, D.T.; Cairns, C.B. Discriminative capacity of biomarkers for acute stroke in the emergency department. J. Emerg. Med. 2011, 41, 333–339. [Google Scholar] [CrossRef]
- Saver, J.L. Time is brain—Quantified. Stroke 2006, 37, 263–266. [Google Scholar] [CrossRef] [Green Version]
- Glushakova, O.; Glushakov, A.; Miller, E.; Valadka, A.; Hayes, R. Biomarkers for acute diagnosis and management of stroke in neurointensive care units. Brain Circ. 2016, 2, 28. [Google Scholar] [CrossRef]
- Xiong, L.; Yang, Y.; Zhang, M.; Xu, W. The use of serum glial fibrillary acidic protein test as a promising tool for intracerebral hemorrhage diagnosis in Chinese patients and prediction of the short-term functional outcomes. Neurol. Sci. 2015, 36, 2081–2087. [Google Scholar] [CrossRef]
- Ren, C.; Kobeissy, F.; Alawieh, A.; Li, N.; Li, N.; Zibara, K.; Zoltewicz, S.; Guingab-Cagmat, J.; Larner, S.F.; Ding, Y.; et al. Assessment of serum UCH-L1 and GFAP in acute stroke patients. Sci. Rep. 2016, 14, 24588. [Google Scholar] [CrossRef] [Green Version]
- Llombart, V.; García-Berrocoso, T.; Bustamante, A.; Giralt, D.; Rodriguez-Luna, D.; Muchada, M.; Penalba, A.; Boada, C.; Hernández-Guillamon, M.; Montaner, J. Plasmatic retinol-binding protein 4 and glial fibrillary acidic protein as biomarkers to differentiate ischemic stroke and intracerebral hemorrhage. J. Neurochem. 2016, 136, 416–424. [Google Scholar] [CrossRef] [PubMed]
- Foerch, C.; Niessner, M.; Back, T.; Bauerle, M.; De Marchis, G.M.; Ferbert, A.; Grehl, H.; Hamann, G.F.; Jacobs, A.; Kastrup, A.; et al. Diagnostic accuracy of plasma glial fibrillary acidic protein for differentiating intracerebral hemorrhage and cerebral ischemia in patients with symptoms of acute stroke. Clin. Chem. 2012, 58, 237–245. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Luger, S.; Witsch, J.; Dietz, A.; Hamann, G.F.; Minnerup, J.; Schneider, H.; Sitzer, M.; Wartenberg, K.E.; Niessner, M.; Foerch, C. Glial fibrillary acidic protein serum levels distinguish between intracerebral hemorrhage and cerebral ischemia in the early phase of stroke. Clin. Chem. 2017, 63, 377–385. [Google Scholar] [CrossRef] [PubMed]
- Eng, L.F.; Ghirnikar, R.S.; Lee, Y.L. Glial Fibrillary Acidic Protein: GFAP-Thirty-One Years (1969–2000). Neurochem. Res. 2000, 25, 1439–1451. [Google Scholar] [CrossRef]
- Kumar, A.; Misra, S.; Yadav, A.K.; Sagar, R.; Verma, B.; Grover, A.; Prasad, K. Role of glial fibrillary acidic protein as a biomarker in differentiating intracerebral haemorrhage from ischaemic stroke and stroke mimics: A meta-analysis. Biomarkers 2020, 25, 1–8. [Google Scholar] [CrossRef]
- Zhou, S.; Bao, J.; Wang, Y.; Pan, S. S100β as a biomarker for differential diagnosis of intracerebral hemorrhage and ischemic stroke. Neurol. Res. 2016, 38, 327–332. [Google Scholar] [CrossRef]
- Luger, S.; Jæger, H.S.; Dixon, J.; Bohmann, F.O.; Schaefer, J.H.; Richieri, S.P.; Larsen, K.; Hov, M.R.; Bache, K.G.; Foerch, C. Diagnostic Accuracy of Glial Fibrillary Acidic Protein and Ubiquitin Carboxy-Terminal Hydrolase-L1 Serum Concentrations for Differentiating Acute Intracerebral Hemorrhage from Ischemic Stroke. Neurocrit. Care 2020, 33, 39–48. [Google Scholar] [CrossRef]
- Montaner, J.; Mendioroz, M.; Delgado, P.; García-Berrocoso, T.; Giralt, D.; Merino, C.; Ribó, M.; Rosell, A.; Penalba, A.; Fernández-Cadenas, I.; et al. Differentiating ischemic from hemorrhagic stroke using plasma biomarkers: The S100B/RAGE pathway. J. Proteom. 2012, 75, 4758–4765. [Google Scholar] [CrossRef] [PubMed]
- Senn, R.; Elkind, M.S.V.; Montaner, J.; Christ-Crain, M.; Katan, M. Potential role of blood biomarkers in the management of nontraumatic intracerebral hemorrhage. Cerebrovasc. Dis. 2014, 38, 395–409. [Google Scholar] [CrossRef] [Green Version]
- Bustamante, A.; López-Cancio, E.; Pich, S.; Penalba, A.; Giralt, D.; García-Berrocoso, T.; Ferrer-Costa, C.; Gasull, T.; Hernández-Pérez, M.; Millan, M.; et al. Blood Biomarkers for the Early Diagnosis of Stroke: The Stroke-Chip Study. Stroke 2017, 48, 2419–2425. [Google Scholar] [CrossRef] [PubMed]
- Walsh, K.B.; Unruh, D.; Hart, K.; Roll, S.; Sperling, M.; Davidson, W.S.; Lindsell, C.J. Apolipoprotein A-I and Paraoxonase-1 Are Potential Blood Biomarkers for Ischemic Stroke Diagnosis. J. Stroke Cerebrovasc. Dis. 2016, 25, 1360–1365. [Google Scholar] [CrossRef] [Green Version]
- Lopez, M.F.; Sarracino, D.A.; Prakash, A.; Athanas, M.; Krastins, B.; Rezai, T.; Sutton, J.N.; Peterman, S.; Gvozdyak, O.; Chou, S.; et al. Discrimination of ischemic and hemorrhagic strokes using a multiplexed, mass spectrometry-based assay for serum apolipoproteins coupled to multi-marker ROC algorithm. Proteom. Clin. Appl. 2012, 6, 190–200. [Google Scholar] [CrossRef] [PubMed]
- Qin, C.; Zhao, X.L.; Ma, X.T.; Zhou, L.Q.; Wu, L.; Shang, K.; Wang, W. Proteomic profiling of plasma biomarkers in acute ischemic stroke due to large vessel occlusion. J. Transl. Med. 2019, 17, 214. [Google Scholar] [CrossRef]
- Kavalci, C.; Genchallac, H.; Durukan, P.O.L.A.T.; Cevik, Y. Value of biomarker-based diagnostic test in differential diagnosis of hemorrhagic-ischemic stroke. Bratisl. Lek. Listy 2011, 112, 398–401. [Google Scholar]
- Vanni, S.; Polidori, G.; Pepe, G.; Chiarlone, M.; Albani, A.; Pagnanelli, A.; Grifoni, S. Use of biomarkers in triage of patients with suspected stroke. J. Emerg. Med. 2011, 40, 499–505. [Google Scholar] [CrossRef] [PubMed]
- Koch, P.M.; Kunz, A.; Ebinger, M.; Geisler, F.; Rozanski, M.; Waldschmidt, C.; Weber, J.E.; Wendt, M.; Winter, B.; Zieschang, K.; et al. Influence of Distance to Scene on Time to Thrombolysis in a Specialized Stroke Ambulance. Stroke 2016, 47, 2136–2140. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, R.; Ye, X.; Cui, T. Recent progress of biomarker detection sensors. Research 2020, 2020, 7949037. [Google Scholar] [CrossRef]
- Block, D.R.; Ouverson, L.J.; Wittwer, C.A.; Saenger, A.K.; Baumann, N.A. An approach to analytical validation and testing of body fluid assays for the automated clinical laboratory. Clin. Biochem. 2018, 58, 44–52. [Google Scholar] [CrossRef]
- Dincer, C.; Bruch, R.; Kling, A.; Dittrich, P.S.; Urban, G.A. Multiplexed Point-of-Care Testing—xPOCT. Trends Biotechnol. 2017, 35, 728–742. [Google Scholar] [CrossRef] [Green Version]
- Harpaz, D.; Eltzov, E.; Seet, R.C.S.; Marks, R.S.; Tok, A.I.Y. Point-of-care-testing in acute stroke management: An unmet need ripe for technological Harvest. Biosensors 2017, 7, 30. [Google Scholar] [CrossRef] [Green Version]
- Abreu, C.M.; Soares-dos-Reis, R.; Melo, P.N.; Relvas, J.B.; Guimarães, J.; Sá, M.J.; Cruz, A.P.; Pinto, I.M. Emerging biosensing technologies for neuroinflammatory and neurodegenerative disease diagnostics. Front. Mol. Neurosci. 2018, 11, 164. [Google Scholar] [CrossRef]
- Clerico, A.; Franzini, M.; Masotti, S.; Prontera, C.; Passino, C. State of the art of immunoassay methods for B-type natriuretic peptides: An update. Crit. Rev. Clin. Lab. Sci. 2015, 52, 56–69. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Wang, R.; Luo, F.; Wang, P.; Lin, Z. Miniaturized electrochemical sensors and their point-of-care applications. Chin. Chem. Lett. 2020, 31, 589–600. [Google Scholar] [CrossRef]
- Development of Stroke Point of Care Immunoassay for Cellular Fibronectin. Available online: https://www.sbir.gov/sbirsearch/detail/280110 (accessed on 4 July 2021).
- Fernandes, E.; Sobrino, T.; Martins, V.C.; Lopez-loureiro, I.; Campos, F.; Germano, J.; Rodríguez-Pérez, M.; Cardoso, S.; Petrovykh, D.Y.; Castillo, J.; et al. Point-of-care quantification of serum cellular fibronectin levels for stratification of ischemic stroke patients. Nanomed. Nanotechnol. Biol. Med. 2020, 30, 102287. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Vega, G.; García-Robaina, A.; Ben Ismail, M.; Pasamar, H.; García-Berrocoso, T.; Montaner, J.; Zourob, M.; Othmane, A.; del Campo, F.J.; Baldrich, E. Detection of plasma MMP-9 within minutes. Unveiling some of the clues to develop fast and simple electrochemical magneto-immunosensors. Biosens. Bioelectron. 2018, 115, 45–52. [Google Scholar] [CrossRef] [PubMed]
- Cruz, A.; Queirós, R.; Abreu, C.M.; Barata, C.; Fernandes, R.; Silva, R.; Ambrósio, A.F.; Soares-Dos-Reis, R.; Guimarães, J.; Sá, M.J.; et al. Electrochemical Immunosensor for TNFα-Mediated Inflammatory Disease Screening. ACS Chem. Neurosci. 2019, 10, 2676–2682. [Google Scholar] [CrossRef] [PubMed]
- Kuo, Y.C.; Lee, C.K.; Lin, C.T. Improving sensitivity of a miniaturized label-free electrochemical biosensor using zigzag electrodes. Biosens. Bioelectron. 2018, 103, 130–137. [Google Scholar] [CrossRef]
- Harpaz, D.; Koh, B.; Marks, R.S.; Seet, R.C.S.; Abdulhalim, I.; Tok, A.I.Y. Point-of-Care Surface Plasmon Resonance Biosensor for Stroke Biomarkers NT-proBNP and S100β Using a Functionalized Gold Chip with Specific Antibody. Sensors 2019, 19, 2533. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Agostini, M.; Amato, F.; Vieri, M.L.; Greco, G.; Tonazzini, I.; Baroncelli, L.; Caleo, M.; Vannini, E.; Santi, M.; Signore, G.; et al. Glial-fibrillary-acidic-protein (GFAP) biomarker detection in serum-matrix: Functionalization strategies and detection by an ultra-high-frequency surface-acoustic-wave (UHF-SAW) lab-on-chip. Biosens. Bioelectron. 2021, 172, 112774. [Google Scholar] [CrossRef]
- Eltzov, E.; Guttel, S.; Low Yuen Kei, A.; Sinawang, P.D.; Ionescu, R.E.; Marks, R.S. Lateral Flow Immunoassays—From Paper Strip to Smartphone Technology. Electroanalysis 2015, 27, 2116–2130. [Google Scholar] [CrossRef]
- Sinawang, P.D.; Rai, V.; Ionescu, R.E.; Marks, R.S. Electrochemical lateral flow immunosensor for detection and quantification of dengue NS1 protein. Biosens. Bioelectron. 2016, 77, 400–408. [Google Scholar] [CrossRef] [PubMed]
- Sinawang, P.D.; Harpaz, D.; Fajs, L.; Seet, R.C.S.; Tok, A.I.Y.; Marks, R.S. Electrochemical impedimetric detection of stroke biomarker NT-proBNP using disposable screen-printed gold electrodes. EuroBiotech J. 2017, 1, 165–176. [Google Scholar] [CrossRef] [Green Version]
- Eltzov, E.; Marks, R.S. Colorimetric stack pad immunoassay for bacterial identification. Biosens. Bioelectron. 2017, 87, 572–578. [Google Scholar] [CrossRef]
- Tsai, T.T.; Huang, T.H.; Chen, C.A.; Ho, N.Y.J.; Chou, Y.J.; Chen, C.F. Development a stacking pad design for enhancing the sensitivity of lateral flow immunoassay. Sci. Rep. 2018, 8, 17319. [Google Scholar] [CrossRef] [Green Version]
- Eltzov, E.; Marks, R.S. Miniaturized Flow Stacked Immunoassay for Detecting Escherichia coli in a Single Step. Anal. Chem. 2016, 88, 6441–6449. [Google Scholar] [CrossRef] [PubMed]
- Majdinasab, M.; Mitsubayashi, K.; Marty, J.L. Optical and Electrochemical Sensors and Biosensors for the Detection of Quinolones. Trends Biotechnol. 2019, 37, 898–915. [Google Scholar] [CrossRef]
- Li, H.; Liu, X.; Li, L.; Mu, X.; Genov, R.; Mason, A.J. CMOS electrochemical instrumentation for biosensor microsystems: A review. Sensors 2017, 17, 74. [Google Scholar] [CrossRef]
- Gencoglu, A.; Minerick, A.R. Electrochemical detection techniques in micro- and nanofluidic devices. Microfluid. NanoFluid. 2014, 17, 781–807. [Google Scholar] [CrossRef]
- Hemochron Jr Signature + Coagulation Analyzer—Point-of-Care Coagulation Tests on Fresh whole Blood. Available online: https://www.somatechnology.com/Medical-Products/Coagulation-Analyzers/hemochron-jr-signature-coagulation-analyzers.aspx (accessed on 4 July 2021).
- pocHi-Hematology-Analyzer. Available online: https://www.sysmex.com/us/en/Products/Hematology/3PartDiff/Pages/pocHi-Hematology-Analyzer.aspx (accessed on 4 July 2021).
- i-STAT System. Available online: https://www.pointofcare.abbott/us/en/offerings/istat (accessed on 4 July 2021).
- Reflotron® Plus System and Reflotron® Sprint System. Available online: https://diagnostics.roche.com/global/en/products/instruments/reflotron-plus.html (accessed on 4 July 2021).
- Comparison of Abbott AxSYM and Roche Elecsys 2010 for measurement of BNP and NT-proBNP. Available online: https://www.sciencedirect.com/science/article/abs/pii/S000989810600074X (accessed on 4 July 2021).
- PI, Alere Triage BNP Test for the Beckman Coulter Access Family of Immunoassay Systems. Available online: https://www.quidel.com/immunoassays/triage-test-kits/triage-bnp-test-beckman-coulter-access-systems/tw (accessed on 4 July 2021).
- BNP Test Cartridge | Abbott Point of Care. Available online: https://www.pointofcare.abbott/int/en/offerings/istat/istat-test-cartridges/BNP#:~:text=Thei-STAT (accessed on 4 July 2021).
- TBICheckTM: A Portable Device to Detect Mild Traumatic Brain Injury. Available online: https://tbicheck.com/technology#:~:text=TBICheckTM%3A (accessed on 4 July 2021).
- Prediction Sciences, LLC. Available online: https://www.sbir.gov/sbc/prediction-sciences-llc (accessed on 4 July 2021).
- Rapid Test for Cellular Fibronectin—Prediction Biosciences S.A.S. Available online: https://www.freepatentsonline.com/y2015/0362502.html#google_vignette (accessed on 4 July 2021).
- Intellectual Property & Publications. Available online: http://valtaribio.com/intellectual-property-publications/ (accessed on 4 July 2021).
- Immune Biomarkers for the Diagnosis of Mild Traumatic Brain Injury. Available online: https://content.iospress.com/articles/neurorehabilitation/nre1437 (accessed on 4 July 2021).
- SMARTChip for Early Diagnosis of Suspected Stroke. Available online: http://www.io.nihr.ac.uk/wp-content/uploads/migrated/SMARTChip-Horizon-Scanning-alert-FINAL.pdf (accessed on 4 July 2021).
- Sun, J.; Zhao, Y.; Hou, Y.; Li, H.; Yang, M.; Wang, Y.; Sun, B. Multiplexed electrochemical and SERS dual-mode detection of stroke biomarkers: Rapid screening with high sensitivity. New J. Chem. 2019, 43, 13381–13387. [Google Scholar] [CrossRef]
- Tanak, A.S.; Muthukumar, S.; Krishnan, S.; Schully, K.L.; Clark, D.V.; Prasad, S. Multiplexed cytokine detection using electrochemical point-of-care sensing device towards rapid sepsis endotyping. Biosens. Bioelectron. 2021, 171, 112726. [Google Scholar] [CrossRef]
- Wei, F.; Patel, P.; Liao, W.; Chaudhry, K.; Zhang, L.; Arellano-Garcia, M.; Hu, S.; Elashoff, D.; Zhou, H.; Shukla, S.; et al. Electrochemical sensor for multiplex biomarkers detection. Clin. Cancer Res. 2009, 15, 4446–4452. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Park, S.Y.; Kim, M.H.; Kim, O.J.; Ahn, H.J.; Song, J.Y.; Jeong, J.Y.; Oh, S.H. Plasma heart-type fatty acid binding protein level in acute ischemic stroke: Comparative analysis with plasma S100B level for diagnosis of stroke and prediction of long-term clinical outcome. Clin. Neurol. Neurosurg. 2013, 115, 405–410. [Google Scholar] [CrossRef] [PubMed]
- Zinkstok, S.M.; Engelter, S.T.; Gensicke, H.; Lyrer, P.A.; Ringleb, P.A.; Artto, V.; Putaala, J.; Haapaniemi, E.; Tatlisumak, T.; Chen, Y.; et al. Safety of thrombolysis in stroke mimics: Results from a multicenter cohort study. Stroke 2013, 44, 1080–1084. [Google Scholar] [CrossRef] [Green Version]
- Vibha, D.; Misra, S. Blood Biomarkers for Stroke Differentiation. In Stroke Biomarkers; Peplow, P.V., Dambinova, S.A., Martinez, B., Eds.; Humana Press: New York, NY, USA, 2020; pp. 79–111. ISBN 9781493996810. [Google Scholar]
- Martí-Fàbregas, J.; Borrell, M.; Cocho, D.; Belvís, R.; Castellanos, M.; Montaner, J.; Pagonabarraga, J.; Aleu, A.; Molina-Porcel, L. Hemostatic markers of recanalization in patients with ischemic stroke treated with rt-PA. Neurology 2005, 65, 366–370. [Google Scholar] [CrossRef]
- Zang, N.; Lin, Z.; Huang, K.; Pan, Y.; Wu, Y.; Wu, Y.; Wang, S.; Wang, D.; Ji, Z.; Pan, S. Biomarkers of Unfavorable Outcome in Acute Ischemic Stroke Patients with Successful Recanalization by Endovascular Thrombectomy. Cerebrovasc. Dis. 2020, 49, 583–592. [Google Scholar] [CrossRef]
- Kim, S.; Yi, H.J.; Lee, D.H.; Sung, J.H. Association of High-sensitivity C-reactive Protein with Patient Prognosis Following Mechanical Thrombectomy for Acute Ischemic Stroke. Curr. Neurovasc. Res. 2020, 17, 402–410. [Google Scholar] [CrossRef]
- Castellanos, M.; Brea, D.; Dorado, L.; Sobrino, T.; Milla, M.; Serena, J.; Lez, R.R.G.; Rodri, M. Association of growth factors with arterial recanalization and clinical outcome in patients with ischemic stroke treated with tPA. J. Thromb. Haemost. 2010, 1, 1567–1574. [Google Scholar]
- Powers, W.J.; Rabinstein, A.A.; Ackerson, T.; Adeoye, O.M.; Brown, M.; Demaerschalk, B.M.; Leslie-mazwi, T.M.; Ovbiagele, B.; Scott, P.A.; Sheth, K.N.; et al. Guidelines for the Early Management of Patients With Acute Ischemic Stroke: 2019 Update to the 2018 Guidelines for the Early Management of Acute Ischemic Stroke A Guideline for Healthcare Professionals From the American Heart Association/American. Stroke 2019, 50, 344–418. [Google Scholar] [CrossRef] [PubMed]
- Romoli, M.; Paciaroni, M.; Tsivgoulis, G.; Agostoni, E.C.; Vidale, S. Mothership versus drip-and-ship model for mechanical thrombectomy in acute stroke: A systematic review and meta-analysis for clinical and radiological outcomes. J. Stroke 2020, 22, 317–323. [Google Scholar] [CrossRef] [PubMed]
- Stinear, C.M.; Byblow, W.D.; Ackerley, S.J.; Smith, M.C.; Borges, V.M.; Barber, P.A. PREP2: A biomarker-based algorithm for predicting upper limb function after stroke. Ann. Clin. Transl. Neurol. 2017, 4, 811–820. [Google Scholar] [CrossRef]
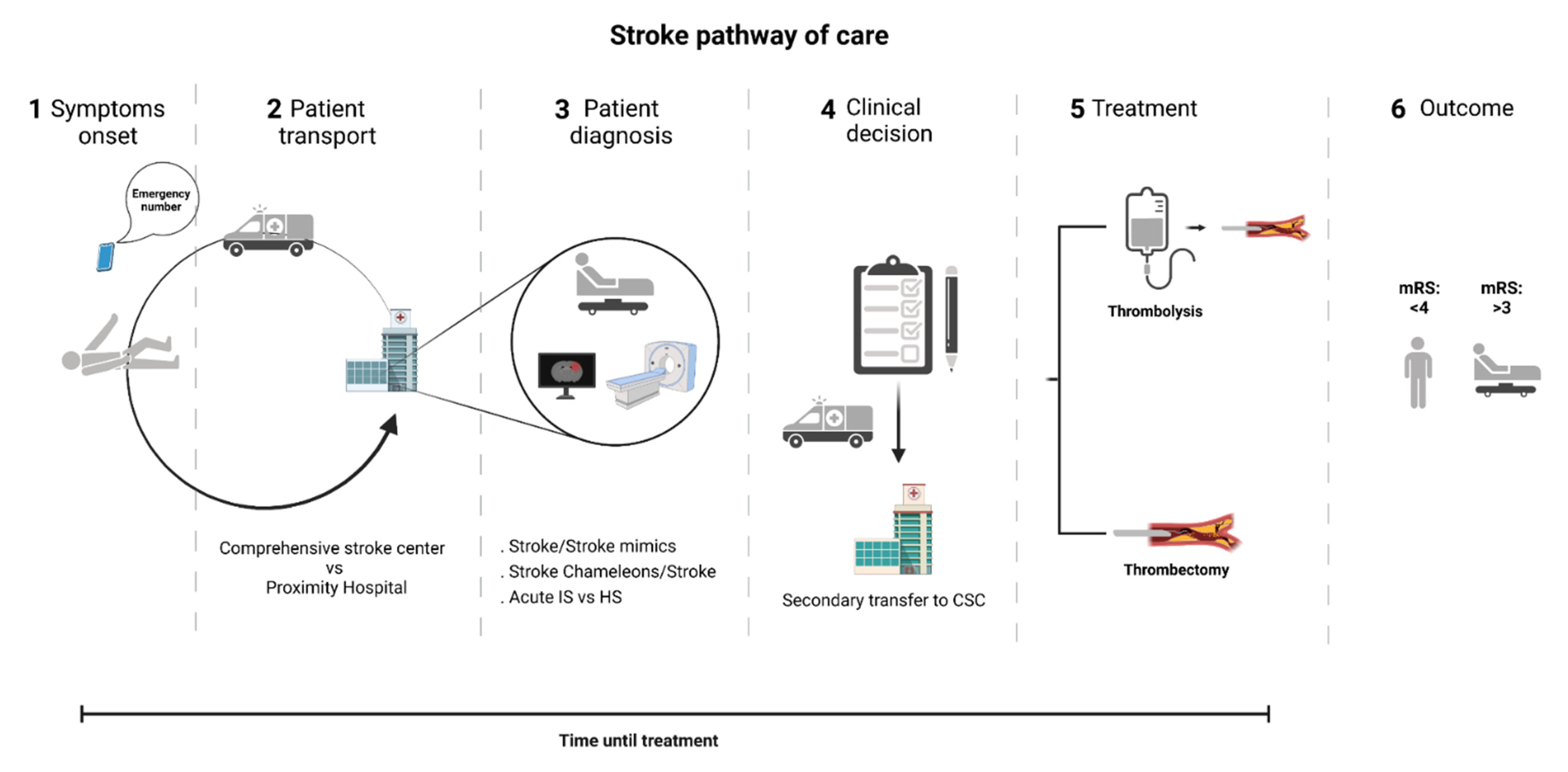
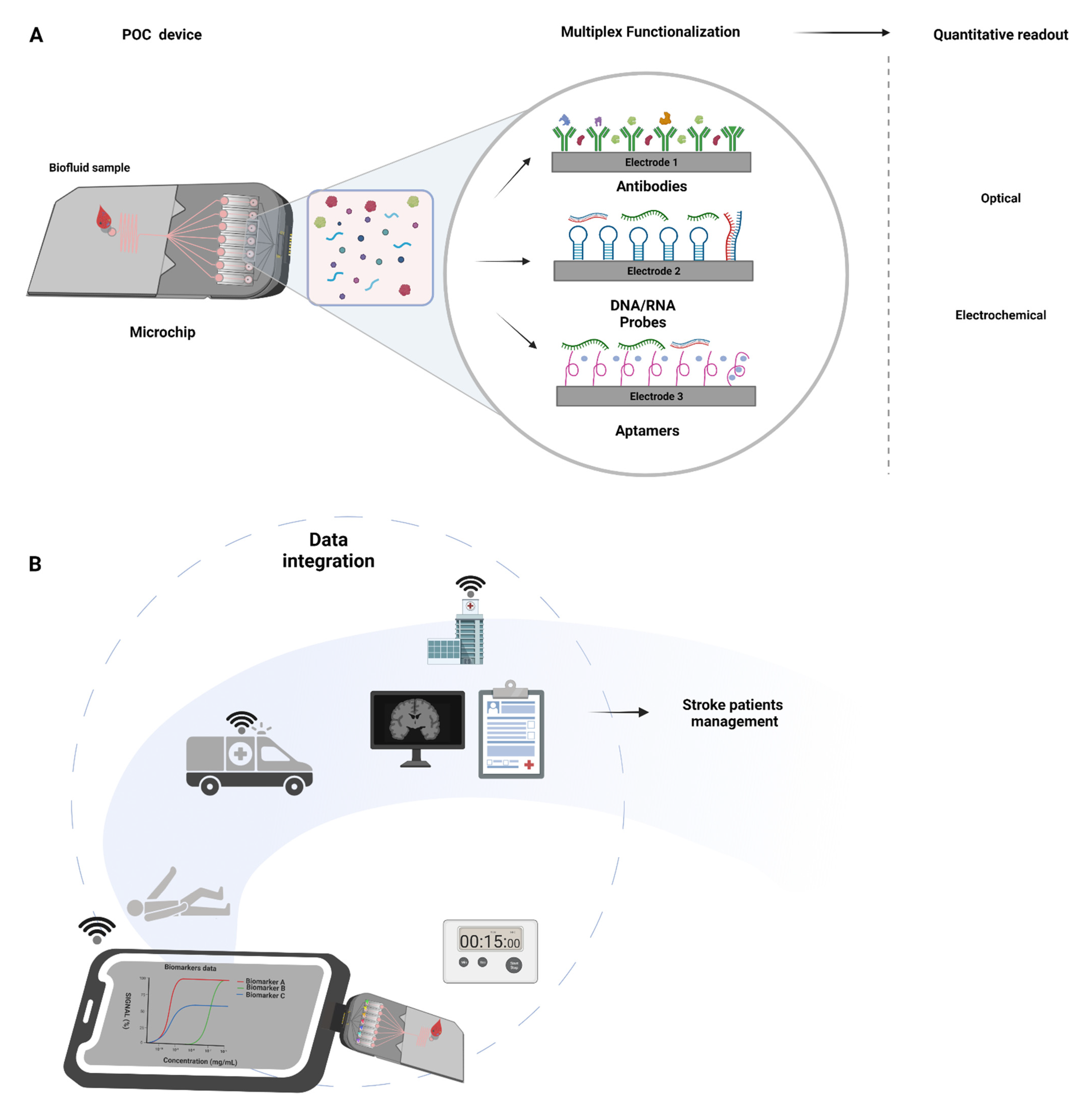
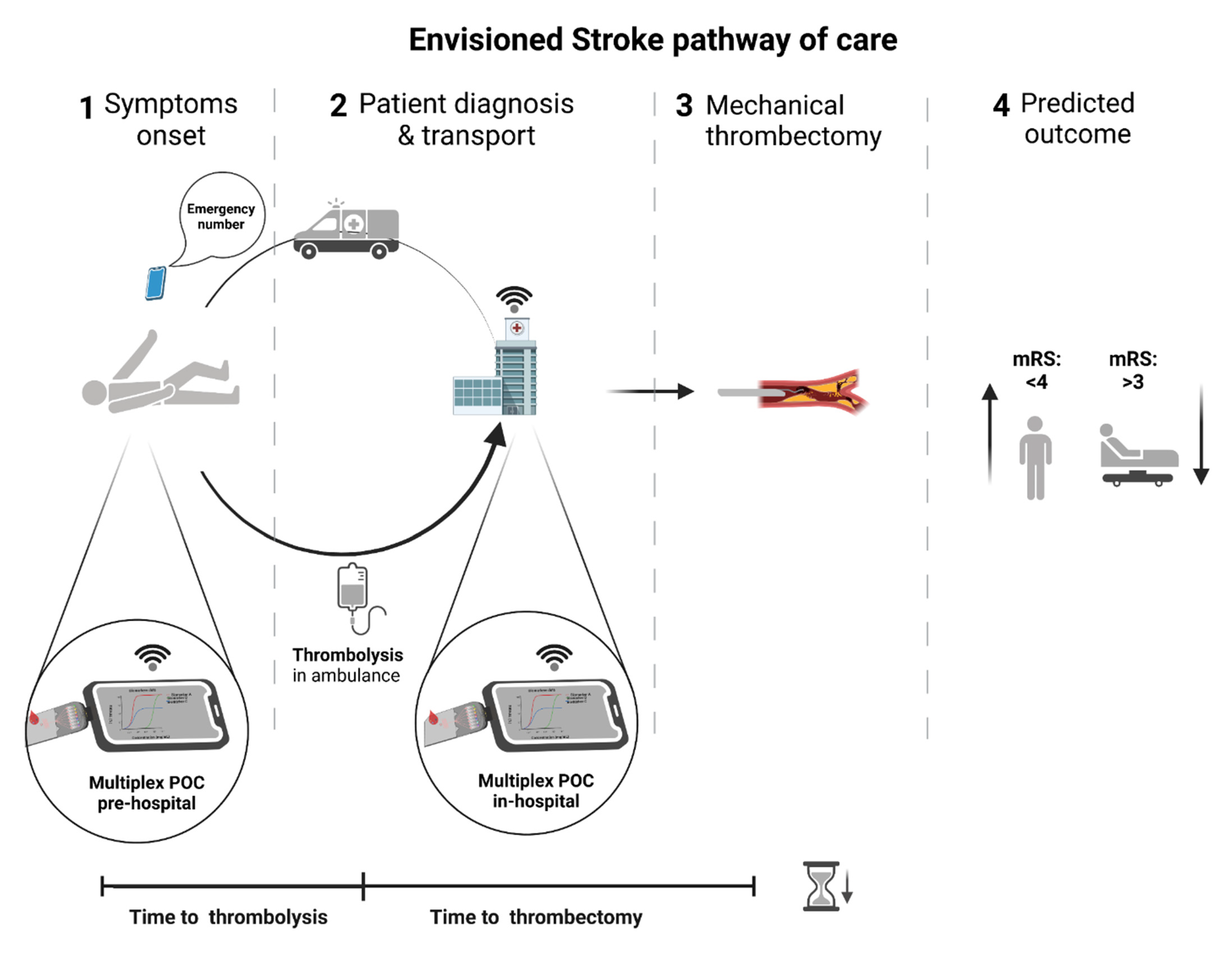
| Biomarker Origin | Protein | Biomarker Level in IS | Biomarker Level in Control | Cut-Off Point | Sensitivity | Specificity | AUC | Study Sample | Reference |
|---|---|---|---|---|---|---|---|---|---|
| Brain Cells | NR2 | 5.4 (0.1–62.7) ng/mL | 0.3 (0.02–1.1) ng/mL | 1 ng/mL | 92.1% | 96.5% | 0.92 | Combined stroke mimics and healthy controls | [41] |
| S100B | N/A | N/A | 39.9 pg/mL | 76.5% | 82.7% | 0.87 | Non-stroke controls | [42] | |
| GPBB | 46.3 (±38.6) ng/mL | 4.1 (±7.6) ng/mL | 7.0 ng/mL | 93.0% | 93.0% | 0.96 | Non-stroke controls | [43] | |
| BNP | 90.8 (±156.4) pg/mL | 11.3 (±6.1) pg/mL | N/A | N/A | N/A | 0.69 | Healthy and stroke mimics | [34] | |
| Anti-NMDA (NR2A/2B ab) | 5.0 (3.2–7.2) ng/mL | 1.5 (1.0–1.9) ng/mL | 2.0 ng/mL | 97.0% | 98.0% | 0.99 | Healthy controls | [44] | |
| Brain Cells, Endothelium/Matrix, Blood | MMP-9 | 242.1 (±242.6) ng/mL | 211.2 (±184.8) ng/mL | N/A | N/A | N/A | 0.55 | Healthy and stroke mimics | [34] |
| PARK 7 | N/A | N/A | 14.2 ng/mL | 58.0% | 90.0% | 0.88 | Healthy controls | [45] | |
| NDKA | N/A | N/A | 22.5 ng/mL | 67.0% | 89.9% | 0.94 | Healthy controls | [45] | |
| Blood | APOA1-UP/LRP | 1.3 (IQR 0.4) | 2.1 (IQR 0.4) | <1.8 | 90.6% | 97.1% | 0.98 | Non-stroke controls | [46] |
| Biomarker Origin | Proteins | Biomarker Level in IS | Biomarker Level in Control | Cut-Off Point | Sensitivity | Specificity | AUC | Study Sample | Reference |
|---|---|---|---|---|---|---|---|---|---|
| Brain Cells, Endothelium/Matrix, Blood | MMP9 | N/A | N/A | N/A | 91.7% | 93.0% | 0.99 | Healthy controls | [36] |
| BNGF | |||||||||
| vWF | |||||||||
| MCP-1 | |||||||||
| S-100B | |||||||||
| Brain Cells, Endothelium/Matrix | Eotaxin | N/A | N/A | N/A | N/A | 0.92 | Stroke mimics | [22] | |
| EGFR | |||||||||
| S100A12 | |||||||||
| TIMP-4 | |||||||||
| Prolactin | |||||||||
| Brain Cells, Endothelium/Matrix, Blood | BNP | 90.8 (±156.4) pg/mL | 11.3 (±6.1) pg/mL | N/A | 91.0% | 21.5% | N/A | Healthy controls and stroke mimics | [34] |
| D-dimer | 888.1 (±1289) ng/mL | 188.6 (±113.8) ng/mL | |||||||
| MMP9 | 242.1 (±242.6) ng/mL | 211.2 (±184.8) ng/mL | |||||||
| S100B | 103.1 (±13.6) pg/mL | 188.6 (±147.1) pg/mL | |||||||
| Brain Cells, Endothelium/Matrix, Blood | IL-6 | 4.0 (0.8–12.3) pg/mL | 1.2 (0.0–2.4) pg/mL | - | N/A | N/A | 0.75 | Stroke mimics | [48] |
| S100B | 63.3 (29.7–122.8) ng/mL | 33.8 (15.4–60.8) ng/mL | |||||||
| MMP-9 | 30.4 (0–115.2) pg/mL | 2.3 (0.0–20.6) pg/mL | |||||||
| Biomarker Origin | Protein | Biomarker Level in IS | Biomarker Level in HS | Cut-Off Point | Sensitivity | Specificity | AUC | Reference |
|---|---|---|---|---|---|---|---|---|
| Brain Cells | GFAP | 0.08 (0.02–0.14) ng/mL | 1.91 (0.41–17.7) ng/mL | 0.30 ng/mL | 84.2% | 96.3% | 0.91 | [56] |
| S100B | 61.7 (±37.3) pg/mL | 161.2 (±79.7) pg/mL | 67.0 pg/mL | 95.7% | 70.4% | 0.90 | [60] | |
| UCH-L1 | 338.0 pg/mL | 401.0 pg/mL | 291.0 pg/mL | 73% | 45.0% | 0.59 | [61] | |
| Endothelium/Matrix | sRAGE | 1.0 ng/mL | 0.8 ng/mL | <0.97 ng/mL | NA | NA | NA | [62] |
| Blood | RBP4 | 59.8 (±12.3) µg/mL | 36.9 (±14.7) µg/mL | 61.0 µg/mL | 68.4% | 84.0% | NA | [55] |
| Biomarkers Origin | Proteins | Biomarker Level in Ischemic Stroke | Biomarker Level in Hemorrhagic Stroke | Cut-Off Point | Sensitivity | Specificity | AUC | Reference |
|---|---|---|---|---|---|---|---|---|
| Brain Cells, Endothelium/Matrix, Blood | RBP-4 | 29.2 (25.1–35.7) μg/mL | 34.4 (26.0–40.0) μg/mL | 38.0 μg/mL | 51.5% | 100% | N/A | [27] |
| NT-proBNP | 0.8 (0.2–2.4) ng/mL | 0.4 (0.2–0.7) ng/mL | 1.3 ng/mL | |||||
| GFAP | 186.3 (132.8–280.2) pg/mL | 1699.6 (411.1–10,145.4) pg/mL | 325 pg/mL | |||||
| sRAGE | 1.0 ng/mL | 0.8 ng/mL | <0.9 ng/mL | 22.7% | 80.2% | 0.76 | [62] | |
| S100B | 58.7 pg/mL | 107.7 pg/mL | 96.0 pg/mL |
| POC Device | Analytical Platform | Blood Biomarkers | Application | Reference |
|---|---|---|---|---|
| Hemochron® Junior | Optical | ACT-LR, ACT, PT, Citrate PT, APTT, and Citrate APTT | Pre- and In-hospital | [94] |
| PocH-100i Hematology Analyzer | Hydrodynamics/Impedance | Full blood cell count | Pre- and In-hospital | [95] |
| i-STAT | Electrochemical | Blood gases, electrolytes, metabolites, and coagulation | Pre- and In-hospital | [96] |
| Reflotron® plus analyzer | Optical | c-glutamyltransferase, p-amylase, glucose | Pre- and In-hospital | [97] |
| AxSYM® BNP | Optical | BNP | In/Post-hospital | [98] |
| Triage® BNP | Optical | BNP | In/Post-hospital | [99] |
| iSTAT BNP | Electrochemical | BNP | In/Post-hospital | [100] |
| TBI Check® | N/A * | H-FABP and GFAP | Pre- and In-hospital | [101] |
| Prediction Sciences LLC | Optical | c-Fn | In-hospital | [102,103] |
| ReSTTM | N/A * | Immune response | In-hospital | [104,105] |
| SMARTChip | Electrochemical | Purines | In-hospital | [106] |
| POC | Modality | Analytical Platform | Multiplex Capacity * | On-Site Analysis | Reference |
|---|---|---|---|---|---|
| µPADs | Paper-based system | Optical | ≥2 biomarkers | Yes | [73] |
| Stack Pad | Paper-based system | Optical | >2 biomarkers | Yes | [88,89,90,106] |
| ELFI | Paper-based system | Electrochemical | >2 biomarkers | Yes | [85,86,87] |
| EIS-SERS | Paper-based system | Electrochemical and surface-enhanced Raman spectroscopy | ≥2 biomarkers | Yes | [107] |
| EIS | Array-based system | Electrochemical | ≥5 biomarkers | Yes | [108,109] |
| MuitiLab | Microfluidic-based system | Electrochemical | ≥8 biomarkers | Yes | [73] |
| mChip | Microfluidic-based system | Optical | ≥5 biomarkers | Yes | [73] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dias, A.; Silva, I.; Pinto, I.M.; Maia, L.F. Timely and Blood-Based Multiplex Molecular Profiling of Acute Stroke. Life 2021, 11, 816. https://doi.org/10.3390/life11080816
Dias A, Silva I, Pinto IM, Maia LF. Timely and Blood-Based Multiplex Molecular Profiling of Acute Stroke. Life. 2021; 11(8):816. https://doi.org/10.3390/life11080816
Chicago/Turabian StyleDias, Alexandre, Isabel Silva, Inês Mendes Pinto, and Luís F. Maia. 2021. "Timely and Blood-Based Multiplex Molecular Profiling of Acute Stroke" Life 11, no. 8: 816. https://doi.org/10.3390/life11080816






